CHAPTER 111 Total Laryngectomy and Laryngopharyngectomy
Total Laryngectomy
Historical Development
Although Patrick Watson of Edinburgh often is credited with performing the first total laryngectomy in 1866, there is no recorded proof that the operation actually was done by him. Research into Watson’s own article1 apparently answers this question; Watson stated he performed only a tracheotomy while the patient was alive and then did a postmortem laryngectomy of the syphilitic larynx. Billroth of Vienna, on December 31, 1873, carried out the first total laryngectomy for a patient with laryngeal cancer. One month earlier, he had performed a vertical cricothyrotomy and local intralaryngeal excision of this patient’s lesion. Gross pathologic recurrence necessitated further radical ablation,2 but the total laryngectomy was punctuated by considerable bleeding, coughing, and arousal from the anesthetic. The patient died 7 months after surgery,3 but Bottini of Turin in 1875 performed a total laryngectomy on a patient who survived for 10 years. In 1880 the operative or early postoperative mortality rates were approximately 50%.
This high mortality rate led Gluck of Germany to develop a two-stage procedure in which the tracheal separation was performed first, such that a healed tracheocutaneous stoma was present when the laryngectomy and pharyngeal closure were performed 2 weeks later. In the 1890s, with his pupil Sorenson, he then developed a successful single-stage operation, similar to contemporary techniques, in which the larynx was removed from above downward. Solis-Cohen, advancing from partial laryngectomies in the 1860s, reported at the 1892 Philadelphia County Medical Society Meeting carrying out a total laryngectomy by use of similar Gluck-Sorenson techniques, although Frederick Lange of New York apparently reported performing the first total laryngectomy in the United States in 1879.3,4 Radiation therapy was popular for treatment of patients with laryngeal cancer during the first half of the twentieth century, although with improvements in surgical and anesthetic technique and a recognition of radiation therapy’s limitations, surgery (including total laryngectomy) still maintained a prominent role in the second half of the century. In the past two decades, there has been an increased focus on organ preservation. For intermediate and advanced-stage tumors, concurrent chemotherapy and radiotherapy have become part of the standard of care,5 particularly for those patients who are in some way unsuitable for organ preservation surgery such as transoral laser or robotic excision. However, total laryngectomy as a primary treatment modality is still relevant for many patients. The attention on chemoradiation has resulted in an increase in salvage total laryngectomy and laryngopharyngectomy procedures, complicated by a greater potential for complications due to the tissue damage following chemoradiotherapy.
Indications for Total Laryngectomy
Malignant Disease
With the spectrum of organ preservation surgery that is now available,6 the need for total laryngectomy as the only surgical option for those with laryngeal cancer has decreased. Organ preservation surgery includes specific procedures for supraglottic and glottic cancer. There are two major organ preservation surgical strategies, transoral (endoscopic) and open. Transoral procedures range from the very limited endoscopic resections that most head and neck surgeons can accomplish to extensive laser procedures that require greater surgical experience and specialized instrumentation,7,8 covered in Chapter 109. Open procedures include the more conventional vertical partial laryngectomy group of procedures and horizontal supraglottic partial laryngectomy group. In the early 1990s, a group of open operations called the supracricoid partial laryngectomies became more popular. The supracricoid partial laryngectomy with cricohyoidopexy (SCPL-CHP) for selected supraglottic carcinomas and the supracricoid partial laryngectomy with cricohyoidoepiglottopexy (SCPL-CHEP) for selected glottic carcinomas have extended the spectrum of tumors that can be managed surgically while avoiding total laryngectomy.9 More information about organ preservation surgery and nonsurgical organ preservation are covered in Chapters 110 and 112. In addition to the increased number and success of open procedures for organ preservation, even in the setting of prior radiotherapy, the continually improving instrumentation and experience of endoscopic transoral resections continues to improve, which reduces the need for total laryngectomy.10–14 Organ preservation surgery can safely be used after radiotherapy but with a higher complication rate.15
Another operation that avoids total laryngectomy is the “near-total laryngectomy.”16 This procedure is not strictly an organ preservation operation because it does not maintain an airway without a tracheostome. Essentially, it can be viewed as a total laryngectomy with an epithelialized voice fistula.17 Nonsurgical treatment for patients with laryngeal malignancy also is established as primary treatment in some centers. Management includes definitive radiation alone with surgery for salvage for patients with primary lesions up to T3 or neoadjuvant chemotherapy and radiotherapy, with total laryngectomy reserved for those with unresponsive tumors. The frequently cited Veterans Affairs study18,19 randomly assigned patients with advanced laryngeal cancer to chemotherapy and radiotherapy or total laryngectomy. Although present, the increased survival in the surgical arm was not significant. Criticisms of the study prompted a head and neck intergroup trial that randomly assigned patients to three groups and compared radiation alone versus concurrent versus sequential chemotherapy with radiotherapy. This study has recently been published and demonstrates that concurrent chemotherapy with radiotherapy is superior to sequential chemotherapy followed by radiotherapy or radiation alone in terms of local control and organ preservation.20 However, there was no surgical arm to this study, thereby falling short of establishing a new standard of care. Furthermore, using primary concurrent chemoradiation therapy, the 5-year results for both disease-free survival (39%) and laryngectomy-free survival (46%) show that this method leaves the majority of patients either dead or absent their larynx at 5 years.21 Therefore, this leaves much room for improvement in treatment of laryngeal cancer. Other studies have shown that if nonsurgical organ preservation is to be used, concurrent chemotherapy with hyperfractionated radiotherapy is more effective than radiotherapy alone22 and that hyperfractionated radiotherapy is more effective than standard radiation alone.23 Newer trials are investigating the impact of induction chemotherapy to assess tumor response. A complete (or near complete) response to chemotherapy then selects the patients for concurrent chemotherapy and radiotherapy.24 No biologic marker yet identified can reliably predict which patients are better treated surgically. This is also the topic of investigations in concurrent studies.25 Because the survival of the two treatment strategies is equivalent, quality-of-life issues are of paramount importance. Recently, a study demonstrated that in the intergroup trial, patients who had total laryngectomy after radiotherapy had a high complication rate including a risk of pharyngocutaneous fistula of up to 30%, highest in the concurrent chemotherapy group.26 Studies that have evaluated the quality of life after nonsurgical management of advanced laryngeal cancer do suggest that organ preservation is associated with improved quality of life.27,28 Although the details of such studies are beyond the scope of this chapter, the results indicate that more extensive analysis of the quality of life is important. These studies are difficult to accomplish because many patients are not available after the study, and the quality of life of the survivors might be significantly different than those of the patients who have died, particularly if they died of local complications related to uncontrolled primary site disease. Total laryngectomy still remains a viable option as the primary surgery for advanced cancer or as salvage surgery after radiotherapy and finally for a group of less common conditions. Overlap between these indications occurs. For example, a patient with radionecrosis of the larynx may be found to have residual tumor on permanent sections and, as such, the surgery was salvage in hindsight. Similarly, a tumor that fails radiotherapy may also be causing chronic aspiration and may be too large to allow for any sort of organ preservation surgery. This sort of patient has three (or more) of the following indications for undergoing the procedure.
Indications
Patient Selection and Workup
The following patient requirements should be met before a total laryngectomy is performed.
Surgical Technique (see Key Indicator Video on website)
Resection
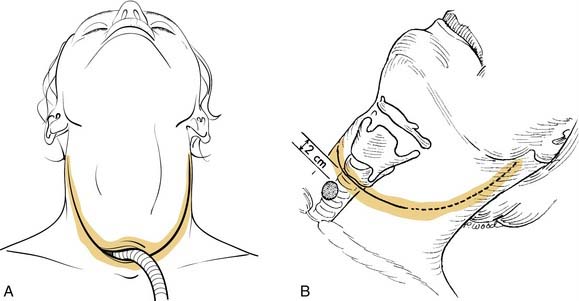
Proper patient positioning provides access to the anterior part of the neck for the surgeon and the assistant. Positioning is best achieved by placing the patient on a table fitted with a head holder, allowing the head to be cantilevered out but remain well supported. This also assists bilateral neck dissection, which is often performed “in-continuity” and allows surgeons to position themselves all the way around the head. The table is turned 180 degrees from the anesthesiology team to further assist access to the patient. Before the operation day, airway management is planned with the anesthesiologist so that an agreement is reached regarding timing of tracheotomy and intubation. In the unobstructed larynx the anesthesiologist may pass an orotracheal tube with anesthetic induction, which may be removed at subsequent tracheotomy or left in situ until tracheal transection is performed at the end of the laryngectomy. With an obstructed airway or in a case in which intubation may displace malignant tissue into the lower airway, a preliminary tracheotomy with the patient under local anesthesia is performed. The tracheotomy skin incision is made at the intended site of the final stoma. The stoma can be placed in the line of the incision (long flap) or 2 to 3 cm inferior to the incision (Fig. 111-1, A and B). The advantage of the former is that creation of the stoma is more reliable and avoids a bipedicled bridge of skin between the flap incision and the tracheostomy incision, which can result in stenosis of the stoma. The advantage of the latter is that in the event of pharyngocutaneous fistula, which requires diversion, the stoma is less likely to be involved in the resulting wound. The latter consideration is perhaps more important in previously irradiated patients because the risk of fistula in primary total laryngectomy is low. This configuration also avoids the technical nuances of bilateral, three-point closures at the stoma necessitated by exteriorizing the lower cut tracheal end through the laryngectomy skin incision. In the long flap approach, the three-point closure is performed after the inferior aspect of the stoma is completed, elongating it with the technique shown in Fig. 111-6. This is somewhat more difficult to accomplish with the stoma created 2 to 3 cm into the inferior flap because the trachea is not anchored to the lateral aspect of the dissection. Ultimately, the comfort level of the surgeon determines the choice of incision.
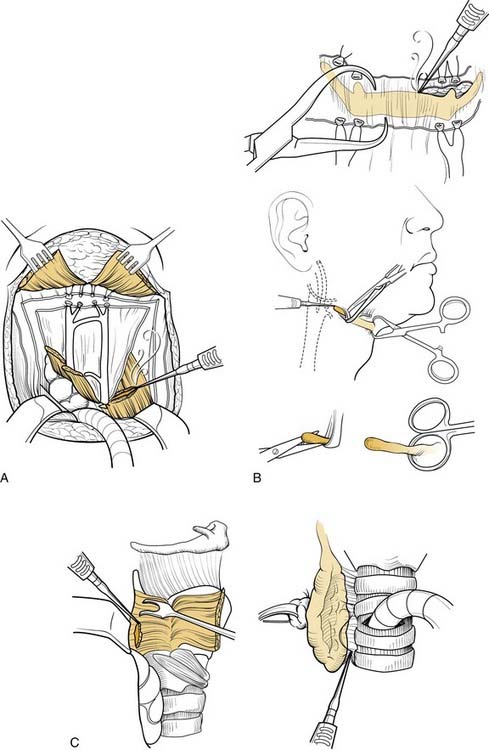
For access to the larynx itself, a curved, horizontal neck skin incision is preferred because of its minimal intersection with the pharyngeal closure and its potential for extension laterally into a neck dissection incision. Once the incision is deepened through, but not beyond, the platysma, flaps are elevated superiorly and inferiorly in the subplatysmal plane until there is exposure above to the upper border of the hyoid bone and below to the cervical trachea. The anterior jugular veins and the prelaryngeal Delphian node are left undisturbed on the specimen, as are the strap muscles. The sternocleidomastoid muscle then is identified along its anterior border on each side. The investing layer of cervical fascia is incised longitudinally from the hyoid above to the clavicle below. The omohyoid then is divided, which allows entry to the loose areolar compartment bounded laterally by the sternomastoid muscle and carotid sheath and medially by the pharynx and larynx contained in the visceral compartment of the neck. Appropriate neck dissections are performed. The extent of neck dissection is beyond the scope of this chapter, but a few points merit mention. For supraglottic carcinoma, the minimal neck dissection to be performed is selective neck dissection, stations II and III bilaterally. It is extended to level IV and possibly IB and V if lymphadenopathy indicates the need. For advanced glottic carcinoma with supraglottic involvement, the same procedure is recommended. Unilateral (ipsilateral) neck dissection can be considered for T3 glottic carcinoma. For patients who have previously been irradiated, evidence still supports selective neck dissection.30,31 In general, both neck dissections can be pedicled at the thyrohyoid membrane area and the resection can easily be done en bloc.
The strap muscles then are divided inferiorly from their sternal origins and elevated to expose the thyroid gland (Fig. 111-2, A). Controversy exists with regard to thyroidectomy as part of a total laryngectomy. Studies note that involvement of the thyroid gland is rare.32 However, when there is nodal disease in the jugular chain from glottic or subglottic cancer, the thyroid is at high risk because of the paratracheal and parapharyngeal lymphatics associated with the thyroid. Also, in patients who have invasion of the thyroid or cricoid cartilage in close proximity to the thyroid gland, the gland may become directly involved by the tumor. The risk to the thyroid is highest with transglottic cancers that extend more than 1 cm subglottic.32–34 In some cases, both lobes of the thyroid should be removed because of these risk factors. When the lobe of the thyroid is to be removed, the superior and inferior thyroid vascular pedicles are therefore ligated and divided, as is the middle thyroid vein. The lobe(s) to be preserved then is (are) dissected off the laryngotracheal skeleton from medial to lateral, thereby preserving blood supply to the remaining thyroid and parathyroid parenchyma by way of inferior thyroid vessels (the superior thyroid artery can be preserved also if oncologically sound, but this is not necessary). An effort should be made to preserve the vascularity to the parathyroid glands, but if a parathyroid gland is noted that is poorly vascularized after the procedure, this can be reimplanted into the neck musculature as is done for parathyroidectomy in thyroid cancer. Next, after dividing and ligating the upper and lower extent of the anterior jugular veins, the superior aspect of the hyoid bone is skeletonized by detaching the mylohyoid, geniohyoid, digastric sling, and hyoglossus in sequence from medial to lateral (see Fig. 111-2, B). “Cold” knife dissection should be used lateral to the lesser cornu, avoiding excess electrical stimulation or direct damage to the hypoglossal nerve. Caution is advised for supraglottic cancers that involve the aryepiglottic fold, pyriform sinus, or vallecula because too much skeletonization of the hyoid bone and thyrohyoid membrane area may put the surgeon in close proximity to the deep extent of the tumor, which might result in a close or positive margin histologically. This is not a concern with completely endolaryngeal cancers. The sternohyoid and thyrohyoid muscle attachments on the lower border of the hyoid bone remain undisturbed. Further laryngeal cartilage skeletonization now is performed if the tumor does not extend outside the pyriform fossa. The posterior border of the thyroid cartilage lamina is rotated anteriorly by upward traction, allowing sharp release of the constrictor muscles from the inferior to the superior cornu (see Fig. 111-2, C). Above the superior cornu, the laryngeal branch of the superior thyroid artery should be identified, ligated, and divided before it penetrates the thyrohyoid membrane.
The pharyngotomy incisions and definitive laryngeal removal now are performed. To avoid contact with the neoplasm or cutting through its submucosal extensions, the pharynx is entered contralateral to the tumor. If superior extension to the tongue base is present, lateral pharyngotomy behind the thyroid cartilage is performed, and by use of a headlamp, the extent of tumor is inspected. A safe 2-cm margin of normal-looking mucosa then is preserved with further cuts from below, progressing superiorly behind the thyrohyoid membrane, around the hyoid bone, and then transversely across the vallecula or tongue base. By contrast, if the disease is confined below the level of the hyoid, entry by way of the vallecula is feasible with a direct anteroposterior approach (Fig. 111-3) in the horizontal plane of the upper hyoid border. Strict maintenance of this plane, avoiding excessive inferior traction on the hyoid itself, precludes violation of the pre-epiglottic space. Once the mucosa is breached, the epiglottis’ tip is identified, grasped with an Allis forceps if tumor free, and gently pulled anteriorly out of the pharyngotomy. A view of the endolarynx and pharynx now is possible to assess tumor extent and to plan appropriate mucosal cuts (see Fig. 111-2, D).
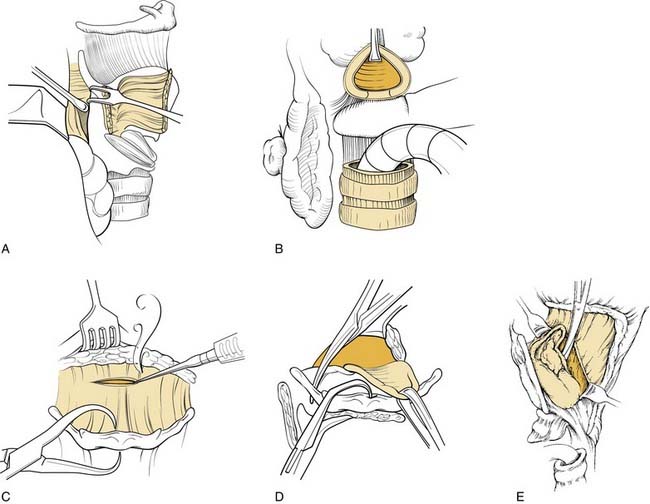
Figure 111-3. Entry into larynx. A, Use of a Freer elevator to mobilize the pyriform sinus and internal perichondrium from the thyroid cartilage. This should not be performed if the pyriform sinus is likely to be involved by the tumor. B, The trachea is transected, and the ligamentous attachments are divided permitting dissection of the trachea away from the upper esophagus up to the level of the posterior cricoarytenoid musculature. This cut is beveled as shown in Figure 111-6. This step is delayed if there is concern about significant subglottic extension. C, Dissection follows the hyoepiglottic ligament to the epiglottis and vallecula to avoid entry into the pre-epiglottic space. D, If clinically uninvolved, the vallecula is entered on the nontumor side, and if it is not involved, the tip of the epiglottis is grasped. E, The pharyngoepiglottic fold cuts are extended. If uninvolved, the previously preserved pyriform sinus mucosa is preserved by transecting the mucosa close to the aryepiglottic fold. This is accomplished by placing one scissor blade in the lumen and the other between the previously released internal perichondrium and the thyroid cartilage. This leaves the larynx pedicled only on the mucosa of the anterior esophageal inlet, which can be transected under direct vision preserving as much mucosa as is oncologically sound. If the trachea was not previously transected, it is done at this time to release the specimen.
With a Mayo scissors, bilateral, inferiorly direct cuts are made, releasing the lateral pharynx from the larynx. The inside scissors blade is on mucosa, and the outside blade is on constrictor musculature. As these vallecula-to-pyriform sinus incisions are made, the larynx is further angled anteriorly out of the wound until it is released to the apices of the pyriform sinuses. The postcricoid mucosa is exposed and incised sharply in the transverse direction connecting the inferior extent of both lateral incisions across the lower half of the cricoid lamina. A plane of blunt dissection initially behind the posterior cricoarytenoid muscle, but from there down between the trachea and longitudinal esophageal muscle, is then opened until the desired level of tracheal transection is reached. Optimum exposure of the trachea at this stage is achieved by further lateralization of the preserved thyroid lobe and then followed by knife transection of the trachea itself. This cut is bevelled upward from anterior to posterior (see Fig. 111-6, A), with special care not to encroach on any subglottic tumor extension. If the latter is present, a 1.5- to 2-cm margin of healthy-appearing trachea should be resected in continuity with the larynx to avoid stomal recurrence.35
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree