Vaccinia
R. Mark Buller
Jay S. Pepose
INTRODUCTION
Poxviruses infect the eye during endogenous viremia or after exogenous inoculation from another site. Historically, in unvaccinated individuals, smallpox and human monkeypox were associated with (corneal) blindness at a frequency of 0.45% to 0.9% and 1.7%, respectively.1,2 With the eradication of smallpox in 1979, and the paucity of human monkeypox outside of Central Africa, this complication is rarely seen. The majority of poxvirus-associated ocular disease is caused by molluscum contagiosum virus after a natural infection3 and by vaccinia virus as a complication of the smallpox vaccine.
Ocular vaccinial infections result from autoinoculation from a smallpox vaccination site before scab formation or from contact with a recently vaccinated individual. Ocular lesions are characterized as pustular blepharitis, preseptal cellulitis, conjunctivitis, epithelial keratitis, and stromal keratitis sometimes accompanied by iritis.4 These complications are currently lower than the historic average because of enhanced clinical care associated with smallpox vaccination—especially the use of occlusive bandages over the vaccination site until lesion resolution. In the event of a bioterrorist release of variola virus and the need to vaccinate many citizens quickly, it is conceivable that ocular vaccinial complications could revisit or exceed previous rates if vaccination precautions are not implemented.
VIROLOGY
CLASSIFICATION
The earliest classification of poxviruses was based on disease symptoms and gross pathological manifestations. Later criteria included morphological characterization of virions and staining patterns of cytoplasmic inclusion bodies in infected cells. The poxvirus family is divided into two subfamilies: Entomopoxvirinae (poxviruses of insects) and Chordopoxvirinae (poxviruses of vertebrates). The Chordopoxvirinae are further classified into genera through studies of cross-protection in animals and cross-neutralization in tissue culture. Species of the same genera cross-protect or cross-neutralize each other and share similar biological properties. Current taxonomic classification is based on viral genomic DNA sequences. Vaccinia virus is the prototypic member of the orthopoxvirus genus along with closely related cowpox, ectromelia, monkeypox, and variola (smallpox) viruses.
VIRION MORPHOLOGY
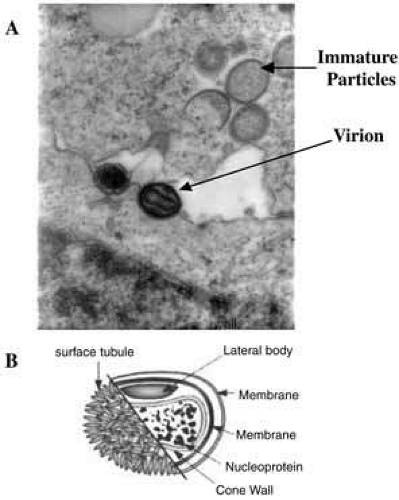
Poxviruses are the largest of all animal viruses and can just be visualized by light microscopy. High-resolution electron microscopy shows virions to be oval or brick-shaped structures 200 to 400 nm in length, with axial ratios of 1.2 to 1.7 (Fig. 1A). Images of electron microscopic thin sections show the surface membrane to enclose a biconcave or cylindrical core that contains genomic DNA.
The vaccinial genome comprises a linear molecule of double-stranded DNA with covalently closed termini. Terminal hairpins constitute two isomeric, imperfectly paired, “flip-flop” DNA forms consisting of inverted complementary sequences. Variably sized, tandem repeat sequence arrays are present near the ends. Each virion contains one DNA molecule associated with proteins and organized in a nucleoprotein core. One or two lateral bodies are present in the concave region between the core wall and a single membrane, which contains a number of virus-encoded proteins. This virion form is known as the intracellular mature virus (Fig. 1B).
REPLICATION CYCLE
The intracellular replication cycle of vaccinia virus involves a sequence of events (Fig. 2).5 The vaccinia virion containing early RNA transcription machinery attaches to and fuses with the plasma membrane. Within 15 minutes the virion transcription machinery is activated (uncoating I). Early genes are expressed that code for a variety of functions that modify the host cell for optimal virus replication, attenuate the host response to infection, and mediate viral synthetic processes. After further uncoating (uncoating II), between 1.5 to 6 hours postinfection, the viral genome is replicated via concatamers.
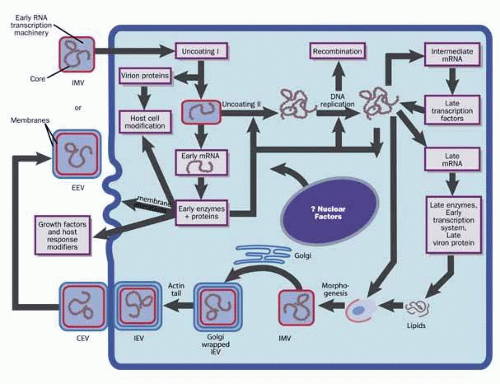 Fig. 2 The cytoplasmic vaccinia virus replication cycle. IMV, intracellular mature virus; IEV, intracellular enveloped virus; CEV, cell-associated enveloped virus; EEV, extracellular enveloped virus. |
From progeny DNA templates, late transcription factors are expressed from intermediate genes, and late gene RNA is synthesized. Late genes encode the early transcription system, enzymes, and structural proteins necessary for virion assembly. By 4 hours postinfection, virion morphogenesis commences with the formation of membrane structures in the intermediate compartment and the packaging of resolved unit-length genomic DNA. The intracellular mature virus (IMV) has one membrane derived from the intermediate compartment. Some IMVs acquire an additional double layer of intracellular membrane derived from the trans Golgi network that contains unique virus proteins, the intracellular enveloped virus (IEV).
IEVs are transported to the periphery of the cell where fusion with the plasma membrane ultimately results in release of extracellular enveloped virus (EEV) or, if attached to the exterior surface of the plasma membrane, as cell-associated enveloped virus (CEV). While IMV and CEV/EEV are infectious, the external antigens on the two virion forms are different. Thus, each virion type probably binds to different cellular receptors and are likely uncoated by different mechanisms. EEVs are thought to be the most important form involved in cell-to-cell spread and systemic disease.
PREDISPOSING FACTORS
Exposure can occur by accidental autoinoculation of the eye or eyelid with virus from the inoculation site or from close physical contact with a vaccinee shedding virus before eschar detachment.6 The majority of ocular manifestations of smallpox vaccination in immunocompetent patients are self-limiting. Selective cases may require treatment with topical trifluridine, topical corticosteroids, and vaccinia immune globulin (VIG). More severe ocular and periocular manifestations reflect a higher viral inoculum, increased local viral replication, and host immune response.
RISK FACTORS
Several factors predispose to serious systemic complications that can follow vaccination or contact with a vaccinated person. In patients with impaired T-cell function, the primary vaccination site may not heal and the infection can spread both locally and by viremia. This condition, known as vaccinia necrosum (i.e., progressive vaccinia), produces necrotic lesions, most commonly in individuals who have HIV infection, cancer, or congenital T-cell deficiency, or who are on immunosuppressive chemotherapy.7 Cutaneous manifestations of HIV infection or other skin conditions, such as seborrheic dermatitis, impetigo, scabies, burns, and pemphigoid foliaceus, may also predispose to secondary contact acquisition of vaccinia. Smallpox vaccination is contraindicated in subjects with eczema or a history of eczema or atopic dermatitis, once they are susceptible to developing eczema vaccinatum. Eczema vaccinatum manifests with fever, lymphadenopathy, and generalized (often confluent) lesions in areas other than the vaccination site, frequently the face and limbs.
PREVENTIVE PRACTICES
Because autoinoculation is the most frequent mode for developing ocular vaccinia, the vaccination site should be covered until the scab detaches (which can be up to 21 days).8 A semi-permeable polyurethane dressing may be useful.9 Vaccinees should avoid touching the site when bathing. Patients with inflammatory eye disease requiring steroid treatment might be at increased risk of inadvertent inoculation as a result of touching or rubbing the eye.10
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree