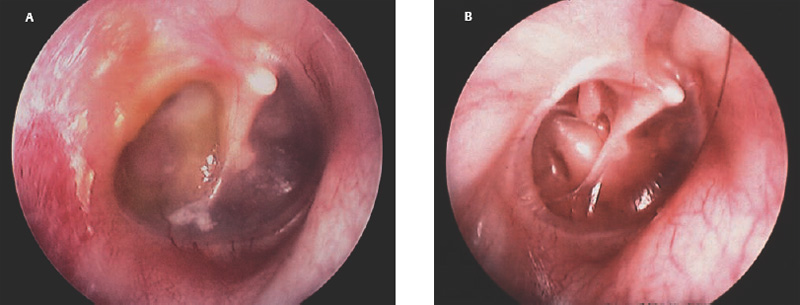
2 Tympanoplasty: Indications and Technique Today the goal of successful tympanoplasty is to create a mobile tympanic membrane or graft with an aerated mucosal-lined middle ear space and a sound-conducting mechanism between the mobile membrane and the inner ear fluids. A review of the literature reveals that many techniques have been developed and employed successfully, and there is a rich history of the evolution of techniques to produce this end. With this review the authors hope to describe a brief history of the evolution of the over–under tympanoplasty, with indications and technical aspects of the technique. Embryologically, the tympanic membrane is derived from the fusion of the ectodermal meatal plugs from the first branchial cleft and the endodermally derived first branchial pouch (tubotympanic recess). The tympanic membrane and middle ear cavity make up the area of contact between these two structures. The tympanic membrane separates the delicate middle and inner ear structures from the external environment. It measures ∼10 mm in diameter and is conically shaped with the apex of the cone at the umbo. Histologically, the tympanic membrane has three layers. This structure contains an outer ectodermal layer composed of keratinizing squamous epithelium, a middle mesodermal fibrous layer, and an inner endodermal mucosal layer. The outer epidermal layer is composed of stratum corneum, granulosum, spinosum, and basale. This layer has cell growth and migratory properties responsible for the self-cleaning and replacement function of the tympanic membrane.1,2 Studies have demonstrated the presence of epidermal growth factor and fibroblast growth factor, which are thought to promote healing of membrane perforations and contribute to the success of tympanoplasty procedures.3 The mesodermal fibrous layer is the intermediate layer or the lamina propria. There are different compositions depending on the location along the tympanic membrane.4 In the pars tensa the lamina propria has a subepidermal loose connective tissue layer containing the internal blood vessels and nerves and a fibrous layer made of outer radial and inner circular fibers. These fibers are made of collagen. This should contribute to the vibratory functions of the tympanic membrane.5,6 The pars flaccida or Shrapnell membrane has elastic fibers and accounts for the flaccidity of this variably sized area.4 There are two major blood supplies of the tympanic membrane. An external plexus from the tympanic branch of the deep auricular artery sends large manubrial branches along the Shrapnell membrane and the manubrium and numerous radial branches into the tympanic membrane from along its circumference.7 The malleal artery is the major blood supply of the posterior half of the tympanic membrane, which is better perfused than the anterior half.8 The anterior half is supplied from smaller radial branches that enter around the annulus derived from the internal plexus from the stylomastoid branch of the postauricular artery.7 The middle ear (mesotympanum) is the air-containing space that is bounded laterally by the tympanic membrane. The medial wall of the middle ear is formed by the promontory, which is the lateral bony wall of the cochlea, and which is covered by a thin layer of mucosa (mainly nonkeratinizing low cuboidal epithelium or ciliated columnar epithelium in places). The anterior portion of the middle ear, termed the protympanum, contains the bony opening of the eustachian tube. The inferior middle ear (hypotympanum) contains air cells; posteriorly the jugular bulb may be present. The superior middle ear is confluent with the epitympanum, or attic, which contains the head of the malleus and body of the incus. Posteriorly, the middle ear is bounded by the bony ear canal, the mastoid segment of the facial nerve, and the pneumatized cell tract called the facial recess. Inferior to the facial nerve is a space named the sinus tympani, important in cholesteatoma surgery because it is a frequent site for recurrence. The middle ear contains the ossicles—malleus, incus, and stapes—and the tympanic segment of the facial nerve, chorda tympani nerve, tensor tympani, and stapedius muscles. The mucosa of the middle ear envelops the ossicles like a mesentery and forms a very fine layer that partitions the air-containing space and determines the direction that disease may grow (as discussed in Chapter 1). The middle ear contains the ossicular chain with its ligaments and the tendons of the tensor tympani and stapedius muscles. The attic or epitympanum is almost completely separated from the mesotympanum by the ossicles and their folds except for two small but constant spaces. The Prussak space is bounded superiorly by the lateral malleolar fold arising from the junction of the malleus head and neck and radiates out to insert on the entire bony rim of the notch of Rivinus. The tympanoplasty operation is a surgical procedure to eradicate disease in the middle ear and to reconstruct the hearing mechanism, with or without mastoid surgery, with or without tympanic membrane grafting. This was defined by the American Academy of Ophthalmology and Otolaryngology’s Committee on Conservation of Hearing in 1964.9 Hippocrates recognized that “acute pain of the ear, with continued strong fevers, is to be dreaded, for there is danger that the man may become delirious and die.”10 Early surgery for the draining ear was basically a mastoid operation and was life saving. The first attempt to repair a tympanic membrane was performed in 1640 when Banzer used a pig’s bladder stretched across an ivory tube and placed in the ear, which gave temporary hearing improvement.11 In 1853, Toynbee placed a rubber disc attached to a silver wire over a perforation with hearing improvement.12 In 1877, Blake placed a paper patch over a perforation, and in many patients a hearing improvement was noted.13 The concept of tympanoplasty is credited to Berthold who in 1878 was thought to have performed the first true tympanoplasty. His technique involved deepithelializing the tympanic membrane by applying plaster against the membrane for 3 days, removing the epithelium, and then placing a skin graft over the defect.14 The “modern era” of tympanoplasty occurred in the 1950s because of many developments in antibiotics, instrumentation, and the operating microscope. Following the introduction of tympanoplasty by Wullstein and Zollner in the early 1950s, all surgeries used an overlay graft.15,16 Wullstein’s paper, “Tympanoplasty as an Operation to Improve Hearing in Chronic Otitis Media and Its Results” set the stage for this operation to improve hearing and protect the middle ear from the outside environment. At that time, this consisted of full-thickness and split-thickness skin grafts. By the end of the decade, graft eczema, desquamation and poor long-term take rate had prompted many surgeons to seed alternate grafting materials and techniques.17 In 1956, Sooy had reported the use of canal skin pedicle graft to close marginal perforations.18 In 1958, House and Sheehy and Plester working independently began using canal skin as a free overlay graft.19,20 Shea, Austin, and Tabb working independently in 1959 employed vein as an undersurface graft to repair tympanic membrane perforations.21–23 The vein graft tended to atrophy over a few months and occasionally reperforated. In 1961 Storrs described the first undersurface fascia technique to be used in this country.24 With the use of connective tissue most of the problems incurred with free skin grafts were eliminated. The first successful use of homograft tympanic membrane in this country was by Ned Chalat in 1964.25 His experience was reported in the Harper Hospital Bulletin and went unnoticed for several years. Many authors reported promising results with this technique in 1968 by House and Glasscock, and soon to be followed by Perkins, Smith, and Wehrs.26–29 With this technique procurement and sterilization of the donor material have been problematic. Over the years surgeons have used various living or homograft grafting materials already mentioned, including loose areolar connective tissue, perichondrium, cartilage, fat, and periostium.30,31 Fascia has been the preferred material because of the internal structure of this material and because of the plentiful amount in the operative field. The high success rate of this material probably resides in the internal structure of collagen and mucopolysaccharides. It is interesting to note that both collagen and mucopolysaccharides have been implicated as playing a critical role in wound healing. Collagen is felt to contribute to wound tensile strength, and there is evidence that the chemically and biologically complex mucopolysaccharides play a positive role in the healing process, attracting fibroblasts into the wound area through chemotaxis.32 With a dry perforated eardrum the surgeon needs to assess the size, location, and middle ear spacing (retracted ossicular chain, narrow middle ear cleft, lateral promontory, or high jugular bulb). Depending on the particular situation encountered, one may then choose underlay, overlay or over–under tympanoplasty technique (Fig. 2.1). Fig. 2.1 Tympanic membrane perforation with tympanosclerosis. Fig. 2.2 (A) Chronic suppurative otitis media. (B) Previous ear, treated with oral and topical antibiotics. With chronic suppurative otitis media (CSOM) one tries to dry the drainage if possible with general medical procedures such as topical and oral antibiotics, improvement of eustachian tube dysfunction, or allergy management. In some patients the drainage may not be controlled prior to surgery. The principles outlined earlier are followed, with the possible addition of an aerating mastoidectomy in a select patient group (Fig. 2.2A,B). One must determine whether the perforation caused by the cholesteatoma is confined to the middle ear or extends into the epitympanic space or mastoid (Fig. 2.3). Fig. 2.3 Cholesteatoma with total perforation. A review of the literature reveals that many techniques have been developed and employed successfully. Tympanoplasty techniques have employed approaches such as transcanal, endaural, or postauricular. The grafts have been placed over and under the tympanic membrane or the malleus, and the biological material used has been full-thickness skin, partial-thickness skin, fascia, perichondrium, cartilage, and periosteum. We will describe the various techniques and approaches commonly used in the past and a personal evolution of technique for the majority of surgery utilizing the over–under tympanoplasty. The underlay or undersurface technique employs the use of grafting material medial or under the remnant tympanic membrane typically under the malleus when the perforation extends to that area. The free edges of the perforation are prepared using a right-angled hook, and in some instances, a fine scissors is used to resect the perforation margins. The intent is to separate the outer cutaneous layer from under-turned epithelium or separate the inner mucosal layer from the external tympanic membrane epithelium. This develops a fresh edge for healing. This technique, originally described by Shea in 1957, was used initially for iatrogenic tympanic membrane perforations caused at the time of middle ear surgery.28 Austin and Shea in 1961 published their combined experience with this technique stating that vein was an ideal material because it did not substitute for the missing squamous layer of the drum, but as a replacement for the fibrous layer across which normal epithelium will grow.33 The great hardiness of the vein as a free graft was also thought to be desirable. Their approach began as a transcanal or transmeatal procedure. The postauricular approach was used as a surgeon’s decision at the time of surgery. Long-term results with vein proved to be fraught with atrophy over a few months and occasionally re-perforated. These results were improved with the use of fascia, loose areolar connective tissue, or perichondrial tissue and have been the preferred material over the past 40 years (Fig. 2.4A–D). The overlay technique as practiced in the 1960s and occasionally used today, consisted of accessing the drum head through the surgeon’s preferred incision. The epithelium of the surface of the drum was removed, a fascia graft was placed on the perforation, and the ear was packed with various materials. Sheehy and Glasscock replaced the pedicled canal skin grafts earlier described by authors with temporalis fascia overlay grafts.34 After comparing cases of canal skin and fascia grafts the following conclusions were made: (1) fascia grafts (97.5% take rate) were generally superior to canal skin grafts (91.8% take rate); (2) fascia grafts were also superior (91%) to canal skin grafts (70%) in closing total perforations; and (3) fascia was an excellent material to close ears that were draining at the time of surgery as evidenced by higher success rate (98%) than dry perforations using canal skin. This technique was to be performed from the postauricular approach. Once the vascular strip incision is dissected out of the ear canal, it is held anteriorly with the retractor. The medial superior, anterior, and inferior canal wall skin is incised near the bony–cartilaginous junction and then carefully elevated medially to the annulus. The entire cuff of skin is then cut free leaving the annulus in its physiological position. This skin is then removed from the operative site and placed into saline for the duration of the case. Canal wall widening is performed with a small drill as needed to visualize the anterior portion of the annulus, and marked manipulation of anterior ear canal wall skin and drilling of the bone is necessary. The squamous layer of the tympanic membrane remnant, fascia placed in the physiological position of the drum, and then the ear canal skin replaced over the fascia. The vascular strip is replaced, and the ear canal is packed with pledgets of absorbable gelatin sponge moistened in an antibiotic solution. With this technique, there is obvious marked bony and soft tissue dissection necessary, and ear drum blunting common, with frequent conductive hearing loss. By combining the benefits of both techniques, the over–under tympanoplasty has become the preferred technique for various approaches to tympanoplasty.35–37 This technique places the tympanic membrane fascia graft lateral to the malleus but medial to the remnant of tympanic membrane and fibrous annulus and medial to the bony annulus anteriorly and inferiorly. This allows excellent exposure to the anterior middle ear space and prevents medialization of the graft to the promontory. Ossicular reconstruction may be placed directly to the underside of the malleus. Over–under tympanoplasty has become a preferred technique for perforations that abut the malleus (Fig. 2.5), large or near total perforations, cases of significant malleus retraction (making the classic underlay technique impractical) (Fig. 2.6), significant anterior tympanosclerosis, or anterior middle ear cholesteatoma (Table 2.1). The over–under tympanoplasty has been popularized recently as a technique for certain perforations. Previous authors have demonstrated the technique of placing a graft lateral to the malleus but medial to the remnant of the tympanic membrane. As early as 1972, Austin stated that the graft may be secured either over or under the malleus tip, depending on the ease of positioning, and then some of the skin covering the malleus is dissected and replaced on the graft surface.38 Glasscock, Wehrs, and Hough also made similar inferences when describing underlay grafting in particular situations, usually with significant retraction of the malleus, large perforations, or anterior middle ear disease.39–41 In 2001, Kartush et al presented their results with this technique, including excellent long-term results for closure of perforations, hearing improvement or stabilization, and low incidence of complications.42 The authors used this with and without mastoidectomy and with and without ossicular reconstruction. Fig. 2.5 Tympanic membrane perforation that abuts the malleus in the retraced middle ear space. Fig. 2.6 Tympanic membrane with malleus retraction. Postauricular over–under tympanoplasty technique is begun with appropriate preoperative decisions common in all tympanoplasty surgery previously described elsewhere. Most patients will undergo general anesthesia with endotracheal intubation for this type of surgery. After anesthesia is secured, the patient is turned 180 degrees. The surgeon will sit at the surgical side of the patient, with the scrub nurse across the patient from the surgeon. An area of hair is removed from around the ear for ∼2 cm above and behind the auricle. The natural oils of the skin are removed with alcohol solution or acetone, adherence material is placed on the edges of the prepped area, and plastic drapes are placed to the shaved area to hold the hair out of the field. The ear is injected with lidocaine-HCl 1% with 1:100,000 epinephrine in a subdermal plane at the postauricular incision site and a four-quadrant external ear location. A cotton ball is placed in the ear canal (to keep the scrub solution from the middle ear space), and the ear is then washed with an iodine solution, the solution being kept in contact with the ear for ∼6 minutes. The ear is then dried from the solution by the scrub nurse, the ear is folded forward, and a plastic drape attached to a waterproof barrier is applied.
♦ Tympanic Membrane and Middle Ear Cleft
♦ Historical Review
♦ Conditions of the Middle Ear in Tympanoplasty Surgery
Perforated Eardrum That Is Dry
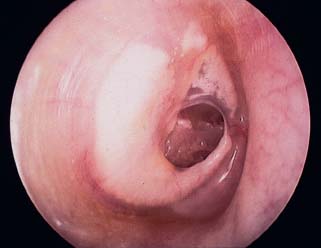
Perforated Eardrum with Chronic Suppurative Otitis Media
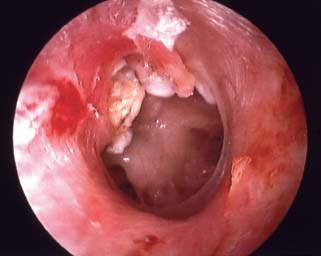
Perforated Eardrum Secondary to Cholesteatoma
♦ Techniques
Evolution of Techniques
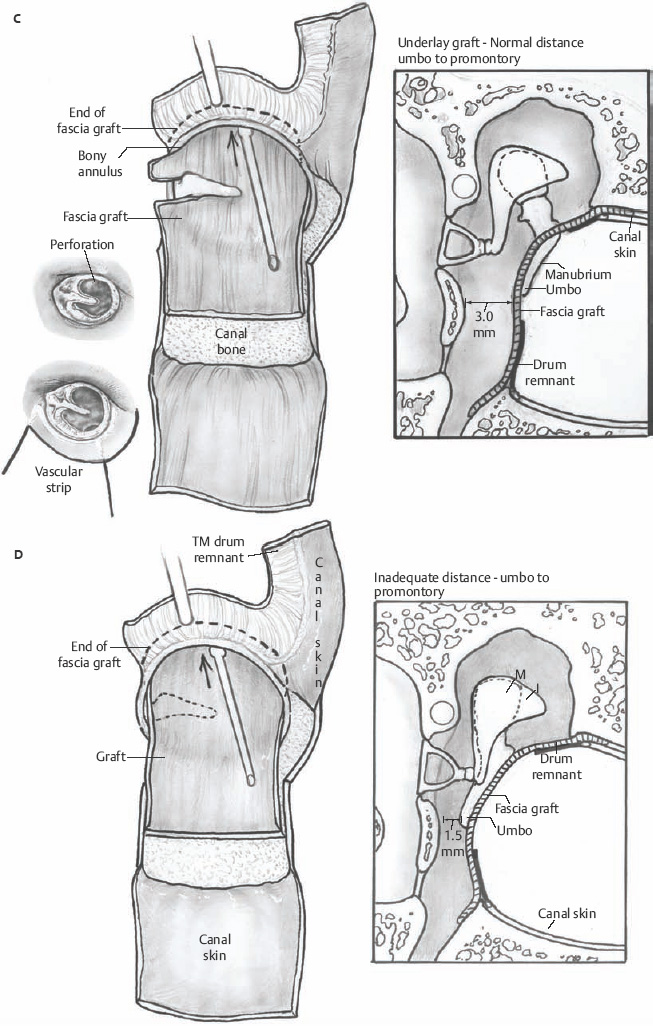
Underlay (Undersurface) Technique
Overlay Technique
Indications for Over–Under Tympanoplasty
Surgical Technique of Over–Under Tympanoplasty
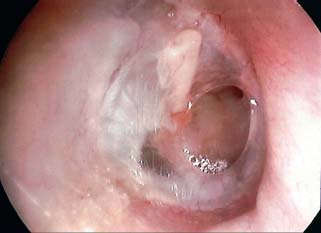
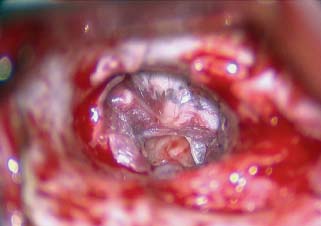
Surgical Preparation
Fascia Periostium incised and elevated Fibrous tissue removed from umbo Graft placement |
The drill cord, irrigation and suction tubes, and cautery lines are wrapped in a towel and placed and secured to the top of the drape over the patient. A second pouch is made to hold the drill handpiece, suction tips, and cautery devices. This prevents the lines from tangling or falling off the surgical field.
The surgeon then cuts the drape directly over the surgical site. A wet sponge is used to clean the external ear of any dried iodine solution, and then the ear is dried.
The ear canal is then suctioned free from any ear canal debris or iodine solution. The vascular strip area is examined and then injected with the same lidocaine–epinephrine solution at the boney cartilaginous junction site. The surgeon is to observe blanching of the skin of the vascular strip skin. The ear is then irrigated with sterile saline solution to further debride and clean the ear canal and examine the tympanic membrane pathology.
Incisions
The auricle is then held by the right-handed surgeon with the left hand, pulling the ear forward and laterally. The incision is made with a No. 15 blade ∼5 mm behind the postauricular fold. Once the area of the loose areolar tissue overlying the temporalis fascia is seen, this bloodless plane is carried to the mastoid tip inferiorly. The nurse holds the auricle, and bleeding points are controlled with an electrocautery. A self-retaining retractor is then placed.
Harvesting Fascia
The scrub nurse holds the superior edge of the wound laterally with a small retractor to help visualize the fascia. We attempt to harvest true fascia for the reasons previously mentioned. Fascia of the temporalis muscle posteriorly is ideal because anteriorly the fascia thickens and occasionally splits with adipose tissue interposed. An incision is made in the fascia superior and parallel to the linea temporalis, and then a delicate iris scissors is used to harvest simply the fascia without areolar tissue laterally or muscle tissue medially. For a subtotal perforation the graft is ∼10 by 15 mm and for larger perforations a 15 by 20 mm graft is harvested. The graft is then placed on a Teflon block, cleaned of any areolar or muscle tissue, and then straightened on the block. The block is then placed onto the scrub nurse’s back table where a lamp is placed near the graft to dehydrate the graft. Once the graft is dehydrated the lamp is turned off to prevent excessive drying of the graft.
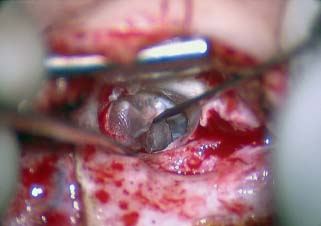
Fig. 2.7 Beaver blade tympanomeatal cut.
Exposing the Ear Canal and Middle Ear
The area of the linea temporalis is palpated, and then an incision is made along this line between the temporalis muscle and the ear canal. A “T” incision is made through the tissue overlying the mastoid to the mastoid tip. A Lempert periosteal elevator is used to expose the mastoid bone, and the spine of Henle is visualized.
The self-retaining retractor is placed in this deeper plane. Now with the use of the operating microscope, a smaller periosteal elevator in the surgeon’s right hand and a fine 20-gauge suction in the left hand are used to elevate the soft tissue of the bony external ear canal. Care is taken not to tear the delicate skin of the medial portion of the ear canal skin. The tympanomastoid suture line and the tympanosquamous suture line are visualized. The surgeon then utilizes a No. 6400 Beaver blade to make the superior and inferior incisions of the vascular strip, and a No. 7200 Beaver blade is used to make a parallel to annulus cut ∼4 to 5 mm lateral to the annular rim (Fig. 2.7). The vascular strip is then held anteriorly in the self-retaining retractor. This exposure now allows the surgeon to visualize the pathology of the tympanic membrane more clearly.
Now with the 20-gauge suction in the surgeon’s left hand and a small round dissector in the right hand, the cuff of medial ear canal skin is elevated to the annulus. Generally a relaxing incision is made on the medial ear canal wall skin of the tympanic bone, 4 to 5 mm lateral and parallel to the inferior annulus. The middle ear is entered by elevating the cuff of skin until the fibrous annulus is visualized inferior to the chorda tympani nerve, and then elevating the annulus inferiorly and anteriorly. At this point, the middle ear may be well visualized (Fig. 2.8).
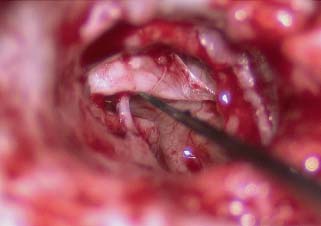
Fig. 2.8 Exposing the middle ear.
Control of Disease
These initial steps are performed in all cases. Once the middle ear is well exposed, the decision for which type of tympanoplasty technique is determined (underlay, overlay, or over–under). The indications for over–under tympanoplasty technique are outlined in Table 2.2.
Middle ear cholesteatoma, granulation tissue, and diseased mucosa are dealt with at this time. If a mastoidectomy is required to remove tissue safely from the middle ear or if there is disease in the mastoid then an intact canal mastoidectomy with facial recess approach is employed. Further treatment decisions are made as described in other chapters of this book.
Preparation of Tympanic Membrane Remnant
When the disease process is under control, the tympanic membrane remnant is prepared for grafting. A rim of tissue is removed from the perforation edge to remove diseased tissue or mucosal thickening and encourage migration of healthy epithelium and the mucosal layer. Now for those patients where an over–under tympanoplasty is to be employed, the malleus is clearly visualized by elevating the tympanic membrane. The periosteum of the malleus is incised with a fine sharp needle (Fig. 2.9A,B). This periosteal cuff is then used to elevate the tympanic membrane off the short process of the malleus, the neck of the malleus, and the long process of the malleus to the fibrous umbo area (Fig. 2.10). At this point either a microalligator-type scissors or the laser is used to remove the remnant from the malleus (Fig. 2.11).
Perforations or retractions that abut the malleus Large or near-total perforations Severe malleus retraction Significant anterior tympanosclerosis Anterior middle ear cholesteatoma Ossicular reconstruction |
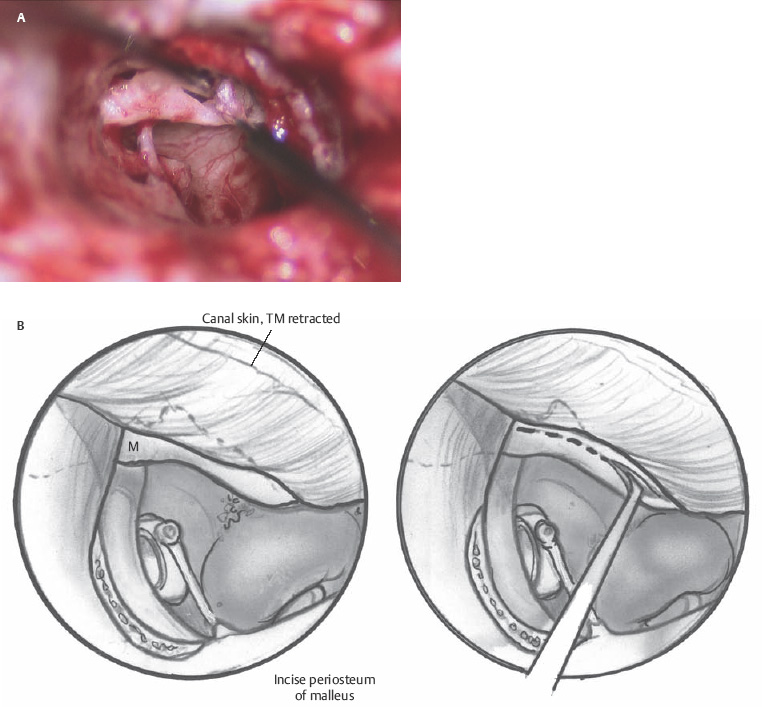
Fig. 2.9 (A,B) Incising the periosteum of the malleus.
Internal Packing
When there is significant middle ear mucosal disease or severely retracted malleus, a fitted piece of silicone sheeting (Silastic, Dow Corning, Midland, MI) is placed on the entire promontory area (0.25 mm thickness). Generally the sheeting is fashioned to ∼10 to 12 mm in diameter with a small cutout where the stapes is located. Gelfoam (Pharmacia, Peapack, NJ) is cut into 8 mm discs, saline soaked, and dried on a Silastic cutting block. One or two discs are used to pack the eustachian tube. Another disc is placed on the Silastic sheet medial to the malleus. If more is necessary to fill the middle ear cleft an 8 mm disc cut in half is used to fill the space.
Placement of Graft
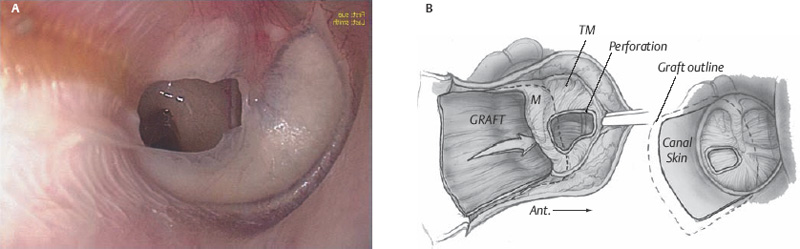
The parchment-like fascia graft is now removed from the Teflon block and trimmed to the appropriate size. Generally, for near total perforations, the fascia is trimmed to a tongue shape with the anterior edge ∼10 mm in diameter. The graft is then made ∼15 mm in length. This allows the graft to be tucked under the anterior, inferior, and superior bony annulus, over the malleus, and then onto the posterior bony canal wall (Fig. 2.12). The remnant of tympanic membrane is then placed onto the lateral surface of the graft, and the remainder of the ear canal skin flaps is replaced into the normal position (Fig. 2.13). A disc of EpiFilm (bioactive lamina composed of an ester of hyaluronic acid, Xomed-Medtronics, Jacksonville, FL) may be placed onto the lateral surface of the tympanic membrane, especially in large perforations. A Gelfoam 8 mm disc is placed lateral to the EpiFilm (or primarily) and then antibiotic ointment applied through a 14-gauge soft catheter on a 5 mL syringe is placed onto the Gelfoam, filling the anterior sulcus of the ear canal.
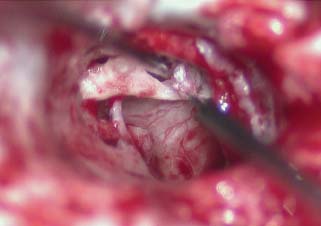
Fig. 2.10 Elevation of the tympanic membrane to the umbo and cutting from the malleus.
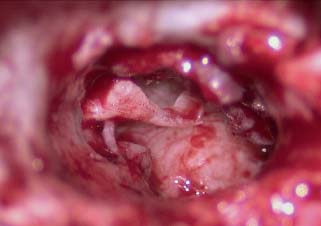
Fig. 2.11 Tympanic membrane off the malleus.
The self-retaining retractors are removed, hemostasis is secured, and the T incision is closed with an absorbable suture. The postauricular incision is sutured with an absorbable suture in a subcuticular manner. No skin sutures are necessary.
The ear canal is then visualized under the operating microscope and evacuated of blood, making sure not to suction onto the graft, but placing the vascular strip skin onto the fascia graft. This is held into position with a nonabsorbable sponge moistened with an antibiotic steroid suspension. A mastoid dressing is placed.
Postoperative Care
The mastoid dressing is removed the first postoperative day. The ear canal sponge is moistened with the antibiotic steroid suspension a couple of times each day. The pack is removed on the first postoperative appointment in about a week.
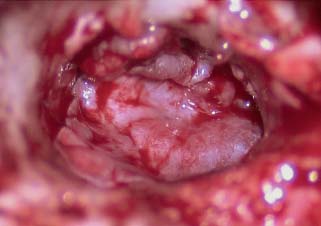
Fig. 2.12 Fascia graft over the malleus, under the bony annulus and onto posterior boney canal wall.
♦ Cartilage Tympanoplasty
The cartilage graft has been utilized in certain tympanoplasties for over 40 years. Situations that may benefit from the use of cartilage with or without perichondrium are for scutum defects, for repair of posterior retraction pockets, or for total tympanoplasties, for the recurrent retracted middle ear cleft, and for recurrent perforations after previous failed tympanoplasties. Although similarly used as fascia for reconstruction, the rigid quality of cartilage tends to resist resorption and retraction. The anticipation of conductive hearing loss when using cartilage to reconstruct the tympanic membrane is of concern, but most reports state that there is not a statistically significantdiff erence in advanced tympanic membrane reconstruction.37,43 A modification of Dornhoffer’s technique is described here. Others have used “palisade” techniques as well, in which the cartilage is scored.
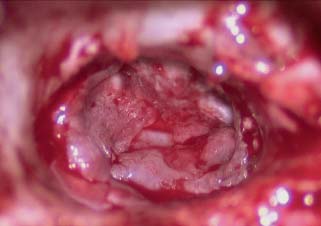
Fig. 2.13 TM remnant over fascia graft.
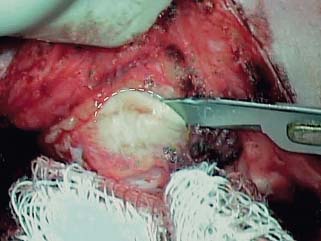
Fig. 2.14 Harvesting cartilage and perichondrium from the cimbum concha. With the surgeon’s finger in the cimbum, the loose tissue is cleared from the plane of the perichondrium, a curvilinear incision is made through the cartilage.
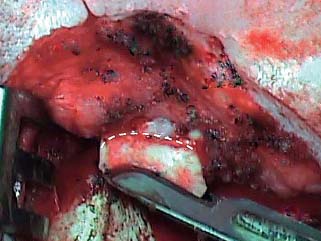
The graft may be taken from the cimbum concha through the postauricular wound. The surgeon’s finger placed in the cimbum can be helpful in defining its boundaries. The loose tissue over the cimbum is incised with a No. 15 blade, then the plane of the perichondrium is defined and exposed. The cartilage is then incised in a semicircular shape (Fig. 2.14), and the subdermal tissue is dissected away from the perichondrium on the lateral (concave) side, care being taken not to buttonhole the thin auricular skin. The incision is then completed to form a circle ∼1 cm in diameter (Fig. 2.15), and the cartilage graft, with perichondrium on both surfaces, is removed. The graft is prepared by elevating the perichondrium away from the convex surface with a duckbill elevator, leaving it hinged at one end (Fig. 2.16). The cartilage itself is then trimmed to the size of the tympanic membrane, and a groove can be created to accommodate the malleus handle. The final graft should be an island of cartilage on a much larger piece of perichondrium.
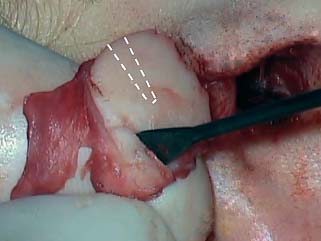
The tympanic membrane remnant is prepared as previously described. The cartilage graft is then used to reconstruct the entire surface area of the tympanic membrane, rotating it into position until the groove is aligned with the malleus handle (Fig. 2.17A,B). If the graft is of proper size, the edge of the cartilage will snap up against the posterior bony annulus and fit snugly like a manhole cover (the use of Gelfoam in the middle ear is optional with this technique). The free perichondrium is then laid against the bony canal. The tympanomeatal flap is then draped over the composite graft (Fig. 2.18A,B). If the perforation is large, the flap can be incised at its narrowest point, usually inferiorly, and its edges advanced and rotated over the area of the perforation, to provide maximal epithelial coverage (this maneuver borrows skin from the ear canal to cover the tympanic membrane defect, which rarely poses a problem in healing).
The cartilage graft with or without perichondrium is most frequently used in the patient with a scutum defect from cholesteatoma disease (Fig. 2.19). It should be noted that when cartilage is used, an intact canal wall mastoidectomy has been performed either at this operation or previously. Cartilage-perichondrial grafts are also selectively used in canal wall down procedures. We advise the use of perichondrium attached to the cartilage as the graft of choice. The cartilage is then trimmed from the perichondrium as needed to repair the particular defect.
The tragus is an alternative place for harvesting cartilage, with or without perichondrium, with over 1 cm of cartilage available. The surgeon may harvest the cartilage with perichondrium on both sides of the cartilage if tissue is needed. This series of photos is incision, identification of perichondrium, then incision through the cartilage with preservation of the anterior perichondrium in its anatomical position (Figs. 2.20, 2.21, and 2.23). Removal of cartilage from the perichondrium is then accomplished, with the necessary size determined by the pathology. The perichondrium is then draped over the malleus with the cartilage to fit the defect of the tympanic membrane and scutum if necessary, then the remaining perichondrium is laid onto the posterior bony canal wall (Figs. 2.23 and 2.24).
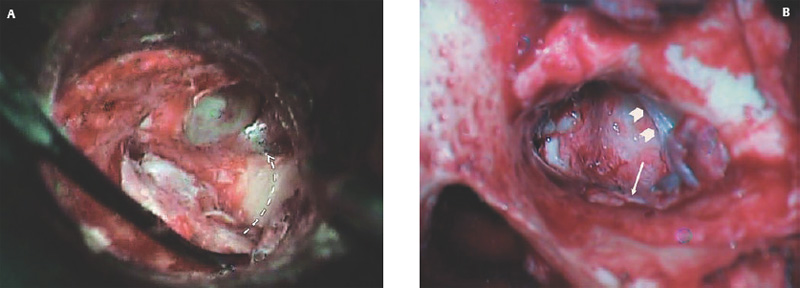
Fig. 2.17 (A) The cartilage graft is inserted into the ear and used to reconstruct the tympanic membrane (TM) in an underlay fashion, rotating into position until it is cinched against the bony annulus. (B) Cartilage graft rotated into position, with cartilage tucked under the anterior TM remnant (arrowheads), and the free edge of the perichondrium draped over the bony canal (arrow).
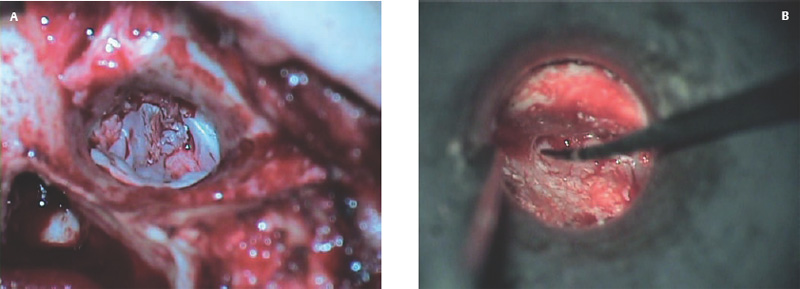
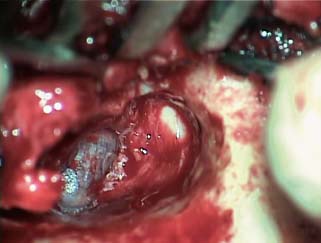
Fig. 2.19 Cartilage graft reconstruction of scutum, as well as posterosuperior tympanic membrane.
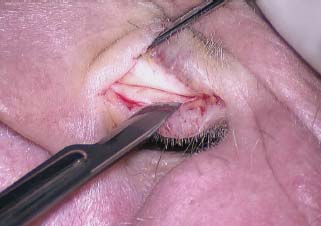
Fig. 2.20 Scissor dissection of the anterior perichondrium from the tragus cartilage.
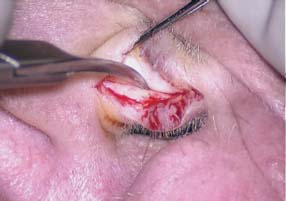
Fig. 2.21 Scissors dissection of the posterior tragal soft tissue from the tragus perichondrium and cartilage.
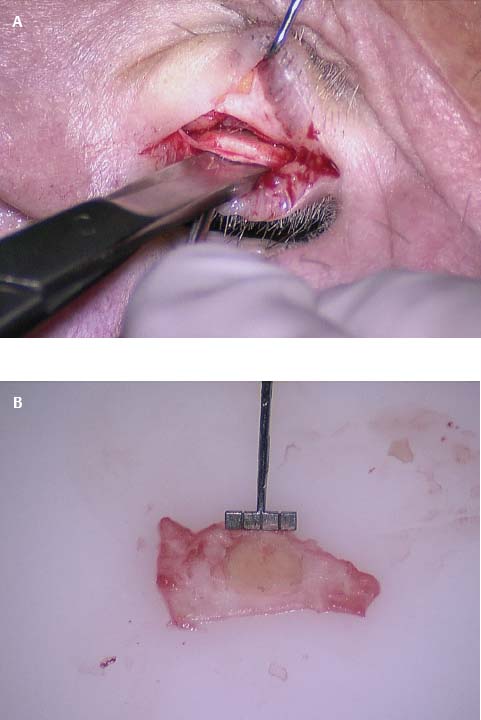
Fig. 2.22 Trimmed cartilage block attached to the perichondrial tissue.
♦ Laser-Assisted Myringoplasty
Laser-assisted myringoplasty is a minimally invasive surgical technique for a select group of patients, using the CO2 laser to reduce or eliminate tympanic membrane atelectasis. Our study revealed a significant reduction in the redundant tympanic membrane. Tympanic membrane atelectasis is the loss of normal contour and elasticity of the tympanic membrane as a result of persistent negative middle ear pressure. Atelectasis occurs in advanced chronic otitis media with or without effusion and often with chronic otorrhea. Atelectasis predisposes the patient to adhesive otitis, tympanic membrane atrophy, cholesteatoma formation, ossicular erosion, hearing loss, dizziness, and perforation. Atelectasis is one of the most difficult problems encountered with the tympanic membrane due to its chronicity and progression of severity over time. If identified in the early stages of retraction, atelectasis can be halted or even reversed by placing ventilation tubes to eliminate negative pressure in the middle ear. We have observed that even after placement of middle ear ventilation tubes to alleviate the effect of eustachian tube dysfunction, the tympanic membrane will often remain atelectatic or “floppy,” with persistence of a retraction pocket (Fig. 2.25).
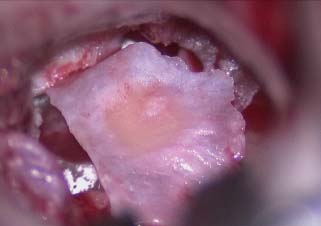
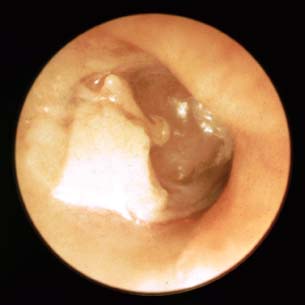
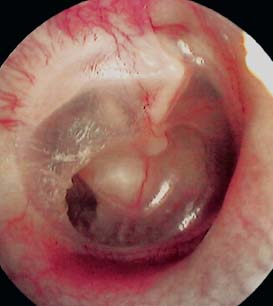
Fig. 2.25 Atelectatic tympanic membrane with a small retraction pocket.
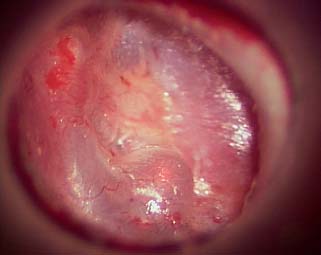
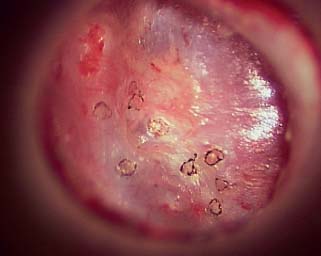
Fig. 2.27 Laser contracted tympanic membrane.
The redundancy and weakening of the tympanic membrane must be addressed. Traditionally, the approach is formal tympanoplasty with resection of the atelectatic portion of the membrane and reconstruction with a fascia graft. We sought to develop a minimally invasive clinical technique in which a CO2 laser is used to cause a thermal contraction of the tympanic membrane. This allows for tissue tightening and reduction in size of the atelectatic pocket without tympanoplasty. The laser can also be used to lyse fibrous scar bands arising on the perimeter of the atelectatic region of the tympanic membrane. Lysis of the fibrous bands also promotes reduction in the size of the atelectatic region of the membrane (Figs. 2.26, 2.27, and 2.28).
Our conclusion has been consistent, reproducible descriptions of tympanic membrane atelectasis are necessary. Attention must be made to size, location, and depth of the atelectatic region, and this information must be conveyed in the literature via a detailed tympanic membrane atelectasis grading system. This will permit a standardized reporting format.
♦ Conclusion
Over–under tympanoplasty with fascia or perichondrium and cartilage is the preferred method of tympanoplasty for the indications previously noted. Flexibility of technique is insured by the experience of the surgeon, and with any technique the surgeon should review long-term results in success of take rate, hearing improvement, and low complication rate. This technique should be included in the repertoire of the otologic surgeon.
References
1. Litton WB. Epithelial migration over tympanic membrane and external canal. Arch Otolaryngol 1963;77:254–257
2. Boxall JD, Proops DW, Michaels L. The specific locomotive activity of tympanic membrane and cholesteatoma epithelium in tissue culture. J Otolaryngol 1988;17 (4):140–144
3. Mondain M, Ryan A. Epidermal growth factor and basic fibroblast growth factor are induced in guinea-pig tympanic membrane following traumatic perforation. Acta Otolaryngol 1995;115 (1):50–54
4. Shimada T, Lim DJ. The fiber arrangement of the human tympanic membrane: a scanning electron microscopic observation. Ann Otol Rhinol Laryngol 1971;80 (2):210–217
5. Decraemer WF, Dirckx JJ, Funnell WR. Shape and derived geometrical parameters of the adult, human tympanic membrane measured with a phase-shift moiré interferometer. Hear Res 1991;51 (1):107–121
6. Hussl B, Timpl R, Lim D, Ginsel M, Wick GG. Immunohistochemical analysis of connective tissue components in tympanosclerosis. In: Lim DJ, Bluestone CD, Klein JO, Nelson JD, eds. Recent Advances in Otitis Media: Proceedings of the Fourth International Symposium. New York: BC Decker; 1988:402–406
7. Wilson JG. Nerves and nerve endings in the membrana tympani of man. Am J Anat 1911;11:101–115
8. Applebaum EL, Deutsch EC. Fluorescein angiography of the tympanic membrane. Laryngoscope 1985;95 (9 Pt 1):1054–1058
9. Committee on Conservation of Hearing of the American Academy of Ophthalmology and Otolaryngology: Standard classification for surgery of chronic ear disease. Arch Otolaryngol 1964;81:204–205
10. Hippocrates. De Carnibus [in German and Greek]. Teubren BB, trans. Leipzig, Berlin, Germany; 1935
11. Banzer M. Disputatio de Audiotone Laesa. Wittenbergae: Johannis Rohrerei; 1651
12. Toynbee J. On the Use of an Artificial Membrane Tympani in Cases of Deafness Dependent Upon Perforations or Destruction of the Natural Organ. London: J Churchill & Sons; 1853
13. Blake CJ. Transactions of the first Congress of the International Otological Society. New York: D Appleton & Company; 1887
14. Berthold E. Ueber myringoplastik. Wier Med Bull 1878;1:627
15. Wullstein H. The restoration of the function of the middle ear, in chronic otitis media. Ann Otol Rhinol Laryngol 1956;65 (4):1021–1041
16. Zollner F. The principles of plastic surgery of the sound-conducting apparatus. J Laryngol Otol 1955;69 (10):637–652
17. Plester D. Myringoplasty methods. Arch Otolaryngol 1963;78:310–316
18. Sooy FA. A method of repairing a large marginal tympanic perforation. Ann Otol Rhinol Laryngol 1956;65 (4):911–914
19. House WF, Sheehy JL. Myringoplasty: use of ear canal skin compared with other techniques. Arch Otolaryngol 1961;73:407–415
20. Plester D. Myringoplasty methods. Arch Otolaryngol 1963;78:310–316
21. Shea JJ Jr. Fenestration of the oval window. Ann Otol Rhinol Laryngol 1958;67 (4):932–951
22. Austin DF, Shea JJ Jr. A new system of tympanoplasty using vein graft. Laryngoscope 1961;71:596–611
23. Tabb HG. Closure of perforations of the tympanic membrane by vein grafts: a preliminary report of twenty cases. Laryngoscope 1960;70:271–286
24. Storrs LA. Myringoplasty with the use of fascia grafts. Arch Otolaryngol 1961;74:45–49
25. Chalat NI. Tympanic membrane transplant. Harper Hosp Bull 1964;22: 27–34
26. Glasscock ME III, House WF. Homograft reconstruction of the middle ear: a preliminary report. Laryngoscope 1968;78 (7):1219–1225
27. Perkins R. Human homograft otologic tissue transplantation buffered formaldehyde preparation. Trans Am Acad Ophthalmol Otolaryngol 1970;74 (2):278–282
28. Smith MF. Viable homograft reconstruction of the middle ear transformer mechanism and posterior osseous external canal. Trans Pac Coast Otoophthalmol Soc Annu Meet 1972;53:63–69
29. Wehrs RE. Homograft tympanic membrane in tympanoplasty. Arch Otolaryngol 1971;93 (2):132–139
30. Goodman WS. Tympanoplasty: areolar tissue graft. Laryngoscope 1971;81 (11):1819–1825
31. Goodhill V. Tragal perichondrium and cartilage in tympanoplasty. Arch Otolaryngol 1967;85 (5):480–491
32. Patterson ME, Lockwood RW, Sheehy JL. Temporalis fascia in tympanic membrane grafting: tissue culture and animal studies. Arch Otolaryngol 1967;85 (3):287–291
33. Sheehy JL, Glasscock ME III. Tympanic membrane grafting with temporalis fascia. Arch Otolaryngol 1967;86 (4):391–402
34. Bojrab DI, Causse JB, Battista RA, Vincent R, Gratacap B, Vandeventer G. Ossiculoplasty with composite prostheses: overview and analysis. Otolaryngol Clin North Am 1994;27 (4):759–776
35. McFeely WJ Jr, Bojrab DI, Kartush JM. Tympanic membrane perforation repair using AlloDerm. Otolaryngol Head Neck Surg 2000;123 (1 Pt 1):17–21
36. Jackson CG, Glasscock ME III, Nissen AJ, Schwaber MK, Bojrab DI. Open mastoid procedures: contemporary indications and surgical technique. Laryngoscope 1985;95 (9 Pt 1):1037–1043
37. Dornhoffer J. Cartilage tympanoplasty: indications, techniques, and outcomes in a 1,000-patient series. Laryngoscope 2003;113 (11):1844–1856
38. Austin DF. Transcanal tympanoplasty. Otolaryngol Clin North Am 1972;5 (1):127–143
39. Glasscock ME III. Tympanic membrane grafting with fascia: overlay vs. undersurface technique. Laryngoscope 1973;83 (5):754–770
40. Wehrs RE. Grafting techniques. Otolaryngol Clin North Am 1999;32 (3): 443–455
41. Hough JV. Tympanoplasty with the interior fascial graft technique and ossicular reconstruction. Laryngoscope 1970;80 (9):1385–1413
42. Kartush JM, Michaelides EM, Becvarovski Z, LaRouere MJ. Over-under tympanoplasty. Laryngoscope 2002;112 (5):802–807
43. Gerber MJ, Mason JC, Lambert PR. Hearing results after primary cartilage tympanoplasty. Laryngoscope 2000;110 (12):1994–1999
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


