Local therapy of uveitis consists of topical drops, periocular injections, and intravitreal modalities, including injections and implants.
TOPICAL THERAPY
PREDNISOLONE AND DIFLUPREDNATE
 Prednisolone acetate 1% ophthalmic suspension is a commonly used steroid eye drop marketed under the brand names Pred-Forte, Econopred, and Omnipred. Numerous generic formulations are also available.
Prednisolone acetate 1% ophthalmic suspension is a commonly used steroid eye drop marketed under the brand names Pred-Forte, Econopred, and Omnipred. Numerous generic formulations are also available.
 Difluprednate ophthalmic emulsion 0.05% (Durezol) is a newer alternative that is currently FDA approved for the treatment of inflammation and pain associated with ocular surgery and is being evaluated for the treatment of uveitis.
Difluprednate ophthalmic emulsion 0.05% (Durezol) is a newer alternative that is currently FDA approved for the treatment of inflammation and pain associated with ocular surgery and is being evaluated for the treatment of uveitis.
 It is thought to be at least as effective as prednisolone acetate 1%, and some evidence suggests that it is approximately twice as effective.
It is thought to be at least as effective as prednisolone acetate 1%, and some evidence suggests that it is approximately twice as effective.
 Unlike prednisolone acetate, which is a suspension, difluprednate does not require shaking, and due to its higher potency, it may allow for a less frequent dosing regimen.
Unlike prednisolone acetate, which is a suspension, difluprednate does not require shaking, and due to its higher potency, it may allow for a less frequent dosing regimen.
Indication and Dosing
 Topical drops should be used to treat anterior uveitis. It may also be used to treat the anterior component of uveitis that also affects the intermediate or posterior compartments. Drops should not be used as monotherapy for intermediate uveitis, posterior uveitis, or panuveitis.
Topical drops should be used to treat anterior uveitis. It may also be used to treat the anterior component of uveitis that also affects the intermediate or posterior compartments. Drops should not be used as monotherapy for intermediate uveitis, posterior uveitis, or panuveitis.
 The drops should be dosed according to disease activity and can be used as frequently as every half hour while awake.
The drops should be dosed according to disease activity and can be used as frequently as every half hour while awake.
 Brand name Pred-Forte is felt to be more effective than generic prednisolone acetate 1%.
Brand name Pred-Forte is felt to be more effective than generic prednisolone acetate 1%.
 The goal is to eliminate the anterior chamber cell activity whenever possible.
The goal is to eliminate the anterior chamber cell activity whenever possible.
 A common regimen is to taper by 1 drop per week, but tapering should not occur until the eye is inflammation free (if able to be achieved within a reasonable period of time).
A common regimen is to taper by 1 drop per week, but tapering should not occur until the eye is inflammation free (if able to be achieved within a reasonable period of time).
Complications and Side Effects
 The main side effects of topical steroids are an elevated intraocular pressure and cataract formation. This risk may be higher with difluprednate.
The main side effects of topical steroids are an elevated intraocular pressure and cataract formation. This risk may be higher with difluprednate.
 Secondary infections, such as reactivated herpetic keratitis, bacterial keratitis, or fungal keratitis are also possible side effects.
Secondary infections, such as reactivated herpetic keratitis, bacterial keratitis, or fungal keratitis are also possible side effects.
 Patients should not use contact lenses while using topical steroid therapy.
Patients should not use contact lenses while using topical steroid therapy.
CYCLOPLEGICS AND MYDRIATICS
 Cycloplegics and mydriatics such as tropicamide, cyclopentolate, scopolamine, homatropine, and atropine dilate the pupil and can prevent the development of posterior synechiae and may ameliorate pain associated with ciliary body inflammation and spasm.
Cycloplegics and mydriatics such as tropicamide, cyclopentolate, scopolamine, homatropine, and atropine dilate the pupil and can prevent the development of posterior synechiae and may ameliorate pain associated with ciliary body inflammation and spasm.
Indication and Dosing
 It can be used for all cases with significant anterior chamber inflammation or eye pain suggestive of ciliary body spasm.
It can be used for all cases with significant anterior chamber inflammation or eye pain suggestive of ciliary body spasm.
 Usually shorter acting agents such as tropicamide provide a few hours of dilation followed by subsequent pupillary constriction. Three times a day dosing allows several cycles of dilation and constriction during a 24-hour period; by keeping the pupil moving, posterior synechiae development is reduced.
Usually shorter acting agents such as tropicamide provide a few hours of dilation followed by subsequent pupillary constriction. Three times a day dosing allows several cycles of dilation and constriction during a 24-hour period; by keeping the pupil moving, posterior synechiae development is reduced.
 Eyes with darker irides or higher levels of inflammation may require stronger/longer acting agents such as scopolamine, homatropine, or atropine, both to facilitate the dilation/constriction cycle and to relieve ciliary pain.
Eyes with darker irides or higher levels of inflammation may require stronger/longer acting agents such as scopolamine, homatropine, or atropine, both to facilitate the dilation/constriction cycle and to relieve ciliary pain.
 Some pupillary motility should be encouraged. If the pupil is immobile while dilated, posterior synechiae can still form.
Some pupillary motility should be encouraged. If the pupil is immobile while dilated, posterior synechiae can still form.
Complications and Side Effects
 These medications are anticholinergics; therefore they may induce dryness of the mouth, facial flushing, headache, and rarely vasomotor or cardiorespiratory disturbances. Patients at risk of such complications may be advised to perform nasolacrimal occlusion for 1 minute after drop instillation.
These medications are anticholinergics; therefore they may induce dryness of the mouth, facial flushing, headache, and rarely vasomotor or cardiorespiratory disturbances. Patients at risk of such complications may be advised to perform nasolacrimal occlusion for 1 minute after drop instillation.
PERIOCULAR THERAPY
TRIAMCINOLONE
 Triamcinolone acetonide is an injected long-acting steroid suspension, obtained under the brand names Kenalog or Kenalog-40.
Triamcinolone acetonide is an injected long-acting steroid suspension, obtained under the brand names Kenalog or Kenalog-40.
 Kenalog has been used for periocular injections in an off-label fashion for decades.
Kenalog has been used for periocular injections in an off-label fashion for decades.
Indication and Dosing
 It is useful for all types of intraocular inflammation: Anterior, intermediate, posterior, and panuveitis, as well as uveitic macular edema.
It is useful for all types of intraocular inflammation: Anterior, intermediate, posterior, and panuveitis, as well as uveitic macular edema.
 Transconjunctival, transdermal, anterior sub-Tenon’s, and posterior sub-Tenon’s (Nozik style) injection techniques may be used.
Transconjunctival, transdermal, anterior sub-Tenon’s, and posterior sub-Tenon’s (Nozik style) injection techniques may be used.
 Typically 40 mg (usually in 1 mL) is given, although 20 mg/0.5 mL may be used for an anterior sub-Tenon’s injection.
Typically 40 mg (usually in 1 mL) is given, although 20 mg/0.5 mL may be used for an anterior sub-Tenon’s injection.
Complications and Side Effects
 The most feared complication is inadvertent ocular penetration.
The most feared complication is inadvertent ocular penetration.
 Patients may develop ptosis, even after a single injection.
Patients may develop ptosis, even after a single injection.
 Septal atrophy with protuberance of the lower eyelid may also occur.
Septal atrophy with protuberance of the lower eyelid may also occur.
Figure 14-1. This patient developed a posterior subcapsular cataract due to both her uveitis and periocular steroid injections. (Courtesy of Julia Monsonego, CRA.)
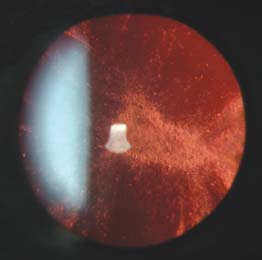
 Increased intraocular pressure and cataract may occur months or even years later, so patients need to have regular intraocular pressure measurements (Fig. 14-1).
Increased intraocular pressure and cataract may occur months or even years later, so patients need to have regular intraocular pressure measurements (Fig. 14-1).
 A triamcinolone injection may increase the risk of secondary infections and contact lens use should be discouraged.
A triamcinolone injection may increase the risk of secondary infections and contact lens use should be discouraged.
 Chorioretinal scars suggestive of previous Toxoplasma infection and a positive Toxoplasma serum IgG titer are a strong relative contraindication to steroid injection.
Chorioretinal scars suggestive of previous Toxoplasma infection and a positive Toxoplasma serum IgG titer are a strong relative contraindication to steroid injection.
INTRAVITREAL THERAPY
TRIAMCINOLONE
 Triamcinolone acetonide is an injectable long-acting steroid suspension.
Triamcinolone acetonide is an injectable long-acting steroid suspension.
 Trivaris (Allergan) and Triesence (Alcon) are preservative-free and are FDA-labeled for intraocular use in uveitis and ocular inflammatory conditions.
Trivaris (Allergan) and Triesence (Alcon) are preservative-free and are FDA-labeled for intraocular use in uveitis and ocular inflammatory conditions.
 Preservative-free triamcinolone can be obtained from some compounding pharmacies.
Preservative-free triamcinolone can be obtained from some compounding pharmacies.
Indication and Dosing
 It can be used in cases of intermediate, posterior, and panuveitis, as well as in cases of uveitic cystoid macular edema, in cases in which systemic therapy is insufficient and/or contraindicated.
It can be used in cases of intermediate, posterior, and panuveitis, as well as in cases of uveitic cystoid macular edema, in cases in which systemic therapy is insufficient and/or contraindicated.
 The traditional dose is 4 mg. However, a lower dose of 1 to 2 mg may be considered, particularly for uveitic macular edema. One mg may also be associated with a lower rate of side effects.
The traditional dose is 4 mg. However, a lower dose of 1 to 2 mg may be considered, particularly for uveitic macular edema. One mg may also be associated with a lower rate of side effects.
Side Effects
 Patients may develop:
Patients may develop:
 True infectious endophthalmitis
True infectious endophthalmitis
 A paradoxical noninfectious inflammatory response to the drug (“sterile endophthalmitis”)
A paradoxical noninfectious inflammatory response to the drug (“sterile endophthalmitis”)
 A “pseudoendophthalmitis” due to triamcinolone particles settling in the anterior chamber and appearing as a hypopyon
A “pseudoendophthalmitis” due to triamcinolone particles settling in the anterior chamber and appearing as a hypopyon
 Increased intraocular pressure and cataract may occur months or years later.
Increased intraocular pressure and cataract may occur months or years later.
 As with sub-Tenon’s steroid injection, when possible, periocular steroid injection should be avoided in eyes with a previous Toxoplasma infection and a positive Toxoplasma serum IgG titer.
As with sub-Tenon’s steroid injection, when possible, periocular steroid injection should be avoided in eyes with a previous Toxoplasma infection and a positive Toxoplasma serum IgG titer.
RANIBIZUMAB AND BEVACIZUMAB
 Ranibizumab (Lucentis) and bevacizumab (Avastin) are genetically engineered monoclonal antibodies that bind to and inhibit the biologic activity of human vascular endothelial growth factor (VEGF). Ranibizumab is FDA approved for intraocular injection to treat wet macular degeneration and macular edema associated with retinal vein occlusion. Bevacizumab is FDA approved for IV use for various forms of cancer; the intraocular use of bevacizumab is off label, but its use is well established both in the ophthalmic literature and in clinical practice.
Ranibizumab (Lucentis) and bevacizumab (Avastin) are genetically engineered monoclonal antibodies that bind to and inhibit the biologic activity of human vascular endothelial growth factor (VEGF). Ranibizumab is FDA approved for intraocular injection to treat wet macular degeneration and macular edema associated with retinal vein occlusion. Bevacizumab is FDA approved for IV use for various forms of cancer; the intraocular use of bevacizumab is off label, but its use is well established both in the ophthalmic literature and in clinical practice.
Indication and Dosing
 Anti-VEGF therapy may be used in conjunction with steroid therapy (usually sub-Tenon’s or intravitreal triamcinolone) for inflammatory choroidal neovascularization associated with white dot syndromes, sarcoidosis, and other forms of uveitis. It is also useful for peripheral retinal and anterior segment neovascularization.
Anti-VEGF therapy may be used in conjunction with steroid therapy (usually sub-Tenon’s or intravitreal triamcinolone) for inflammatory choroidal neovascularization associated with white dot syndromes, sarcoidosis, and other forms of uveitis. It is also useful for peripheral retinal and anterior segment neovascularization.
 While anti-VEGF therapy may be considered for patients with uveitic macular edema unresponsive to steroid and steroid-sparing therapy, any effect tends to be mild and transient, with a need for repeated injections over time. Therefore, anti-VEGF treatment should not be considered first-line therapy for uveitic macular edema.
While anti-VEGF therapy may be considered for patients with uveitic macular edema unresponsive to steroid and steroid-sparing therapy, any effect tends to be mild and transient, with a need for repeated injections over time. Therefore, anti-VEGF treatment should not be considered first-line therapy for uveitic macular edema.
 The typical dosing is a 0.05-mL injection of either ranibizumab 0.5 mg or bevacizumab 1.25 mg.
The typical dosing is a 0.05-mL injection of either ranibizumab 0.5 mg or bevacizumab 1.25 mg.
Complications and Side Effects
 The standard potential complications of an intravitreal injection apply.
The standard potential complications of an intravitreal injection apply.
 Rarely, anywhere from days to weeks after an injection, patients may develop a mild inflammatory response with anterior chamber and vitreous cells but without significant vitreous haze.
Rarely, anywhere from days to weeks after an injection, patients may develop a mild inflammatory response with anterior chamber and vitreous cells but without significant vitreous haze.
FLUOCINOLONE IMPLANT (RETISERT)
 The fluocinolone acetonide intravitreal implant is a long-acting implant marketed under the name Retisert. It is inserted through a pars plana incision performed in the operating room (Fig 14-2).
The fluocinolone acetonide intravitreal implant is a long-acting implant marketed under the name Retisert. It is inserted through a pars plana incision performed in the operating room (Fig 14-2).
Indication and Dosing
 The FDA label states that the fluocinolone implant is indicated for the treatment of chronic noninfectious uveitis affecting the posterior segment of the eye. In terms of the SUN classification, the implant can be used to treat significant intermediate, posterior, or panuveitis.
The FDA label states that the fluocinolone implant is indicated for the treatment of chronic noninfectious uveitis affecting the posterior segment of the eye. In terms of the SUN classification, the implant can be used to treat significant intermediate, posterior, or panuveitis.
Figure 14-2. The Retisert implant. The Retisert implant is surgically placed through the pars plana. The anchoring suture is passed through the hole in the anchoring strut. The drug is released over 30 months. The implant can be left in place, and if needed, a second implant can be place elsewhere in the eye. (Courtesy of Bausch and Lomb.)

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


