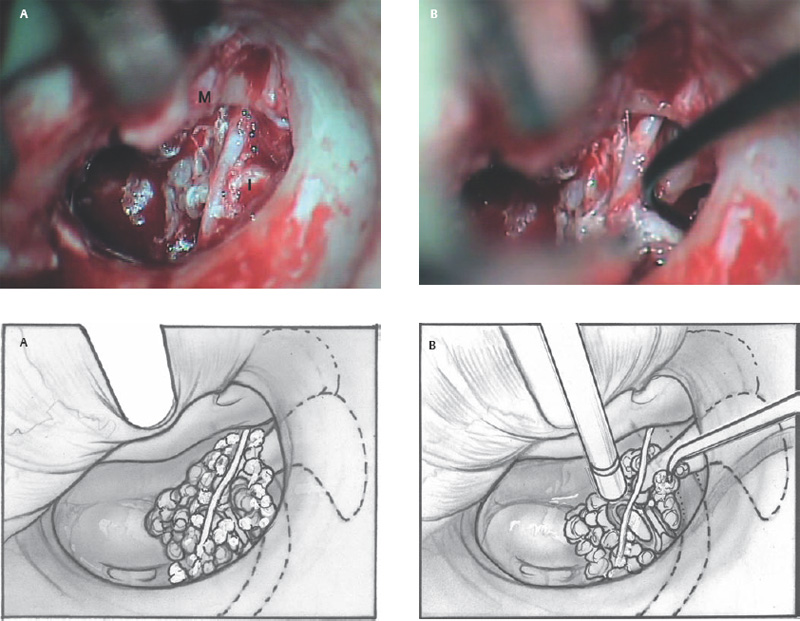
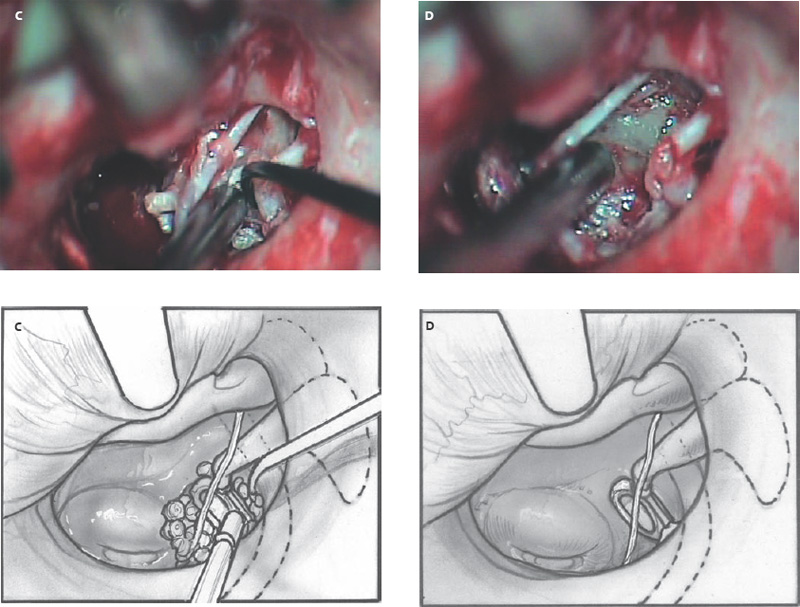
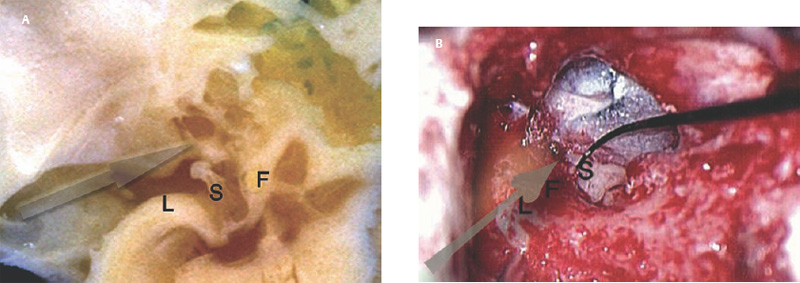
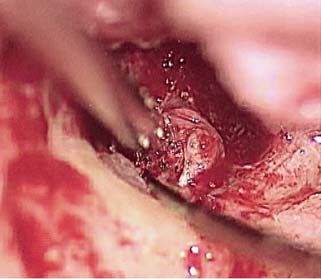
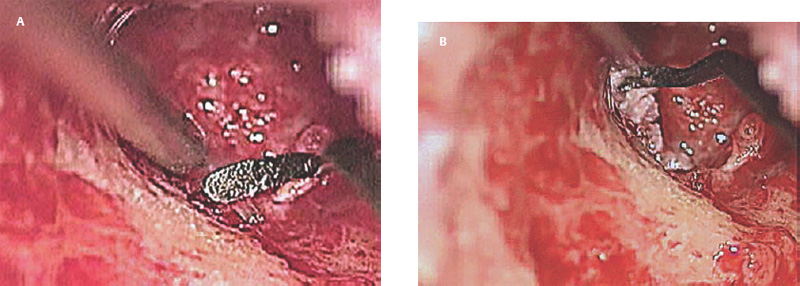
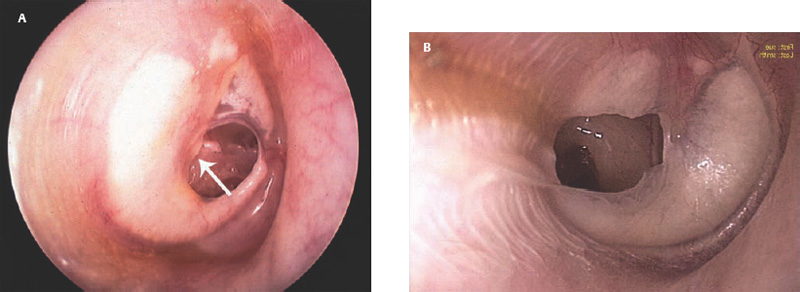
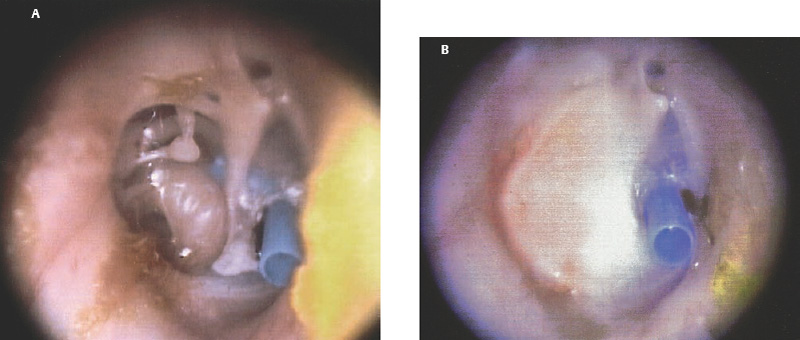
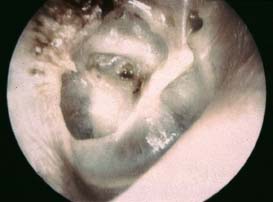
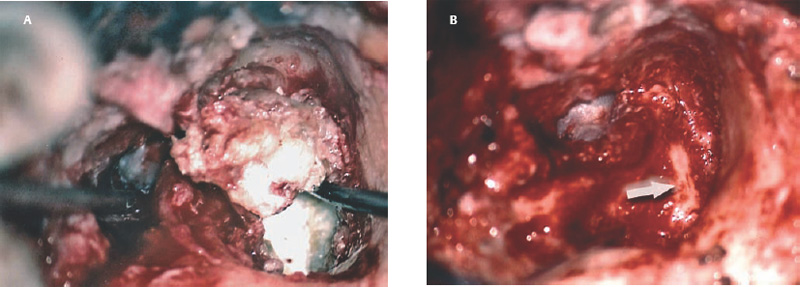
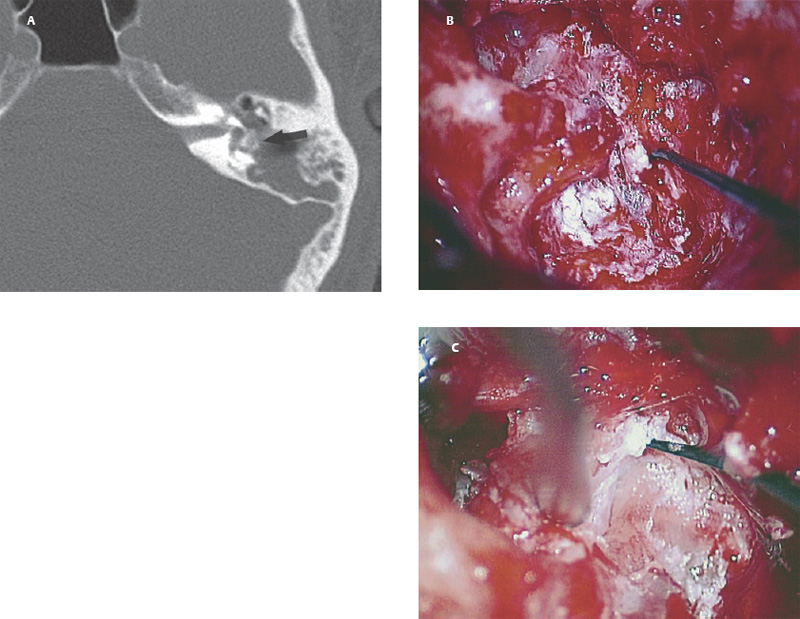
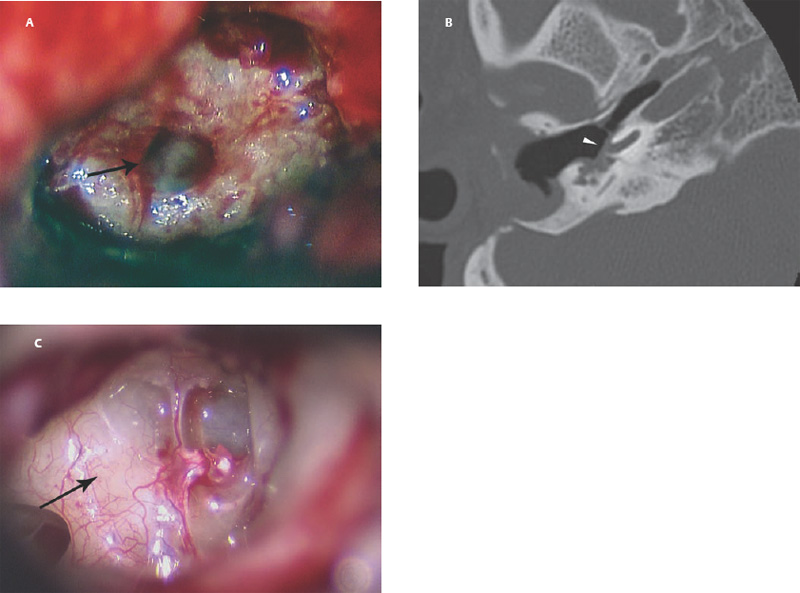
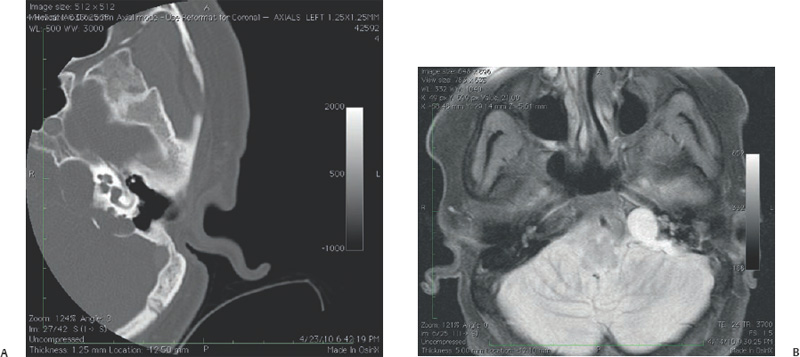
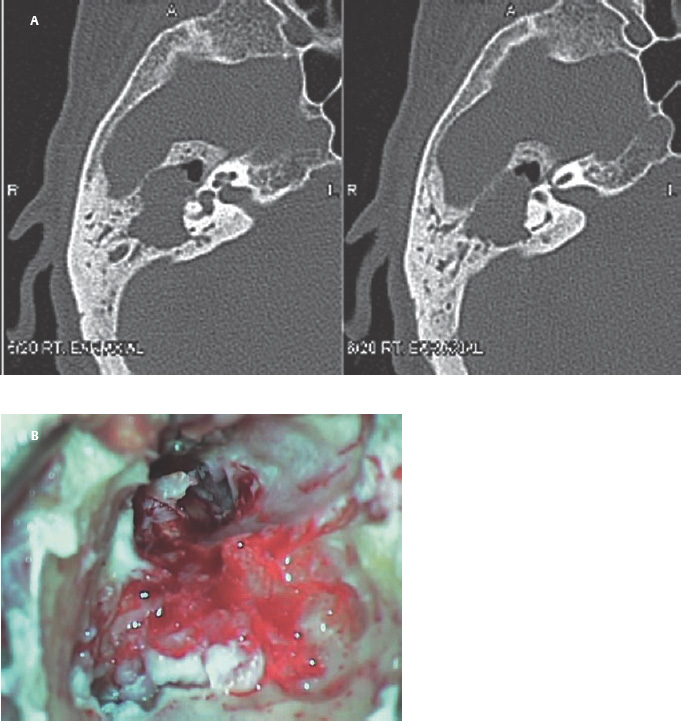
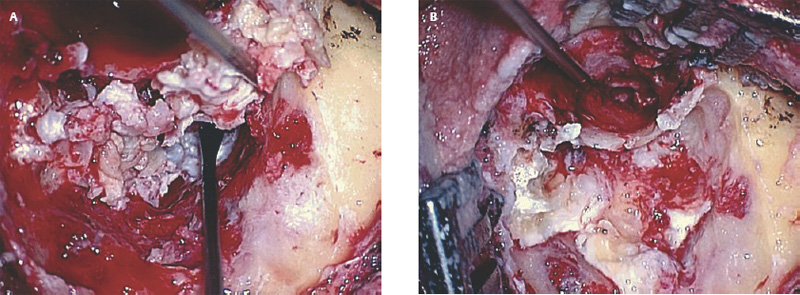
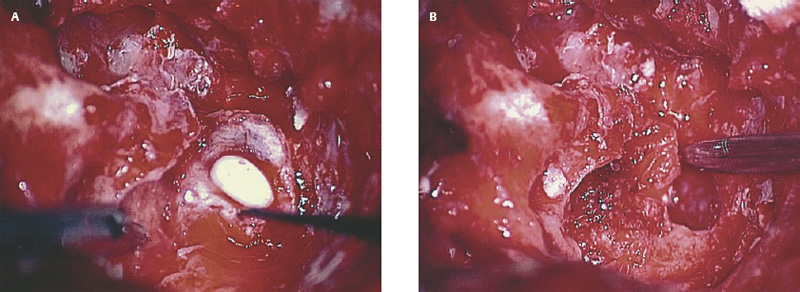
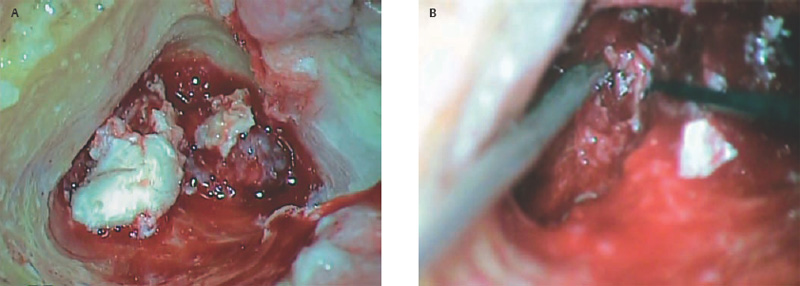
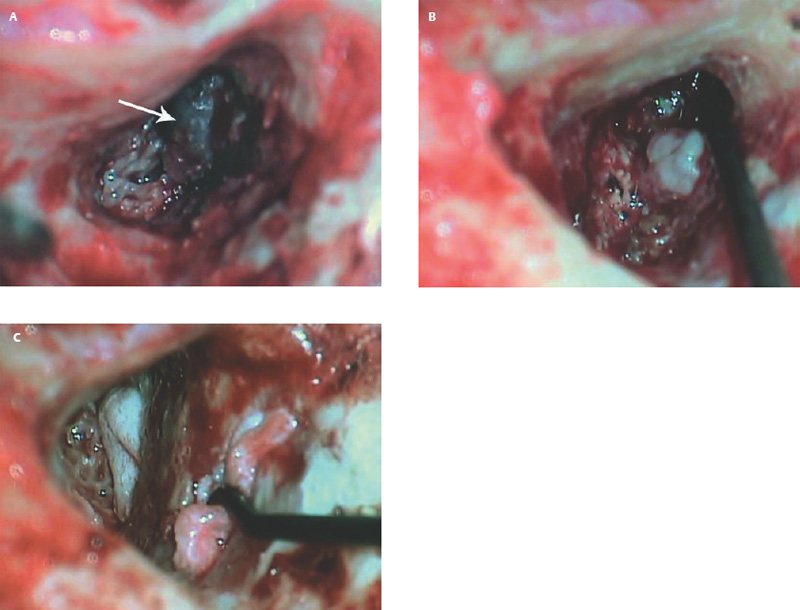
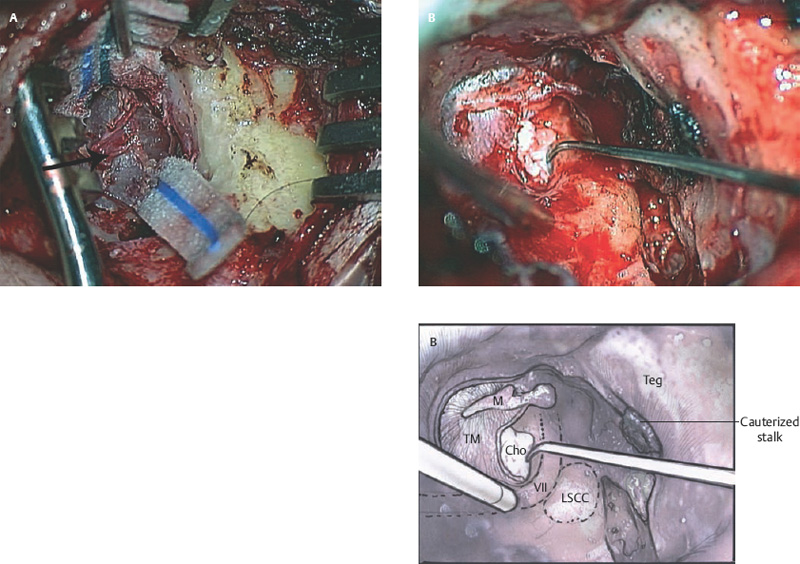
6 Situations That Arise at Surgery: Anatomical Issues The unique biological behavior of cholesteatoma and the intricate anatomy of the temporal bone create challenges during surgery. These situations are discussed individually or together. In this chapter we will address some of the more common anatomical situations that may arise. These include disease on the oval window; disease in the sinus tympani (the most frequent site for recurrence); disease on the bare facial nerve; lateral semicircular canal fistula; cholesteatosis/giant cholesteatoma; and cerebrospinal fluid (CSF) leakage/encephalocele. In the next chapter, we will discuss hearing issues that arise at surgery, namely, intact ossicular chain, disease in the normal, better, or only-hearing ear, and bilateral cholesteatoma. Dissecting disease from the stapes and oval window is a common event at surgery. Cholesteatoma in the middle ear can insinuate itself in and around the stapes. This is particularly true of retraction pocket cholesteatoma of the pars tensa that forms in the posterosuperior quadrant and travels medially toward the promontory and posteriorly into the sinus tympani. The stapes superstructure and lenticular process are contained within a mucosal envelope that can act as a conduit for the vertical spread of disease from the tympanic membrane to the promontory (Fig. 6.1).1 Attic cholesteatoma can also involve the stapes by growing medially from the posterior epitympanic space along the long process of the incus. Surgical access to cholesteatoma of the middle ear is gained through the external ear canal and medial to the remainder of the tympanic membrane when intact. Surgical dissection around the stapes is best approached in a stepwise, superior to inferior direction (Fig. 6.2A). If the long process of the incus is still intact, it can be followed inferiorly and sometimes preserved. If the lenticular process of the incus has been eroded (a common occurrence), the incus can be lifted laterally or, if necessary, removed for better exposure. This is necessary when disease extends into the attic in the plane medial to the incus. Once the incus is fully disarticulated, curettage of the scutum provides access to the epitympanum, which facilitates removal of the incus. Cholesteatoma that extends superiorly will require an atticotomy to access the disease. Fig. 6.2 Dissection of a left middle ear cholesteatoma from the stapes, left ear. (A) Cholesteatoma enveloping stapes. M, malleus; I, incus, eroded. (B) Dissection proceeds in a downward direction, exposing eroded incus, and freeing disease from capitulum while stabilizing crura with suction. (C) Matrix is then peeled away from inferior aspect crura. (D) Completed dissection. The stapes can be difficult to identify visually when it is covered or encased by granulation tissue and cholesteatoma. Gentle palpation should be performed to locate the superstructure before lifting the cholesteatoma from this area. In some instances, the stapes may be deflected inferiorly toward the promontory and can be unintentionally fractured or mobilized if this is not recognized. Once the stapes head is identified by palpation and inspection, disease can be peeled off the crura in medial-to-lateral and posterior-to-anterior directions using the trailing edge of a sharp pick (Fig. 6.2B–D). The stapedial tendon is often present and provides countertraction during the posterior to anterior dissection. The side of a 3F Baron suction tip can be used to steady the crura during this maneuver. The matrix can then be sharply dissected off the footplate and from between the crura. This maneuver requires patience and a steady hand. If this proves to be difficult, raising concern that continued dissection may lead to stapes mobilization and extraction, consideration should be given toward staging the procedure and returning at a future date. Occasionally the crura are eroded and may become dislocated from the footplate.2 Using a sweeping motion will ensure that the crura are not forcibly broken because the footplate may then fracture or become mobilized. If a perilymph leak does develop through a fractured footplate or a torn annular ligament, it should be immediately covered with a graft of fascia or areolar tissue. Traumatic luxation of the stapes can result in vertigo and permanent sensorineural hearing loss, and so this aspect of the dissection must be done with great care.3–5 Not uncommonly, the entire superstructure will have been eroded by the disease, and the matrix will be adherent to the footplate. Complete removal can be difficult because the defined matrix layer is often lacking in this location, and the footplate may be covered with inflammatory granulation tissue.2 In these cases, applying firm but gentle pressure with the tip of a curved needle or hockey stick (whirlybird) dissector will allow an edge of the matrix to be elevated off the stapes footplate, still working in a superior to inferior direction. If the matrix breaks apart, the same technique is used but the disease will be delivered piecemeal. It is important not to leave residual matrix in the sulcus between the oval window and the facial nerve. If additional exposure to the sinus tympani is needed, the canal wall down approach provides a less encumbered view of the oval window and better access to the sinus tympani. From the surgeon’s vantage point—looking from the mastoid toward the middle ear—the lateral semicircular canal, facial nerve, and stapes fall in a line and lie at the same depth (Fig. 6.3A,B). The lateral semicircular canal is the most consistent landmark, located at the base of the mastoid antrum. Cholesteatoma matrix should be lifted off the dome of the lateral canal, working in a posterior-to-anterior direction from the antrum toward the middle ear. Once the dense ivory bone of the lateral semicircular canal is exposed, the matrix is peeled away from the lateral aspect of the facial nerve, looking carefully for any dehiscence and avoiding pressure from the instrument against the facial nerve. The sulcus between the facial nerve (fallopian canal) and the lip of the oval window forms an acute angle, so the surgeon should not disrupt the matrix in this area. The disease should then be lifted off the stapes superstructure, taking care not to rock the stapes excessively (Fig. 6.4). Finding the superstructure may be tricky—it may be covered by disease, displaced inferiorly, or absent—but it can usually be located by palpating through the matrix with a pick, stripping disease from posterior to anterior using a cupped forceps or sharply cutting away the inflammatory/cholesteatoma mass with a fine microscissors. The pyramidal process can be a good landmark for finding the stapes in disease; it transmits the stapedius tendon that attaches to the posterior crus of the stapes. Once the superstructure is located, the disease can be dissected off , working from the base of the posterior crus toward the capitulum, while stabilizing the crura with a suction (Fig. 6.4). The stapes crura may be eroded, and the superstructure can occasionally become detached from the footplate during the dissection. Sometimes erosion of the crura will not be apparent until after they are exposed. Fig. 6.3 (A) The lateral semicircular canal (L), facial nerve (F), and stapes (S) lie at the same depth and in a straight line, as shown in this cross-sectional view of a cadaver temporal bone, and in (B) a surgical photograph of a left ear mastoidectomy. The surgeon’s line of sight is shown (arrow) when looking from the mastoid toward the middle ear. In canal wall up surgery, the entire oval window is not as easy to expose. A purely transcanal, trans–middle ear approach can be used, but if the disease extends back toward the sinus tympani or superiorly toward the attic the bony canal will have to be widened. If a mastoidectomy is done first, a transmastoid atticotomy plus facial recess approach will provide exposure to the posterior middle ear and give better access to the sinus tympani. The route to the oval window through the facial recess is the same as in canal wall down. However, the angle of visualization is more acute from the perspective of the mastoid. Again, the lateral semicircular canal, facial nerve, and stapes fall in a line. In most cases, when disease is present in the posterior epitympanic space, the incus will have to be removed. It is important to thin the posterior bony canal wall to its cortical layer with a diamond drill so as to gain exposure as anteriorly as possible. It is useful when drilling to conceptualize the shape of the bony canal wall as a truncated cone rather than a cylinder. The incus buttress can be maintained, but more often it is helpful to drill it away, allowing better access to the area around the stapes. In these surgical situations, the incus is frequently eroded, justifying removal of the diseased incus. If the incudostapedial joint is still intact it should be disarticulated and the incus pushed superiorly into the attic. In canal wall up surgery, the strategy should be to mobilize the matrix from behind forward, dissecting it off the lateral canal and facial nerve superiorly, and out of the sinus tympani posteriorly, toward the stapes superstructure. By rotating the operating table toward the surgeon, the middle ear dissection can be accomplished through the ear canal. In difficult cases, it may be necessary to leave matrix attached to the footplate. With this situation, Silastic sheeting can be placed in the middle ear and the tympanic membrane grafted. At the second stage, the cholesteatoma (frequently a pearl remnant) is resected, then a footplate to malleus or footplate to tympanic membrane (type V) reconstruction is performed. Alternatively, it may be necessary to leave matrix attached to the footplate. This situation requires that the middle ear be left open, and a second procedure is done at a later stage to remove the residual. Ossicular reconstruction can be performed after removing disease from the footplate, provided the surgeon is confident that there is no residual disease. A partial prosthesis can be used if the stapes superstructure is intact, or a total prosthesis if the superstructure is absent. Sometimes a total prosthesis can be placed against the footplate between the crura if the superstructure is tenuous or leaning forward, which helps to maintain stability of the prosthesis. The sinus tympani, or tympanic sinus, is a triangular space that communicates with the mesotympanum. It is bounded superiorly and inferiorly by two ridges of bone, the ponticulum and subiculum, and laterally by the fallopian canal of the facial nerve. The presence of the facial nerve prevents the sinus tympani from being unroofed, which prohibits complete visualization of its contents. It is often not possible to fully inspect the confines of this space, raising the risk of leaving residual disease. It is important to attempt to keep the entire sac or retraction pocket intact to insure its total removal. Sinus tympani cholesteatoma arises most often from a retraction pocket of the posterosuperior quadrant of the tympanic membrane, which, when it develops in this area, can grow medialward and become invasive.6 It is also encountered in attic cholesteatomas that extend downward toward the middle ear, and in middle ear atelectasis, where a retraction pocket can extend into the sinus and be very difficult to remove. A marginal perforation at the posterior annulus may also give rise to keratin matrix that migrates into the sinus tympani. The anatomy of the sinus tympani is shown in Fig. 6.5. The ponticulum, which forms the superior limit, is a bridge of bone found at the level of the inferior lip of the oval window. The subiculum, which forms the inferior boundary, is at the level of the superior lip of the round window. The sinus tympani extends medial to the mastoid segment of the facial nerve. Its size is variable, and it can be quite deep and extensive. A related space, termed the posterior sinus, lies superior to the sinus tympani and just posterior to the stapes. At times this space is contiguous with the sinus tympani, if the ponticulum is rudimentary in size. The anatomy of these spaces has been well demonstrated and beautifully illustrated in recent publications by Holt and Marchioni et al.7,8 Disease enters the sinus tympani by migrating medially along the lenticular process and stapes superstructure, then posteriorly along the plane of the promontory. This direction of growth can be appreciated in cases of retraction pockets arising from posterior middle ear collapse (Fig. 6.6A). Surgical access to the sinus tympani is made challenging by the presence of the facial nerve. The sinus can be accessed either through the middle ear via a tympanoplasty approach, enlarging the posterior bony canal and scutum, or through the mastoid via facial recess or canal wall down approaches. Both approaches require that the bone anterior to the facial nerve be thinned aggressively to provide as much exposure as possible. Even so, clearing disease from the apex of the sinus tympani is a partly blind maneuver. In the tympanoplasty approach, a generous bony canalplasty should be done first, removing bone posteriorly to the mastoid segment of the facial nerve.9 This is done by saucerizing the posterior canal bone until a large, concave contour results, identifying the facial nerve at the second genu and following it inferiorly. To fashion a round canal, it is necessary to remove additional bone from the inferior and superior canal wall. The latter initiates a transcanal atticotomy. In cases where disease involves the attic and antrum, it may be safer to assure exteriorization performing a canal wall down mastoidectomy. In the facial recess approach, the facial recess opening must be sufficient to allow maximal visualization as well as to permit instrumentation. This requires creating an adequate vertical opening (∼3 mm in height) by thinning the posterior canal wall aggressively, at least up to the chorda tympani, and sometimes transecting the chorda to expose the fibrous annulus. The facial nerve should then be skeletonized and the bony ledge anterior to the nerve removed as well (Fig. 6.6B). This requires using a diamond burr to define the circumference of the nerve, as previously discussed in Chapter 4. Sometimes the pyramidal process will also need to be removed. Once the bony approach is completed, the patient’s head can be tilted (by airplaning the operating table) so as to gain the best possible view into the sinus. Because the facial nerve is the limiting factor, the exposure obtained through the facial recess approach should be similar to the transcanal approach already described (Fig. 6.6C). If still greater access is needed, the canal wall should be taken down, initially to the chorda tympani inlet; and if this does not provide enough exposure then the canal wall is taken down to the level of the facial nerve. Fig. 6.6 Dissection of a cholesteatoma from the sinus tympani via the facial recess, left ear. (A) Middle ear cholesteatoma viewed through the ear canal, extending posteriorly toward the sinus tympani (arrow). (B) Mastoidectomy with a facial recess approach exposes the posterior limit of the cholesteatoma sac. (C) Skeletonizing the mastoid segment of the facial nerve allows access to the sinus tympani (arrow). (D) After dissecting the sac from the sinus tympani, a mirror is used to confirm that there is no residual disease. Once adequate surgical exposure is obtained, the disease can be dissected using blunt right angle instruments. A whirlybird dissector (equivalent to the Hough excavator) or Derlacki elevator is well suited for this task. The object is to create a plane between the bone and the outside of the epithelial capsule to deliver the sac in toto. The matrix should be held together as a shell if possible (Fig. 6.7A,B). Piecemeal dissection should be avoided. If the capsule starts to break, a new point of dissection should be started. If the sac cannot be held together, then the debris should be evacuated, and a peeling action should be used to deliver the sac. A mirror or endoscope is very helpful to inspect the sinus tympani after dissection to ascertain that no residual disease remains (Fig. 6.6D). Residual cholesteatoma has a pearlescent appearance and is usually easy to discern from mucosa. A mirror is simpler to set up than an endoscope because the microscope does not have to be moved; however, it is tricky to use and requires a bit of practice. The otoendoscope has gained increasing attention for enhanced visualization to facilitate complete removal of sinus tympani disease.10 The endoscope offers the benefit of superb optics and provides a panoramic view, allowing eradication of the cholesteatoma, often through a purely transcanal approach. The endoscope requires a modification of technique, where the scope must be steadied during the surgical dissection. If the operating surgeon holds the endoscope while viewing the TV monitor, only one hand is available for surgical dissection. An alternative method requires two sets of hands as well as access from both sides of the operating table. When using the endoscope, care must be taken to avoid traumatizing the stapes crura. The instrument is compromised by the presence of blood given that it may be difficult to navigate a suction tip into the sinus cavity. Chemical defogging agents are also generally needed. In cases of very deep sinus tympani, a retrofacial approach has been proposed. This requires complete circumferential skeletonization of the nerve and a superior angle of drilling. This approach should only rarely be needed.11 Cases of middle ear atelectasis with sinus tympani retraction pockets are particularly difficult to manage. Because the sac is empty, it can fragment easily requiring great care and patience to remove the sac completely. Leonetti et al have reported on a technique of exteriorizing the sinus tympani and dissecting the sac in toto in a series of cases, with very good control over long-term follow-up.12,13 The technique they describe requires that a myringotomy tube be maintained long term to prevent re-formation of the sac. Tympanic membrane cholesteatoma is a special situation that may arise during surgery and is occasionally unexpected by the surgeon. In the office, a perforation may be noticed with a whitish discoloration of the remaining tympanic membrane. On first otoscopic examination, one may believe that this is tympanic membrane tympanosclerosis. Occasionally, there may be migratory epithelium as seen in the figure, which will alert the surgeon to the presence of this same type of migratory epithelium on the undersurface of the tympanic membrane (Fig. 6.8A,B). The usual approach to tympanic membrane surgery is undertaken. When the tympanomeatal flap is elevated, the undersurface of the tympanic membrane is found to have a thin layer of squamous epithelium lacing the medial layer. Occasionally there may be free cholesteatoma in the middle ear and on the promontory. The epithelial matrix may extend along the tympanic membrane to the fibrous annulus and onto the mucosal layer of the middle ear cleft. The matrix may also wrap around the malleus and tensor tympani muscle. A 30-degree telescope is useful to inspect the undersurface of the tympanic membrane to define the extent of involvement. The operation must eradicate the disease and may need to be quite extensive. If the malleus and tensor muscle are involved, then the surgeon must remove most of the tympanic membrane and perform either an underlay or over–under tympanoplasty to make certain of total removal of this process. It may even be necessary to disarticulate the incudostapedial joint to gain enough access to totally remove the epithelial matrix around the malleus and tensor muscle. Ossicular reconstruction with an incus interposition graft is then performed. Close patient follow- up is necessary for years to make sure there is no recurrence. Posterior collapse of the tympanic membrane that reduces the middle ear space presents a challenge. Many times one will find a contracted middle ear space where the annulus is closer to the promontory (especially inferiorly) than in other diseased ears. Dissecting the squamous epithelium from the oval window, sinus tympani, and round window is difficult to do without leaving disease behind, as discussed in the previous sections. Creating and maintaining a pneumatized middle ear space is an additional challenge because a severely retracted tympanic membrane (or narrow middle ear cleft) has the propensity to re-retract over time. A tympanostomy tube will probably not correct the problem once middle ear adhesions have formed (Fig. 6.9A). There are two possible ways to deal with a collapsed middle ear space—either the tympanic membrane can be elevated and the middle ear space reconstituted with or without an intact canal wall mastoidectomy,14 or a canal wall down mastoidectomy can be performed, exteriorizing the disease process. Preoperatively, an estimate can be made of the potential for reconstituting the middle ear space by trying to lift the tympanic membrane away from the promontory with pneumatic otoscopy or by having the patient perform a forceful Valsalva maneuver. It is helpful to judge whether there is aeration anterior to the malleus—if so, it is possible that the posterior disease is old and there is now adequate eustachian tube function. Even in patients with complete atelectasis of the tympanic membrane, eustachian tube function may still be adequate to re-create an aerated middle ear space, provided that the patient does not have severe allergies, gastroesophageal reflux, history of smoking, sleep apnea, or severe nasal and sinus disease. If the middle ear can be partially or totally inflated, then one can perform a tympanoplasty (with or without an intact canal wall mastoidectomy), placing one or two sheets of medium-thick silicone (Silastic) sheeting on the promontory to maintain the space.15 When this is done in conjunction with a canal wall up mastoidectomy, the Silastic sheeting can be placed through the facial recess. Cartilage may be a better grafting material than fascia in these cases because it provides stiffness and greater resistance to retraction (Fig. 6.9B).16 If there is erosion of the incus and stapes superstructure, an ossiculoplasty with a total replacement prosthesis can be performed at the same time. If the superstructure is present, a partial replacement prosthesis can be used or the drum or graft can be attached directly to the stapes head (“myringostapediopexy,” or type III ossicular reconstruction). In the short term, a ventilating tube may be helpful to prevent recurrent middle ear collapse,17 but if a cartilage tympanoplasty is done this may not be necessary.18 In many cases, however, the tympanic membrane will be firmly adherent to the promontory mucosa and the middle ear space cannot be re-created. In such a situation, it is better to leave the tympanic membrane epithelium in place and perform a canal wall down (modified radical) mastoidectomy or atticotomy (in a mastoid with limited pneumatization) so as to exteriorize the disease (Fig. 6.10). This will have the effect of creating an open cavity with a lower chance of recurrence. Middle ear function will be compromised, but the tympanic membrane will remain stable in this position over time. Hearing results will result in a moderate air–bone gap, but a maximum conductive hearing loss may be avoided if there is isolation between the round window and oval window by having the tympanic membrane tented over the promontory leaving a residual air space over the round window (this is known as a cavum minor or type IV ossicular reconstruction). The technique of re-creating a middle ear is illustrated here. At surgery, the ear is first inspected, and a suction can be used to assess whether the tympanic membrane is adherent to the promontory mucosa (Fig. 6.11A). The tympanomeatal flap is elevated up to the fibrous annulus. The tympanic membrane epithelium will travel medial and posterior to the bony annulus and can be difficult to separate. One should try to find a pocket of pneumatization to begin dissection. This can often be found inferiorly toward the hypotympanum or anteriorly at the eustachian tube entrance. Curetting the bony annulus is helpful to gain exposure in the posterior mesotympanum. Care should be taken not to tear the flap because the thin epithelium will be difficult to retrieve from the posterior middle ear space once it becomes separated. The technique requires a curved (Rosen) needle or right angle dissector, delicate technique, and patience. Once the middle ear space is entered, the sac is bluntly dissected from the annulus, working posteriorly toward the sinus tympani (Fig. 6.11B). One should attempt to remove disease in continuity from this area. If the disease tracks posteriorly into the sinus tympani, a canal wall up mastoidectomy with facial recess may be necessary to gain adequate exposure (Fig. 6.11C). Dissection is continued anteriorly and superiorly, separating the tympanic membrane from the promontory while attempting to leave an intact layer of mucosa (tympanolysis). The resulting flap will be thin, and there will be redundant epithelium. An underlay graft of cartilage, taken from the tragus, cimbum, or cavum concha can be used to repair the tympanic membrane and to provide some stiffness (Fig. 6.11D). Silastic sheeting can be placed on the promontory to prevent recurrent adhesions. If a mastoidectomy is required, a canal wall up procedure with facial recess should be performed. In that case, the Silastic can be placed over the promontory with an extension of it going through the facial recess. Despite this being a staged procedure with the intention of returning to remove the Silastic, the interim hearing results may be surprisingly good due to conduction of sound energy through the Silastic on the stapes. The status of the stapes determines the alternatives for ossicular reconstruction. If the superstructure is eroded attempts should be directed toward removing disease off the footplate. Ossicular reconstruction using sculpted incus interposition or total prosthesis can then be performed. If the superstructure is present, the matrix is dissected off the crura using a meticulous and careful technique. If disease is very adherent to the crura, sharp dissection should be used to remove it, or a potassium titanyl phosphate (KTP) or argon laser can be used to vaporize the disease. If the tympanic membrane is attached to the stapes head but not the crura, it can be left intact as a type III reconstruction (myringostapediopexy). Placing an interface of cartilage between the tympanic membrane and head of the stapes provides a greater surface area for transmission of sound vibrations. If there is no pneumatization and no possibility of separating the tympanic membrane from the stapes footplate, then a canal wall down (modified radical) mastoidectomy with type IV tympanoplasty (cavum minor) can be performed. There is a risk that a retraction pocket may re-form after surgery. If recurrent retraction develops, one should consider inserting a small ventilating tube. This can usually be done postoperatively in the office. Alternatively, a subannular ventilating tube can be placed at the original surgery to provide long-term ventilation. In both attic and middle ear cholesteatomas, the matrix is frequently found covering the tympanic segment of the facial nerve. Familiarity with the anatomy of the nerve in the temporal bone is essential because its position must be anticipated before removing the soft tissue overlying it. Live intraoperative facial nerve monitoring and stimulation are helpful but are not a substitute for anatomical knowledge or proper technique. The facial nerve normally has a thin bony covering (fallopian canal), which is dehiscent in at least 15% of cases (the percentage is probably higher in cholesteatoma because of its erosive nature).19–22 These dehiscences usually occur along the inferior aspect of the nerve superior to the stapes but may often involve the dorsum of the nerve between the processus cochleariformis and second genu. The processus is an excellent landmark for locating the tympanic segment of the nerve before exposing it. Knowledge of the anticipated location of the facial nerve dehiscence is key to avoiding surgical injury to the nerve. The facial nerve is best approached in a superior-to-inferior, posterior-to-anterior direction, using the lateral semicircular canal, stapes, and cochleariform process as landmarks. The facial nerve is approached as the sac is dissected off the medial wall of the attic and lateral semicircular canal. In canal wall up surgery, this requires an adequate (transmastoid) atticotomy. The incus and malleus head will usually be lined by or eroded by disease and will need to be removed. The contents of the sac should be decompressed before the sac itself is lifted or peeled off the medial attic wall. By lifting the matrix in a continuous sheet from the medial attic wall toward the dorsum of the nerve, a plane is developed that allows the disease to be lifted cleanly and without pressure or direct contact with the nerve. This maneuver is illustrated in Fig. 6.12. Generally a sharp instrument such as a needle dissector is more effective than a blunt excavator for achieving this. In removing cholesteatoma from around the tympanic segment of the facial nerve, the matrix should be swept off the nerve, toward the stapes, without turning the tip of the instrument toward the facial nerve itself. The technique of safely dissecting disease from an exposed facial nerve becomes familiar as one gains experience. Dehiscence of the bony fallopian canal should be expected along its inferior surface, the most common location of the exposed nerve. At times, the lateral surface or even the entire circumference of the nerve may be dehiscent in the tympanic segment. The safest strategy is to approach the nerve from superior to inferior beginning along the lateral surface, lifting the matrix away from the nerve sheath while attempting to keep the matrix intact. In cases in which the bony canal is dehiscent, the epineurium may be covered by a layer of slightly inflamed mucosa. The corresponding “edema plane” also helps to protect the nerve from being pulled up with the matrix. Again, using the trailing point of a sharp pick allows this to be done safely and efficiently (Fig. 6.13A,B). The enclosed video that illustrates the removal of residual cholesteatoma from a bare facial nerve in a canal wall down cavity demonstrating the concept of the “edema plane.” A facial nerve monitor was in use during the dissection. A stimulating probe tip instrument was also used during this task. Stimulating instruments allow a small electric current (typically 0.05 to 1.0 mA) to be delivered during the critical part of the dissection, giving audible feedback to the surgeon about the proximity of the instrument tip to the bare nerve. Intraoperative facial nerve monitoring and stimulation can be useful adjuncts while dissecting disease off a bare nerve by providing feedback to the surgeon about the location and integrity of the nerve. The use of routine facial nerve monitoring during surgery of chronic ear disease remains controversial, but its benefit is convincing during a situation such as this one. The monitor can only be effective if it is present and working at the moment that it is needed. There continues to be disagreement about whether the facial nerve monitor should be used routinely and whether its use should constitute the standard of care for surgery of chronic ear disease. This issue is addressed more fully in Chapter 8. It should be remembered that if one encounters questions about the facial nerve dehiscence during surgery, the stimulator-monitor can be connected at that time, testing for nerve integrity prior to continuing facial nerve dissection. Lateral semicircular canal fistula is one of the more common situations encountered during cholesteatoma surgery and is often unrecognized beforehand. Cholesteatoma is an erosive disease. The bony covering of the lateral semicircular canal is susceptible to osteoclasis by the cholesteatoma matrix. Fistulas may be encountered in a significant percentage of cholesteatomas.23 The usual presentation of lateral semicircular canal fistula is vertigo, often brought on by the patient sneezing, coughing, spontaneous Valsalva (nose blowing), changing head position, or by pressing on the tragus. The expected clinical finding is a positive fistula test (Politzer sign or Hennebert sign), deviation of the eyes away from the affected ear with positive, or toward the affected ear with negative, pressure in the external canal. The fistula sign can be elicited with the examiner’s cupped hand over the external meatus with a pneumatic (Siegle) otoscope or Politzer bag. It should be noted, however, that many lateral semicircular canal fistulas are asymptomatic and are only discovered at surgery. Vertigo may be absent in one third of patients, and the fistula test may be negative 50% of the time.24 Sensorineural hearing loss may or may not be present. A preoperative computed tomographic (CT) scan is desirable when a fistula is suspected, either by a history of dizziness or by the finding of unilateral sensorineural hearing loss. Erosion of bone is typically observed in the axial images through the plane of the lateral semicircular canal. Fistulas can also be observed in the coronal plane, although an imprecise cut may miss the finding, and volume averaging can give a false-positive finding (Fig. 6.14A–C). CT may be falsely negative in 40% of cases.24 When a fistula is suspected at surgery, care should be taken to avoid aggressively manipulating disease over the lateral semicircular canal. A canal wall up procedure should be performed initially to debulk the contents of the sac. The matrix should be left over the lateral canal until most of the disease has been removed and the surgeon is ready to deal with the fistula. How the fistula should be handled remains a matter of debate. Classical teaching maintains that a canal wall down procedure should be done to exteriorize the disease, and a thin layer of matrix should be left in place over the fistula. The residual matrix provides a protective covering for the inner ear, and exteriorization of the cavity limits the potential for infection or continued bone erosion.25 The residual matrix is used as part of the lining of the new mastoid cavity. If cholesteatoma and granulation tissue are too firmly attached to the matrix to be removed safely, these are better left intact. Postoperative ototopical drops are used with great success to later control this process. Others have argued for completely removing the matrix over the fistula and resurfacing the labyrinth with fascia, cartilage, or bone pAte. The rationale is to avoid leaving any disease behind, which has the potential for regrowth or re-infection (Fig. 6.15A,B).23,26 It is believed that the inner ear can be safely unroofed without causing permanent loss of cochlear and vestibular function as long as the membranous portion is not violated The evidence for this comes from experimental research on surgical entry into the inner ear,27 from clinical experience with fenestration surgery for otosclerosis,26 and, more recently, with posterior semicircular canal occlusion for benign positional vertigo.28 As a result, some surgeons consider it safe to remove the remaining matrix from over the lateral canal and immediately seal the fistula with autologous material or even bone wax. Some have recommended that this be done in two stages,29 although the general experience of others suggests that this is not routinely necessary. Kobayashi et al30 went so far as to advocate that the lateral semicircular canal be intentionally transected, as a method to ablate the canal without creating sensorineural hearing loss. Even with this evidence, we believe it preferable to leave the matrix intact over the fistula whenever there is an infectious component to prevent potential invasion with bacteria. The results of cholesteatoma surgery in the presence of labyrinthine fistula are generally good, with about an 85% rate of postoperative hearing preservation and a < 10% rate of dead ears.24 The risk to the hearing appears to be similar whether or not the matrix is removed. Facial nerve exposure may be more common when fistula is present because there is erosive disease nearby. The long-term results of fistula repair have not been systematically studied or reported. Postoperatively, vertigo and sensitivity to tragal pressure may not disappear because the fistula remains covered by soft tissue and not bone. Vertigo may occur more readily than usual during postoperative cleaning as it does in patients who have had fenestration operations. The ear may be susceptible to delayed sensorineural hearing loss because of labyrinthine sclerosis or because the open area may allow bacterial toxins to enter the inner ear (Fig. 6.16A–C). Fistulas may also occur in recurrent cholesteatoma. Hakuba et al31 presented 25 labyrinthine fistulae occurring an average of 20 years after canal wall down mastoidectomy surgery. Leaving an open cavity does not guarantee that fistula will never form, especially in patients who have poorly saucerized mastoid cavities. These patients need routine follow-up for years. Common sense would dictate that labyrinthine fistula be suspected in every case of extensive or recurrent cholesteatoma (since preoperative vertigo is not always present, and CT does not always reveal the fistula). Whenever a fistula is suspected, the lateral semicircular canal should be approached in a careful and controlled manner. The core of the cholesteatoma (debris) should be completely debulked and the matrix removed everywhere except over the lateral semicircular canal. The remaining matrix is then slowly lifted under saline irrigation. If the matrix is unusually adherent or if infection is present, it is wise to leave the matrix intact and perform an open-cavity mastoidectomy. Postoperative antibiotics and steroids may be beneficial in these cases. Fistulae can also occur into the round or oval windows or by erosion of the promontory. Although these are far less common than lateral semicircular canal fistulae, they can occur nevertheless. Cholesteatoma that has eroded into the vestibule must be either removed completely or exteriorized, even though this has the potential to result in a dead ear. Figure 6.17 illustrates a case that presented with normal hearing. The cholesteatoma was dissected off the promontory revealing a fistula (Fig. 6.17A,B) that was repaired with fascia. Cochlear function was maintained. Six months later, the ear was reexplored for ossicular reconstruction, and the fistula was found to have closed with the new bone (Fig. 6.17C). Giant cholesteatomas are those that occur in well-developed mastoids and replace all the cellular bone. Cholesteatosis refers to cholesteatoma that burrows into all the pneumatized spaces. Petrous temporal bone cholesteatoma is a term given to cholesteatoma that invades or spreads medial to the otic capsule. These are biologically aggressive forms of cholesteatoma that will recur unless they are completely removed. The cause of this aggressive behavior is unknown: these lesions do not differ histologically from routine cholesteatoma. These cholesteatomas can be associated with labyrinthine erosion, facial nerve exposure, dural exposure, and extension to the petrous apex. The surgical procedure will almost always be a canal wall down mastoidectomy (even when the mastoid volume is large) with exenteration of all visible air cell tracts (“subtotal petrosectomy”). Occasionally a translabyrinthine route will be needed to access the petrous apex, or a middle fossa approach to reach the supralabyrinthine tract.32,33 Figure 6.18 shows such a case. Preoperative facial nerve paralysis, an exceptionally rare occurrence, may require primary nerve repair with grafting.33–35 CSF leakage will require primary dural repair. The best strategy to avoid complications such as these is to create a widely exteriorized cavity with the matrix left intact. Giant cholesteatomas can develop at any age. In older patients, these may be neglected cholesteatomas that grew to a large size over many years. In younger patients, they may be biologically aggressive lesions that readily erode soft bone. Figure 6.19A shows a giant cholesteatoma in an elderly woman. This probably grew insidiously for many years until it reached a large size and was first diagnosed when it became infected. Because of her age, this lesion was initially managed conservatively but eventually required surgery because of persistent infection. The appearance of the ear at surgery is shown in Fig. 6.19B. Figure 6.20A,B shows a giant cholesteatoma in a 40-yearold man who presented with vertigo and purulent otorrhea. He had surprisingly good hearing preoperatively. At surgery, he had extensive disease with profuse green pus, a lateral semicircular canal fistula, and exposed facial nerve. Postoperatively he developed a Pseudomonas infection of the mastoid cavity, with suppurative labyrinthitis. He now has a large but stable, dry cavity without evidence of recurrence 3 years postoperatively. Cholesteatosis is a descriptive term for cholesteatoma that insinuates itself into all the recesses of the temporal bone. It is an aggressive process that occurs in well-developed pneumatized mastoid bones and fortunately is rare. The treatment demands a thorough surgical operation that opens all the involved spaces. This can be a long and arduous procedure. The concept is to remove all the cellular spaces of the mastoid until all that is left is the labyrinth, facial nerve, and tegmen and posterior fossa dural bony plates. This is the procedure that Fisch termed “subtotal petrosectomy.” If the disease extends to the petrous apex, it may be impossible to remove entirely without performing a labyrinthectomy as well. Figure 6.21A,B shows cholesteatoma pushing through the subarcuate cell tract, between the crura of the superior semicircular canal. CSF leakage is an infrequent occurrence in cholesteatoma surgery. It can occur iatrogenically if the dura is accidentally lacerated, or spontaneously, from a brain herniation (meningoencephalocele) that erodes through its dural covering. Iatrogenic CSF leaks can occur when drilling near the tegmen, usually when using a cutting burr. The tegmen has an undulating contour, and it is possible to perforate it when skeletonizing its bony surface. The underlying dura is vascular and has a pink appearance when seen through a thin layer of bone. Once exposed, the dural surface will often bleed; the small epidural vessels are easily controlled with bipolar cautery. This epidural bleeding is an important warning to the surgeon because dura will usually withstand one brush with the burr, but not a second one. Spontaneous tegmen defects are common, occurring in ∼20% of temporal bone cadavers, and usually have no serious consequence unless the dura is injured. Cholesteatoma matrix can adhere to bare dura and can be difficult to remove safely (Fig. 6.22A). Good technique requires that the margins of the bony tegmen defect first be defined visually. Then the disease can be elevated away from the dura with a blunt dissector using a peeling motion (Fig. 6.22B). Blind dissection along the tegmen, on the other hand, may yield an unhappy outcome. Encephaloceles and meningoceles are herniations of dura with or without brain tissue. The dural covering is often very thin and therefore more vulnerable than a simple area of dural exposure. Small, sessile meningoencephaloceles can be left alone if they are not involved with disease. Larger, broad-based meningoencephaloceles can be reduced and the tegmen defect repaired with cartilage, banked bone, split calvarial bone, hydroxyapatite discs, or synthetic commercially sold dura substitutes. Larger, pedunculated encephaloceles should be excised. The stalk can be fulgurated with bipolar cautery, and the lesion amputated. A CSF leak will often ensue and will need to be controlled with a multilayer repair, as described forthwith. Spontaneous CSF leaks of the temporal bone are usually associated with intracranial hypertension, arachnoid granulations,36 or previously undetected meningoencephaloceles with dural erosion (in children, spontaneous leaks are often associated with congenital anomalies of the inner ear). They can present with otorhinorrhea or be first discovered following myringotomy tube placement or an episode of meningitis. CSF leaks should always be repaired. The presence of CSF leakage and the site of the leak may be difficult to verify preoperatively. When a leak is suspected, a fluid sample should be collected if possible. Testing for glucose (two thirds of the serum value) or chloride (higher than serum) content is suggestive but not definitive. Beta-2-transferrin assay is the most specific confirmatory test for CSF. It takes but a few drops of fluid to run the assay. It takes ∼3 hours to complete, but this laboratory test is not routinely available at many laboratories. Shipping the aliquot of fluid to a designated laboratory requires a turnaround time of at least a few days. Beta-trace protein, a newer assay, may be just as sensitive, easier, and cheaper to run. Once the presence of CSF is established, imaging studies are used to try to localize the site of the leak.37 The single most useful study is a high-resolution CT scan. It may demonstrate the area of bony erosion of the tegmen or, less commonly, the posterior fossa dural plate; however, the bony defect may not correspond to the underlying dural defect that gave rise to the leak. Multiple tegmen defects and bilateral tegmen thinning are not uncommonly found and may introduce additional uncertainties. Magnetic resonance imaging (MRI) scanning in the coronal or sagittal plane will demonstrate an encephalocele but possibly not a meningocele. Radionuclide cisternography can definitively establish the presence of an active leak by showing pooling of intrathecally injected material in the mastoid. This imaging technique has low resolution and will not clearly demonstrate the location of the dural defect. The sensitivity of radionuclide testing can be enhanced by placing cottonoid pledgets in the nasal cavity and ear canal (if there is otorrhea) to look for radioactivity with a gamma counter; a positive test is diagnostic for CSF leakage but does not localize the site of the leak. CT cisternography with intrathecal contrast is the most precise method of localizing a leak, but it depends on a high rate of flow; slow or intermittent leaks will likely yield a negative study. Ultimately, once the diagnosis of CSF leakage is made, surgical repair will be needed. At surgery, the site and size of the leak can be accurately determined, and a meningoencephalocele if present will usually be readily apparent. The use of intrathecal dyes or fluorescein is rarely necessary to visualize an occult leak in the mastoid. CSF leaks can be repaired from “above” (intracranially, using a middle cranial fossa approach), or from “below” (transmastoid).38,39 The transmastoid approach is preferable for leaks that coexist with chronic middle ear disease when the defect is within the mastoid tegmen. Defects that are more anterior in the epitympanum, especially at or beyond the head of the malleus, are difficult to access without disarticulating the ossicular chain. A middle fossa approach may be necessary. The transcranial approach may also be more effective for controlling leaks that involve a larger area of dura, that involve multiple defects, or that recur after an unsuccessful transmastoid repair.40 The transcranial approach is performed through a middle cranial fossa (transtemporal) craniotomy. An extradural repair can be done by elevating the dura from the bony floor of the middle fossa and sliding a layer of fascia against the dural defect, then cartilage or split calvarial bone against the bony defect. An intradural repair is the most secure method of repair and may be needed if the dura is attenuated. A dural incision is created along the lateral surface of the temporal lobe, then the brain is elevated and fascia, synthetic dura (Duragen Integra LifeSciences, Plainsboro, NJ), acellular dermis (AlloDerm, LifeCell Corp., Branchburg, NJ), or cadaveric pericardium placed between brain and dura. As the brain reexpands, the graft will be held firmly in place. The transmastoid approach is performed by creating a canal wall up mastoidectomy and transmastoid atticotomy, exposing and skeletonizing the entire length of the tegmen (Fig. 6.23A). Once the leak is visualized, the surrounding bone is thinned with a diamond burr to maximize exposure. The tegmen can have an undulating contour so it should be skeletonized to a constant thickness. The healthy dura is carefully separated from the surrounding bone using a blunt elevator. If an encephalocele is present, it is either reduced or excised, depending on its size (Fig. 6.23B). A multilayer repair is undertaken by placing tissue against the dural defect (fascia, perichondrium, or AlloDerm can be used), then using stiffer material (cartilage, cortical bone, or banked bone) to repair the tegmen defect (Fig. 6.23C). The graft materials can be held in place by sandwiching them in the epidural space. As the intracranial contents expand they prevent the repair from migrating. Additional security can be gained by using fibrin glue over the repair and using an abdominal fat graft to fill the mastoid cavity. When a CSF leak occurs in a canal wall down cavity, the mastoid cavity cannot be isolated from the ear canal as it can when the canal wall up is maintained. In this situation, the ear canal will usually have to be oversewn and the mastoid cavity obliterated with fat. The eustachian tube should also be sealed by packing muscle into the protympanum. If the area of leak is small, however, the area can sometimes be effectively sealed with hydroxyapatite bone cement (Bone-Source, Stryker, Kalamazoo, MI; or Norian, Synthes, West Chester, PA). The bone cement should be covered with soft tissue, such as a skin flap or Palva flap, because it will become desiccated and behave as a foreign body if left exposed to the outside. This material should not be used in an infected cavity. A case example is provided. A 45-year-old man with a history of bilateral cholesteatomas had undergone bilateral, staged, canal wall down mastoidectomies. The left ear, the better-hearing ear, developed decreasing hearing, and a soft tissue mass was found partially covering the eardrum and emerging from posterosuperiorly in the mastoid cavity. The mass was soft to palpation and did not contain any squamous elements. CT scan revealed a small breach in the tegmen mastoideum. The patient was brought to the operating room for surgical treatment with a preoperative differential diagnosis of encephalocele, residual cholesteatoma, or inflammatory cyst. At surgery, a large encephalocele was confirmed. The lesion was dissected from its mucosal attachments, and the neck was isolated (Fig. 6.24A). The neck was fulgurated with bipolar electrocautery, and the mass was excised. A small CSF leak developed intraoperatively. The bony tegmen was thinned with a diamond drill. The dural edges were bluntly elevated from the surrounding bone. A multilayer repair was performed using a temporalis fascia graft in the epidural space, and bone cement (Norian, Stryker) to repair the tegmen (Fig. 6.24B). A conchomeatal skin flap was developed to cover the bone cement. The CSF leak did not recur, but the skin flap retracted, and the bone cement became exposed and desiccated, with inflamed granulation tissue around its edges. Nine months later, with the inflammation controlled and the cavity perfectly dry, the meatoplasty was revised, drilling the bone cement down to a thin eggshell, and turning a thicker Palvatype (fibromuscular) flap over it. The cavity has remained dry with no evidence of recurrent cholesteatoma. The patient is able to wear a hearing aid effectively. In certain instances, the leak will not be adequately controlled via a transmastoid approach, and a combined intra-and extracranial route will be needed. Some have advocated performing a “minicraniotomy,” performing a mastoidectomy initially, then creating a narrow bony aperture through the squamosa through which the dura can be elevated off the bony floor of the middle fossa and an extradural repair performed. Cholesteatoma can be an aggressive disease that finds its way into places within the middle ear and mastoid that can be difficult to dissect and remove. The oval window, the sinus tympani, the exposed facial nerve, the fenestrated lateral semicircular canal, the petrous apex, and exposed or dehiscent dura are all dangerous places that can become involved with disease. The need for complete disease removal may create situations that can jeopardize inner ear and facial nerve function. These situations demand clear judgment, honed surgical skills, and experience to achieve successful surgical outcomes. 1. Jackler RK. The surgical anatomy of cholesteatoma. Otolaryngol Clin North Am 1989;22 (5):883–896 2. Savi 3. HÜttenbrink KB. Manipulating the mobile stapes during tympanoplasty: the risk of stapedial luxation. Laryngoscope 1993;103 (6):668–672 4. Jahrsdoerfer RA, Johns ME, Cantrell RW. Labyrinthine trauma during ear surgery. Laryngoscope 1978;88 (10):1589–1595 5. Smyth GD, Gormley PK. Preservation of cochlear function in the surgery of cholesteatomatous labyrinthine fistulas and oval window tympanosclerosis. Otolaryngol Head Neck Surg 1987;96 (2):111–118 6. Sudhoff H, Tos M. Pathogenesis of sinus cholesteatoma. Eur Arch Otorhinolaryngol 2007;264 (10):1137–1143 7. Holt JJ. Posterior sinus of the middle ear. Ann Otol Rhinol Laryngol 2007;116 (6):457–461 8. Marchioni D, Alicandri-Ciufelli M, Grammatica A, Mattioli F, Presutti L. Pyramidal eminence and subpyramidal space: an endoscopic anatomical study. Laryngoscope 2010;120 (3):557–564 9. Lau T, Tos M. Treatment of sinus cholesteatoma: long-term results and recurrence rate. Arch Otolaryngol Head Neck Surg 1988;114 (12):1428–1434 10. Marchioni D, Mattioli F, Alicandri-Ciufelli M, Presutti L. Transcanal endoscopic approach to the sinus tympani: a clinical report. Otol Neurotol 2009;30 (6):758–765 11. Pulec JL. Sinus tympani: retrofacial approach for the removal of cholesteatomas. Ear Nose Throat J 1996;75 (2):77, 81–83, 86–88 12. Leonetti JP, Buckingham RA, Marzo SJ. Retraction cholesteatoma of the sinus tympani. Am J Otol 1996;17 (6):823–826 13. Leonetti JP, Marzo SJ, Beauchamp MM, Jellish WS. Long-term results with operated sinus tympani retraction cholesteatoma. Otolaryngol Head Neck Surg 2006;135 (1):152–154 14. Spielmann P, Mills RP. Surgical management of retraction pockets of the pars tensa with cartilage and perichondrial grafts. J Laryngol Otol 2006;120 (9):725–729 15. Paparella MM, Jung TT. Experience with tympanoplasty for atelectatic ears. Laryngoscope 1981;91 (9 Pt 1):1472–1477 16. Milewski C. Composite graft tympanoplasty in the treatment of ears with advanced middle ear pathology. Laryngoscope 1993;103 (12):1352–1356 17. Avraham S, Luntz M, Sadé J. The influence of ventilating tubes on the surgical treatment of atelectatic ears. Eur Arch Otorhinolaryngol 1991;248 (5):259–261 18. Elsheikh MN, Elsherief HS, Elsherief SG. Cartilage tympanoplasty for management of tympanic membrane atelectasis: is ventilatory tube necessary? Otol Neurotol 2006;27 (6):859–864 19. Bayazit YA, Ozer E, Kanlikama M. Gross dehiscence of the bone covering the facial nerve in the light of otological surgery. J Laryngol Otol 2002;116 (10):800–803 20. Moody MW, Lambert PR. Incidence of dehiscence of the facial nerve in 416 cases of cholesteatoma. Otol Neurotol 2007;28 (3):400–404 21. Lin JC, Ho KY, Kuo WR, Wang LF, Chai CY, Tsai SM. Incidence of dehiscence of the facial nerve at surgery for middle ear cholesteatoma. Otolaryngol Head Neck Surg 2004;131 (4):452–456 22. Selesnick SH, Lynn-Macrae AG. The incidence of facial nerve dehiscence at surgery for cholesteatoma. Otol Neurotol 2001;22 (2):129–132 23. Parisier SC, Edelstein DR, Han JC, Weiss MH. Management of labyrinthine fistulas caused by cholesteatoma. Otolaryngol Head Neck Surg 1991;104 (1):110–115 24. Copeland BJ, Buchman CA. Management of labyrinthine fistulae in chronic ear surgery. Am J Otolaryngol 2003;24 (1):51–60 25. Vartiainen E. What is the best method of treatment for labyrinthine fistulae caused by cholesteatoma? Clin Otolaryngol Allied Sci 1992;17 (3):258–260 26. Ostri B, Bak-Pedersen K. Surgical management of labyrinthine fistulae in chronic otitis media with cholesteatoma by a one-stage closed technique. ORL J Otorhinolaryngol Relat Spec 1989;51 (5): 295–299 27. Smouha EE, Namdar I, Michaelides EM. Partial labyrinthectomy with hearing preservation: an experimental study in guinea pigs. Otolaryngol Head Neck Surg 1996;114 (6):777–784 28. Parnes LS, McClure JA. Effect on brainstem auditory evoked responses of posterior semicircular canal occlusion in guinea pigs. J Otolaryngol 1985;14 (3):145–150 29. Vanden Abeele D, Offeciers FE. Management of labyrinthine fistulas in cholesteatoma. Acta Otorhinolaryngol Belg 1993;47 (3):311–321 30. Kobayashi T, Shiga N, Hozawa K, Hashimoto S, Takasaka T. Effect on cochlear potentials of lateral semicircular canal destruction. Arch Otolaryngol Head Neck Surg 1991;117 (11):1292–1295 31. Hakuba N, Hato N, Shinomori Y, Sato H, Gyo K. Labyrinthine fistula as a late complication of middle ear surgery using the canal wall down technique. Otol Neurotol 2002;23 (6):832–835 32. Grayeli AB, Mosnier I, El Garem H, Bouccara D, Sterkers O. Extensive intratemporal cholesteatoma: surgical strategy. Am J Otol 2000;21 (6):774–781 33. Moffat D, Jones S, Smith W. Petrous temporal bone cholesteatoma: a new classification and long-term surgical outcomes. Skull Base 2008;18 (2):107–115 34. Bartels LJ. Facial nerve and medially invasive petrous bone cholesteatomas. Ann Otol Rhinol Laryngol 1991;100 (4 Pt 1):308–316 35. Magliulo G, Terranova G, Sepe C, Cordeschi S, Cristofar P. Petrous bone cholesteatoma and facial paralysis. Clin Otolaryngol Allied Sci 1998;23 (3):253–258 36. Gacek RR, Gacek MR, Tart R. Adult spontaneous cerebrospinal fluid otorrhea: diagnosis and management. Am J Otol 1999;20 (6):770–776 37. Brown NE, Grundfast KM, Jabre A, Megerian CA, O’Malley BW Jr, Rosenberg SI. Diagnosis and management of spontaneous cerebrospinal fluid-middle ear effusion and otorrhea. Laryngoscope 2004;114 (5):800–805 38. Sanna M, Fois P, Russo A, Falcioni M. Management of meningoencephalic herniation of the temporal bone: personal experience and literature review. Laryngoscope 2009;119 (8):1579–1585 39. Wootten CT, Kaylie DM, Warren FM, Jackson CG. Management of brain herniation and cerebrospinal fluid leak in revision chronic ear surgery. Laryngoscope 2005;115 (7):1256–1261 40. Pelosi S, Bederson JB, Smouha EE. Cerebrospinal fluid leaks of temporal bone origin: selection of surgical approach. Skull Base Surg 2010;20:253–259
♦ Oval Window Disease
♦ Sinus Tympani Disease
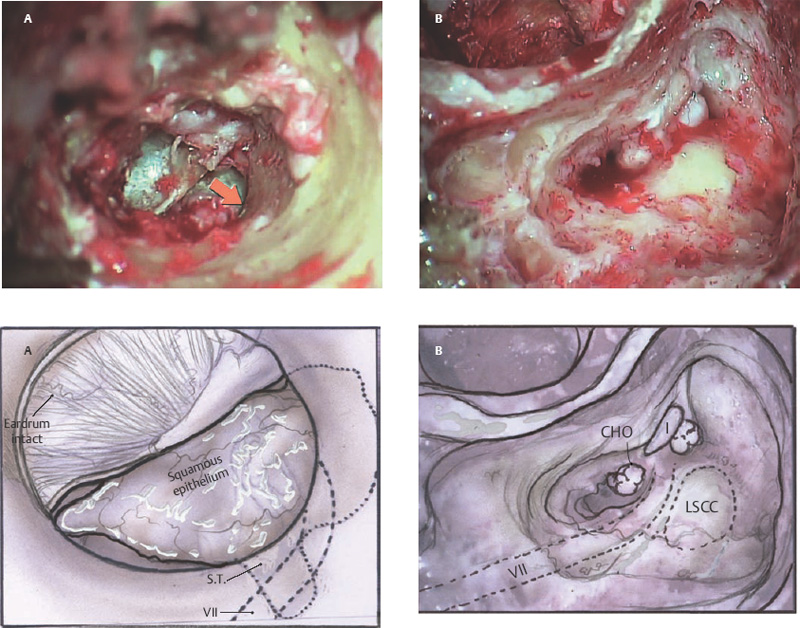
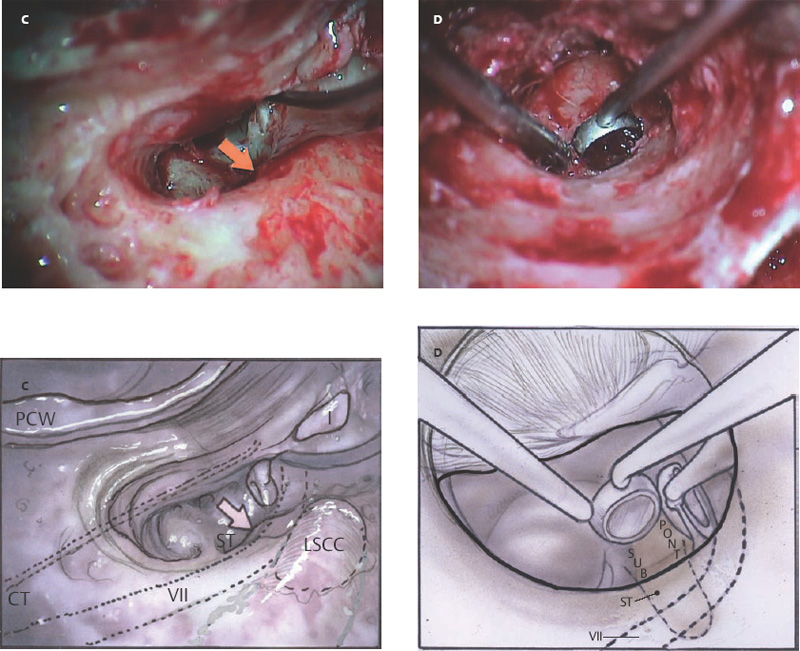
Tympanic Membrane Cholesteatoma
♦ Collapsed Middle Ear Space
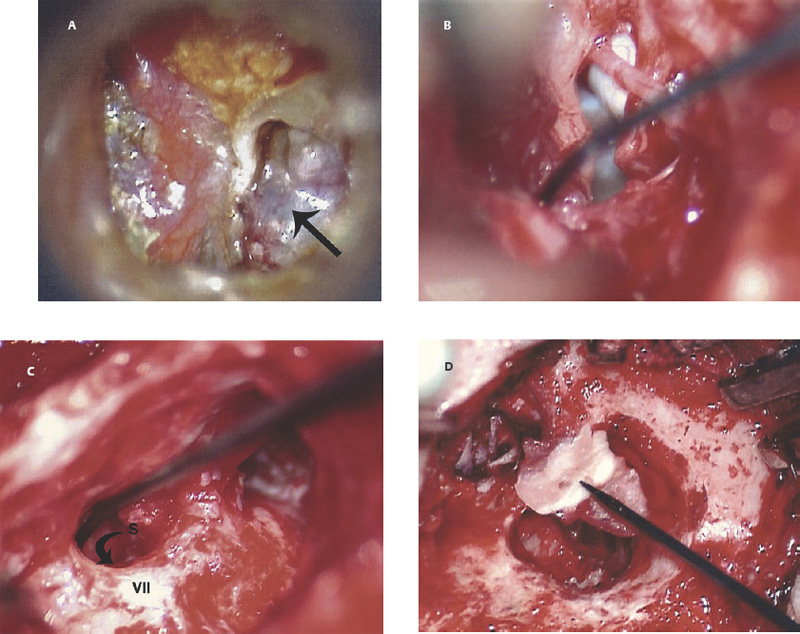
♦ Dehiscent Facial Nerve Canal
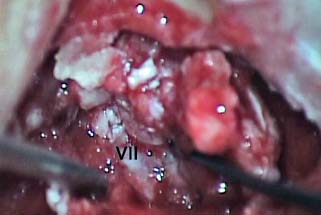
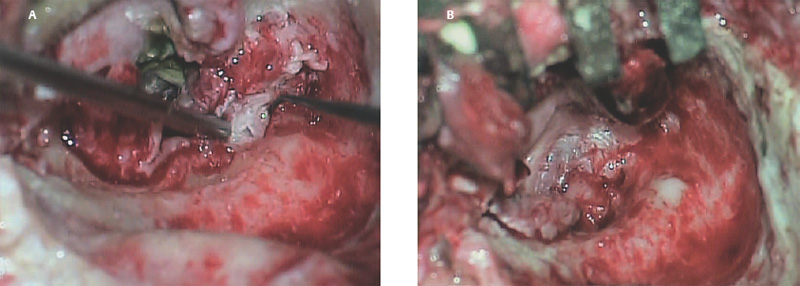
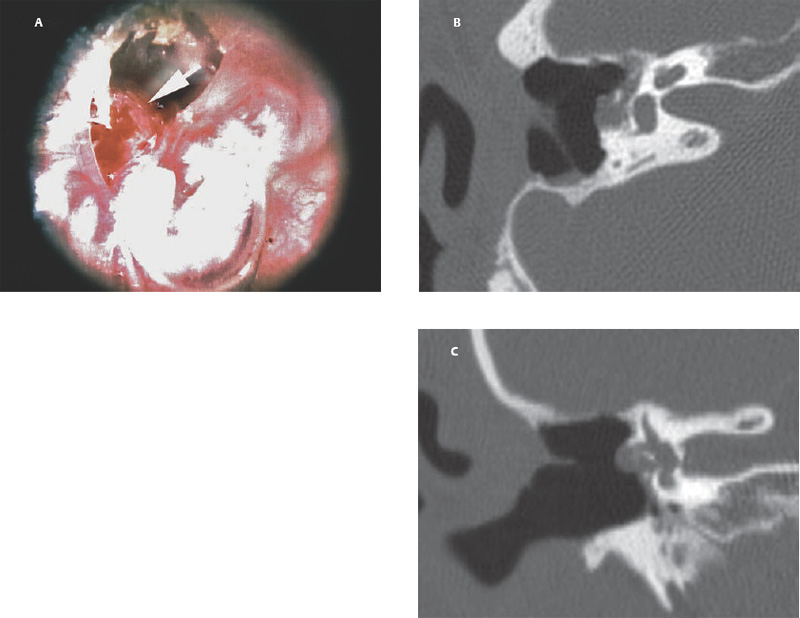
♦ Lateral Semicircular Canal Fistula
♦ Cholesteatosis/Giant Cholesteatoma
♦ Cerebrospinal Fluid Leak/Encephalocele
♦ Conclusion
References
 D, Djeri
D, Djeri D. Cholesteatoma in the oval window niche. Eur Arch Otorhinolaryngol 1990;247 (2):81–83
D. Cholesteatoma in the oval window niche. Eur Arch Otorhinolaryngol 1990;247 (2):81–83
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


