Systemic Hypertension and the Eye
Jaclyn L. Kovach
Stephen G. Schwartz
Susan Schneider
Richard B. Rosen
Introduction
Systemic hypertension affects approximately 30% of adults. According to the seventh report by the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7), stage 1 hypertension is defined as 140 to 159 systolic or 90 to 99 diastolic blood pressure; stage 2 hypertension is at least 160 systolic or at least 100 diastolic; and pre-hypertension is 120 to 139 systolic or 80 to 89 diastolic (Table 13.1). Pharmacologic treatment is recommended for patients with stage 1 or 2 disease, and lifestyle modifications are suggested for patients that meet pre-hypertension criteria. Elevated blood pressure increases the risk for stroke, coronary artery disease and death.1 Early identification of this usually asymptomatic disease is vital. Retinal examination and direct visualization of the retinal vasculature can aid in the diagnosis of systemic hypertension and risk stratification for secondary sequelae, including stroke.
TABLE 13.1 | |
|---|---|
|
Hypertension and the Eye
Hypertension can have profound direct and indirect effects on the eye. Direct effects of elevated blood pressure on ocular tissues include hypertensive retinopathy, choroidopathy and optic neuropathy. Hypertension is associated with worsening of diabetic retinopathy and also increases the risk of secondary ocular diseases including retinal vein and artery occlusion, ischemic optic neuropathy, and retinal artery macroaneurysm.
Hypertensive Retinopathy
Hypertensive retinopathy has been found in 2% to 14% of the nondiabetic population age 40 years and older.2 It is typically a bilateral condition, but unilateral retinopathy has been described.3 Hypertensive retinopathy is more common in blacks relative to whites,4,5 which can be explained mostly by higher levels of blood pressure in blacks. The prevalence of hypertensive retinopathy is higher in women than in men.4 There is also a strong association between smoking an advanced hypertensive retinopathy.6,7,8,9,10 A mutation in the angiotensin converting enzyme11,12 or apolipoprotein E gene places affected patients at an increased risk of developing hypertensive retinopathy.13
Liebreich first described hypertensive retinopathy in 1859,14 and then in 1898 Marcus Gunn described a series of patients with hypertension and renal disease. Gunn noted generalized and focal arteriolar narrowing, arteriovenous nicking, flame-shaped and blot-shaped hemorrhages, cotton wool spots, and swelling of the optic disc.15 Keith et al. showed, in 1939, that signs of retinopathy were predictive of death in patients with hypertension.16
There have been numerous revisions to the classification scheme for hypertensive retinopathy. The current grading system defines mild hypertensive retinopathy as arteriolar narrowing and arteriovenous nicking (Fig. 13.1); moderate hypertensive retinopathy as hemorrhages, cotton-wool spots, microaneurysms, and/or hard exudates (Fig. 13.2); and malignant hypertensive retinopathy as optic nerve swelling accompanied by moderate hypertensive retinopathy (Fig. 13.3).17 A patient’s stroke risk increases with the severity of their hypertensive retinopathy. Reversal of malignant hypertensive retinopathy can occur with normalization of blood pressure.18
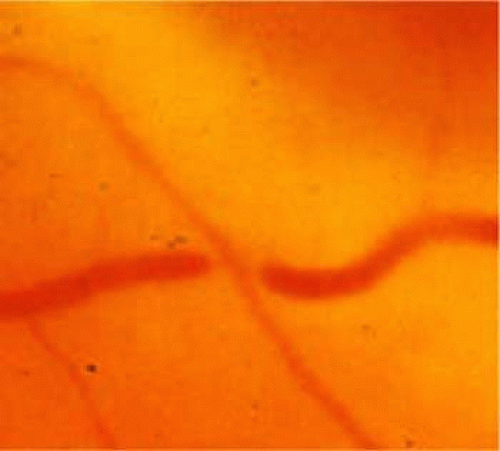 FIGURE 13.1 Arteriovenous nicking and arteriolar narrowing are features of mild hypertensive retinopathy. |
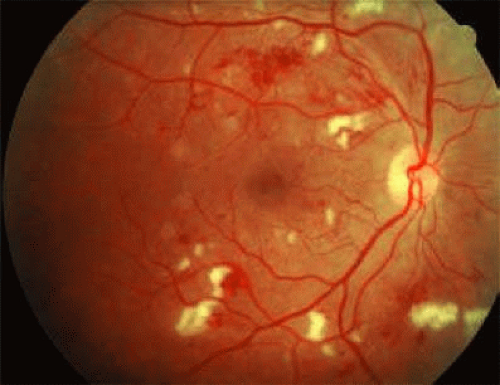 FIGURE 13.2 Moderate hypertensive retinopathy includes scattered microaneurysms, hemorrhages, and cotton-wool spots. |
