Surface
Right adrenal gland
Left adrenal gland
Anterior surface
Superior: “bare area” of the liver
Inferior: peritoneum and first part of the duodenum (occasionally)
Medial: inferior vena cava (IVC)
Lateral: “bare area” of the right lobe of the liver
Superior: peritoneum and the stomach
Inferior: body of the pancreas
Posterior surface
Superior: diaphragm
Inferior: anteromedial aspect of the right kidney
Medial: left crus of the diaphragm
Lateral: medial aspect of the left kidney
13.1.1 Vascular Supply
The adrenal and the thyroid and parathyroid glands are the viscera with the greatest blood supply per gram of tissue. The arterial supply of the adrenal glands comes from three main sources:
The superior adrenal arteries, arising from the inferior phrenic arteries. One artery may be larger than the others, or all may be of similar size.
The middle adrenal artery arises from the aorta just proximal to the origin of the renal artery. It can be single, multiple, or absent.
One or more inferior adrenal arteries arise from the renal artery, an accessory renal artery, or a superior polar artery.
A total of 50–60 arteries penetrate the capsule over the entire surface as all of the abovementioned arteries branch freely before entering the adrenal gland [4]. The pattern of the arterial supply has been shown to vary between each individual [5–7]. In the majority of cases (61 % of individuals), the supply by the middle or inferior adrenal arteries may be lacking, while the superior adrenals are absent in only about 2 % of cases. In a small subset of the population (5 % of individuals), the arterial supply is derived entirely from one source, a singular vessel supplying the superior, middle, and inferior branches.
The adrenal venous drainage is much simpler with a single vein emerging at the hilum and draining the entire adrenal gland. The left vein passes downward over the anterior surface of the gland and is joined by the left inferior phrenic vein before entering the left renal vein. The right vein is typically very short and opens into the posterior side of the inferior vena cava. The right adrenal vein does not usually have any tributaries other than from the adrenal gland. Accessory veins occur in 5–10 % of patients; on the right, these vessels may drain into the right hepatic vein or the right renal vein; on the left, accessory veins may drain directly into the left renal vein [2].
13.1.2 Lymphatic Drainage
The lymphatics of the adrenal gland drain into the renal hilar nodes, lateral aortic nodes, and nodes of the posterior mediastinum above the diaphragm by way of the diaphragmatic orifices for the splanchnic nerves. Rouvière stated that lymphatics from the upper pole of the right adrenal gland may enter the liver [8]. The majority of capsular lymphatic vessels pass directly to the thoracic duct without the intervention of lymph nodes [9].
13.1.3 Innervation
The adrenal cortex appears to have only vasomotor innervation. Most of the fibers reaching the gland from the splanchnic nerves, the lumbar sympathetic chain, the celiac ganglion, and the celiac plexus enter the medulla. These fibers are preganglionic and end on the medullary chromaffin cells [10]. This arrangement is not as anomalous as it might appear; chromaffin cells arise from the same embryonic source as do the postganglionic neurons elsewhere. Most of these preganglionic fibers in humans are nonmyelinated [11].
13.2 Applied Physiology
The adrenal cortex appears yellow because of its high lipid content and accounts for approximately 80–90 % of the gland’s volume, while the adrenal medulla constitutes up to 10–20 % of the gland’s volume and is reddish-brown in color [2].
The adrenal cortex is composed of three zones (from outside to inside): the zona glomerulosa, the zona fasciculata, and the zona reticularis [3]. In all three zones, all cells produce steroids. Zona glomerulosa also secretes the mineralocorticoid aldosterone; zona fasciculate produces the carbohydrate-active steroid, cortisol, and the adrenal sex steroids. The innermost layer, zona reticularis, secretes cortisol, androgens, and estrogens.
The adrenal medulla cells secrete epinephrine and are called chromaffin cells or pheochromocytes. Distributed throughout the medulla, but few in number, are postganglionic sympathetic neurons. Most medullary cells secrete epinephrine, but some secrete norepinephrine instead.
Cholesterol is the common precursor of all steroid hormones synthesized in the adrenal cortex. Steroid synthesis begins with cholesterol being transported in the mitochondria by the acute regulatory protein (StAR) [12]. Cholesterol then undergoes a series of oxidative reactions catalyzed predominantly by cytochrome P-450 yielding the hormonally inactive compound pregnenolone. Further oxidation by the enzyme CYP17, confined to the zona fasciculata and zona reticularis, converts pregnenolone and progesterone into the major adrenal sex steroids dehydroepiandrosterone (DHEA) and androstenedione [13]. Oxidation of pregnenolone and 17-hydroxypregnenolone by 3b-hydroxysteroid dehydrogenase results in the production of the glucocorticoids, corticosterone, and cortisol, with only the latter being active in humans. Aldosterone is generated by oxidation of corticosterone by the enzyme CYP11B2 localized exclusively in the zona glomerulosa.
13.2.1 Glucocorticoids
Glucocorticoids are under the control of a complex mechanism that starts by the release of corticotropin-releasing factor (CRF) by the hypothalamus resulting in secretion of adrenocorticotropic hormone (ACTH) by the anterior pituitary [13]. ACTH is able to act on the adrenocortical cell surface and stimulate glucocorticoid secretion. The release of ACTH displays a circadian rhythm and is higher on waking with levels gradually declining throughout the day to reach a nadir in the early evening. Glucocorticoids can downregulate the production of both CRF and ACTH via a negative feedback mechanism.
Glucocorticoid hormones have a wide range of effects on almost all organ systems in the body. They cause an increase in blood glucose concentrations, through upregulation of gluconeogenesis, inhibition of glucose uptake by peripheral tissues, lipolysis stimulation, and a general state of insulin resistance. In the cardiovascular system, glucocorticoids maintain the cardiac contractility and peripheral vascular by sensitizing arterial smooth muscle cells to β-adrenergic stimulation [14]. Finally, glucocorticoids are potent anti-inflammatory and immunosuppressive agents that act at many levels.
13.2.2 Mineralocorticoids
Angiotensin II and blood potassium levels are the primary regulators of the release of aldosterone from the zona glomerulosa. The renin-angiotensin-aldosterone axis is sensitive to the delivery of sodium to the distal convoluted tubule of the kidney; in states such as hypovolemia, shock, renal artery vasoconstriction, and hyponatremia where there is a low sodium delivery, the release of renin from the juxtaglomerular apparatus is stimulated [13]. Aldosterone release is also highly responsive to small changes in the blood potassium level, as previously mentioned. Hypokalemia reduces aldosterone release by suppressing renin secretion and also by acting directly at the zona glomerulosa, while hyperkalemia acts on the opposite way.
Aldosterone’s role is to regulate the circulating fluid volume and electrolyte balance by promoting sodium and chloride retention in the distal tubule of the kidney. On the other hand, potassium and hydrogen ions are secreted into the urine. As in the case of glucocorticoids, there is also a negative feedback mechanism that allows suppressing the release of renin whenever there is an increase in sodium delivery to the distal tubule.
13.2.3 Adrenal Sex Steroids
Secretion of the adrenal androgens androstenedione and DHEA is regulated by ACTH and other incompletely understood mechanisms. The physiologic effects of adrenal sex steroids are generally weak in comparison to the gonadal sex steroids, particularly in males.
13.2.4 Catecholamines
Tyrosine is the substrate used for the synthesis of catecholamines in the adrenal medulla. Dihydroxyphenylalanine (l-dopa) is produced by the hydroxylation of tyrosine, and decarboxylation of l-dopa generates dopamine, which is then taken up by neurosecretory granules and β-hydroxylated to form norepinephrine [13]. Epinephrine is created by the action of phenylethanolamine N-methyl-transferase (PNMT), which is localized to the chromaffin cells of the adrenal medulla and organ of Zuckerkandl.
The stored catecholamines are released in the circulation whenever sympathetic stimulation of the adrenal medulla results in depolarization of the chromaffin cell membrane. Basal levels of adrenal catecholamine secretion are normally low, although major physiologic or psychological stressors can cause large increases in their plasma levels.
13.3 Cortical Tumors (Adenoma-Carcinoma)
13.3.1 Primary Hyperaldosteronism (Conn’s Syndrome)
Primary hyperaldosteronism (PHA) is the autonomous aldosterone secretion from one or both adrenal glands and was first described by Jerome Conn in 1954. It is classically manifested as resistant hypertension with hypokalemia, depletion of potassium, retention of sodium, and suppression of plasma/renin activity (PRA). However, the majority of patients may be actually normokalemic [15].
The prevalence of PHA between patients with hypertension was traditionally thought to be around 1 % of the patients with hypertension [16]. The more liberal definition of hyperaldosteronism with the introduction of the concept of normokalemic PHA and the wide application of screening of aldosterone/renin ratio in all hypertensive patients has caused an increase in the prevalence of PHA. Primary hyperaldosteronism usually occurs in individuals between the ages of 30 and 50 years with a mild male predilection.
Most patients are asymptomatic, although those with significant hypokalemia may complain of muscle cramps, weakness, paresthesias, polydipsia, polyuria, nocturia, headaches, and fatigue. Patients typically have moderate to severe hypertension that is refractory to medical therapy.
The most common causes of PHA are unilateral aldosterone-producing adenoma (aldosteronoma) and bilateral adrenal hyperplasia (BAH). Aldosteronoma is present in 30–60 % of cases depending on the pattern of screening (selective versus nonselective) [13]. Adrenocortical carcinoma and glucocorticoid-suppressible hyperaldosteronism are rare, each accounting for less than 1 % of cases [17].
Familial hyperaldosteronism type 1 is a rare autosomal dominant condition which results in abnormal regulation of aldosterone synthesis by ACTH. Patients have a family history of early-onset hypertension, and this condition can be treated medically. Familial hyperaldosteronism type 2 is also a rare condition with autosomal dominant inheritance where patients have autonomous aldosterone hypersecretion, which is not suppressible by dexamethasone. The causes of PHA are shown in Table 13.2 [18].
Table 13.2
Causes of primary hyperaldosteronism
Etiology | Selective screening | Nonselective screening |
|---|---|---|
Aldosterone-producing adenoma | 60 % | 30 % |
Bilateral adrenal hyperplasia (idiopathic hyperaldosteronism) | 35 % | 65 % |
Aldosterone-producing adrenocortical carcinoma | <1 % | <1 % |
Familial hyperaldosteronism | ||
Type 1 (glucocorticoid-remediable aldosteronism) | <1 % | <1 % |
Type 2 (non-glucocorticoid-remediable aldosteronism) | <1 % | <1 % |
The ultimate goal of preoperative diagnostic testing is to identify and lateralize aldosteronomas. The first biochemical test performed is to determine the ratio of plasma aldosterone to plasma renin activity. This test is performed after discontinuation of interfering medications such as spironolactone, angiotensin-converting enzyme inhibitors, diuretics, and β-adrenergic blockers. Variable cutoff values have been used in the literature, but a cutoff of 30 yields a sensitivity of approximately 90 % [19]. It is important to note that a positive screening result with A/R ratio is not diagnostic and requires a confirmatory test.
Confirmatory biochemical testing is aimed at demonstrating lack of suppression of aldosterone levels after salt loading. Patients with PHA fail to suppress aldosterone levels with sodium loading. This is done with IV saline loading (2–3 L of isotonic saline given over a 4–6-h period, followed by measurement of plasma aldosterone) or oral salt loading (200 mEq = 5000 mg sodium daily over a 3-day period, followed by measurement of 24-h urine aldosterone excretion) (Fig. 13.1).
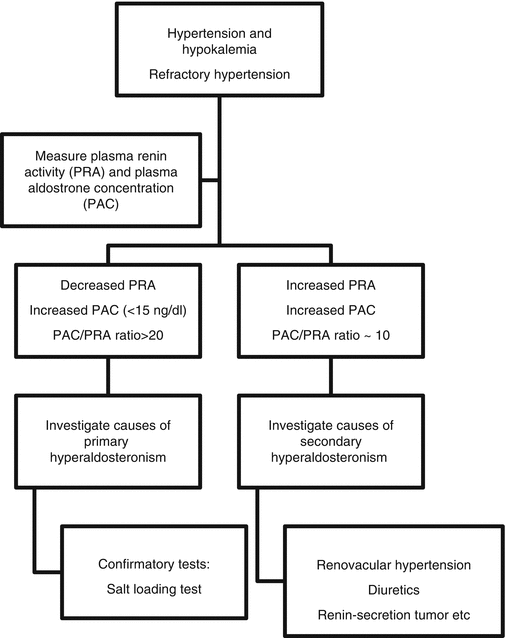

Fig. 13.1
Algorithm for diagnosing aldosteronism with the use of plasma renin activity (PRA) and plasma aldosterone concentration (PAC) and their ratio (PAC/PRA) [20]
Once diagnosis of PHA is confirmed, the next step is to lateralize the source of aldosterone excess production. Anatomic imaging and adrenal venous sampling (AVS) are the initial tests performed, and they help differentiate between adenoma and hyperplasia. Thin-cut (3 mm) adrenal computed tomography (CT) is most accurate for nodules that are larger than 1 cm in diameter [21]. If a solitary adrenal mass 1 cm or greater in size is identified with a normal contralateral adrenal gland and the patient is younger than 40 years old, there is sufficient confidence to proceed to unilateral adrenalectomy surgery [18].
Magnetic resonance imaging (MRI) scans are less sensitive, but more specific, particularly if opposed-phase chemical shift images are obtained. Such scans are very useful whenever CT is contraindicated such as pregnant patients, and in patients who are unable to tolerate intravenous iodine contrast.
New scans currently under evaluation in the armamentarium to lateralize the site of excess aldosterone secretion include 11C-metomidate (MTO), a potent inhibitor of the adrenocortical enzyme 11β-hydroxylase that has been advanced as a positron emission tomography (PET) radiotracer. In a small study, MTO was shown to offer a noninvasive technique to visualize subcentimeter adrenal adenomas and differentiate functional tumors from incidentalomas [22].
In cases where the adrenal CT results are equivocal or the presence of bilateral adrenal nodules is suspected, the next step is to perform AVS, which is 95 % sensitive and 90 % specific in localizing the aldosteronoma in expert hands [2]. This test relies on simultaneous measurement of cortisol, aldosterone, and renin levels in the peripheral circulation, as well as the left and right adrenal veins [13]. Furthermore, cortisol levels are used to correct for differences in dilution secondary to nearby veins, and this has been shown to produce higher accuracy of the test [23].
There are no generally accepted AVS diagnostic cutoffs, largely due to the range of different AVS techniques between centers, and the small number of procedures performed globally. The Endocrine Society guidelines suggest that a cortisol-corrected aldosterone ratio of >4 in the aldosterone to cortisol ratios between the adrenal veins indicates the presence of a unilateral tumor [24]. Ratios between 2 and 3 should be correlated with other clinical, laboratory, and radiographic findings. Aldosterone to cortisol ratios that differ by less than twofold point toward bilateral adrenal hyperplasia if the aldosterone levels are increased bilaterally [23, 25] (Fig. 13.2a). In patients with BAH, AVS allows the identification of the dominant side in case that unilateral adrenalectomy is indicated.
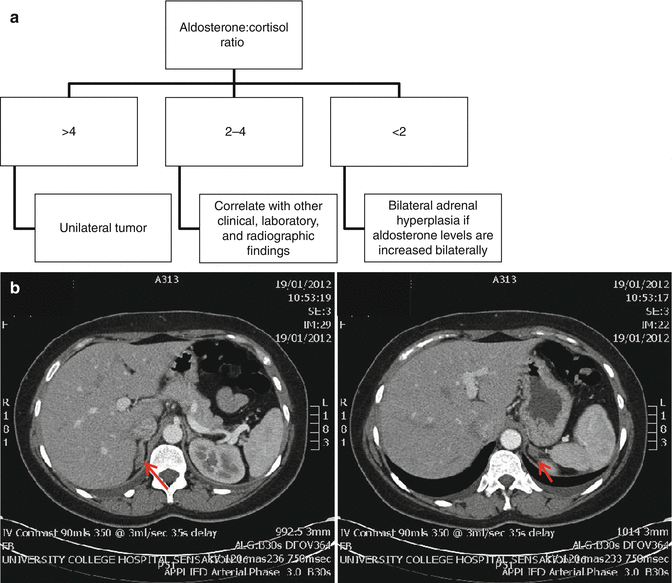

Fig. 13.2
(a) Algorithm for the management of adrenal tumors based on adrenal venous sampling results. (b) CT scan showed bilateral nodules in the adrenal glands (Red arrow) hence the need for AVS. AVS showed adequate cannulation of the left adrenal vein with adrenal/peripheral vein cortisol ratio = 16.7; cannulation of the right adrenal vein was less optimal with adrenal/peripheral vein cortisol ratio = 1. There was a significant aldosterone/cortisol ratio toward the left adrenal vein = 25.3 and the patient had a left laparoscopic adrenalectomy
Figure 13.2b shows a CT scan and AVS performed for a patient with a clinical and biochemical diagnosis of PAS to localize the adenoma.
AVS | Aldosterone | Cortisol | Adrenal/peripheral vein cortisol ratio | Aldosterone/cortisol ratio |
|---|---|---|---|---|
IVC | 3820 | 446 | ||
Left adrenal vein | 189400 | 7470 | 16.7 | 25.3 |
Right adrenal vein | 798 | 482 | 1 | 1.6 |
There is still considerable controversy over which patients should undergo this study as it is an invasive procedure with a 90 % technical success rate in experienced hands [25]. The most common cause for adrenal venous sampling failure to produce valuable results is the failure to cannulate the right adrenal vein due to its very short length (5–8 mm) [26].
Functional scans such as scintigraphy with 131I-6-iodomethyl norcholesterol (NP-59) with dexamethasone suppression have a sensitivity of approximately 90 % [1]. Like cholesterol, this compound is taken up by the adrenal cortex, but unlike cholesterol, it remains in the gland without undergoing further metabolism. Corticosteroids inhibit the uptake of radiolabeled cholesterol by hyperplastic adrenal glands, and thus dexamethasone increases tracer uptake by aldosterone-producing adenomas thereby improving the sensitivity of their detection. Adrenal adenomas appear as “hot” nodules with suppressed contralateral uptake, whereas hyperplastic glands show bilaterally increased uptake. Disadvantages of NP-59 scanning include higher radiation doses, the need to block the thyroid to prevent radioiodine uptake, and the length of the study, which can take 5–7 days to complete. Functional isotope scanning has largely been abandoned in the UK.
Laparoscopic adrenalectomy (LA) is the treatment of choice for aldosterone-producing adenomas and most other adrenal tumors and has numerous advantages over the conventional open approach, including fewer wound complications, shorter hospitalization, and quicker recovery [27]. Several studies confirmed the superiority of LA over open adrenalectomy (OA), and a recent systematic review largely confined the use of OA for oncological resections in the setting of adrenocortical carcinoma (ACC) [28–31]. Open surgical approaches to the adrenal glands include the posterior, flank, and transabdominal and are usually reserved for large adrenal tumors that harbor a suspicion of adrenocortical carcinoma.
The retro-peritoneoscopic approach evolved as an alternative minimally invasive technique to LA and was popularized by Walz et al. in 2006 [32]. Advantages of this approach include direct access to the adrenal gland without the need for mobilization of intra-abdominal viscera, avoidance of adhesions from previous abdominal surgery, and the ability to perform bilateral adrenalectomy without repositioning the patient. Disadvantages may include the limited working space of the retroperitoneum that can make dissection challenging and limit the size of tumors that can be resected with this technique to less than 6 cm.
Adrenalectomy is greater than 90 % successful in improving hypokalemia and approximately 70 % successful in correcting the hypertension [2]. In patients who continue to be hypertensive in the short term, medications may be added back temporarily as needed until blood pressure gradually reaches a new equilibrium over time.
All patients undergoing adrenalectomy for primary aldosteronism require preoperative medical optimization with control of hypertension and adequate potassium supplementation to keep potassium levels normal. Patients are generally treated with spironolactone (an aldosterone antagonist), amiloride (a potassium-sparing diuretic that blocks sodium channels in the distal nephron), nifedipine (a calcium channel blocker), or captopril (an ACE inhibitor).
13.3.2 Cushing’s Syndrome
Cushing’s syndrome is a constellation of diseases encompassing obesity, diabetes, arterial hypertension, muscular weakness, and adrenal hyperplasia. It was described by Harvey Cushing in 1932, and the term syndrome includes all hypercortisolism states; one of the most frequent causes of this syndrome is the homonymous disease, Cushing’s disease, which is caused by an excess production of pituitary adrenocorticotropic hormone (ACTH) from microadenoma of the adrenal gland [18].
There is a strong female-to-male predilection with the female-to-male ratio in Cushing’s syndrome ranging from 3 to 15:1, and the prevalence of Cushing’s disease is about 39/million people [33, 34].
The causes of Cushing’s syndrome have been traditionally classified as ACTH-dependent Cushing’s syndrome, ACTH-independent Cushing’s syndrome, and pseudo-Cushing’s syndrome (Table 13.3). The most common cause of Cushing’s syndrome (85 % of cases) is excessive production of ACTH, mainly due to autonomous pituitary adenomas. Non-adrenal (ectopic) secretion of ACTH from tumors of the lung, thymus, or pancreas constitutes about 10 % of the ACTH-dependent Cushing’s syndromes [34]. ACTH-independent Cushing’s syndrome can be due to an adrenal adenoma, carcinoma, or nodular adrenal hyperplasia. Finally, pseudo-Cushing’s syndrome is caused by major depressive disorders or alcoholism.
Table 13.3
Causes of Cushing’s syndrome
Causes | Proportion (%) |
|---|---|
ACTH dependent | 80–85 |
Cushing’s disease | 70 |
Non-adrenal ACTH syndrome | 10 |
Other source of ACTH | 5 |
ACTH independent | 15–20 |
Adrenal adenoma | 10 |
Adrenal carcinoma | 5 |
Macronodular adrenal hyperplasia | <2 |
Primary pigmented nodular adrenal disease | <2 |
McCune-Albright syndrome | <2 |
The most common clinical manifestations of Cushing’s syndrome are round facies, “buffalo hump” caused by increased fat in the dorsal neck, thin skin, hirsutism, easy bruisability, purple striae, depression, osteoporosis, hypertension, and glucose intolerance [1].
Laboratory investigations used to confirm the diagnosis include:
24-h urine collection for measurement of free cortisol
Overnight 1-mg dexamethasone suppression test which is performed by administration of 1 mg of dexamethasone at 11 pm and determination of a fasting plasma cortisol level between 8 and 9 am the following day [18]
Late-night salivary cortisol test
Low-dose dexamethasone suppression test (either as 1 mg p.o. before bedtime or as 0.5 mg p.o. every 6 h for 48 h) [18]
The finding of a raised 24-h urine free cortisol level or a failure to suppress plasma cortisol by dexamethasone signifies loss of physiologic negative feedback and is suggestive of Cushing’s syndrome with 95 % accuracy.
The next step is to distinguish between ACTH-dependent and ACTH-independent disease. ACTH-independent Cushing’s syndrome is caused by autonomous adrenal cortisol production and is associated with an undetectable ACTH level (<5 pg/mL). The underlying pathology is variable, with a solitary adrenal adenoma found in approximately 90 % of cases, adrenocortical carcinoma in less than 10 %, and bilateral micronodular or macronodular hyperplasia in less than 1 % [35]. Computed tomography with thin sections (3–5 mm) can distinguish between a unilateral adenoma, carcinoma, or bilateral symmetric or asymmetric hyperplasia. Carcinomas tend to be larger than adenomas (>5 cm), inhomogeneous, with an irregular border [1]. Distinguishing adenoma from carcinoma in large tumors can be aided by magnetic resonance imaging. Carcinomas appear brighter than the liver on T2-weighted images.
Hyper-cortisolemia associated with normal or elevated ACTH levels points to ACTH-dependent Cushing’s syndrome, which is most commonly caused by a pituitary corticotroph microadenoma (Cushing’s disease) [34]. Computed tomography (CT) imaging or magnetic resonance imaging (MRI) of the pituitary gland and a high-dose dexamethasone suppression testing (serum or urine cortisol measurement after the administration of 2 mg of dexamethasone every 6 h over a 48-h period) will confirm Cushing’s disease. Corticotroph adenomas will be suppressed in response to high-dose dexamethasone administration, whereas ectopic ACTH sources are completely lacking in feedback inhibition. In case where a clear-cut pituitary tumor is not demonstrable in CT/MRI, bilateral inferior petrosal sinus ACTH sampling with CRF stimulation is used. Other tests to localize an ectopic ACTH source include CT imaging of the chest/abdomen and occasionally somatostatin receptor scintigraphy [2].
The treatment of Cushing’s syndrome depends on the subtype causing the hypercortisolism; Cushing’s disease requires surgery of the pituitary gland, while adrenal and ectopic tumors call for surgical removal of the adrenal gland and the ectopic tumor, respectively. Adrenalectomy has been shown to be more than 90 % effective in the treatment of primary adrenal Cushing’s syndrome [34]. Pituitary microsurgery for Cushing’s disease, typically performed via a trans-nasal transsphenoidal approach, is approximately 75 % successful in experienced hands. In case of a failed operation, pituitary irradiation can help in providing remission of the disease. Laparoscopic bilateral adrenalectomy can be considered for patients in whom pituitary surgery has failed, and the disease symptoms are not controlled [18].
Perioperative and postoperative glucocorticoid administration is a critical part of the care of patients with Cushing’s syndrome. All adrenalectomy patients for Cushing’s syndrome should receive a perioperative dose of hydrocortisone (100 mg i.v. every 8 h for 24 h) [34]. Whenever adrenalectomy is performed for a solitary adenoma, hydrocortisone can usually be tapered to physiologic replacement levels over the course of several weeks. It must be noted that a subset of patients with Cushing’s syndrome might have significant HPA axis suppression depending on the duration and the duration of the underlying disease. These patients might require glucocorticoid supplementation for a longer period than usual. Perioperative antibiotics as a single dose or for 24 h to patients undergoing adrenalectomy for Cushing’s syndrome can be administered due to their elevated risk for surgical site infection.
13.3.3 Adrenocortical Cancer (ACC)
Cancers of the adrenal gland are rare constituting only 0.05–0.2 % of all cancer deaths [36]. Adrenocortical cancer (ACC) has a bimodal age distribution with the first peak prior to age 5 and the second peak between the ages of 40 and 50 with no significant gender predilection [37]. Even though many of these tumors are functionally active and secrete steroid hormones, they usually present in an advanced stage, and as a result their prognosis is poor.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


