Chapter 74 Sympathetic Ophthalmia
Introduction
Sympathetic ophthalmia, also known as sympathetic ophthalmitis and sympathetic uveitis, is a rare bilateral diffuse granulomatous uveitis that occurs a few days to several decades after penetrating accidental or surgical trauma to an eye. Both the traumatized eye, commonly referred to as the “exciting” eye, and the fellow eye, referred to as the “sympathizing” eye, are affected. Injury to and/or incarceration of uveal tissue has been a feature of nearly all cases of sympathetic ophthalmia. The clinical signs and symptoms are usually detected in the sympathizing eye within the first 3 months after trauma to the fellow eye.1,2
The notion that injury to one eye may have repercussion in the contralateral eye probably dates back to ancient times, with Hippocrates (460–370 bc), but has also been reported by Agathias, in a compilation from Constantius Cephalis (1000 ad).1,3 However it was William Mackenzie, in 1830, who provided the first comprehensive clinical description and pioneered the use of the term “sympathetic ophthalmia” to describe this entity.1,3 Ernst Fuchs, in 1905, thoroughly detailed the characteristic histopathologic features.4 The pathogenesis of sympathetic ophthalmia has, however, remained an enigma despite years of study, although the two most commonly held views were that it represented an autoimmune response or was infectious. There is now some experimental evidence implicating the development of an autoimmune delayed hypersensitivity reaction to melanocytes or tyrosinase peptide antigen as a possible pathogenetic mechanism.
Epidemiology
Sympathetic ophthalmia is a relatively rare disease, although exact figures are difficult to determine because the onset or the diagnosis, or both, are often delayed for months to years after the initial injury. In addition, definite histopathologic confirmation of the diagnosis is made in only about one-third of suspected cases, but may be established histopathologically in others not suspected clinically.2 In 1972, Liddy and Stuart5 reported an incidence of sympathetic ophthalmia of 0.19% following penetrating injuries and 0.007% following intraocular surgery. Among the general population, sympathetic ophthalmia has been estimated to affect 0.03 per 100 000 persons per year6 and possibly corresponds to 1–2% of all uveitis cases.7 Surgical procedures that may lead to sympathetic ophthalmia include cataract extraction, iridectomy, paracentesis, cyclodialysis, synechialysis, retinal detachment repair, keratectomy, vitrectomy, evisceration, laser cyclophotocoagulation, and others.2,3
Although advances in modern surgical techniques may be contributing to a lower incidence of sympathetic ophthalmia, this is probably being partially offset by more aggressive surgical management of severely traumatized eyes, which in the past would have been promptly enucleated. In fact, the epidemiology of sympathetic ophthalmia has been changing, and more cases are now being associated with intraocular surgery,6–8 in contrast to accidental penetrating trauma.3 Pars plana vitrectomy is one of the leading surgical procedures associated with sympathetic ophthalmia,9 with an estimated risk of around 0.01%;10 cases have been recently reported even after the advent of small-gauge sutureless vitrectomy surgery.11,12 For these reasons, sympathetic ophthalmia should not be considered a disappearing disease and thus should not be neglected.
Some studies have shown a male preponderance, but this is believed to be a reflection of the higher incidence of accidental trauma in males. Indeed, when only cases of surgical trauma are considered, the ratio is similar. In Winter’s series of 257 cases of sympathetic ophthalmia, there was no difference in age incidence.13 Other authors have reported relative peaks in childhood and early adult years, thought to reflect a higher incidence of accidental trauma in these ages, and an additional peak in the sixth and seventh decades, thought to represent an increased incidence of surgical procedures among persons in this age group.14
Pathogenesis
The exact cause of sympathetic ophthalmia is unknown. Clinical studies have shown that the predominant predisposing factors are accidental penetrating trauma, accounting for approximately 60–70% of cases, and penetrating surgical trauma, accounting for nearly 30%. Recent studies have pointed to an inversion of this proportion, with many cases now being associated with surgical rather than accidental trauma.6,7,9 A small percentage of cases are the result of contusion injuries with occult scleral rupture and perforating corneal ulcers. The common denominator in the overwhelming majority of cases is the presence of a penetrating injury in which wound healing is complicated by incarceration of the iris, ciliary body, or choroid.2
Historically, the pathogenesis of sympathetic ophthalmia has been suspected to be infectious. However no organisms have ever been isolated from cases of sympathetic ophthalmia, and infective agents have not induced the disease in laboratory animals. Cases associated with severe infectious keratitis in the absence of a corneal perforation are probably associated with exposure of the uveal antigens to the immune system, through a disrupted hemato-ocular barrier.15,16 Sympathetic ophthalmia may also develop in the setting of posttraumatic or postoperative infectious endophthalmitis and it is possible that such infection may potentiate the development of sympathetic ophthalmia in these cases17 and not prevent it, in contrast to what had previously been suggested.1,18,19 Most cases of sympathetic ophthalmia however develop in the absence of intraocular infection.14
Several investigators have proposed an immunologic basis for sympathetic ophthalmia, in which a T-cell-mediated autoimmune response against an antigenic protein from the uvea, particularly a uveal tyrosinase peptide, may be involved.20–24 Marak25,26 and Wong and colleagues20 demonstrated enhanced transformation of peripheral lymphocytes from patients with histologically confirmed sympathetic ophthalmia when exposed to homologous uveal-retinal extracts in tissue culture, suggesting that these patients have lymphocytes that are sensitized to some component(s) of uveal-retinal antigen. When antigens extracted from the retina are injected into guinea pigs, intraocular inflammation similar to sympathetic ophthalmia develops in these animals.26 Some studies indeed suggest that sympathetic ophthalmia may result from altered T-cell response to one of the soluble proteins associated with the retinal photoreceptor membranes, particularly the retinal S antigen, or to other retinal or choroidal melanocyte antigens.21–23,26,27 However, recent studies reveal that T-cell immune response to tyrosinase peptide may play a role in the development of sympathetic ophthalmia, as in Vogt–Koyanagi–Harada disease.22,28–30
There may be a genetic predisposition to the development of sympathetic ophthalmia. Human leukocyte antigen (HLA) association has been reported in sympathetic ophthalmia, including HLA-A11, HLA-B40, HLA-DR4/DRw53, and HLA-DR4/DQw3 haplotypes.31,32 Studies from Asia and Europe found a significant correlation between HLA-DRB1*4 and HLA-DQB1*04 and the development of sympathetic ophthalmia.33–35 A similar association was also seen in patients with Vogt–Koyanagi–Harada disease. Finally, genetic background may also correlate to disease severity, either through some of those predisposing HLA haplotypes such as HLA-DRB133 or through cytokine polymorphisms such as in interleukin-10, modifying the immune response and the risk of disease recurrence.36
Immunopathology
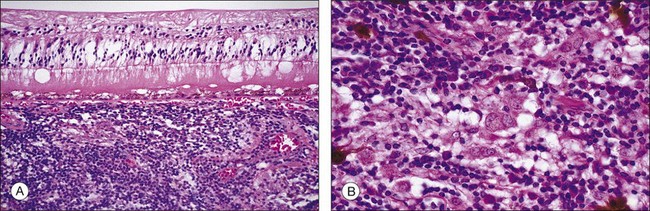
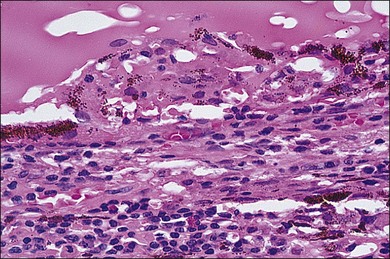
In sympathetic ophthalmia the immunopathologic alterations are similar in both the exciting and sympathizing eyes and typically consist of a diffuse granulomatous inflammation of the uveal tract, made up of lymphocytes, plasma cells, and nests of epithelioid histiocytes; pigment is often present within these epithelioid cells and also within giant cells (Fig. 74.1). In the majority of cases, the inflammatory process does not involve the choriocapillaris or the retina. Absence of necrosis is another characteristic feature. The choroid is diffusely involved and thickened by an infiltration of predominantly lymphocytes, collections of epithelioid cells, and a few giant cells; neutrophils are rarely seen. Plasma cells may be present, particularly in patients treated with corticosteroids. Eosinophils can also be found and are frequently concentrated in the inner choroid, particularly in heavily pigmented individuals. Nodular clusters of epithelioid cells containing pigment are often seen lying between the retinal pigment epithelium (RPE) and Bruch’s membrane; these appear clinically as the drusen-like, yellow-white dots known as Dalen–Fuchs nodules (Fig. 74.2).1,2,4,13,14,37,38 Infiltration of the pars plana of the ciliary body occurs early in the course of disease, and the inflammatory cells from this site may spill over into the vitreous cavity. Similar inflammatory cell infiltration in the iris may result in the clinical appearance of a thickened iris.
