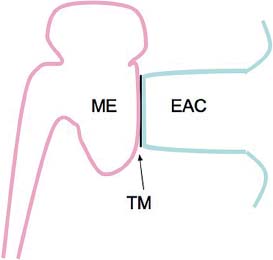
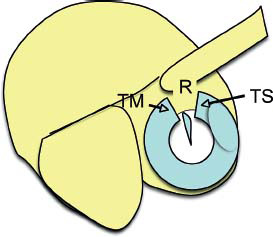
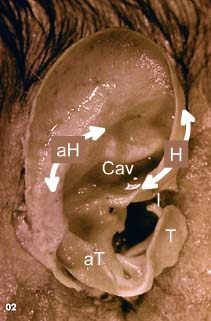
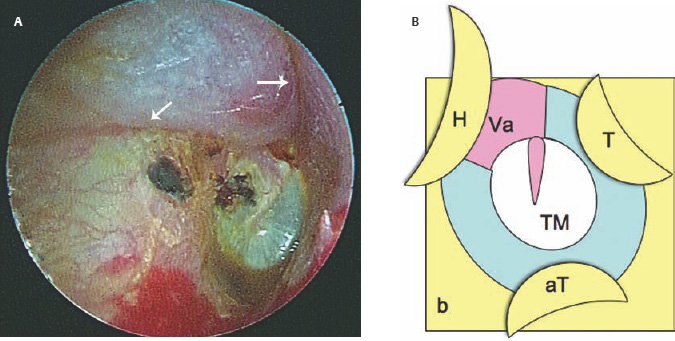
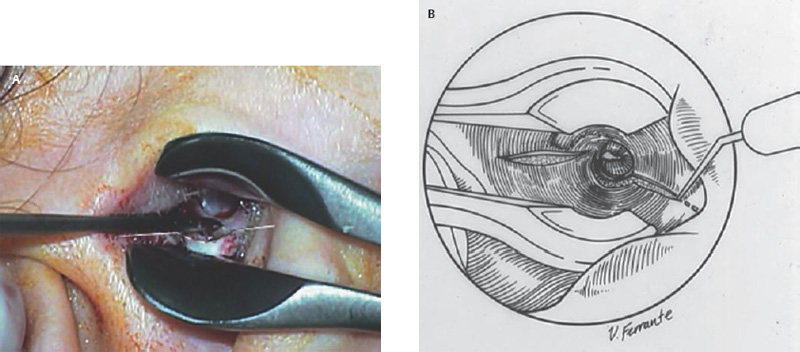
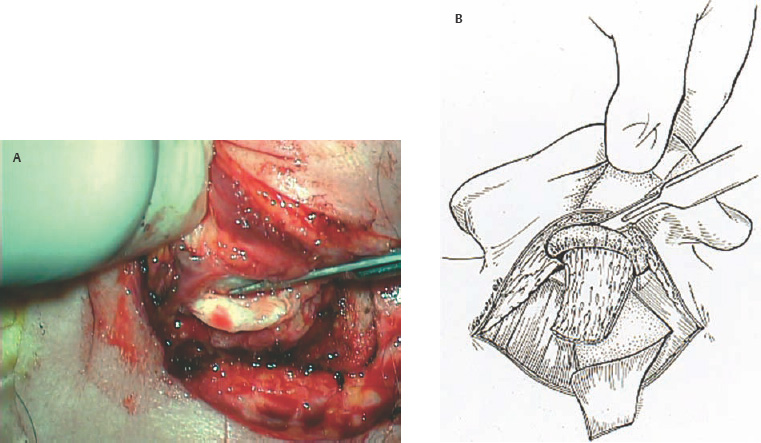
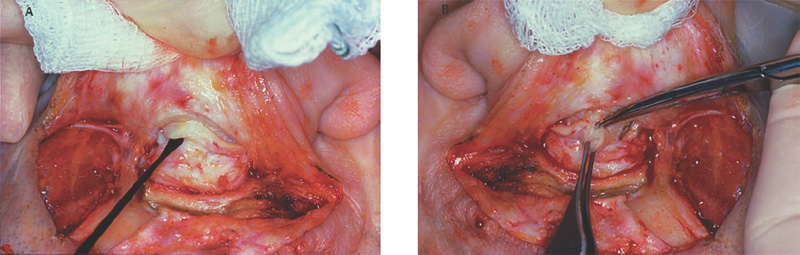
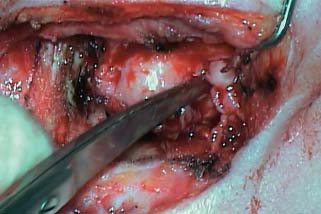
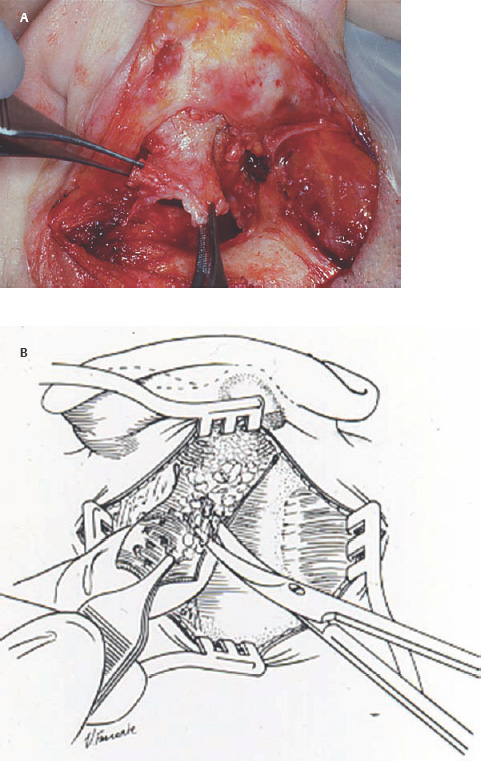
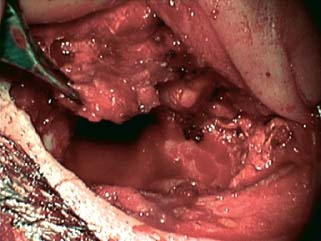
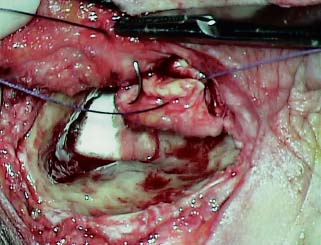
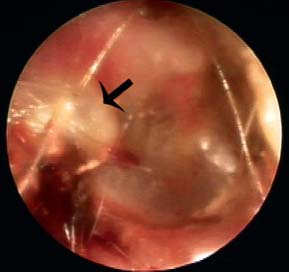
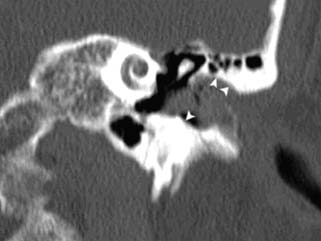
5 The Meatus and Cavity Management The external auditory meatus is often not given its due importance in discussions of ear disease and ear surgery. The meatus is the point of physical entry to the ear and allows the doctor a visual portal for diagnosis of cholesteatoma and, for that matter, every form of ear pathology. Obstruction of the meatus by cerumen, disease, or foreign material causes hearing loss and discomfort, and these are often the first symptoms that alert the patient to a problem. Surgical management of the meatus has great importance in controlling the functional outcome after otologic surgery because the meatus allows for proper surveillance in the postoperative period, and for detecting and managing recurrent disease. Primary cholesteatoma of the external canal and a related condition, keratosis obturans, also merit special discussion. The external auditory canal (EAC) is unique in that it is lined by keratinizing squamous epithelium, which does not penetrate the skin surface anywhere else in the body. This fact is what makes cholesteatoma possible. Embryologically, the EAC derives from the first branchial cleft, an invagination from the external surface of the face, which meets the derivative of the first pharyngeal pouch, the middle ear cleft, at the plane of the tympanic membrane (the tympanic membrane is formed by an outer ectodermal layer, a middle fibrous mesodermal layer, and an inner mucosal endodermal layer, as reviewed in Chapter 2) (Fig. 5.1). The anatomy of the external meatus is not complex. The EAC is formed of bone in its medial two thirds and cartilage in its lateral one third. The bony portion is formed mainly by the tympanic bone. The tympanic ring is horseshoe shaped and is deficient superiorly in the area called the notch of Rivinus. The anterosuperior end of the tympanic bone forms the tympanosquamous suture, and the posterosuperior end forms the tympanomastoid suture (Fig. 5.2). The cartilaginous ear canal forms the meatus itself. The bony–cartilaginous junction is the narrowest part of the EAC and the point at which cerumen often becomes trapped. The cartilage that forms the external meatus is contiguous with the auricular cartilage. Anterosuperiorly, the cartilage is deficient at the incisura, the area between the root of the helix and the tragus. Inferiorly the cartilage can be quite thick. The tragal–antitragal notch is at the 6 o’clock position of the meatus (Fig. 5.3). The skin that covers the cartilaginous ear canal is relatively thick and contains specialized apocrine glands that produce cerumen. The skin that covers the tympanic bone is very thin, not hair bearing or glandular, and very sensitive to pain or pressure. The skin that covers the notch of Rivinus superiorly, between the two suture lines, is looser, and is referred to as the vascular strip because it has its own blood supply (Fig. 5.4A,B). The tight ear canal skin is contiguous with the epithelial lining of the pars tensa of the tympanic membrane. The vascular strip skin is contiguous with the pars flaccida and the skin overlying the manubrium up to the umbo. A hydroplane injection of local anesthetic, often used to effect hemostasis at the start of ear surgery, will not cross the suture lines; therefore at least two injections are needed, one in the vascular strip and one in the inferior canal skin (injecting the four quadrants of the ear canal, as is commonly done, is superfluous). Fig. 5.2 Schematic drawing of a right temporal bone, showing the tympanic bone (light blue) forming the bony external auditory canal. The bone is horseshoe shaped, with a deficiency superiorly, the notch of Rivinus (R), and communicating anteriorly at the tympanosquamous (TS) and posteriorly at the tympanomastoid (TM) sutures. The meatoplasty is an essential step in canal wall down mastoidectomy. It is important for postoperative cavity maintenance—an adequate meatus ensures that all areas of the mastoid cavity can be inspected, and that keratin cysts or areas of inflammation can be dealt with in the office setting. The meatoplasty also ensures that the mastoid bowl is aerated, important to prevent the formation of recurrent cholesteatoma. On the other hand, an inadequate meatus may allow squamous debris to reaccumulate in the mastoid bowl, which may lead to cholesteatoma recurrence. A narrow meatus may make inspection and cleaning difficult and uncomfortable for the patient, which may discourage regular follow-up. Also, recurrent or persistent drainage may result from inadequate aeration, frequent water contamination, and insufficient hygiene. Fig. 5.5 (A) The meatal incisions are made through skin and cartilage, at 6 and 12 o’clock, as shown in the photo and (B) drawing of the right ear. The 12 o’clock incision is made in the incisura, the space between the root of the helix and the tragus. The 6 o’clock incision will be made between the tragus and antitragus. The goal of the meatoplasty is to create a meatus that is widely patent, but not disfiguring. The overall goal of the surgery is to create a shallow, well-pneumatized cavity that is widely exteriorized to the outside. Exteriorization of the cavity is what prevents the normal skin lining from forming another cholesteatoma. The meatoplasty is often left until the end of the surgical procedure, and if the operation is lengthy or difficult, the surgeon may try to complete this segment of the procedure hastily. However, the meatoplasty affects the surgical outcome as much as any other portion of the procedure and may be a key step in preventing cholesteatoma recurrence; thus it deserves the proper attention. Any of the steps of the meatoplasty can be performed at the start of the operation, and this may be more convenient to gain surgical exposure during the procedure. There are many individual variations on the technique of meatoplasty, but some common elements exist.1 Meatal incisions should be made through skin and cartilage and must be adequately long so as to create a conchomeatal (laterally based skin) flap that can be oriented posteriorly and into the mastoid cavity. The conchal cartilage at the meatal rim must be either scored or excised, to make the meatus larger. Stay sutures should be placed to ensure that the meatus remains patent. These concepts were developed by Lempert,2 in an era when endaural approaches to the mastoid were popular. The technique shown here incorporates those elements. The first step is to create long meatal incisions, extending laterally through skin and cartilage. A Lempert speculum can be used, with a No. 15 knife blade (or a disposable Beaver-type blade) to be sure that the full thickness is traversed (Fig. 5.5A,B). As this is done, the meatus can be splayed open with the leaves of the speculum. These incisions should be located superiorly at 12 o’clock, in the incisura, the skin between the root of the helix and the tragus, where there is a natural absence of cartilage; and inferiorly at 6 o’clock, between the tragus and the antitragus (these are analogous to the No. 1 and No. 2 endaural incisions described by Lempert; extending the superior incision into the preauricular sulcus can potentially allow access to the entire mastoid). The next step is to break the spring of the conchal cartilage.3 The cartilage that forms the meatal rim is dense—removing this allows the meatal opening to relax backward. This step is best accomplished with the surgeon’s nondominant index finger in the meatal opening to serve as an anchor as well as to gauge the size of the opening. Working through the postauricular wound, the subcutaneous tissue is sharply dissected away from the conchal cartilage, staying external to the plane of the perichondrium. Once the cartilage is exposed, a crescent-shaped incision is made through the cartilage at the meatal rim (Fig. 5.6A,B), and this cartilage is dissected from the subcutaneous tissues and excised (Fig. 5.7A,B). The index finger should be easily accommodated at this point; otherwise additional cartilage can be removed with a knife or sharp scissors. The cartilage that forms the inferior meatus is often the limiting factor and should be split or excised4 (Fig. 5.8). The cartilage that forms the root of the helix is also quite dense, and scoring this will often allow the meatus to be effectively enlarged. A useful measure when first learning this technique is to pass two sterile needles (25 gauge) through the conchal skin, to see where the corresponding cartilage needs to be removed. Fig. 5.6 (A,B) A crescent-shaped piece of cartilage is excised from posteriorly. Fig. 5.8 The inferior canal cartilage is dissected. Fig. 5.9 (A,B) The meatal skin is thinned, to create a flap that will re-line the base of the mastoid cavity. In this example, the skin flap is thinned down to the subdermal tissue plane. (Courtesy of Dr. Simon Parisier.) The next step is to thin the meatal skin. There are two methods that can be employed. The first excises all the fibrous subcutaneous tissue and attached cartilage to form a thin conchomeatal skin flap that can be draped over the posterior wall and floor of the mastoid cavity (Fig. 5.9A,B). The second method, the “musculoperiosteal flap” described by Palva (Fig. 5.10), leaves the fibrous tissue pedicled to the distal end of the conchomeatal flap, to provide some bulk for cavity obliteration.5 The second method helps limit the size of the cavity but carries the risk of burying mucosal disease. Whichever method is selected, the conchomeatal skin flap should be tacked back with stay sutures to the conchal perichondrium to create a widely patent meatal opening (Fig. 5.11). The flap may be directed posteriorly and somewhat superiorly toward the sinodural angle, where the cavity is largest. Variations exist on how to make the meatal incisions. Sheehy created “vascular strip” incisions at the start of the case, pedicled superiorly, and following the tympanomastoid and tympanosquamous sutures. Others have described a bifid flap, where the conchomeatal skin is split longitudinally, with one limb directed upward and the other downward, in an effort to create a large, round meatus.6 Z-plasties and M-plasties have also been described.7,8 Most surgeons will use packing at the end of the procedure to stent the meatus open. Resorbable and nonresorbable materials have been used. Gelfoam (Pfizer, New York, NY) (absorbable gelatin sponge) can be placed in the medial end of the cavity to hold the tympanomeatal flap and tympanic membrane grafts in place. Gelfoam can usually be easily removed with suction or alligator forceps in the postoperative period, even in children, or it can be left in place to dissolve gradually. Bacitracin ointment can also be placed at the base of the cavity, although it will liquefy more quickly than Gelfoam. Merogel (Medtronic Corp., Mystic, CT) (derived from hyaluronic acid) has also been adapted for this purpose. Nonresorbable packing materials include thin silicone sheeting, Owen’s silk, or expandable sponges (eg, Merocel, Medtronic Corp.). A rosebud packing can be fashioned and filled with Gelfoam, antibiotic-impregnated gauze, or cottonoid discs. Our own preference is to use Gelfoam medially, followed by a thick cushion of bacitracin ointment, and to fold a strip of Xeroform gauze (Covidien, Mansfield, MA) into the meatus. The Xeroform layer is bacteriostatic and nonadherent and can usually be removed painlessly and without difficulty at the first postoperative visit. The remaining Gelfoam and bacitracin ointment can be gently suctioned or simply left to dissolve. The packing is removed at 7 to 10 days postoperative. The patient is started on antibiotic eardrops (eg, ofloxacin solution, 4 drops twice a day), and told to keep the ear dry. If granulation tissue forms in the zone between the skin edges, it can be lightly cauterized with topical silver nitrate. Polypoid granulation tissue can be debrided after cauterizing its base. By 4 to 6 weeks postoperative, a large, stable meatus usually results (Fig. 5.12). Fig. 5.12 Postoperative appearance of the meatus. (Courtesy of Dr. Simon Parisier.) External canal cholesteatoma is much less common than cholesteatoma of the middle ear and mastoid. This lesion typically presents with otorrhea and chronic, dull pain from local invasion into the bony EAC. Clinically, it has the same appearance as middle ear cholesteatoma, a pearlescent ball of skin, often with surrounding inflammation or granulation tissue (Fig. 5.13). External canal cholesteatoma always involves the inferior aspect of the ear canal, although it may grow to fill the entire canal.9 On computed tomographic (CT) imaging, bony erosion of the floor of the ear canal will usually be seen.10 Fig. 5.14 Computed tomographic scan, in coronal plane, showing keratosis obturans. The soft tissue lesion has filled and widened the bony canal (arrowheads), without causing bone necrosis. Primary and secondary forms have been identified. The primary form is idiopathic, although there is evidence that repeated minor trauma inflicted by the patient might be necessary for its formation. The secondary form is usually due to obstruction of the canal (eg, by a bony osteoma) or to focal injury of the EAC by prior surgery or radiation. Very occasionally, a middle ear cholesteatoma will grow exophytically into the EAC; this should not be mistaken for an EAC cholesteatoma and should be treated according to the principles covered elsewhere in this book. The treatment of external canal cholesteatoma is wide surgical excision of the lesion with a cuff of normal skin.9,11 The bone is then recontoured with a diamond burr, attempting to avoid opening the mastoid air cells. A split-thickness skin graft may be applied for soft tissue coverage if a significant area of exposed bone remains. Keratosis obturans is a similar but separate condition. Keratosis obturans is a plug of cerumen and keratin that occludes the entire ear canal, and that causes widening of the bony EAC. In contrast to EAC cholesteatoma, which begins in the inferior part of the bony EAC, keratosis involves the entire circumference of the ear canal, and it is frequently bilateral. It occurs in younger patients, and is associated with sinusitis and bronchiectasis. It presents with painful occlusion and hearing loss.12,13 CT will reveal a nonerosive soft tissue lesion with circumferential bony expansion of the EAC (Fig. 5.14). The absence of bone necrosis and skin ulceration are the most important features that distinguish keratosis obturans from EAC cholesteatoma.14 This condition is treated by complete removal of the abnormal plug by curettage, which may require local anesthetic injection (or general anesthesia in children or uncooperative adults), followed by recleaning every few months to prevent reaccumulation. Keratosis obturans will tend to recur if left unattended. 1. Paparella MM, Meyerhoff WL. “How I do it”—otology and neurology: a specific issue and its solution. Meatoplasty. Laryngoscope 1978;88 (2 Pt 1):357–359 2. Lempert J. Lempert endaural subcortical mastoidotympanectomy for the cure of chronic persistent suppurative otitis media. Arch Otolaryngol 1949;49 (1):20–35 3. Raut VV, Rutka JA. The Toronto meatoplasty: enhancing one’s results in canal wall down procedures. Laryngoscope 2002;112 (11):2093–2095 4. Eisenman DJ, Parisier SC. Meatoplasty: the cartilage of the floor of the ear canal. Laryngoscope 1999;109 (5):840–842 5. Saunders JE, Shoemaker DL, McElveen JT Jr. Reconstruction of the radical mastoid. Am J Otol 1992;13 (5):465–469 6. Donaldson JA, Duckert LG. “How I do it”—otology and neurotology: a specific issue and its solution. Meatoplasty. Laryngoscope 1981;91 (10):1757–1758 7. Fagan P, Ajal M. Z-meatoplasty of the external auditory canal. Laryngoscope 1998;108 (9):1421–1422 8. Mirck PG. The M-meatoplasty of the external auditory canal. Laryngoscope 1996;106 (3 Pt 1):367–369 9. Dubach P, Häusler R. External auditory canal cholesteatoma: reassessment of and amendments to its categorization, pathogenesis, and treatment in 34 patients. Otol Neurotol 2008;29 (7):941–948 10. Heilbrun ME, Salzman KL, Glastonbury CM, Harnsberger HR, Kennedy RJ, Shelton C. External auditory canal cholesteatoma: clinical and imaging spectrum. AJNR Am J Neuroradiol 2003;24 (4):751–756 11. Lin YS. Surgical results of external canal cholesteatoma. Acta Otolaryngol 2009;129 (6):615–623 12. Sismanis A, Huang CE, Abedi E, Williams GH. External ear canal cholesteatoma. Am J Otol 1986;7 (2):126–129 13. Piepergerdes MC, Kramer BM, Behnke EE. Keratosis obturans and external auditory canal cholesteatoma. Laryngoscope 1980;90 (3):383–391 14. Persaud RA, Hajioff D, Thevasagayam MS, Wareing MJ, Wright A. Keratosis obturans and external ear canal cholesteatoma: how and why we should distinguish between these conditions. Clin Otolaryngol Allied Sci 2004;29 (6):577–581
♦ Cholesteatoma
♦ Anatomy and Considerations
♦ Surgical Technique of Meatoplasty

♦ External Canal Cholesteatoma and Keratosis Obturans
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