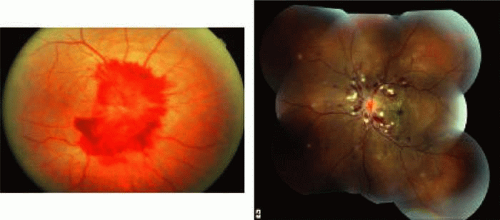 FIGURE 13.3 Malignant hypertensive retinopathy features optic nerve swelling in addition to moderate hypertensive retinopathy. |
The pathophysiology of hypertensive retinopathy corresponds to these specific stages. The first response to an elevation in blood pressure is vasospasm and an increase in vasomotor tone. This is seen clinically as diffuse retinal arteriolar narrowing. Chronic atherosclerotic changes develop including intimal thickening, media-wall hyperplasia, and hyaline degeneration. These abnormalities can be appreciated on exam as diffuse and focal areas of arteriolar narrowing, opacification of arteriolar walls (copper or silver wiring), and compression of venules by arterioles at their common adventitial locations (arteriovenous nicking).2
With more dramatic elevation in blood pressure, there is breakdown of the blood–retinal barrier. This allows the exudation of blood (hemorrhages), lipid and cholesterol (hard exudates), and ischemia of the nerve fiber layer (cotton-wool spots).2 Nerve fiber layer hemorrhages found in the distribution of radial peripapillary capillaries are more common than dot/blot hemorrhages, which likely reflects the arteriolar character of the radial peripapillary capillary bed.19 Hard exudates are typically found in the posterior pole in a circinate ring or macular star configuration. Macular edema can form secondary to an increase in plasma transudation from the capillaries into the surrounding retina and from retinal ischemia.20,21 Cotton wool spots are areas of acute inner retinal ischemia caused by occlusion of terminal arterioles. These small, fluffy white areas resolve funduscopically in 4 to 6 weeks and leave a corresponding nerve fiber layer defect.
Severe hypertension can lead to increased intracranial pressure and concomitant optic nerve swelling or hypertensive optic neuropathy.
Hypertensive Choroidopathy
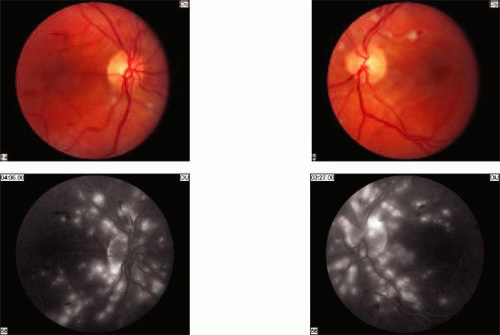
The choroid is very sensitive to blood pressure changes. Circulating vasoactive factors and neural stimulation can cause constriction of the choroid and choriocapillaris, which leads to choroidal ischemia, and damage to the overlying retinal pigment epithelium and outer retina. The outer blood–retinal barrier may also be compromised. Clinical clues to choroidal ischemia include Elschnig spots and Seigrist’s streaks. Elschnig spots22 are ischemic infarcts of the RPE that correspond to underlying areas of choroidal hypoperfusion. They appear as focal subretinal lesions with yellowish halo within 24 hours of accelerated hypertension. Histopathologic examination of these lesions shows hyalinization and necrosis of choroidal vessels and RPE atrophy. Linear, equatorial ischemic infarcts are called Siegrist’s streaks. Fluorescein angiography of new Elschnig spots demonstrates hyperfluorescence because of leakage at sites of compromise of the outer blood-brain barrier (Fig. 13.4). Chronic cases reveal hyperfluorescence secondary to RPE window defects. Damage to the outer blood–retinal barrier allows fluid from the choroid to gain access to the subretinal space. Ischemic RPE cells are slow to pump fluid from the subretinal space and serous retinal detachments form (Fig. 13.5). Resolution of such detachments can follow the restoration of blood pressure control. Spontaneous, massive suprachoroidal hemorrhages are also associated with hypertension.23
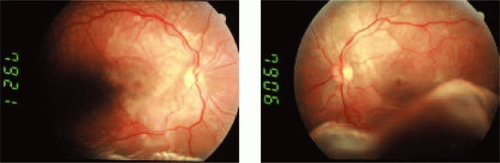 FIGURE 13.5 Bilateral serous retinal detachments caused by hypertensive choroidopathy.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|