The retina is usually free of inflammatory infiltrates. However, few enucleated eyes of sympathetic ophthalmia show collections of mononuclear cells around the blood vessels, and occasional involvement in the areas overlying the Dalen–Fuchs nodules, and in the pars plana region. Other pathologic changes include scleral involvement with inflammatory infiltrates around the emissary veins and extension of the granulomatous process into the optic nerve and surrounding meningeal sheaths, the sites where melanocytes are also present.1,4,14,39 Some of the eyes with characteristic histologic features of sympathetic ophthalmia but also with breaks in the lens capsule may additionally reveal features of phacoanaphylaxis, with zonal granulomatous inflammation around the lens material.39 Even though typical features of sympathetic ophthalmia include nonnecrotizing granulomatous uveitis, there are cases exhibiting atypical features such as nongranulomatous choroiditis or chorioretinal adhesions with the inflammatory process involving the choriocapillaris, as seen in chronic Vogt–Koyanagi–Harada disease.13,39
Immunohistochemical studies have revealed infiltration of predominantly T lymphocytes in the uveal tract.37,40 B lymphocytes may also be present, especially in longstanding disease.41,42 Among the T lymphocytes, both helper (CD4+) and suppressor/cytotoxic (CD8+) cells have been observed,37,40 driving a Th1 response with secretion of proinflammatory cytokines such as interferon-γ and interleukin-2.43 These cells are probably recruited to the eye by the selective expression of intercellular adhesion proteins in the uveal tract (particularly some integrins),44 as well as some other molecules in macrophages and RPE cells, including chemokines, such as monocyte chemotactic protein-1 (CCL2/MCP-1) and stromal cell-derived factor-1 (CXCL12/SDF-1), and metalloproteinases, such as gelatinase-B.42 Relative preservation of the choriocapillaries in the acute phase of the disease process may be associated with secretion of anti-inflammatory cytokines by the RPE.27 A recent study has interestingly shown CD4+ T lymphocytes and melanin-laden macrophages expressing HLA-DR underneath the conjunctiva in the exciting eye, suggesting that antigen processing and presentation may initially take place at that site, further leading to activation of lymphocytes and to the granulomatous response.45
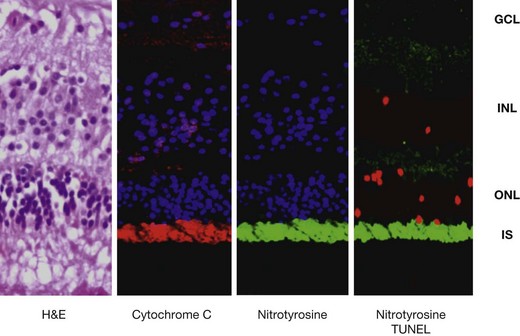
Even though the retina seems to be relatively spared in the pathologic process of sympathetic ophthalmia, tumor necrosis factor (TNF)-α-mediated mitochondrial oxidative stress has recently been localized in the outer retina of enucleated human globes with the disease (Fig. 74.3). This was associated with apoptosis of photoreceptors and probably such photoreceptor damage could be a mechanism leading to vision loss in sympathetic ophthalmia.46
Clinical findings
The clinical onset of sympathetic ophthalmia is typically heralded by the development of an apparently mild intraocular inflammation in the sympathizing eye and worsening of inflammation in the exciting eye. The interval between the time of injury and the onset of inflammation in the sympathizing eye has been reported to be as short as 5 days and as long as 66 years after trauma.1,3,47 In general, however, sympathetic ophthalmia rarely develops sooner than 2 weeks after trauma, with 80% of cases occurring within 3 months and 90% within 1 year of the penetrating injury.2,7,9 The peak incidence occurs between 4 and 8 weeks after accidental trauma, while cases following surgical trauma may have a more delayed onset.48
Symptoms in the sympathizing eye include mild pain, photophobia, and increased lacrimation, blurring of vision, visual fatigue, or even paresis of accommodation. The exciting eye may have a decrease in vision and an increase in photophobia. Moreover, both eyes may show ciliary injection and a partially dilated and poorly responsive pupil.1,2,8,9
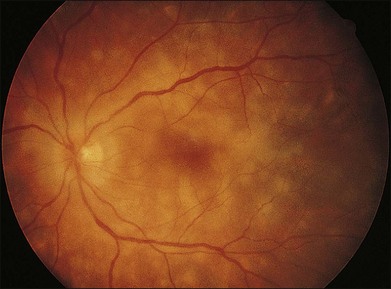
The clinical signs are variable and can be either insidious or fairly rapid in onset. Anterior-segment changes are those of an anterior uveitis, with ciliary injection, keratic precipitates, flare, and inflammatory cells in the anterior chamber. Thickening of the iris and even iris nodules may also be seen and posterior synechiae are common. Posterior-segment findings in sympathetic ophthalmia include inflammatory cell infiltration of the vitreous, hyperemia and edema of the optic disc, diffuse edema and exudative detachment of the retina, as well as small yellow-white deposits beneath the RPE, so-called Dalen–Fuchs nodules.1,2,8,9,13,14 Bullous serous detachments may also be seen in the peripheral retina. Occasionally, multiple deep ill-defined yellowish lesions may be present, corresponding to choroidal granulomas (Fig. 74.4). Inflammatory scleral involvement is rarely seen clinically, but is a common finding on microscopic examination of enucleated eyes. With time, patients may develop depigmentation of the choroid leading to the so-called sunset glow fundus, as well as RPE changes, as seen in individuals with chronic Vogt–Koyanagi–Harada disease.49,50 Although less commonly than in Vogt–Koyanagi–Harada disease, extraocular involvement – including meningismus, dysacusis, vitiligo, poliosis, and alopecia – may also be present in patients with sympathetic ophthalmia.2,49,51
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree