Fig. 10.1
Anatomy of the nasopharynx. The uppermost part of the pharynx
10.1.1 Benign Tumors (Nasopharyngeal Fibroma/Angiofibroma)
Nasopharyngeal fibroma or angiofibroma is a benign tumor, which is highly destructive causing pressure necrosis of the bone. It is composed of immature fibroblasts and blood vessels and is covered by intact mucous membrane unless traumatized.
It commonly affects young males. Usually the boy presents with progressive nasal obstruction, recurrent epistaxis, and nasal discharge. If left untreated, it may expand into the orbit and cause blindness. Physical examination reveals a firm mass in the nasopharynx (Fig. 10.2).
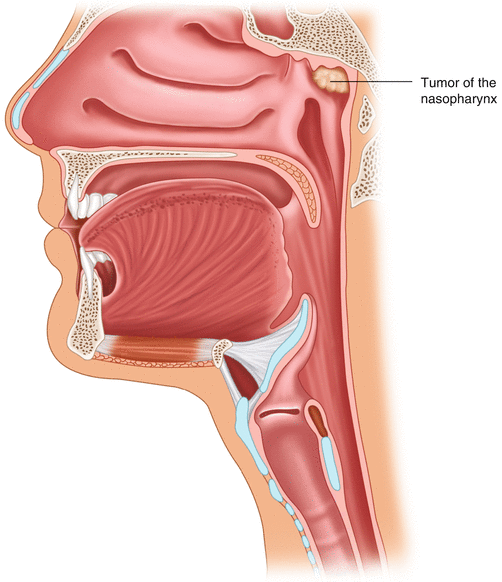

Fig. 10.2
Location of nasopharyngeal tumor
Transpalatal excision is the treatment of choice.
10.1.2 Malignant Tumors
The nasopharynx is called the “blind spot” (Fig. 10.2). Diagnosis is usually delayed for 8–18 months. It is a common site for occult primary tumors. Approximately 80–85 % of cancers are squamous cell carcinoma (SCC), and the remainder is primarily lymphoma, adenocarcinoma, and melanoma [1]. There are two varieties of SCC, keratinizing and nonkeratinizing of which lymphoepithelioma is an important form characterized by islands of lymphocytes scattered through the epithelioid tumor.
In the WHO classification, three histopathological types of nasopharyngeal carcinomas (NPC) are recognized [2]:
Type I: SCC with varying degrees of differentiation
Type II: Nonkeratinizing carcinoma
Type III: Undifferentiated carcinoma (often known as lymphoepithelioma) (Fig. 10.3).
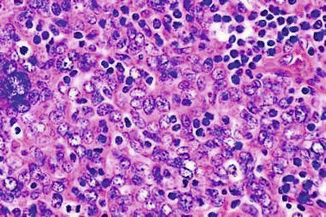
Fig. 10.3
Undifferentiated nasopharyngeal carcinoma (lymphoepithelioma), HE 600×450
Nasopharyngeal cancer is more common among the Chinese, and those patients have a high titer of Epstein-Barr virus antibodies in their serum [3]. In the early course of the disease, the only symptom may be a sense of needing to clear the throat. Later, unilateral nasal obstruction, epistaxis, and/or discharge, cervical adenopathy and cranial nerve palsy may be noted. Unilateral conductive impairment of hearing, with or without tinnitus, is also a common presenting symptom due to obstruction of the Eustachian tube by the primary tumor. The obstruction may lead to serious otitis media. The rich lymphatic network within the parapharyngeal space results in contralateral metastases and affection of retropharyngeal LNs near the jugular foramen as well as possible involvement of the last four cranial nerves [4].
The primary tumor extent should be evaluated by both CT scan and MRI. The MRI is more sensitive than CT for detection of the primary tumor, its direct soft tissue extent, regional nodal metastasis, and perineural extension; however, CT is a better tool for defining bone erosion. Positron emission tomography (PET) scanning in NPC may be useful in detecting both local failures after treatment and distant metastases; however, its definitive role remains to be defined.
Histological confirmation of the diagnosis is essential. The diagnostic sensitivity of biopsy under local anesthesia has been found to be comparable to that obtained by examination under general anesthesia. The biopsy is facilitated by direct visualization of the nasopharynx with a fiber-optic nasopharyngoscope. Patients with advanced stage NPC have been reported to have higher plasma EBV-DNA levels than those with early-stage disease [5]. Further studies demonstrated that EBV-DNA may be a valuable tool for monitoring of NPC patient response during radiotherapy and chemotherapy [6], as well as early detection of tumor recurrence [7].
The treatment of choice for NPC is radiotherapy (RT), except for small early lesions and tumors that recur after RT. With advances in technology, the modern RT for NPC should be that of three-dimensional conformal radiotherapy (3D-CRT) or intensity-modulated radiotherapy (IMRT) with inverse planning. Such techniques yield superior local control when compared to standard 2D methods [8]. The use of adjuvant chemotherapy cannot be recommended as a standard therapeutic approach in patients with locoregionally advanced disease [9–12].
In absence of significant skull-base erosion and intracranial extension or cranial nerve palsy, surgical resection of recurrent or persistent local tumor is the mainstay salvage treatment [13, 14]. There are several approaches to the nasopharyngectomy, namely, transcervical, transoral and transpalatal, posterolateral, trans-maxillary (maxillary swing) [14], and midface deglove [15]. There is no “ideal” surgical approach that suits all cases of local relapses and therefore it should be tailored to the individual patients depending on the disease extent. Cervical LN metastases are usually found in 70 % of patients when first diagnosed, and radical neck dissection (RND) is the treatment of choice in such cases.
The median survival for patients with distant metastases is around 9 months. Wide ranges of chemotherapeutic agents have been used in the treatment of patients with locally recurrent and metastatic NPC. New active agents include paclitaxel and gemcitabine. Cisplatin-containing regimens yielded with encouraging response rates of 50–90 % [16].
10.2 Tumors of the Oropharynx
The oropharynx is the middle part of the throat which includes the base of the tongue, the tonsils, the soft palate, and the walls of the pharynx. Oropharyngeal cancer (OPC) is rather uncommon. It can occur at any age, but is more likely to affect people over the age of 50 years, and is more common in men than in women. Oropharyngeal cancer can be divided into two types, the one related to human papillomavirus (HPV) infection (HPV-positive cancer) [17–20] and HPV-negative cancer, which is usually linked to alcohol or tobacco use [21]. There are several differences between HPV-positive and HPV-negative tumors in many aspects including histological appearance, differentiation, risk factors, and prognosis [22].
There are different histopathological types of OPC, but the most common is the squamous cell carcinoma (SCC) (Fig. 10.4). Other rare types include salivary gland cancer, lymphoma, small-cell cancer, and sarcoma.
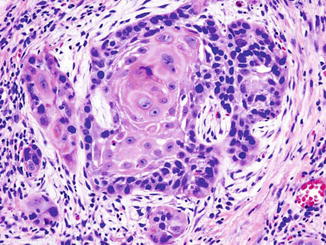

Fig. 10.4
Squamous cell carcinoma (SCC) of the oropharynx, HE 600×450
The factors that can increase the risk of developing OPC include smoking and chewing tobacco, heavy alcohol use, poor nutrition, HPV and EBV infections, Plummer-Vinson syndrome, asbestos exposure, and P53 mutation [23].
The most common symptoms of OPC include a persistent sore throat, dysphagia, unexplained weight loss, voice changes, earache, a lump in the back of the throat or mouth (Fig. 10.5), a lump in the neck, retrosternal dull pain, and cough.
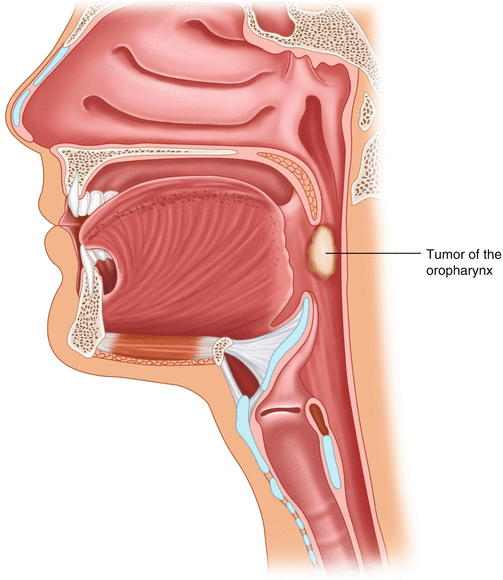

Fig. 10.5
Location of nasopharyngeal tumor
The primary tumor extent should be evaluated by both CT scan and MRI. Histological confirmation of the diagnosis is essential.
Treatment of OPC depends on the stage and grade of the tumor as well as the general condition of the patient. The usual treatments include surgery, radiotherapy (RT), chemotherapy, and targeted therapies.
In early OPC, both surgery and RT are equally effective. The latter is used if surgery is likely to seriously affect the patient’s speech and swallowing. Larger tumors are often treated with a combination of different treatments. Currently, intensity-modulated radiotherapy (IMRT) is used to precisely shape the RT beams to the area of cancer, thereby improving its efficacy and reducing its side effects [24]. Radiotherapy can be used as radical RT for cure or as adjuvant therapy after surgery to reduce the risk of recurrence. For locally advanced OPCs, RT may be given together with chemotherapy (chemoradiation) [25]. It can also be given to cervical LNs.
Surgical resection may also be used, although RT with or without chemotherapy is more commonly used. Surgery can also be used to relieve symptoms or to treat recurrence after RT. Cervical LNs are treated by neck dissection.
Chemotherapy may be given, before RT or surgery (neoadjuvant chemotherapy), at the same time as RT (chemoradiation), after RT or surgery (adjuvant chemotherapy) and for metastatic disease. Chemoradiation may be used instead of surgery in locally advanced disease. This can avoid the effects on speech and swallowing that surgery may cause. The side effects, particularly a sore skin and mouth, are worse when chemotherapy and RT are given together.
Targeted therapies for OPC include “cetuximab” infusion (the most commonly used) which interferes with attachment of epidermal growth factor (EGF) on their receptors in cancer cells, thereby stopping growth of the tumor and may also make it more sensitive to the effects of RT [26].
Several studies have found better prognosis for HPV-positive tumors and reported similar findings in patients treated with either primary surgical or nonsurgical therapies [27–30]. A meta-analysis confirmed that HPV-positive OPC cancer patients had a 28 % lower risk of death than their negative counterparts [31]. The HPV status was also associated with a better response to induction chemotherapy in two recent studies [32, 33].
10.3 Tumors of the Hypopharynx
The hypopharynx is the region between the oropharynx above and the esophageal inlet below. Hypopharyngeal cancers (HPCs) are often named for their location, including pyriform sinus, lateral pharyngeal wall, posterior pharyngeal wall, or post-cricoid pharynx. The pyriform sinus is involved in 65–85 % of cases, the posterior pharyngeal wall in 10–20 %, and the post-cricoid area in 5–15 % [34].
There is a male preponderance with a male-to-female ratio of about 3:1. The incidence of HPC rises in people above the age of 40 years [35]. Patients diagnosed with HPC are typically men aged 55–70 years with a history of tobacco use and/or alcohol ingestion. Asbestos may pose an independent risk for the development of HPC [36]. The role of human papillomavirus (HPV) in HPC is unclear, although it may play more of a role in cancers of the oropharynx and oral cavity. Nutritional (Plummer-Vinson syndrome, iron and vitamin C deficiencies) and metabolic deficiencies are implicated in rare instances.
Symptoms of HPC include dysphagia, chronic sore throat, and FB sensation in the throat or referred otalgia. A metastatic cervical LN is often the presenting symptom in approximately 50 % of cases. Other symptoms, which usually develop later, include weight loss, hemoptysis, laryngeal stridor, and hoarseness of voice due to direct extension of the tumor into the arytenoid cartilage or to the recurrent laryngeal nerve (RLN). Halitosis (fetid breath) may occur due to bacterial overgrowth in a fungating necrotic tumor. Nearly 70 % of patients with HPC have stage III disease at the time of presentation.
More than 95 % of HPCs are SCCs, less than 60 % are keratinizing, 33 % are nonkeratinizing, and all are usually poorly differentiated. Variants include basaloid SCC, superficial spreading cancer, sebaceous cancer, adenosquamous cancer, signet ring, and verrucous types. Uncommon histological types include adenocarcinoma, lymphoma, and sarcoma.
Flexible fiber-optic endoscopic examination is important for localization and staging of the primary tumor. Typically, HPC is advanced at presentation, and an obvious abnormality is usually present in either the pharynx (Fig. 10.6) or the neck. Typical findings of HPC include mucosal ulceration, pooling of saliva in the pyriform fossa, edema of the arytenoids, or fixation of the cricoarytenoid joint and/or true vocal cords. During the flexible laryngoscopy, the assessment of vocal cord is important for staging.
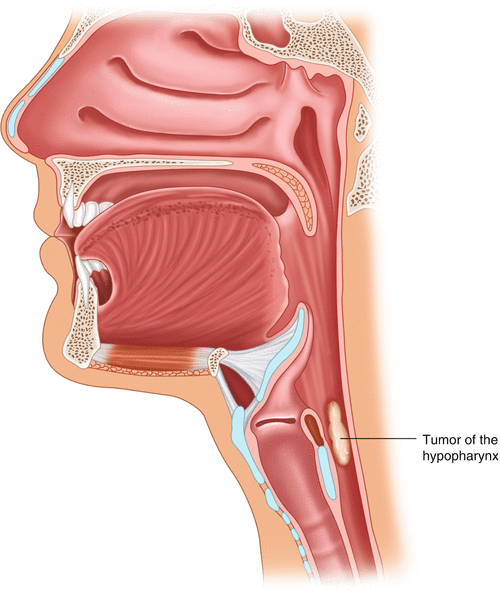

Fig. 10.6
Location of hypopharyngeal tumor
In general, 30 % of patients have local disease at the time of diagnosis, 60 % have local regional disease, and 10 % present with distant metastases. Tumors of the medial wall of the pyriform sinus usually spread to the aryepiglottic folds and may invade into the larynx by involving the paraglottic space, while tumors of the lateral wall and apex commonly invade the thyroid cartilage. Metastasis to the regional LNs is common and should be assessed for size, location, and mobility. Differentials include cat scratch disease, Hodgkin disease, NHL, pharyngitis, and extramedullary plasmacytoma.
Imaging studies should not be considered as a replacement for a flexible fiber-optic examination. Chest X-ray films are important to check for lung metastases, synchronous lung cancer, and comorbid heart or lung disease. Barium swallow is not usually used unless the lesion is too large to introduce a scope. Its findings can help determine the inferior border of the lesion and involvement of the esophageal inlet. In order to visualize the primary tumor and identify regional LNs prior to definitive treatment, contrast-enhanced CT scan and MRI are used; MRI (with gadolinium) is better than CT for delineating soft tissue extension, while CT scan (with bone windows) is superior in detecting bone invasion. If HPC is aggressive (T4, N2-N3, or poorly differentiated), chest CT scan or positron emission tomography (PET) scan is considered for the most sensitive detection of metastases. Integrated PET/CT is helpful in locating and localizing the occult primary tumor and regional disease, as well as in differentiating between malignant disease and posttreatment changes.
Examination under anesthesia (EUA) is critical for defining the anatomic extent of disease and for obtaining a biopsy, which is necessary to establish the diagnosis. Biopsies of all suspicious lesions are usually taken during a triple endoscopy (panendoscopy). Bronchoscopy and esophagoscopy are performed to rule out synchronous cancers. Multiple pharyngeal tumors can be found in nearly 15 % of cases, while synchronous lung or esophageal tumors can be found in approximately 5–10 %.
The goal of management of HPC is to achieve the highest locoregional control with the least functional injury, preserving respiratory function, deglutination, and phonation. Several new treatment options have been introduced recently. These include modern conservative surgical approaches that include robotic assistance and laser dissection, and new RT techniques such as IMRT for increased conformal irradiation, and the use of biologic agents such as the monoclonal antibody cetuximab (Erbitux), which specifically binds and prevents the activation of the epidermal growth factor receptor (EGFR) [37].
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


