CHAPTER 161 Central Neural Auditory Prosthesis
Biocompatibility
Direct stimulation of the central nervous system entails unique safety concerns. The mechanisms of tissue damage resulting from the direct injection of current into brain parenchyma have been extensively studied.1–3 Charge transfer from an electrode to biologic tissue depends on two independent electrochemical mechanisms, as follows:
Safety thresholds of neuroprosthetic stimulation depend on several factors. Yuen and colleagues4 have shown that the extent of neural injury with contemporary electroprosthetic materials producing direct stimulation closely correlates with the charge density per phase. Charge density directly relates to stimulation intensity, stimulation duration, and the effective area of the electrode terminus. The effective area of an electroprosthesis can differ from its geometric area. Brummer and Turner5 measured the real surface area and suggested that surface roughness factors would range from 1.4 to 30.
The threshold for damage to the cerebral cortex found by Agnew and associates6 was 320 µA, with a corresponding charge density of 3200 µcoul/cm2/phase. Niparko and colleagues7 found that the threshold for tissue damage within the cochlear nucleus (with a penetrating electrode) was 150 µA, a current that corresponded with a charge density of 600 µcoul/cm2/phase. Stimulation at intensities of 150 µA and 200 µA (approximately 600 µcoul/cm2/phase and 800 µcoul/cm2/phase) produced significant tissue response at the site of the electrode terminus with neuronal loss, fiber necrosis, and reactive cells present. The injury threshold was found to exceed the threshold for functional activation, however, by a factor of at least five across all animals studied.
History of Clinical Approaches
The concept of auditory brainstem implantation was introduced by House and Hitselberger at the House Ear Institute (HEI, Los Angeles, CA). In 1979, they implanted a two-ball electrode in a woman with NF2 undergoing acoustic neuroma removal. This first ABI was stimulated with a modified hearing aid, and the patient received useful auditory sensations.8 Nonauditory stimulus occurred, which limited use of the electrode. Subsequently, this original electrode was replaced with a specially designed two-electrode surface array, which used a Dacron mesh carrier. This electrode was attached to a percutaneous pedestal connector and was stimulated by a modified 3M-House cochlear implant speech processor. This patient has continued to achieve acoustic percepts with electric stimulation, and has continued to use the device successfully 10 to 12 hours a day.9 Between 1984 and 1992, 24 NF2 patients were implanted at HEI, with the single-channel, initially two-electrode, and subsequently three-electrode, ABI under an investigational device exemption from the U.S. Food and Drug Administration (FDA).
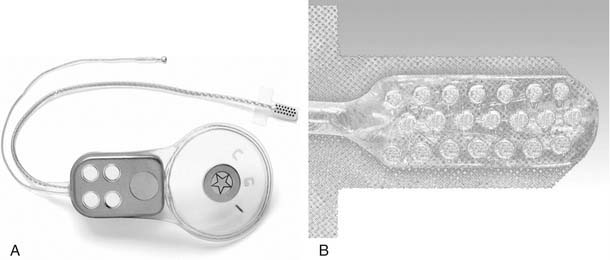
The first fully implantable ABI was manufactured by Cochlear Pty Ltd, based on the CI 22 mini–cochlear implant. The electrode array was modified to an eight-electrode pad, developed by HEI in collaboration with Cochlear Ltd and Huntington Medical Research Institute (Pasadena, CA). After initial trials at HEI, a multicenter clinical trial, based in North America, was undertaken. In Europe, a 21-electrode array was also developed by Cochlear Ltd and used with the same receiver stimulator.10–13 Clinical experience from these trials led to the best features of both electrode arrays being combined to create the current Nucleus ABI electrode, a 21-electrode array connected to the CI 24M receiver stimulator (Fig. 161-1). The system functions similar to a cochlear implant with a fully implantable receiver stimulator package and an externally worn speech processor and transmitting coil. The receiver stimulator package has a removable magnet because many recipients require repeated postoperative cranial magnetic resonance imaging examinations.
Indications
An ABI can be successfully placed as a second-stage procedure after previous vestibular schwannoma surgery. There may be an advantage in delayed implantation if, at the first surgery, there is a large tumor deforming the brainstem and distorting the lateral recess anatomy and position. There is potential, however, for fibrosis and loss of brainstem landmarks occurring after initial tumor surgery that would make placement of the ABI difficult at a second procedure. Prior stereotactic radiotherapy, particularly by gamma knife, is a relative contraindication to ABI placement because of the potential for radiation necrosis of the cochlear nucleus region or fibrous tissue preventing proper device placement. There have been reports where this has been a problem, and where successful ABI use after radiotherapy was achieved.14,15 The use of ABIs in patients without NF2 is discussed later in this chapter.
Anatomy and Surgical Approaches
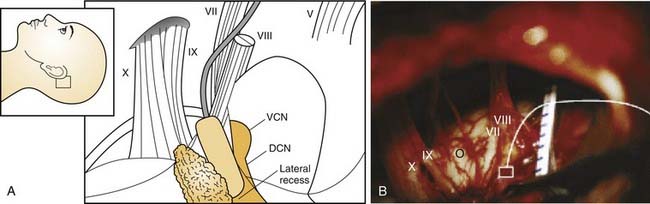
Much of the early ABI research focused on gaining a better understanding of the three-dimensional anatomy of the cochlear nerve root, foramen of Luschka, and cochlear nucleus, and potential surgical approaches.16–23 The cochlear nucleus comprises ventral and dorsal components, situated in the pons where the eighth cranial nerve enters the brainstem at the pontomedullary junction, medial to the cerebellar peduncle. The surfaces of the ventral and dorsal cochlear nuclei are exposed in the anterosuperior aspect of the lateral recess of the fourth ventricle. The ABI electrode is placed into this recess. The foramen of Luschka is the lateral opening of the recess and contains choroid plexus.
The translabyrinthine approach has been favored by most surgeons for ABI placement in NF2 patients undergoing tumor removal.10 Some centers have used a retrosigmoid approach for recipients with NF2 and without NF2.24 The translabyrinthine approach provides a more lateral view of the brainstem and better view into the foramen of Luschka. Possibly this better view assists in atraumatic electrode array placement; however, translabyrinthine and retrosigmoid approaches can be used effectively. Cerebellar retraction is reduced or avoided by the translabyrinthine approach. If a retrosigmoid or lateral suboccipital approach is used, it is important that the exposure is made as far forward and inferiorly as possible, skeletonizing and retracting the sigmoid sinus anteriorly, to allow a low anterior approach to give the most direct access to the region of the foramen, with the least cerebellar retraction.
Positioning the electrode array within the lateral recess is crucial to successful auditory stimulation. If the array is too lateral, stimulation of the glossopharyngeal and facial nerves or other lower cranial nerves and cerebellar flocculus can occur. If the array is too deep, the medial electrode contacts would lie within the fourth ventricle and be ineffective. Similarly, caudal or cranial rotation of the electrode array can significantly affect position over the cochlear nucleus. Figure 161-2 shows a schematic representation of the electrode array position.