CHAPTER 176 Temporal Bone Neoplasms and Lateral Cranial Base Surgery
Benign and malignant lesions (Box 176-1) involving the middle ear and temporal bone are located in one of the most inaccessible areas of the body. Although a variety of histologic types of tumor are found in these areas, each is uncommon, and clinical experience in their diagnosis and management is not easily acquired. Surgical approaches to the skull base are difficult, and the risks of neurologic deficit are significant. The major arteries and veins supplying the brain, intracranial structures, and all of the cranial nerves either exit through or are contiguous with the temporal bone. Modern surgical technique strongly stresses the minimization of cosmetic or neurologic loss in the management of benign and malignant lesions of the temporal bone. Through the years, collaborative approaches to the temporal bone have been developed for treatment of benign lesions and are being expanded to encompass malignant lesions as well. Nevertheless, considerable knowledge of temporal bone anatomy and adequate temporal bone-drilling laboratory experience are essential in adapting these approaches for each patient. Many histologic types are encountered only rarely, accounting for isolated case reports in the literature. Dividing tumors into benign and malignant categories, in combination with knowledge of the particular tumor biology and meticulous preoperative planning, greatly facilitates selection of the appropriate surgical approach and successful outcome.
Box 176-1 Temporal Bone Neoplasms
Skull Base Anatomy
An overview of skull base anatomy may be found in Chapters 127 and 173. With lateral approaches to the skull base, anatomic structures are approached from a vantage point different from that presented in classic anatomic texts in terms of the course of dissection, in that such approaches are designed to enhance exposure while preserving neurovascular structures. The superior boundary of the infratemporal fossa as described by Grant1 consists of the greater wing of the sphenoid the temporal fossa containing the temporalis muscle. The medial limit is the lateral pterygoid plate, and the lateral limit is the mandibular ramus and condyle. The posterior wall of the maxillary sinus marks the anterior limit, and posteriorly and inferiorly, the infratemporal fossa opens into the parapharyngeal space, where fascial bands and ligaments subdivide the enclosed spaces.2–4
The facial nerve exits the stylomastoid foramen to enter the parotid gland and then emerges from the gland distally encased in the superficial muscular aponeurotic system fascia.5 The glenoid fossa may limit superior exposure in lateral approaches.
As low-pressure, valveless channels, the venous system in the skull base is pivotal in lateral approaches. The torcular Herophili is the confluence of the transverse sinus (lateral sinus), superior longitudinal sinus, straight sinus, and occipital sinus. Bisaria6 found that this confluence of sinuses was missing in 24.5% of 110 cadaver dissections. Ligation of the transverse sinus may lead to massive cerebral edema if the torcular Herophili is absent.7 The vein of Labbé, or inferior anastomotic vein, connects the superficial middle cerebral vein with the transverse sinus, draining the superficial posterior temporal and inferior parietal lobes. It occurs on the right 66% of the time and on the left 77% of the time.8 When present, it empties into the transverse sinus just proximal to the sigmoid sinus. Interruption of the vein of Labbé may lead to temporal lobe edema and infarction, unless good collateral drainage is present.7,8 The superior petrosal sinus runs along the posterosuperior edge of the petrous bone from the cavernous sinus to the junction of transverse and sigmoid sinuses, receiving tributaries from the tympanic cavity and from the cerebellar and inferior cerebral veins.9 The jugular bulb, averaging 15 mm in width, usually is separated from the tympanic cavity by bone but has been reported to be dehiscent in 6% of temporal bones.10 The inferior petrosal sinus courses between the jugular bulb and cavernous sinus in the petro-occipital fissure, receiving tributaries from the internal auditory canal, pons, medulla, and inferior cerebellum.9 The inferior petrosal sinus, occasionally the vein of the cochlear aqueduct, and the occipital sinus drain into the jugular bulb. Cranial nerves IX, X, and XI pass through the anterior medial portion of the jugular foramen and may be compressed during maneuvers to obtain hemostasis of the inferior petrosal sinus.11
The internal carotid artery ascends deep to the digastric and styloid muscles to enter the carotid siphon medial to the styloid process, ascending through vertical and horizontal segments. In the vertical segment near the genu, the artery is just deep to the eustachian tube orifice, posteromedial to the glenoid fossa. The osseous eustachian tube orifice is separated from the internal carotid by, on average, 1.5 mm of bone and may have dehiscences.12,13 The midportion of the vertical arterial segment is anteromedial to the basal turn of the cochlea. The distance to the cochlea averages approximately 1.34 mm.14 The average length of the vertical segment is approximately 10 mm (range, 5 to 15 mm).12,15 The horizontal segment travels obliquely in a lateral to medial direction from the genu to the intracranial entry of this vessel at the superior aspect of the foramen lacerum. Its average length is 20 mm (range, 14.5 to 26 mm).12,15 Relevant lymphatic anatomy includes the preauricular and parotid lymphatics that drain the external ear, with more medial aspects of the external auditory canal draining into deep jugular and postauricular nodes. The mucosa of the middle ear and mastoid drains into channels surrounding the eustachian tube, which then drains into the deep superior jugular and retropharyngeal nodes.16
Evaluation of Skull Base Lesions
Accurate diagnosis and evaluation of tumor extent of the middle ear space and temporal bone require appropriate physical examination and audiologic, vestibular, and radiologic assessment. Tumors of the middle ear space frequently can be seen on physical examination. The combination of the frequency of certain lesions and their classic presentations often suggest the tumor type. Tumors of the lateral skull base often manifest with cranial nerve deficits, including hearing loss, pulsatile tinnitus, and eustachian tube dysfunction. A thorough history and physical examination with special attention to the cranial nerves should be undertaken. Audiometry is useful in surgical planning, particularly when removal of the labyrinth will be necessary. Radiographic evaluation provides information regarding location, extensions, surrounding structural relationships, and probable histologic findings.17–25
Computed Tomography and Magnetic Resonance Imaging
Preoperative computed tomography (CT) and magnetic resonance imaging (MRI) are indispensable to modern skull base surgery (see Chapter 135). Hirsch and Curtin26 noted that MRI and CT are “complementary and not competitive in the initial evaluation of skull base lesions.” Modern CT affords exquisite bony detail and fairly good soft tissue information. The strength of CT lies in delineating the effect of the tumor on the bone of the skull base. Soft tissue–soft tissue discrimination is still limited because of the narrow range of electron densities in soft tissues of interest. More soft tissue discrimination is available with CT if iodinated contrast material is administered intravenously. The advent of MRI with its superb soft tissue sensitivity has resulted in fewer indications for use of iodinated contrast CT techniques. Although MRI is able to evaluate alterations in bony structure, it does so only indirectly, because cortical bone has few mobile water protons and hence nearly no intrinsic signal. For this reason, MRI is inferior to CT in the evaluation of subtle bony changes. Such features are important findings in temporal bone masses. The fine soft tissue discrimination of MRI is improved by the intravenous infusion of the MRI contrast medium gadolinium. Another advantage of MRI is its ability to display numerous imaging planes without repositioning the patient, which makes it easier to acquire coronal, sagittal, and even off-axis oblique images. For these reasons, MRI is superior to CT in defining brain or meningeal effects from temporal bone lesions. Because of potentially dangerous magnetic effects, MRI cannot be used in patients with certain cardiac pacemakers or valves, ferromagnetic cerebral aneurysm clips, some vena cava filters, and ferromagnetic intraocular foreign bodies.
Preoperative Carotid Artery Assessment
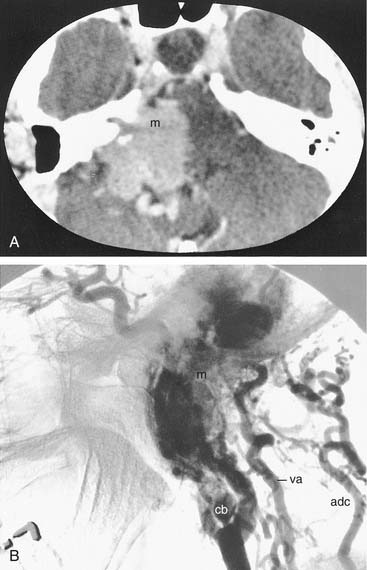
In extirpation of skull base tumors, appropriate management of the internal carotid artery remains paramount (Fig. 176-1). The morbidity associated with carotid artery occlusion is well known. The first documented carotid ligation, for a sword injury, performed in 1585 by Paré, resulted in stroke.27 Literature review reveals that unselected abrupt sacrifice of this vessel resulted in 26% (65 of 254) cerebral infarctions, with a 46% fatality rate (30 of 65).28 Gradual occlusion, as with a Silverstone clamp, does not seem to alter morbidity. Berenstein and associates29 noted that a significant reduction of blood flow or pressure does not occur until a cross-sectional area less than 2 mm2 is attained, regardless of the diameter of the vessel. Late-onset complications from cervical internal carotid artery ligation may result after several hours, as a result of stasis with distal thrombosis and embolic phenomena.
Four-Vessel Arteriography
All patients considered for extensive lateral skull base surgery should undergo a four-vessel carotid angiogram with venous phase (see Chapters 135 and 136). Angiography is only qualitative but may provide critical information (Fig. 176-2).
According to Fisch and colleagues, angiographic evidence of ICA contour irregularity or stenosis at the level of lesion should prompt consideration of elective preoperative ipsilateral carotid balloon occlusion.30 Intraoperative carotid occlusion is associated with a higher rate of permanent neurologic sequelae.
Cerebral Blood Flow Assessment
Some understanding of cerebral blood flow (CBF) is essential to carotid artery management. Normal CBF approximates 50 mL/100 g per minute.31 In alert humans, CBF less than 20 mL/100 g per minute produces failure of brain function, resulting in at least temporary hemiparesis or hemiplegia.32 At this point, electroencephalography (EEG) usually reveals changes reflecting synaptic transmission failure.31 As CBF decreases below 15 mL/100 g per minute, electrical activity is lost.33 Jones and coworkers34 found that long-lasting ischemia in monkeys from local CBF less than 17 to 18 mL/100 g per minute resulted in infarction. The time course of irreversible neuronal damage at various CBF levels in humans is unknown.
Functional assessment of CBF is required in the following clinical circumstances:
Qualitative Cerebral Blood Flow Evaluation
Various methods have been proposed to assess functional collateral circulation in the circle of Willis. Simplest is the Matas test, in which the ipsilateral carotid artery is compressed against a vertebral transverse process while the examiner observes for deterioration of neurologic function.35 Cross-compression carotid arteriography will give a qualitative feel for ipsilateral CBF.36 Direct intraoperative carotid stump pressures can estimate collateral circulation. A back-pressure of 50 mm Hg has been considered to be indicative of adequate CBF,37 but stump pressures have been shown to be unreliable.38,39 Ocular plethysmography and transcranial Doppler velocitometry of the middle cerebral artery40 have similar theoretic limitations. In a series of patients undergoing carotid surgery, intraoperative EEG revealed no change in 18% to 56% of those with CBF levels less than 20 to 24 mL/100 g per minute, depending on the anesthetic used.41 EEG, therefore, may be too insensitive for detection of CBF abnormality in patients who experience prolonged occlusions of the internal carotid artery.
Generally accepted evaluations of CBF all involve the interventional radiologic technique of balloon test occlusion (BTO). The assessment of CBF will be either qualitative or quantitative with BTO with neurologic monitoring. Fisch uses qualitative BTO as the sole method to assess collateral CBF, with the neuroradiologist making an experienced, qualitative judgment regarding which patients should undergo permanent occlusion.30 The collateral circulation may be stressed during BTO with pharmacologically induced hypotension or acetazolamide. Other qualitative techniques for CBF evaluation, such as single photon emission computed tomography (SPECT) and magnetoencephalography, attempt to “quantify” CBF.42–45
Quantitative Cerebral Blood Flow Evaluation
PET has the advantage of measuring CBF and metabolic parameters simultaneously.46 Of the tests mentioned, PET provides the most accurate knowledge regarding cerebral hemodynamics, although it requires expensive infrastructure, is not widely available, and is time consuming.
New advances show that functional MRI (fMRI) has the potential to supplant xenon flow studies.47–51 Such MRI studies have been used to evaluate CBF preoperatively and postoperatively for extracranial-intracranial bypass procedures.52 The potential advantage of fMRI lies in that it requires only software upgrades to current MRI scanners. Disadvantages include the need for transport away from the vascular suite and for special equipment tolerant of the highly magnetic field.
Cerebral perfusion CT with or without acetazolamide challenge can be performed quickly on any standard helical (spiral) CT scanner with perfusion maps generated by software.53,54 Further studies are needed to validate perfusion CT as a quantitative technique comparable to xenon CT.
Xenon 133 scintillation shows excellent correlation with quantitative assessments of CBF in a defined region with a scintillation counter on the scalp41 and also may be used intraoperatively.
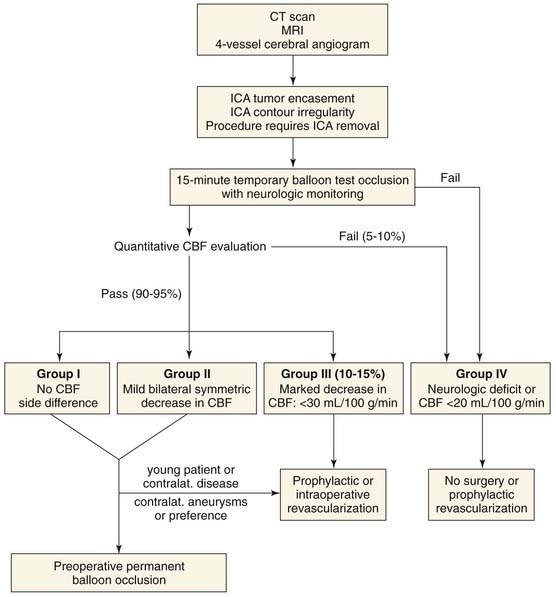
Xenon CT blood flow mapping is the best studied clinical, quantitative measure of CBF.28,42,55–58 Inhaled xenon acts as a contrast agent, whose uptake and diffusion from cerebral tissue is proportional to blood flow. CBF distribution is estimated with specialized computer software before and after BTO of the internal carotid artery during serial CT scans and from which a blood flow map is calculated. An algorithm for carotid artery management has been proposed on the basis of preoperative xenon CT blood flow mapping with temporary carotid balloon occlusion (see Fig. 176-1). Group 3 patients (see Fig. 176-1), although “passing” BTO, have CBF values between 20 and 30 mL/100 g per minute and are thought to be in hemodynamic decompensation without neurologic deficit but entirely dependent on systemic blood pressure.56 As is well recognized, intraoperative or postoperative blood pressure fluctuations after ICA occlusion may send these patients over the precipice. It is estimated that 90% of group 3 patients will have a major stroke after permanent occlusion of the internal carotid artery.59
Carotid Management Algorithms
A decision regarding management of the internal carotid artery is best made preoperatively. Intraoperative carotid occlusion usually is performed to control massive bleeding under suboptimal circumstances, resulting in a higher neurologic complication rate. Intraoperative balloon occlusion or intra-arterial shunt placement may be required if the carotid is lacerated and control is difficult.60–62
Vein graft reconstruction, instead of sacrifice of the artery, may be a consideration regardless of the BTO result, especially in young patients and in cases in which contralateral involvement is a possibility, to restore physiologic circulation and reduce risk of late intracranial aneurysms.33,42 The rationale for preservation of an ICA encased by benign tumor when this is possible, or revascularization when preservation is not possible, is valid.63
For patients with BTO results in the “fail” category, definitive resection may not be possible; alternatively, extracranial-intracranial bypass antecedent to the resection procedure should be considered.42,64–66
Patients who “pass” are further subclassified depending on whether qualitative or quantitative testing has been done. Fisch30 using qualitative BTO, recommends permanent occlusion of the internal carotid artery preoperatively if the neuroradiologist confirms adequate cross-circulation and no neurologic deterioration occurs during the test. In a series reported by Zane and colleagues, only 5% (2 of 46 patients, 1 preoperatively and 1 postoperatively) had permanent neurologic sequelae after occlusion when this algorithm was used.30 If quantitative testing has been performed, transarterial permanent balloon occlusion may be considered for operative candidates in groups 1 and 2 (CBF greater than 30 mL/100 g per minute) (see Fig. 176-1) for the same indications as those that occasioned functional CBF testing. If the decision for occlusion of the internal carotid artery is reached preoperatively, permanent balloon occlusion should be performed 1 to 4 weeks before the resection to lessen thromboembolic sequelae. Placement proximally at the level of the carotid bifurcation and distally just before the ophthalmic artery minimizes late thromboembolic sequelae.67 The long-term sequelae of carotid occlusion are unknown, although xenon CT blood flow mapping after BTO correlates well with medium-term neurologic outcome.28 Direct vein graft reconstruction is indicated to allow complete tumor resection (see Fig. 176-1) in patients assigned to group 3 by quantitative CBF assessment (CBF less than 30 mL/100 g per minute), those with bilateral lesions, and young patients with benign tumors, according to Sen and Sekhar.64 Intraoperative revascularization is associated with increased operative time, graft occlusion, need for transfusion, and potential for postoperative blowout.30 Risk of graft occlusion is especially high among patients with long grafts.
Surgical Approaches
Although the biologic behavior of tumors involving the skull base may vary, certain general principles should be followed in their extirpation. It is essential, if possible, to determine preoperatively whether the lesion is benign or malignant. Small, benign lesions in the middle ear space whose borders are clearly visible through the tympanic membrane can be removed through the external auditory canal. When removing the tumor, the surgeon should clearly differentiate it from vascular anomaly, because injury to the internal carotid artery in its intratemporal portion is hazardous and may require ligation of the artery for control of bleeding. Benign tumors limited to the mesotympanum whose borders are not visible through the tympanic membrane may be approached through a combined transcanal and facial recess or through an extended facial recess approach. Occasionally, the facial recess is narrow, necessitating removal of all or part of the bony posterior wall of the external auditory canal. Basic surgical approaches to the mastoid and mesotympanum are described in Chapters 127 and 142. Malignancies are best removed en bloc, whereas benign lesions may be extirpated piecemeal, although depending on biology, resection may require establishing a cuff of normal tissue. Surgical approaches to lesions deeper within the temporal bone should be chosen either to provide adequate exposure for complete excision or to allow easy access to an exteriorized cavity, to preserve useful residual hearing if possible, to preserve facial nerve and other cranial nerve functions when possible, to avoid injury to the brainstem and internal carotid artery, and to provide for wound closure without cerebrospinal fluid (CSF) leak.
The operative approach and intraoperative technique should maximize safe exposure and minimize morbidity. Surgeons should be well versed in all methods of exposure for various regions of the skull base. Circumferential exposure of tumor margins for benign tumors and en bloc technique for malignant lesions, minimization of blood loss by preoperative selective embolization and intraoperative selective vessel ligation, proximal and distal control of the internal carotid artery around the tumor, and preservation of cranial nerves facilitated with intraoperative monitoring by electromyography (EMG) (see Chapter 178) whenever necessary are essential to a successful outcome.
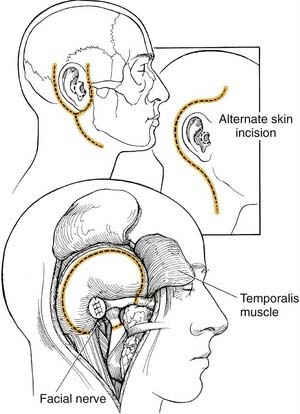
Lateral (Partial) Temporal Bone Resection
A lateral temporal bone resection68–71 removes en bloc the entire osseous and cartilaginous portions of the external auditory canal and tympanic membrane using the extended facial recess approach (Fig. 176-3). This procedure is indicated for patients with malignant neoplasms localized to the osseous canal with no encroachment on the medial mesotympanum. Parotidectomy, neck dissection, and mandibular condylectomy are performed as adjunctive surgical procedures if indicated by preoperative radiologic or intraoperative findings. Intraoperative facial nerve monitoring is useful in this procedure.
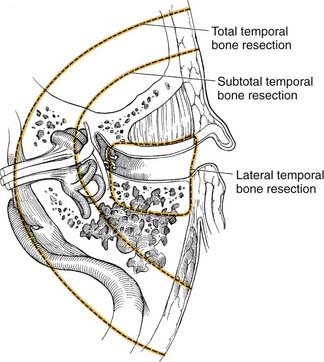
Figure 176-3. Margins of resection for lateral temporal bone, subtotal temporal bone, and total temporal bone resections.
Incisions
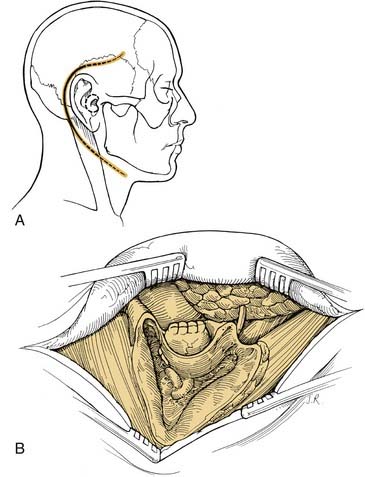
A soft tissue cylinder is designed to include the tragus and conchal bowl. The incision is taken vertically downward, posterior and superior to the temporal bone. The external auditory canal is sutured shut. A postauricular incision is created approximately a fingerbreadth behind the postauricular sulcus (Fig. 176-4A). It may be extended inferiorly into a neck crease for parotidectomy as required. Retractors then are placed.
Mastoidectomy and Facial Recess Approach
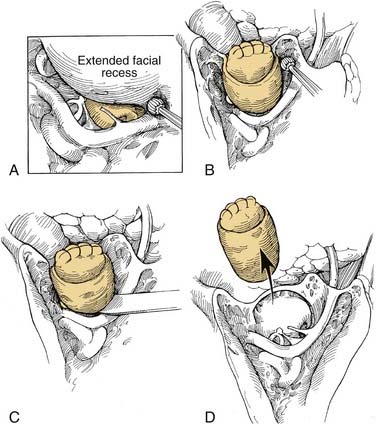
A complete mastoidectomy is performed. The superior mastoid dissection is carried to the root of the zygoma, with care taken not to penetrate the osseous portion of the external auditory canal or the tegmen. Epitympanic dissection in an anterior direction should extend into the temporomandibular joint, as depicted in Figure 176-4B. An extended facial recess approach is then performed by “eggshelling” the vertical segment of the facial nerve from the second genu to the stylomastoid foramen (Fig. 176-5A). The incudostapedial joint is separated, and the pillar of bone between the facial recess approach and the fossa incudis also is removed. The tensor tympani tendon is cut with Bellucci scissors. Hypotympanic dissection is performed by extending the facial recess approach through the chorda tympani nerve. Drilling with either a diamond or cutting burr is continued along the inferior aspect of the hypotympanum medial to the annulus and lateral to the jugular bulb and carotid canal until soft tissue is reached, with care taken not to injure the intraparotid facial nerve (see Fig. 176-5B).
Removal of Specimen
Continuity between the hypotympanic dissection and the anterior epitympanic dissection is established by drilling along the anterior mesotympanum into the temporomandibular joint or by fracturing with a curved osteotome (see Fig. 176-5C). Either cautery or sharp dissection is used to separate the bony anterior canal wall from soft tissue of the temporomandibular joint and the superficial lobe of the parotid. The specimen then is sent en bloc for histopathological examination (see Fig. 176-5D).
Closure
If ancillary procedures such as parotidectomy or mandibular resection are not performed and if postoperative radiotherapy is not anticipated, creation of a large mastoid cavity is acceptable. The eustachian tube orifice should be obliterated with temporalis fascia as in a radical mastoidectomy. The mesotympanum and facial ridge may be covered with temporalis fascia graft to facilitate healing. Further conchal bowl cartilage may need to be removed to create a large meatoplasty. A split-thickness skin graft is then sutured to the edges of the conchal bowl and anterior facial skin or tragal skin and allowed to drape into the mastoid cavity and anterior cavity soft tissue. The skin graft then can be tacked into place with absorbable sutures along the soft tissue areas. Use of a graft will minimize postoperative meatal stenosis.72 Absorbable gelatin then can be placed on the medial aspect of the mastoid cavity, followed by packing with gauze strips.
If postoperative irradiation is anticipated, the mastoid cavity should be obliterated to avoid osteoradionecrosis.73 This may be achieved with use of abdominal fat and rotation of a temporalis graft to cover the defect. Alternately, a superiorly based sternocleidomastoid muscle flap based on the occipital artery may be rotated by incising the sternocleidomastoid muscle at the clavicle. A skin graft then is sutured to the conchal bowl margins and tacked down to underlying muscle.
Subtotal Temporal Bone and Total Temporal Bone Resections
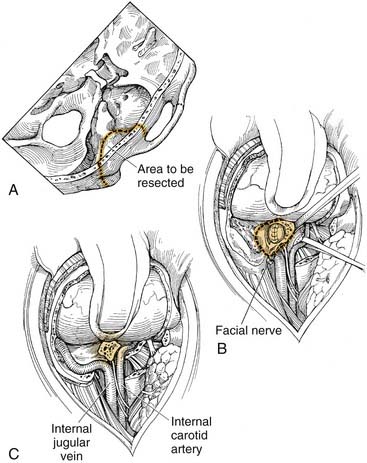
A subtotal temporal bone resection70,74–84 essentially allows en bloc resection of the medial surfaces of the mesotympanum, leaving the air cells of the petrous apex and portions of the bony labyrinth (see Fig. 176-3). This type of resection is indicated for patients with malignancies involving the middle ear.
A total temporal bone resection involves an en bloc resection of the temporal bone, including the petrous apex and the sigmoid sinus (see Fig. 176-3). The petrous internal carotid artery may be included in this resection. Total temporal bone resection is of questionable added benefit if a high-grade tumor involves the petrous apex or internal carotid artery or extends outside of the petrous bone intradurally or toward the cavernous sinus. Total resection may be indicated for low-grade malignancies.83
Incision
A large C-shaped postauricular incision or Y-shaped incision can be designed with a central island removed (Fig. 176-6). Alternately, a preauricular incision may be used for cases with involvement of the external auditory canal. In performing the incision, care should be taken to preserve the greater auricular nerve, because this nerve may be used for facial nerve interposition grafting. Also, care should be taken in elevating the flap off the zygoma, so as to not injure the peripheral frontal branch of the facial nerve.
Subtotal Temporal Bone Resection
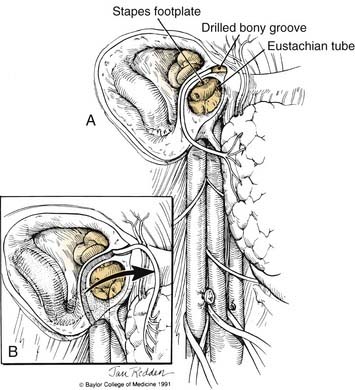
The vertical petrous portion of the internal carotid artery is identified (Fig. 176-7). This resection is performed by drilling with diamond burrs through the glenoid fossa and across the eustachian tube orifice. Mobilization of the carotid is just distal to the first genu. The distal eustachian tube orifice should be either sutured or closed with autologous tissue.
Proximal Facial Nerve Management
Although an osteotomy may be performed at this point, with sectioning of the proximal facial nerve during an often bloody dissection, it probably would cause further stretch injury to the facial nerve. If grafting of the facial nerve is desired, further stretch injury is best avoided. The facial nerve may be delineated from the labyrinthine segment to the internal auditory canal as with routine middle cranial fossa nerve decompression or may be identified by a translabyrinthine approach (see Fig. 176-7A). Nonetheless, clean sectioning of the facial nerve facilitates interposition grafting and later facial nerve function.
Osteotomy
A groove may be drilled along the floor of the middle fossa connecting the glenoid fossa area, lateral internal auditory canal, and posterosuperior mastoid. An osteotome is inserted into the carotid canal just distal to the first genu and pointed toward the internal auditory canal (see Fig. 176-7B). The temporal bone specimen is brought out (see Fig. 176-7C), which often will result in inferior petrosal sinus bleeding, requiring intraluminal packing of the inferior petrosal sinus with oxidized cellulose or control of the jugular bulb and sigmoid sinus.
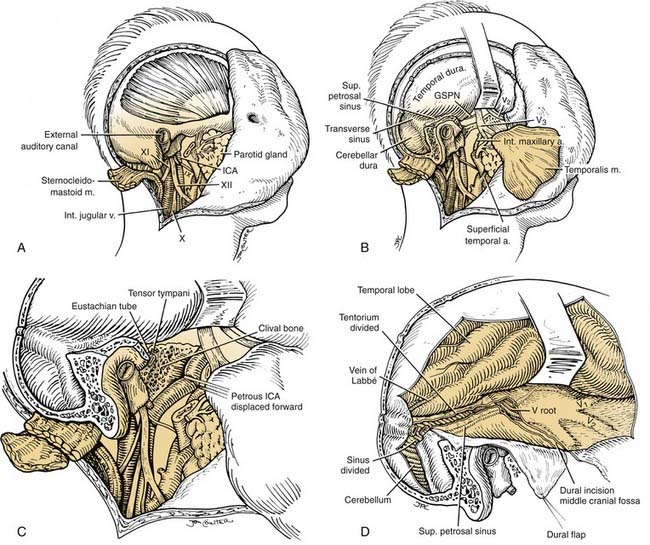
Total Temporal Bone Resection
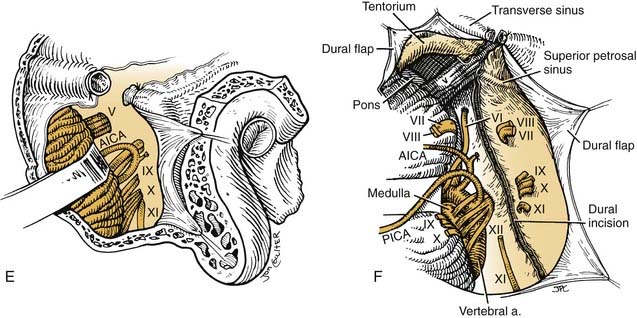
The internal carotid artery is dissected to the foramen lacerum (Fig. 176-8). The vessel may have been permanently occluded perioperatively in accordance with the surgical plan. The internal jugular vein is ligated and cut. A suboccipital craniotomy is performed, extending over the transverse sinus and lateral posterior fossa. The dura is divided over the cerebellum, and the transverse sinus is ligated between the vein of Labbé and the superior petrosal sinus. The tentorium is divided from the lateral sinus across the superior petrosal sinus to the inferior edge of V3 with continued dural incision along the floor of the middle cranial fossa laterally. Cranial nerves VII and VIII and the labyrinthine artery are divided at the internal carotid artery. Cranial nerves IX, X, and XI are divided intradurally. The inferior dural incision along the clivus extends from Meckel’s cave caudal to the lower cranial nerve to the postsigmoid dural edge. An osteotome is inserted just posterior to the foramen ovale and directed slightly posteriorly to avoid entering the foramen lacerum. This will release the temporal bone specimen, with the occasional need for osteotomy just above the jugular bulb to separate the petro-occipital synchondrosis.84,85 Alternatively, a groove is drilled under direct vision through the posterior fossa to connect to an externally drilled groove posterior to the petrous internal carotid artery.83
Infratemporal Fossa Approaches
Attacking various problems of the skull base through a lateral approach transgresses the temporal bone and infratemporal fossa. The postauricular infratemporal fossa approaches as described by Fisch61,86–88 and the preauricular89–92 approaches offer the best access to the jugular bulb, internal carotid artery, petrous apex, clivus, pterygomaxillary fossa, and nasopharynx. Proponents of these approaches have written extensively on the subject, and the techniques described here have been adapted from their work. Anatomic maneuvers may be added as needed to facilitate tumor exposure and extirpation.93,94
Postauricular Approaches to the Infratemporal Fossa
Fisch61 has been the primary innovator of these techniques and should be credited for his work. He has divided these techniques into three basic approaches: Type A dissection entails radical mastoidectomy, anterior transposition of the facial nerve, exploration of the posterior infratemporal fossa, and cervical dissection permitting access to the jugular bulb, vertical petrous carotid, and posterior infratemporal fossa. Type B dissection explores the petrous apex, clivus, and superior infratemporal fossa. Type C allows exposure of the nasopharynx, peritubal space, rostral clivus, parasellar area, pterygopalatine fossa, and anterosuperior infratemporal fossa.
Incisions and Skin Flaps
The planned incision should allow for further extensions without devascularization of the elevated skin flap (Fig. 176-9A). Cervical exposure to a variable extent and anterior extension for dissection toward the clivus and parasellar area should be permitted. Keeping the inferior limb of the incision in a posterior location over the mastoid tip allows extension into the cervical area for dissection while protecting the marginal mandibular nerve. Similarly, extending the incision anterosuperiorly into the frontal area permits elevation over the zygoma to the orbital rim.
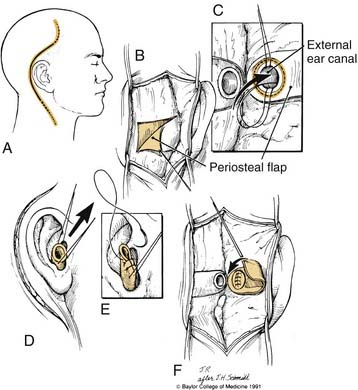
The flap should be elevated superficially to the temporalis and postauricular muscles, leaving a periosteal flap based on the external auditory canal (see Fig. 176-9B). The canal is transected with care to protect the branches of facial nerve anterior to the canal.
Closure of the External Auditory Canal
The external auditory canal is closed in a watertight blind sac. The cartilaginous canal skin is undermined to approximately the level of the conchal bowl with tenotomy scissors. The cartilaginous canal skin then is everted and sutured with absorbable sutures and reinforced medially with the periosteal flap elevated off the mastoid cortex (see Fig. 176-9C to F).96
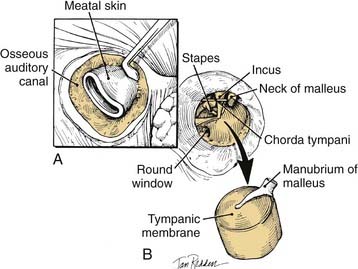
Removal of External Auditory Canal Wall Skin and Tympanic Membrane
The skin of the osseous portion of the external auditory canal wall is elevated circumferentially down to the annulus. With an operating microscope, the tympanic annulus is elevated, the incudostapedial joint is separated, the tensor tympani tendon is cut, and the neck of the malleus is nipped, allowing total removal of the canal wall skin, tympanic membrane, and attached manubrium (Fig. 176-10).
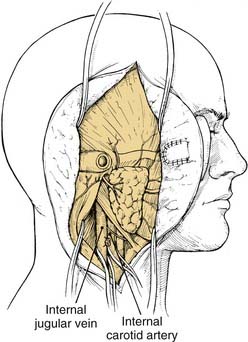
Cervical Dissection
Cervical dissection is done as needed to expose the inferior margins of the tumor. This part of the procedure is not required in most type B and type C approaches, except to gain cervical control of the vascular structures if needed. The greater auricular nerve should be sectioned as distally as possible in the parotid for potential use as an interposition graft if needed. Major structures, including the common, external, and internal carotid arteries, the internal jugular vein, and cranial nerves IX to XII, are identified (Fig. 176-11). Division of the posterior belly of the digastric near the mastoid facilitates identification of these structures up to their entrance in the skull base. Ligation of the occipital artery and ascending pharyngeal artery is indicated when these vessels prove to be the vascular supply to the tumor on arteriography. Transection of the glossopharyngeal nerve often is necessary to follow the carotid artery into the skull base. Vascular loops are placed around the internal carotid artery and jugular vein for quick identification and control in the event of bleeding (see Fig. 176-11).
Extratemporal Facial Nerve Dissection
The extratemporal facial nerve may be located deep to the midpoint of a line between the tragal pointer cartilage and the mastoid tip. After release of parotid tissue from the sternocleidomastoid muscle, the tragal cartilage is dissected medially, identifying the facial nerve just inferior to the pointer. The facial nerve is dissected out to tertiary branches by cutting overlying the parotid gland and freeing it from the underlying parotid tissues. This exposure is required for anterior transposition in the type A approach (see Fig. 176-11). In the type B and type C approaches, facial nerve transposition is not required; only the frontal branch is followed distally to allow its preservation when the zygoma is transected. The parotid gland is bluntly dissected off the masseteric fascia to reduce traction on the facial nerve when the mandible is retracted.
Radical Mastoidectomy
The radical mastoidectomy removes the air cell tracts lateral and adjacent to the otic capsule (Fig. 176-12A). Exenteration of all tracts is important to prevent long-term complications after cavity obliteration.96 The stapes suprastructure is removed to prevent inner ear trauma. The facial nerve is skeletonized in preparation for transposition. The eustachian tube is obliterated with bone wax impregnated with bone dust and a muscle plug. Removal of the external auditory canal, tympanic membrane, ossicles, and air cells of the temporal bone lateral to the otic capsule constitute the radical mastoidectomy.
Type A Approach
FACIAL NERVE TRANSPOSITION
The facial nerve should be skeletonized and freed of its bony canal 180 degrees circumferentially from the geniculate ganglion distal to the stylomastoid foramen. The horizontal semicircular canal is in jeopardy at the second genu, and bone should be removed only on the tympanic side of the facial nerve. The air cells of the mastoid tip are exenterated lateral to the digastric ridge; the cortical shell is removed with a rongeur after incising the attached digastric muscle. At the stylomastoid foramen, the facial nerve is densely adherent to the surrounding fibrous tissue, and these tissues are elevated as a unit to prevent devascularization and stretch injury to the nerve. A new bony canal is drilled in the anterior wall of the epitympanum to receive the nerve. The facial nerve is elevated carefully from the geniculate ganglion to the pes anserinus and is transposed into the new groove and secured within the parotid soft tissue (see Fig. 176-12B).
Intraoperative facial nerve monitoring with EMG is of great benefit during transposition in preventing injury (see Chapter 178).80 Immediate feedback is available to the surgeon during manual manipulations, helping prevent direct trauma and stretch injury, the latter being the most common reason for postoperative dysfunction. With feedback monitoring, the surgeon can remove bone over the facial nerve more aggressively.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree