Hypotony
Jonathan E. Pederson
Hypotony refers to an intraocular pressure below the normal range and is a term lacking strict definition. An intraocular pressure of 6 mm Hg falls three standard deviations from the mean. Below that level, deleterious effects on the eye may occur. However, many eyes with high scleral rigidity (i.e., nonmyopic) may tolerate an intraocular pressure of 2 to 6 mm Hg without visual impairment.
Under normal circumstances, aqueous humor is produced at the rate of about 2.5 μL/minute.1 Most of the aqueous humor passes through the trabecular meshwork and Schlemm’s canal (conventional outflow), driven by the hydrostatic pressure differential between the anterior chamber and the episcleral veins. About 10% of the aqueous humor leaves the anterior chamber by unconventional routes, primarily uveoscleral outflow.2 This latter pathway exits from the anterior chamber, through the intermuscular spaces of the ciliary muscle, into the suprachoroidal space, and finally into the choroidal vessels3 or out through the emissarial channels of the sclera.4 Uveoscleral outflow is largely independent of intraocular pressure.5 If the intraocular pressure falls below the episcleral venous pressure, conventional outflow ceases,6 and uveoscleral outflow alone must drain any aqueous humor being produced.
Theoretically, hypotony could result from reduced aqueous humor formation, increased unconventional outflow, or reduced episcleral venous pressure. Because the normal episcleral venous pressure is 9 mm Hg,7 a large increase in conventional outflow facility would cause the intraocular pressure to fall toward, but not below, that level. Furthermore, the episcleral venous pressure is normal in patients with hypotony, eliminating low episcleral venous pressure as a cause of hypotony.8 Thus, only two plausible mechanisms for hypotony in nonsurgical eyes exist: reduced aqueous humor production, and increased uveoscleral outflow.
As long as aqueous humor production exceeds the rate of uveoscleral outflow, some fluid must pass out through the Schlemm’s canal, and the intraocular pressure will exceed the episcleral venous pressure. Hypotony can occur if aqueous humor production falls below the rate of uveoscleral outflow (i.e., <10% of its normal rate). This rate of aqueous production is unusual except in prephthisical eyes. In the absence of such a profound reduction in aqueous humor formation, only an increase in nontrabecular outflow can account for the presence of hypotony. This is the most common situation. Examples of “unconventional” outflow pathways include uveoscleral outflow, wound leak, and posterior flow of aqueous humor through the vitreous and across the retina and choroid.
ETIOLOGY
The causes of hypotony are listed and are discussed separately in the following sections.
Postoperative hypotony
Wound leak
Overfiltration
Iridocyclitis
Ciliochoroidal detachment
Cyclodialysis
Retinal detachment
Perforation of sclera
Retrobulbar needle
Superior rectus bridle suture
Ciliary body traction from vitreous base
Hypotony after trauma
Iridocyclitis
Retinal detachment
Cyclodialysis
Scleral rupture
Ciliochoroidal detachment
Bilateral hypotony
Osmotic
Dehydration
Diabetic coma
Uremia
Myotonic dystrophy
Miscellaneous forms of hypotony
Vascular occlusive disease
Carotid occlusion
Temporal arteritis
Central retinal artery or vein occlusion
Prephthisis bulbi
WOUND LEAK
Wound leak is a common and frequently overlooked cause of hypotony after surgery of the anterior segment. The leak may be undetected because of the development of a low and diffuse filtering bleb, which is indistinguishable from the normal degree of postoperative chemosis. The only manifestation of the bleb may be microcystic changes in the conjunctival epithelium, without detectable elevation of the conjunctiva.
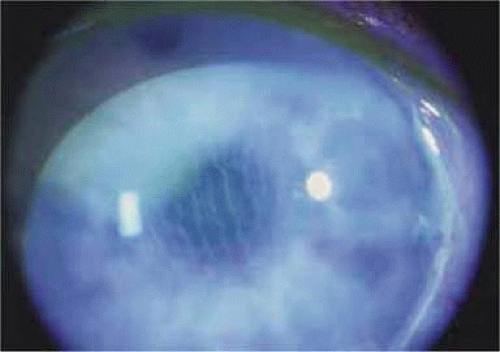
The presence of an external wound fistula is demonstrated by the Seidel test. Streaming of clear aqueous humor into the fluorescein-stained tear film positively identifies the leak (Fig. 58-1). For detection of a small or subtle leak, several aspects of the test are important. First, the concentration of fluorescein in the tear film must be very high. The patient’s lids are held open, and a moistened fluorescein-impregnated paper strip is applied over the area in question. Alternatively, a drop of 2% fluorescein solution may be placed on the eye. Second, because aqueous humor may leak from the wound intermittently when the intraocular pressure is extremely low, digital pressure should be applied if a leak is not immediately apparent.
An external wound fistula should be repaired promptly to prevent endophthalmitis. Occasionally, placement of a pressure patch (and treatment with acetazolamide and timolol to reduce flow through the fistula) will allow the defect to heal. A bandage contact lens or Simmons shell may also be useful. After closure of the fistula, the intraocular pressure may rise to very high levels because the trabecular meshwork may not start functioning immediately. An inadvertent filtering bleb should be repaired if the resulting hypotony is causing visual impairment.
Perforation of the sclera may occur at the time of surgery, which, if undetected, could lead to chronic hypotony. This may occur from the retrobulbar needle, especially in an eye with a posterior staphyloma.9 The superior rectus bridle suture may perforate the sclera, leading to hypotony from external filtration or resultant retinal detachment.10,11,12 This complication is more likely to occur if the eye is very soft just prior to surgery.
OVERFILTRATION
The advent of antimetabolite therapy in glaucoma filtration surgery has resulted in an increased incidence of postoperative hypotony secondary to overfiltration.13 Modification of the surgical technique, such as tighter scleral flap suturing, has reduced the incidence somewhat,14 but the risk of hypotony is still greater than it is without antimetabolites. The usual measures used to treat hypotony secondary to wound leak often do not reverse the problem, because failure of scleral healing is the underlying cause.
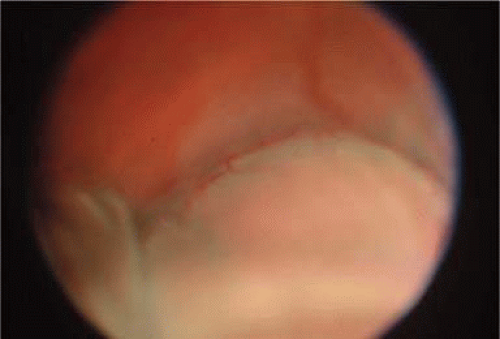
Choroidal detachment may accompany hypotony from overfiltration, but more commonly the fundus exhibits chorioretinal folds and disc edema (Fig. 58-2), with a reduction in visual acuity to the 20/400 range. This was first described by Dellaporta,15 but the term hypotony maculopathy was coined by Gass.16 Presumably, the sclera shrinks from the low intraocular pressure, leaving the choroid wrinkled like an unstretched carpet. Cystoid macular edema may also coexist with hypotony maculopathy.17 Hypotony maculopathy is more common in young myopic patients after filtering surgery.18 It is not known with certainty how long hypotony maculopathy may be allowed to exist in this situation before permanent visual loss occurs. One study found no correlation between duration of hypotony (1–24 months) and visual outcome after revision.19 It is the author’s opinion that 3 months is a reasonable period of time to wait. However, one case of hypotony maculopathy of 7 years’ duration improved from 20/200 to 20/30 after closure of a cyclodialysis cleft,20 and another eye with hypotony maculopathy for 5 years after filtering surgery improved from 20/200 to 20/25 after revision.21 Injection of autologous blood into the bleb has been described22 but is not without risk.23 Resuturing the scleral flap is effective in reversing hypotony and restoring visual acuity while still maintaining some degree of filtration.24,25,26,27,28,29 Transconjunctival suturing may be used.30,31,32,33 A combination of autologous blood and compression suture34 may be effective.35
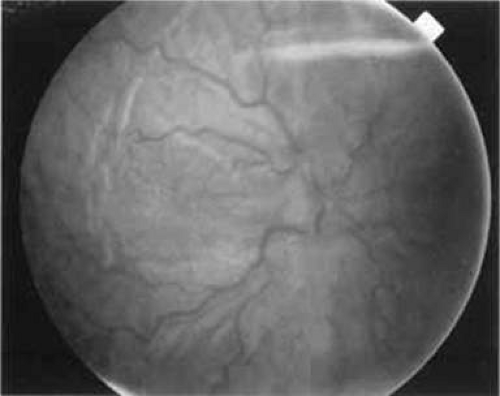 Figure 58-2. Chronic hypotony after filtering surgery with mitomycin C resulting in chorioretinal folds and disc edema. |
A pattern of vertical lines in the corneal epithelium is seen in virtually all eyes with profound hypotony, even in the absence of hypotony maculopathy. Less-prominent horizontal lines may interconnect, creating a chicken wire appearance (Fig. 58-3).
IRIDOCYCLITIS
Mild hypotony may accompany acute anterior uveitis, especially traumatic iritis.36 The blood-aqueous barrier becomes abnormally permeable to protein, as manifested by aqueous flare. The active transport processes of the ciliary epithelium no longer function efficiently because of increased permeability.37 This causes a reduction in the flow of aqueous humor,38 so that the clearance of tracers from the anterior chamber is reduced.39 Uveoscleral outflow is markedly enhanced in iridocyclitis, and aqueous humor flow through the anterior chamber is markedly decreased.40
Iridocyclitis, or at least aqueous flare, is a frequent finding in eyes with marked hypotony from any cause. These eyes are often referred to as having “ciliary body shock.” However, it is difficult to determine clinically whether the underlying disease process itself (e.g., cyclodialysis) alters the production of aqueous humor or whether the concomitant iridocyclitis accounts for the observed aqueous hyposecretion.
CILIOCHOROIDAL DETACHMENT
The clinical and pathologic features of ciliochoroidal detachment are found elsewhere in these volumes. The physiology of suprachoroidal fluid dynamics has been summarized in a review.41
The exact relationship between the ciliochoroidal detachment and hypotony has a long and controversial history. Originally, it was believed that suprachoroidal fluid in ciliochoroidal detachment was derived solely from aqueous humor in the anterior chamber.42 It was postulated that aqueous humor seeps into the suprachoroidal space, creating the detachment and causing hypotony. However, it was later discovered that the protein concentration of suprachoroidal fluid in ciliochoroidal detachment was much higher than that of the aqueous humor, being virtually equal to that of the plasma.43 Electrophoretic analysis of the proteins in suprachoroidal fluid suggests that they originate from the choroidal vessels, with molecular sieving occurring across the capillary endothelium.44
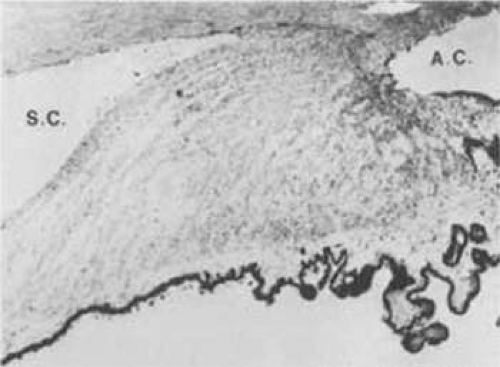
The cause-and-effect relationship between hypotony and ciliochoroidal detachment is also a confusing and controversial subject. It is clearly established that hypotony is a significant factor in the development of a ciliochoroidal detachment (Fig. 58-4).43,44,45,46,47,48 Whether ciliochoroidal detachment alone causes significant hypotony is less clear. An often-cited study evaluated nine patients with hypotony: seven after surgery (three with cyclodialysis clefts) and two with chronic iridocyclitis.49 A sclerotomy was performed in each case, revealing supraciliary fluid in all cases. In addition, four eyes were studied with intravenous fluorescein, revealing stagnation and accumulation of fluorescein in the anterior chamber of eyes with hypotony when compared with fellow eyes. It was concluded that serous detachment of the ciliary body somehow led to reduced aqueous humor formation (hyposecretion), which in turn was the cause of hypotony. However, all eyes exhibited aqueous flare, and the role of concurrent iridocyclitis as a cause for the observed hyposecretion is likely.
Experimental studies in rabbits have encountered the same confusing coexistence of iridocyclitis, making interpretation difficult.50,51 In monkeys, who respond to surgery with minimal inflammation, experimental ciliochoroidal detachment with Ringer’s solution or autologous serum caused a fall in intraocular pressure of 7 mm Hg, but the aqueous humor flow remained normal.52 A detachment created by silicone oil resulted in no lowering of the intraocular pressure despite a high detachment of the ciliary body and choroid. It was postulated that ciliochoroidal detachment results in hypotony because of increased uveoscleral outflow. The pathway from the anterior chamber to the supraciliary space is shortened, and edematous enlargement of the intermuscular spaces of the ciliary body would enhance such fluid movement (Fig. 58-5). Hyposecretion observed clinically must thus be due to concurrent iridocyclitis and not ciliary body detachment, as is commonly believed. In addition, serous choroidal detachment is seen in Harada disease, yet the intraocular pressure is normal in these eyes.53
Proteinaceous suprachoroidal fluid exits the eye by way of the emissarial channels of the sclera54,55 because it cannot be reabsorbed into the choroidal vessels.3 It is unclear why suprachoroidal fluid in the ciliochoroidal space is obstructed. In hypotony, the pressure drop across the sclera is reduced and may delay the outflow of suprachoroidal fluid.
Fortunately, most ciliochoroidal detachments resolve spontaneously without untoward effect. In cases where the detachment persists, no specific nonsurgical therapy exists. The usual clinical practice is to administer topical (and occasionally systemic) corticosteroids and topical cycloplegics to prevent synechiae. If the intraocular pressure could be raised, more rapid resolution of the detachment would occur. Occasionally, increased fluid intake is used to cause increased aqueous formation. Corticosteroid therapy may improve aqueous production and reduce uveoscleral outflow by reducing iridocyclitis. Paradoxically, acetazolamide has been reported to speed absorption of suprachoroidal fluid.56 Perhaps its vasoconstrictive effects57 reduce the transudation of fluid across the choroidal vessels. If nonsurgical efforts fail to resolve the ciliochoroidal detachment and hypotony, and if corneal decompensation develops, prompt drainage of suprachoroidal fluid with reconstitution of the anterior chamber is indicated.58 Improvement of hypotony usually follows removal of suprachoroidal fluid.49,59 Sodium hyaluronate may be used to prevent recurrence of the flat anterior chamber.60,61 In intractable cases, suturing the ciliary body to the sclera may be curative.62
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree