Chapter 52 Transcochlear Approach to Cerebellopontine Angle Lesions
 Videos corresponding to this chapter are available online at www.expertconsult.com.
Videos corresponding to this chapter are available online at www.expertconsult.com.
The transcochlear approach evolved from the inability to excise the base of implantation and control the blood supply of these near-midline and midline tumors with other surgical approaches. Total removal of these lesions through a suboccipital approach is often impossible because of the interposition of the cerebellum and the brainstem.1,2 The transpalatal-transclival approach was attempted for these intradural midline lesions, with little success, during the early 1970s.3 The exposure was often inadequate; the field is relatively far from the surgeon; the blood supply is lateral, away from the surgeon’s view; and the risk of intracranial complications caused by oral contamination is increased. The retrolabyrinthine approach is limited in its forward extension by the posterior semicircular canal. Tumor access with the translabyrinthine approach is limited anteriorly by the facial nerve, which impedes removal of the tumor’s base of implantation, which is anterior to the IAC, around the intrapetrous carotid artery, or anterior to the brainstem. The development of the extended middle fossa approach and combined transpetrous approach enables complete removal of petroclinoid meningiomas, and is used in patients with useful hearing.4–6 The primary limitation with this approach is poor access to tumors with inferior or midline extensions.7,8 The endoscopic-assisted transsphenoidal approach has been gaining acceptance as an alternative approach to access midline intracranial lesions arising from the clivus and petrous apex lesions (Stamm A, personal communication, 2008).
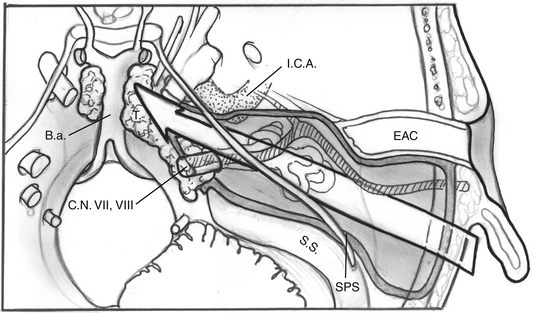
The transcochlear approach was developed by House and Hitselberger2,3 in the early 1970s as an anterior extension of the translabyrinthine approach. It involves rerouting of the facial nerve posteriorly and the removal of the cochlea and petrous apex, which exposes the area of the intrapetrous internal carotid artery. This approach affords wide intradural exposure of the anterior CPA; CN V, VII, VIII, IX, X, and XI; both sixth cranial nerves; the clivus; and the basilar and vertebral arteries, without using any brain retractors. The contralateral cranial nerves and the opposite CPA are also visible.9 This wide exposure affords removal of the tumor base and its arterial blood supply from the internal carotid artery, which is particularly important in the treatment of meningiomas.2 Adding excision and closure of the external auditory canal (EAC), as advocated by Brackmann (personal communication, 1987), increases further the anterior exposure for lesions of the petroclival regions and prepontine cistern. Smaller lesions can be reached without rerouting the facial nerve (transotic approach).
ADVANTAGES OF TRANSCOCHLEAR APPROACH
This approach requires no cerebellar or temporal lobe retraction. Exposure and dissection of the petrous apex and clivus allows excellent exposure of the midline and complete removal of the tumor, its base of implantation, and its blood supply. This is of particular importance in meningiomas.10,11
PATIENT EVALUATION AND PREOPERATIVE COUNSELING
Individuals with tumors that require transcochlear surgery may have minimal symptoms, with tumors that can be quite large at the time of diagnosis.1,12 Petrous apex epidermoids manifest with unilateral hearing loss and tinnitus in 80% of the cases. Facial twitch is also common. Imbalance, ataxia, and parietal or vertex headaches may be the only complaints in 20% of patients.13 Patients with meningiomas and intradural epidermoids may be nearly symptom-free until they present with CN V findings and signs of increased intracranial pressure.14,15 There is a high rate of jugular foramen syndrome in patients with meningiomas.1 Seizures, dysarthria, and late signs of dementia from hydrocephalus were common presenting symptoms in the past.13 Hearing and vestibular functions are frequently normal, and acoustic reflex decay or abnormal auditory brainstem response audiometric results may be the only anomalies.1
Imaging using high-resolution computed tomography (CT) with contrast enhancement, magnetic resonance imaging (MRI), and magnetic resonance angiography (MRA) is essential for diagnosis and surgical planning.16 Petrous apex and intradural epidermoids are expansile, spherical, or oval lesions, with scalloped bone edges on CT. They are isodense to cerebrospinal fluid on CT, with capsular enhancement. On MRI, they are hypointense on T1-weighted images and hyperintense on T2-weighted images. Meningiomas enhance with contrast on MRI, and manifest with a “dural tail.” Evaluation of blood supply of some tumors may also require MRA. In tumors surrounding or invading the intrapetrous carotid artery, patency of the circle of Willis is accessed, and carotid residual pressure is measured; preoperative balloon occlusion of the carotid artery and selective embolization are performed 1 day before surgery. Radioisotope, xenon, or positron emission tomography studies are used to assess cerebral perfusion during occlusion studies.
SURGICAL TECHNIQUE
A wide mastoidectomy and labyrinthectomy are performed, exposing the IAC. When first described by House and Hitselberger in 1976,3 the tympanic ring was not removed, and only the facial recess was opened to permit anterior exposure. Brackmann, after Fisch, modified the approach by removing the entire tympanic ring, malleus, incus and stapes, and blind-sac closing the EAC.17,18
The facial nerve is completely skeletonized, with transection of the greater superficial petrosal and chorda tympani nerves, and is rerouted posteriorly out of the fallopian canal. The cochlea and the fallopian canal are completely drilled out, and the internal carotid artery is skeletonized. A large triangular window is created into the skull base. Its superior boundary is the superior petrosal sinus; inferiorly, it extends below and medial to the inferior petrosal sinus into the clivus. Anteriorly lies the region of the intrapetrous internal carotid artery, and the apex of the triangle is just beneath Meckel’s cave. When the dura is opened, this window gives excellent direct access to the midline without need of any retraction (Fig. 52-1). After tumor removal, the dura is reapproximated with dural silk, the eustachian tube is packed with absorbable knitted fabric (Surgicel) and muscle, and abdominal fat is used to fill the dura and mastoidectomy defects and to cushion the facial nerve.
Mastoidectomy
An extended mastoidectomy (Fig. 52-2) is carried out with microsurgical cutting and diamond burrs and continuous suction-irrigation. Bone removal is started along two lines: one along the linea temporalis and another tangential to the EAC. The mastoid antrum is opened, and the lateral semicircular canal is identified. The lateral semicircular canal is the most reliable landmark in the temporal bone, and allows the dissection to proceed toward delineating the vertical fallopian canal and the osseous labyrinth.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree