Fig. 11.1
Anatomical relations between the recurrent laryngeal nerve (RLN) and inferior thyroid artery (ITA) The RLN may pass posterior (a), anterior (b), or superior (c), to the ITA. The RLN may also make a loop around the ITA (d), or pass posterior to the artery and give off an extralaryngeal division (e)
2.
External branch of the superior laryngeal nerve (SLN)
The external branch of the superior laryngeal nerve (SLN) innervates the cricothyroid muscle. It is important to the pitch of voice as the cricothyroid muscle is tensor of the vocal cords. In most cases, this nerve lies close to the vascular pedicle of the superior pole of the thyroid lobes descending on the fascia of the inferior pharyngeal constrictor. In some patients, the external branch of the SLN lies on the anterior surface of the thyroid lobe, making the possibility of damage during thyroidectomy even greater. In only 15 % of patients is the SLN sufficiently distant from the superior pole vessels to be protected from manipulation by the surgeon. Unfortunately, many surgeons do not even attempt to identify this nerve before ligation of the upper pole vessels of the thyroid [8].
11.2.7 The Parathyroid Glands
The parathyroid glands are small glands that secrete parathyroid hormone (PTH), the major hormone that controls serum calcium homeostasis. Usually four glands are present (in about 80 % of cases), two on each side, but 3–6 glands have been reported. Because of their small size, their delicate blood supply, and their usual anatomic position adjacent to the thyroid gland, these glands are at risk of being accidently removed, traumatized, or devascularized during thyroidectomy.
The superior parathyroid glands arise embryologically from the fourth pharyngeal pouch. They descend only slightly during embryologic development, and their position in adult life remains quite constant. This gland is usually found adjacent to the posterior surface of the middle part of the thyroid lobe, often just anterior to the RLN as it enters the larynx. The inferior parathyroid glands arise from the third pharyngeal pouch, along with the thymus; hence, they often descend with the thymus. Because they travel so far in embryologic life, they have a wide range of distribution in adults, from just beneath the mandible to the anterior mediastinum. Usually, however, these glands are found on the lateral or posterior surface of the lower part of the thyroid gland or within several centimeters of the lower thyroid pole within the thymic tongue [9].
The parathyroid glands can be recognized by their tan appearance, their small vascular pedicle, and the fact that they bleed freely when biopsy is performed, as opposed to fatty tissue with their darkening color of hematoma formation when they are traumatized. With experience, one becomes much more capable of recognizing the parathyroid glands and of differentiating them from either LNs or adipose tissue. Frozen section examination during surgery can be helpful in their identification [10].
11.3 Histology
The thyroid gland is formed of connective tissue stroma and parenchyma of endocrine cells. The connective tissue (true) capsule of the thyroid gives off multiple fibrous septa (trabeculae) passing into the gland, carrying blood vessels, nerves, and lymphatics, to form lobules. The gland is further divided into 20–40 much smaller functional subunits called follicles which store a colloid substance which functions as a hormone store. The colloid is maintained by a single layer of follicular epithelial cells sitting on a basal lamina. These follicles are surrounded by fenestrated capillaries, lymphatics, and so-called parafollicular or C cells.
Microscopically, each lobe or lobule is supplied by an intralobular artery and vein. It is made of two types of secretory cells: follicular cells that secrete T3 and T4 and, in smaller number, the parafollicular or clear cell (C cells), which secrete thyrocalcitonin. The follicles are separated from each other by a highly vascular connective tissue, and each follicle is lined with a single layer of flattened to low columnar epithelium depending on their degree of activity. The basement membrane can only be seen with the electron microscope.
Oncocytes (Hürthle cells, oxyphilic cells, Ashkenazy cells) are large follicular cells with abundant deep eosinophilic granular cytoplasm and numerous mitochondria. They are commonly seen in long-standing Graves’ disease, autoimmune thyroiditis, radiation-induced thyroiditis, follicular-derived neoplasms, and some adenomatoid goiters [11–13].
Solid cell nests (SCN) are thought to represent remnants of the ultimobranchial body, which in turn is derived from the branchial cleft pouch complex 4–5. They are not an uncommon finding in the posterolateral or posteromedial portion of the lateral lobes of the thyroid gland. They can be found in up to 28 % of glands. Histologically, epithelial cells in nests and cords are seen, and small glandular lumina containing a mucinous secretion are often present [14–16].
11.4 Physiology
The function of the thyroid gland is to synthesize, store, and secrete thyroxin (T4) and triiodothyronine (T3). Monoiodotyrosine (MIT) and diiodotyrosine (DIT) are also found in thyroid venous blood.
Inorganic iodide (I−) is absorbed from gastrointestinal tract (GIT) and actively trapped by the acinar cells of the thyroid gland via a transport mechanism that is frequently called the “I− trapping mechanism” or “I− pump.” A transporter named pendrin is located on the luminal surface of the follicular cell and is responsible for allowing passage of I− into the follicle. In the acinar cells, I− is oxidized to iodine (I2) and bound to the 3-position of tyrosine molecules with the aid of the enzyme thyroid peroxidase (TPO) to form MIT and then to the 5-position to form DIT. Two DIT molecules then undergo an oxidative condensation with the release of an alanine residue and the formation of T4. Condensation of MIT with DIT results in T3 formation. These reactions occur while the tyrosine molecules are attached to thyroglobulin. The peptide bonds between the iodinated residues and the thyroglobulin are broken by proteases in lysosomes, and so T4, T3, DIT, and MIT are liberated into the cytoplasm. The iodinated tyrosines are de-iodinated by a microsomal iodotyrosine dehalogenase. Then, T3 and T4 are released into the circulation [17].
Most circulating T3 are derived from peripheral conversion of T4, which is really a prohormone and is significantly less potent than the more metabolically active hormone T3. In the blood, the majority of circulating T3 and T4 are bound to the plasma proteins, mainly thyroxine-binding globulin (TBG) and prealbumin. It is only the “free” unbound forms of hormones that are metabolically active, and T3 is quick acting within few hours, while T4 acts more slowly from 4 to 14 days.
Production of T3 and T4 is regulated by the hypothalamic-pituitary-thyroid axis which is a multiloop feedback circuit. The production of T3 and T4 from the thyroid is stimulated directly by the thyroid-stimulating hormone (TSH), produced by the anterior pituitary. Levels of T3 and T4 are also increased indirectly by thyrotropin-releasing hormone (TRH), which is produced by the hypothalamus in response to low levels of T3/T4 and acts on the pituitary to increase TSH production. Conversely, TRH and TSH production are suppressed by high levels of T3 and T4. Additionally, TRH production is also suppressed by high levels of TSH. In this way, the circulating levels of active thyroid hormones are self-regulating in disease-free individuals [18].
The thyroid also contains parafollicular C cells, which produce the hormone calcitonin, which reduces levels of serum calcium, counteracting the actions of PTH. This is achieved by inhibiting osteoclast activity in bone, inhibiting renal resorption of calcium, and inhibiting absorption of calcium in the intestines. Blood levels of PTH are far more clinically relevant to calcium homeostasis than calcitonin, and no exogenous replacement for calcitonin is required following thyroidectomy.
11.5 Investigating the Enlarged Thyroid
11.5.1 Serological Investigations
11.5.1.1 Thyroid Function Tests
The serum level of TSH should be routinely measured, while T3 and T4 levels are required if TSH level was abnormal (Table 11.1). When hypothyroidism is confirmed, thyroid peroxidase (TPO) antibodies should be requested to check for autoimmune thyroid disease such as Hashimoto’s thyroiditis. Serum thyroglobulin (Tg) level does not help in the initial management of thyroid nodule and is not recommended. Serum Tg levels may be elevated in patients with cancer but are not diagnostic, since similar increases are seen in benign thyroid disorders. However, serum Tg is a useful test in the follow-up of patients who have undergone thyroid resection for differentiated thyroid cancer. A level >10 ng/ml is a reliable indicator of locally recurrent or metastatic disease and predicts the need for ablative dose of 131I [19, 20].
Table 11.1
Serological tests in thyroid disease
TSH | Additional testing | Diagnosis |
|---|---|---|
Normal | Not needed | Euthyroid |
Decreased | Free T4 ↑ Free T4 normal Free T4 ↓ | Hyperthyroid T3 thyrotoxicosis Nonthyroid dis. or drugs |
Increased | Free T4 ↑ Free T4 normal Free T4 ↓ | Thyroid resistance or pituitary tumor Subclinical hypothyroid Hypothyroid |
Basal plasma calcitonin levels may be useful if MTC is suspected (family history of MTC or paraneoplastic syndromes, such as Cushing’s syndrome (ACTH) or carcinoid syndrome with watery diarrhea and vasomotor flushing). In addition, pheochromocytoma is associated with MTC in MEN type II, and patients present with sympathetic nervous system hyperactivity [21].
11.5.2 Imaging
11.5.2.1 Ultrasonography (US)
Ultrasonography (US) is the imaging study of choice for thyroid nodules. It can identify nodules too small to be palpated, the presence of multiple nodules and central or lateral neck lymphadenopathy and provides accurate measurements of nodule diameter allowing serial scans and better assessment of growth. Additionally, it allows characterization of nodules by sonographic features that suggest malignancy. Comet tail sign and coarse calcification suggest very low risk of malignancy. Hypoechoicity and absent halo with indistinct margin are associated with moderate risk of malignancy. The presence of microcalcification is highly suggestive of malignancy, especially papillary thyroid carcinoma (PTC) [22] (Fig. 11.2).
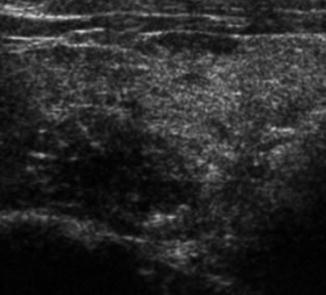

Fig. 11.2
Ultrasonography showing hypoechoic, ill-defined margin and microcalcifications. Biopsy proved to be a papillary thyroid carcinoma (PTC)
Color flow patterns are categorized as (a) type 1, no blood flow; (b) type 2, perinodular flow; and (c) type 3, intranodular blood flow (perinodular vessels may or may not be present). Although nonspecific, thyroid cancers may have internal hypervascularity, whereas benign nodules may have peripheral vascularization. However, type 3 vascularization can be found in both benign and malignant nodules [23]. Completely avascular nodules are more likely to be benign.
There is certainly some subjectivity to sonographic features, and characteristics vary depending on the histology such that US alone cannot reliably distinguish malignant from benign lesions. Although they do not obviate the need for biopsy, these features are extremely useful in selecting the site within a nodule for fine-needle aspiration biopsy (FNAB) in order to improve diagnostic yield or to select appropriate nodules to aspirate within an MNG [24, 25].
11.5.2.2 Magnetic Resonance Imaging (MRI) and Computed Tomography (CT)
Magnetic resonance imaging (MRI) is superior to scintigraphy in evaluating substernal (retrosternal) goiters. Magnetic resonance imaging is noninvasive and easily tolerated but is relatively expensive. Unlike contrast media used with CT, contrast media used in MRI do not influence thyroid function. Computed tomography gives structural information about the gland and its relationship to adjacent structures. Both CT and MRI are relatively expensive and have a limited ability in distinguishing between benign and malignant lesions. However, they are necessary in some cases to determine the staging and extent of the disease and in planning surgery. Indications of CT and/or MRI include the presence of a fixed thyroid mass, hemoptysis indicating pulmonary metastasis, cervical LNs, or when limits of the goiter cannot be determined clinically (retrosternal). They can also show involvement of the larynx, pharynx, trachea, esophagus, or major blood vessels [26].
It is important to avoid iodine contrast media in CT scan to ensure subsequent radioiodine treatment uptake by the remaining thyroid tissue is not compromised. This difficulty may be overcome by requesting for gadolinium-enhanced MRI scan.
11.5.2.3 Thyroid Scintigraphy
The use of radionuclide agents has been helpful in delineating the presence, size, and function of thyroid nodules. Two radioactive iodine (RAI) isotopes have been employed in clinical use. Scanning with 123I has the advantages of low-dose radiation (30 mrad) and a short half-life (12–14 h). This compares favorably with the use of 131I with a higher dose (500 mrad) and a longer half-life (8–10 days). Scanning with 123I is usually used for patients with a suspected lingual thyroid or substernal goiter, whereas 131I is used in patients with well-differentiated thyroid carcinoma (WDTC) to screen for distant metastasis. Thyroid cancers should have little uptake of the radionuclide; however, this deficient area on scanning could be masked by overlying normally functioning tissue. Malignancy has been shown to occur in 15–20 % of “cold” nodules and, additionally, in 5–9 % of nodules with uptake that is “warm” or “hot,” mandating continued aggressive approach to clinically suspicious nodules even if they are not “cold” [27].
Technetium pertechnetate 99 m (99mTc) is also used for the evaluation of thyroid nodules. This substance is trapped by the thyroid but not organified and has a short half-life and a low radiation dose. Screening with 99mTc also shows uptake in salivary glands and major vascular structures and, therefore, requires a higher sophistication of interpretation [20].
11.5.3 Biopsy
11.5.3.1 Fine-Needle Aspiration Cytology (FNAC): Freehand or US Guided
Fine-needle aspiration cytology is the most important step (cornerstone) in the management of thyroid nodules. Freehand or palpation-guided FNAC has a sensitivity of 65–98 % and a specificity of 72–100 % [28]. The US-guided FNAC improves the accuracy of FNAC. The acellular or nondiagnostic (Thy 1) aspirate is reduced from 14 to 8 % with US guidance [28]; sensitivity increases from 92 to 98 % and specificity from 69 to 71 % [29]. Moreover, it can also be used to help localize impalpable nodules and lesions <1 cm or when initial freehand FNAC was nondiagnostic.
Indications and Aims of FNAC
The indications for FNA are all thyroid nodules with a maximal diameter >1 cm and smaller nodules with suspicious findings on US. A lower limit for the maximal diameter does not exist. There are technical difficulties in the sampling procedure in tiny lesions (<0.5 cm), even under US guidance [30]. The aims of the FNAB include the following [30]:
To confirm the benign diagnosis of a nodule justifying the clinicians for a conservative approach avoiding an unnecessary surgery
To confirm the clinical diagnosis of a diffuse goiter like Hashimoto’s thyroiditis or subacute de Quervain thyroiditis
To recognize an aggressive thyroid tumor and to recognize or at least to suspect a clinically relevant low-grade tumor among all nodular enlargements of thyroid
To classify or to suspect some tumor types demanding a special therapeutic approach such as MTC, lymphoma, anaplastic carcinoma, or metastatic carcinoma from a primary site other than the thyroid
To clarify eventual postoperative enlargements in the thyroid region, differentiating mainly between residual and recurrent disease versus granulomas or LN enlargements
To explore various neck enlargements outside the thyroid gland, mainly cystic lesions of the neck, and differentiate between ectopic thyroid cysts, thyroglossal cyst, branchial cyst, and cystic degenerated LN metastases of PTC
To confirm the presence of LN metastases
Limitations of FNAC
The main limitation of the diagnostic approach of the thyroid nodules with FNA is the inadequate and indeterminate smears. Fine-needle aspiration cytology is not successful in (1) the detection of a microscopic focus of PTC, (2) differentiation of follicular adenoma from follicular carcinoma, (3) determination of the extent of the thyroid tumor, (4) exclusion of LN metastases, and (5) the safe recognition of a parathyroidal lesion [31].
Cytology Results of FNAC
Cytology results can be placed in five diagnostic categories (Thy 1–Thy 5) as indicated by the British Thyroid Association (BTA) Guidelines [32]. This will help with subsequent management as summarized in Table 11.2. The probability of a benign thyroid nodule being accurately diagnosed as benign from a single FNAC is 90 %. However, the accuracy of diagnosis increases significantly to 98 % if two separate aspirates were performed on separate occasions [22]. As such, having two aspirates reduces the false-negative rate to only 1.2 % [22].
Category | Description | Recommended action |
|---|---|---|
Thy 1 | Nondiagnostic, insufficient sample Cyst containing colloid or histiocytes only, in absence of epithelial cells | To repeat FNAC (US guidance may help) If the cyst is aspirated to dryness with no residual swelling, clinical/US follow-up alone may be sufficient |
Thy 2 | Benign, nonneoplastic Cyst containing benign epithelial cells | Repeat FNAC in 3–6 months. Two nonneoplastic results 3–6 months apart should exclude neoplasia |
Thy 3 | Follicular or Hürthle cell lesion/suspected folic or Hürthle tumor | MDT discussion – diagnostic lobectomy |
Thy 4 | Suspicious of malignancy | MDT discussion – total thyroidectomy |
Thy 5 | Diagnostic of malignancy | MDT discussion – total thyroidectomy |
The recently issued Bethesda System for Reporting Thyroid Cytopathology (BSRTC) [33], based on an NCI-sponsored conference (2007), is currently considered to be the most suitable for communicating findings from thyroid smears (Table 11.3).
Table 11.3
The Bethesda System for Reporting Thyroid Cytopathology: Recommended Diagnostic Categories [33]
Category | Description |
|---|---|
I | Nondiagnostic or unsatisfactory: Cyst fluid only – virtually acellular specimen, others (obscuring blood, clotting artifact, etc.) |
II | Benign: Consistent with a benign follicular nodule (adenomatoid nodule, colloid nodule, etc.) – consistent with Hashimoto’s thyroiditis – consistent with granulomatous (subacute) thyroiditis |
III | Atypia of undetermined significance or follicular lesion of undetermined significance |
IV | Follicular neoplasm or suspicious for a follicular neoplasm |
V | Suspicious for malignancy |
VI | Malignant |
The Nondiagnostic Smears
All smears lacking specific cellular elements, well preserved and prepared, and sufficient for a diagnosis should be characterized as nondiagnostic. The clinician should manage the patient on the basis of all other’s finding, asking for a re-biopsy, in an interval estimated according to the clinical needs [34].
Accuracy of FNA in Exploring Cervical LNs in the Presence of a Thyroid Carcinoma
A positive finding of a metastasis is principally a safe diagnosis, whereas a negative smear does not exclude the presence of a metastasis since it might be missed by sampling. Cervical LN metastases frequently undergo extensive cystic degeneration, resulting in acellular smears. Cystic foci in LNs remain highly suspicious, even by negative FNA results. The measurement of thyroglobulin (Tg) in the cystic fluid would be a helpful option to confirm a suspicion if the amount of cancer cells are not sufficient for the diagnosis [30].
Core Biopsy (With or Without US Guidance)
A core biopsy, preferably under US guidance, should be considered after two aspiration procedures showing nondiagnostic specimen (Thy 1) or when a thyroid lymphoma was suspected. Thyroid lymphoma typically presents with a rapidly increasing neck swelling in an elderly woman or on a background of autoimmune thyroiditis.
11.5.4 Flexible Laryngoscopy
Indirect laryngoscopy is important to assess vocal cord movements. Patients with difficulty breathing (increased respiratory rate or diminished oxygen saturation) or stridor should be referred as an “emergency.”
11.6 Multinodular Goiter (MNG)
11.6.1 Introduction
Goiter (enlarged thyroid gland) is derived from the Latin word “tumidum gutter,” which means “swollen throat.” Thyroid enlargement may be diffuse or nodular (multinodular or a solitary nodule); hormonal status may be euthyroid, hypothyroid, or hyperthyroid; and histologically, the enlarged thyroid may be benign or malignant.
Multinodular goiter (MNG), defined as an “enlarged thyroid gland with multiple nodules,” is a quite common condition with a marked female preponderance. It affects about 13 % of the world population, ranging from 5 % in the Americas to 32 % in the Eastern Mediterranean area [35]. The cause of MNG is probably multifactorial. Iodine (I2) deficiency, naturally occurring goitrogens, thyroid growth factors (GFs), and heredity have been postulated as possible contributors to goiter development [36]. Thyroid nodules may lead to a variety of clinical sequelae including compressive symptoms, hoarseness, dysphagia, and, importantly, cancer [37, 38], the incidence of which approaches that of patients with a solitary thyroid nodule (STN) [39].
11.6.2 Pathogenesis
The pathogenesis of MNG (Table 11.4) mainly describes two concepts, namely, the I2 deficiency goiters (endemic goiters) and the non-I2 deficiency goiters (sporadic goiters) [40].
Table 11.4
Pathogenetic mechanisms of goiter
Iodine deficiency |
Autonomy |
Immunological thyropathy |
Thyroiditis |
Cyst formation, hematoma, trauma |
Tumors |
Neoplastic production of TSH or TSH analog |
Acromegaly |
Hormonal resistance |
Enzyme deficiency |
Involvement of thyroid gland in extrathyroidal/systemic diseases |
Goitrogenic substances |
In I2 deficiency, less thyroid hormones are produced. A feedback mechanism involving the hypothalamus and pituitary gland leads to increased thyroid-stimulating hormone (TSH) production and consequently to proliferation of thyroid follicles [30], resulting in hypertrophy and hyperplasia of the thyroid gland in a diffuse and homogenous manner [41]. In contrast, in nodular goiter, nodules are surrounded by normal and connective tissue suggesting that they result from heterogeneity of growth [40]. Autonomous growth may occur in toxic as well as in euthyroid nodular goiter depending on whether the gland produces excessive amounts of hormones or not [42].
Other possible factors leading to thyroid proliferation and development of nodules, even in the absence of I2 deficiency, are the epidermal GF and the insulin-like GF [43]. Iodine deficiency alone cannot explain neither the nodularity nor heterogeneity of most goiters. Iodine-independent mechanisms have been attributed to the evolution of thyrotoxicosis and to the poor response of nodular goiters to TSH suppressive therapy, in contrast to diffuse I2 deficiency goiters, which respond well to I2 or T4 (Eltroxin) treatment [40, 44].
For prophylaxis of endemic goiter in I2-deficient areas, a supplementation with 150 μg I2/day is recommended for adults and is increased in pregnancy to 200 μg I2/day. This dose should be adjusted for children to 50 μg for the first year of life, 90 μg for ages 1–6 years, and 120 μg for ages 7–12 years [45, 46].
11.6.3 Clinical Assessment
11.6.3.1 Patient’s History
The patient’s history may be without complaint or may, apart from an awareness of the goiter size, include a globus sensation, dysphagia, dyspnea, choking, or stridor. The rate of growth over time as well as symptoms of hypo- or hyperthyroidism must be evaluated. Symptoms of hyperthyroidism include increased appetite, weight loss, heat intolerance, nervousness, irritability, agitation, palpitation, diarrhea, muscular weakness (myopathy), as well as oligo-/dysmenorrhea. On the other hand, the main symptoms of hypothyroidism are weight gain (myxedema), depression, concentration weakness, cold intolerance, fatigue, constipation, and oligo-/amenorrhea [45].
11.6.3.2 Physical Examination
Palpation of the thyroid gland is performed from the back of the patient, asking them to swallow. Typically, it moves up with deglutition. The size of the gland is evaluated, nodules are palpated (Fig. 11.3), and signs of local compression are assessed. Retrosternal goiter (RSG) may not be visible on clinical examination and may be unrecognized for many years. It may cause superior vena caval obstruction. Additionally, cervical lymph nodes (LNs) should be examined for enlargement, which may indicate malignancy.
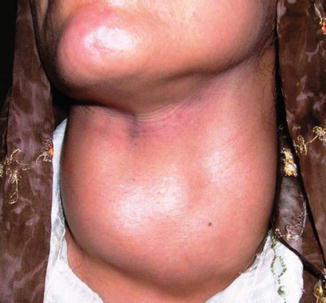

Fig. 11.3
A 28-year-old lady with an enlarged thyroid gland. Note the nodular appearance
Signs of hyperthyroidism may include tachycardia, tachyarrhythmias (extrasystoles, atrial fibrillation or flutter), hyperreflexia, fine tremors, warm and moist hands, soft and fine hair, as well as hair loss. Thyrotoxic crisis/coma is a severe condition of untreated exacerbated hyperthyroidism that may occur in Graves’ disease, autonomous adenoma, or multinodular toxic goiter. It presents with tachycardia, tachyarrhythmia, hyperthermia, diarrhea, vomiting, dehydration, muscular weakness, excitation (grade 1), disorientation, hallucination, somnolence (grade 2), and coma (grade 3).
Signs of hypothyroidism include bradycardia; hypotension; cardiac insufficiency; slow tendon reflexes; dry, pale, cold, rough, and doughy skin (myxedema); rough hair; and a hoarse voice. Myxedema coma is a severe condition that frequently occurs after chronic untreated hypothyroidisms with acute exacerbation due to infection, operation, severe general disease, cold, or sedative. It presents with somnolence, severe hypothermia, hypotension, bradycardia, hypoventilation, hyponatremia, hypoglycemia, and possible pericardial and pleural effusion.
11.6.4 Complications of MNG
Neglected or untreated MNGs may present with complications, which include:
Toxicity
Secondary thyrotoxicosis may develop in 10–20 % of cases, usually above the age of 30 years. Toxic nodular goiter is treated by surgery after patient preparation.
Malignant Transformation
Development of papillary thyroid cancer (PTC) or follicular thyroid cancer (FTC) is the most important complication, which usually occurs in long-standing cases. Clinical criteria of malignant transformation are listed in Table 11.5.
Table 11.5
Clinical criteria of malignant transformation of an MNG
Glandular criteria | Extra-glandular criteria |
|---|---|
Rapid recent growth | Compression symptoms become more evident |
Fixation of the swelling – restricted mobility with deglutition | Vocal cord paralysis due to infiltration of the recurrent laryngeal nerve (RLN) |
Consistency becomes harder | Horner’s syndrome due to infiltration of the cervical sympathetic trunk |
Edges become ill defined | Cervical lymph node (LN) enlargement |
Onset of pain | Unequal carotid pulsations |
Compression Manifestations
The pressure manifestations may result from the rapidly enlarging thyroid, retrosternal extension, malignancy, or hemorrhage. The sequelae will depend on the structures compressed (or infiltrated):
Trachea: Dyspnea results from displacement of the trachea to one side by an enlarging unilateral goiter; compression from both sides, in bilateral goiters (scabbard trachea); or tracheomalacia (softening of trachea) in long-standing cases.
Esophagus: Dysphagia, however, usually results from infiltration rather than compression and should alert the physician to the possibility of cancer.
Neck veins: The patient presents with mediastinal syndrome and congested face.
RLN: Hoarseness of voice mostly occurs due to malignant infiltration than just pressure on the nerve.
Calcification
In long-standing cases, calcification may occur either in the capsule or in the septa. Plain radiography is diagnostic. It results in a “hard” nodule and may be mistaken clinically for malignancy.
Cyst Formation
Development of cysts/pseudocysts in MNGs results from rupture of neighboring acini, hemorrhage, infection, or degeneration of nodules.
Hemorrhage
Bleeding may occur suddenly, precipitated by straining, causing sudden increase in the size of the gland, pain in the neck, and sudden compression of the trachea with impending suffocation augmented by reflex spasm of pretracheal muscles. Urgent treatment is necessary to relief compression immediately by aspiration of the cyst, division of the pretracheal fascia, and incision and evacuation of the hematoma. Urgent thyroidectomy is performed to remove the hemorrhagic nodule.
Infection
Multinodular goiters rarely get infected, but infection has been reported to occur in an MNG more commonly than in a normal gland.
11.6.5 Investigations
11.6.5.1 Laboratory Findings
The most important parameter is the basal TSH serum level. It is normal in the euthyroid state. If not, fT4 and fT3 should be performed. If an autoimmune process is suspected, thyroid autoantibodies should be tested such as the anti-thyroperoxidase (TPO) and anti-TSH receptor antibodies. It must be noted that they may also be positive in healthy individuals or in patients with goiter or autonomy [43].
11.6.5.2 Imaging Findings
Ultrasonography (US)
All patients scheduled for thyroid (or parathyroid) surgery should undergo a preoperative ultrasound (US). It is the most precise tool for evaluating the thyroid and nodule size [43]. The normal volume of the thyroid is 7–20 ml, and nodules larger than 2 mm in diameter may be identified [47]. Besides the size, US provides valuable information regarding echogenicity (the normal thyroid is isoechogenic or slightly hyperechogenic), nodular composition as it allows differentiation between solid nodules and simple or complex cysts, presence of calcifications (micro, i.e., 1 mm or less, or macro), as well as shape and margins. Moreover, US may differentiate extrathyroidal structures from the thyroid gland and may give information on regional lymphadenopathy [48]. Color flow Doppler US gives further information on vascular flow and velocity, and US examination is combined with an FNA cytological sample in cases of suspected malignancy.
Scintigraphy
Scintigraphy has become rare due to the progress in US techniques. It should be performed only if it has an impact on the therapeutic plan, for instance, in a young patient with an STN, possibly a carcinoma, or in case of hyperthyroidism [43]. According to scintigraphy, thyroid nodules may be “hot” in the presence of autonomously functioning thyroid tissue (rarely malignant) or “cold” in which case the incidence of malignancy is 10–20 % [49].
Radiography and Tomography
Plain chest X-rays may show a substernal goiter (Fig. 11.4), and CT scan and MRI are indicated for large tumors extending to adjacent structures such as the mediastinum or the retropharyngeal region [49].
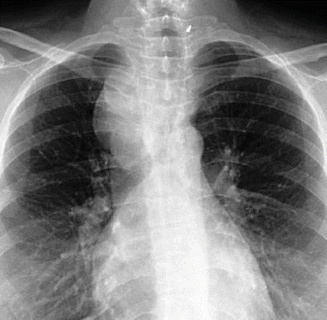

Fig. 11.4
Plain X-ray of the chest showing a mediastinal soft tissue shadow; retrosternal goiter
11.6.5.3 Fine-Needle Aspiration (FNA)
For the evaluation of the potential malignancy of a nodule, US-guided FNAC may give further information. Indications are suspected malignancy with the following findings: young patient, previous radiation exposure of the neck, rapid growth, cold in scintigraphy, and US findings of hypoechogenicity, loss of halo, size >1 cm diameter, ill-defined margin, and the presence of microcalcifications [45].
11.6.5.4 Airway Assessment
Signs of significant airway obstruction are stridor, labored breathing, intercostal retractions, and agitation in case of RSG vena caval obstruction [50]. Indirect laryngoscopy may be helpful and should be a routine examination [51], particularly in repeat surgery for recurrent goiter or if there is evidence of recurrent laryngeal nerve (RLN) dysfunction. A chest X-ray is evaluated for tracheal deviation and compression [52]. Other examinations, such as CT and MRI, are not routinely performed, but may give additional information especially in cases of RSG [52]. Respiratory function tests are debatable [53]. In patients with evidence of a compromised airway, the airway is assessed using fiberoptic laryngoscopy after application of topical anesthesia and oxygen [50].
11.6.6 Management of MNG
11.6.6.1 Nonoperative Treatment
Conservative treatment of MNG with L-thyroxin may be effective or at least partially in reducing the volume of relatively small, benign, solitary, solid thyroid nodules [54–58]. Low TSH suppression is effective in reducing nodule volume [31]. However, some authors reported a volume reduction without treatment, probably due to spontaneous regression [55, 59].
Alternatively, radioactive iodine (RAI) therapy may be used in elderly patients or those with contraindications for surgery [60]. The lifetime risk of cancer due to RAI is negligible in patients over 65 years old.
In Graves’ disease, surgery, RAI therapy, and treatment with antithyroid drugs (ATDs) are all options, whereas autonomy is a classical indication for radiotherapy except in solitary autonomous nodules where surgery is equally effective. Thyroid neoplasms are an indication for surgery as are I2-induced hyperthyroidism and intractable hyperthyroidism not responsive to conservative management [43].
11.6.6.2 Surgical Treatment
Indications and Extent of Surgery
Indications for surgery of the thyroid gland vary depending on the pathology: in euthyroid MNG the main indications are goiter size, compression symptoms, and suspected malignancy. Various surgical options exist, ranging from hemithyroidectomy (lobectomy) to total thyroidectomy (if the thyroid gland contains nodules throughout).
Because of a reportedly high frequency of complications in some series, controversy exists about the routine use of total thyroidectomy (TT) for the management of “benign” MNG [61–63]. However, it is noteworthy that, when performed by experienced hands, TT, compared with subtotal resection, does not increase morbidity in benign pathologies [64–69]. In cases of retrosternal goiter (RSG), TT is preferred owing to the malignant potential and in order to reduce recurrence rate (RR) [70].
It is important to emphasize that proper training and surgical experience are significantly associated with low complication rates in thyroid surgery [71, 72].
Multinodular Goiter (Benign/Thy 2)
Patients with MNG, diagnosed with FNA as being benign (Thy 2), could receive either definitive surgery or annual monitoring with TSH levels (Table 11.6).
Table 11.6
Indications of surgery and annual TSH monitoring for patients with MNG (Thy 2)
Indications of surgery | Indications of annual TSH monitoring |
|---|---|
Compression syndrome | The goiter is small or modest in size |
Cosmetic reasons | The patient is euthyroid with normal TSH |
Follicular or Hürthle cell lesion (Thy 3) | Clinically asymptomatic without signs of compression |
Concern about malignancy (Thy 4) | FNAC of all suspicious nodules is benign |
Persistent troublesome toxic nodules |
Thyroid Nodules Associated with Hypo-/Hyperthyroidism
These nodules are very unlikely to be cancer. They are more likely to be benign toxic nodule or Hashimoto’s thyroiditis. The frequency of malignancy in cold nodules is 10–20 % and only 4 % in hot nodules [73, 74]. These nodules should still be aspirated and if confirmed to be benign (Thy 2) after two aspirates 3–6 months apart, with no other suspicious features, can be safely managed by an endocrinologist and referred back for reevaluation if there was any change in the swelling.
Dominant Nodule in MNG
Patients with hyper- or hypothyroidism associated with MNG with no other suspicious features in history and clinical examination have a low risk of thyroid cancer [32]. They are referred to an endocrinologist. When a dominant nodule is noted to be growing and become suspicious, it should be aspirated and treated accordingly depending on cytology results. Low-risk patients who are euthyroid with MNG of long duration and slow growth have a very low risk of thyroid cancer [32]. Patients can be observed at intermediate or long intervals.
Thyroid Cystic Swelling
It should be clearly stated that in order to help the pathologist in interpreting FNAC, the cyst should be aspirated to dryness under US guidance and any residual mass should be noted and subjected to FNAC immediately as a separate specimen [32]. For a thyroid cyst that is shown to be benign on FNAC and does not recur at follow-up, clinical observation alone may be sufficient. A recurrent thyroid cyst should be re-aspirated during follow-up and the sample sent for cytology. Patients with high risk factors in history and clinical examination can be considered for diagnostic lobectomy. Some surgeons would consider diagnostic lobectomy for a cyst that has recurred for three times or more. Surgery can also be considered at patient’s request.
Results of Surgical Treatment of MNG
The rate of secondary hemorrhage is approximately 1 %, whereas the rate of persistent RLN paresis and of hypoparathyroidism has dropped to <1 % in the last two decades [75, 76]. Adequate surgery is part of the prophylaxis of recurrence [77]. In a case-control study, young age and multiple nodules at initial surgery have been identified as independent risk factors for recurrence [63]. Despite suppressive thyroxin treatment postoperatively, 14 % of patients develop recurrence after subtotal thyroidectomy after a median follow-up of 14.5 years [78]. Without suppression the RR reaches up to 41 % [79, 80]. Since TT can be performed with a minimal complication rate, this option is increasingly being accepted and recommended for the treatment of MNG [66, 81].
Prophylaxis of Recurrence
In addition to adequate surgery, postoperative suppression therapy with L-thyroxin is important for prophylaxis of recurrence [77] provided that the proper dose is given. In I2 deficiency goiter with no substitution, 25 % of patients will have a recurrence. The aim should be a TSH in the lower normal range (0.3–1 mU/l), in contrast with malignancies where the TSH should be suppressed to <0.1 mU/l. Strong TSH suppression, however, increases the risk of cardiac complications and accelerates osteoporosis [82, 83].
Treatment of Recurrent Goiter
Surgery for recurrent goiter has a higher complication rate than in the primary setting [38, 70]. Temporary RLN palsy was found in 5 % and permanent in 3 %, both significantly higher than at primary operation [84]. The indication is therefore restricted to third-degree goiters or suspicion of malignancy. Preoperatively, indirect laryngoscopy for documentation of the RLN function is essential. Intraoperative nerve monitoring may be helpful in the identification of the nerve, which may be altered in position [45]. If preoperative unilateral RLN paresis is present, if possible, only ipsilateral hemithyroidectomy should be considered [77].
11.7 Retrosternal Goiter (RSG)
11.7.1 Introduction
Extension of the goiter beyond the confines of the neck into the thoracic cavity with more than 50 % of the mass inferior to the thoracic inlet is called retrosternal goiter (RSG) or intrathoracic goiter. Most of these goiters are slow growing and often do not cause symptoms until the mass reaches a critical size and causes compression symptoms. Although the majority of these lesions are benign in nature [85–88], approximately 6–16 % may be malignant, and a few cases may exhibit thyrotoxicosis.
11.7.2 Incidence
11.7.3 Anatomical Classification
Retrosternal goiters are located in the superior mediastinum, either anteriorly or posteriorly. They are classified as either primary (Fig. 11.5) or secondary (Fig. 11.6) goiters. Primary goiters account for <1 % of all RSGs. They originate from ectopic embryonal thyrocytes that have descended into the chest with the arch of the aorta. Secondary goiters are cervical goiters that have migrated to the intrathoracic location favored by the negative mediastinal pressure and fixation of the soft tissues. The main differences between the primary and secondary RSGs goiter are shown in Table 11.7.


Fig. 11.5
Primary retrosternal goiter: intrathoracic goiter originating in the chest

Fig. 11.6
Secondary retrosternal goiter: originating from the left thyroid lobe
Table 11.7
Differences between primary and secondary RSGs
Differences | Primary RSG | Secondary RSG |
|---|---|---|
Origin | From ectopic thyroid tissue in the chest | Extension of cervical thyroid into the chest |
Blood supply | Intrathoracic aorta | Inferior thyroid artery |
Connection with cervical thyroid gland | None | Usually contiguous with the cervical gland or connected by a fibrous band |
Cervical mass | None | Yes in 80–90 % of patients |
Secondary RSGs in the anterior mediastinum usually originate from the lower pole of the thyroid gland and descend along the anterolateral aspect of the trachea and anterior to the RLN and carotid vessels. Occasionally, a goiter originating from the posterolateral aspect of the thyroid may descend into the posterior mediastinum pushing the esophagus to the opposite side and causing forward displacement of the trachea and tilting of the larynx. In this case, the RLN, inferior thyroid artery, and the carotid vessel lie anterior to the goiter. DeAndrade [91] reported that 9.4 % (128/1300) of RSGs occurred in the posterior mediastinum.
11.7.4 Clinical Manifestations
11.7.4.1 Symptoms
Patients with RSG are usually in their fifth or decade of life, with a female to male ratio of 3:1. Approximately, 20–35 % of patients are asymptomatic. A neck lump is present in 40–50 % of patients. Other symptoms are the result of compression of intrathoracic structures. Compression of the airway occurs in about 50 % of patients and may present with dyspnea, stridor, or a choking sensation [89, 90] and may require urgent intubation or a semi-urgent operation in 22 % as reported by Newman and Shaha [85]. In some patients, dyspnea is experienced only when the head is turned toward one side or by lying down flat. Dysphagia is present in about 30–40 %, being more common with RSGs located in the posterior mediastinum. Hoarseness of voice is reported in 13 % of patients [86]. Unusual symptoms result from vascular compression causing downhill upper gastrointestinal bleeding from esophageal varices, effort axillary vein thrombosis, transient ischemic attacks (TIA), and cerebral edema.
11.7.4.2 Physical Examination
A palpable lump in the neck, with impalpable lower border and dull sternal percussion, may be demonstrated in about 80–90 % of patients [86]. Other signs include dilated neck veins and tracheal deviation. Raising the arms or hyperextension of the neck may cause dilatation of cervical veins and flushing of the face or even respiratory difficulty/stridor (Pemberton’s sign). Common signs and symptoms of RSG are listed in Table 11.8.
Table 11.8
Clinical symptoms and signs of patients with RSG
Symptoms | Signs |
|---|---|
Asymptomatic Neck lump Compression Airway: dyspnea, stridor, raspy cough, wheezing, chocking Esophageal: dysphagia Nerve: hoarseness of voice, Horner’s syndrome Vascular: SVC syndrome, TIA, cerebral edema, GI bleeding | Cervical mass Dilated veins of the neck Tracheal deviation Flushing of skin Pemberton’s sign Horner’s syndrome |
11.7.5 Diagnosis
11.7.5.1 Plain Chest X-Ray (CXR)
Plain CXR is the most cost-effective method for diagnosis of RSG. Findings include deviation and/or compression of the trachea (Fig. 11.7), soft tissue density or a mass, and occasionally calcifications (Fig. 11.8) and reflection of the mediastinal pleura below the trachea.
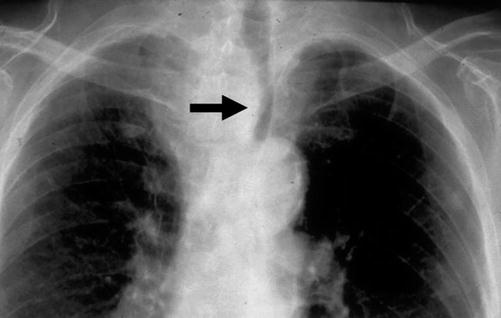
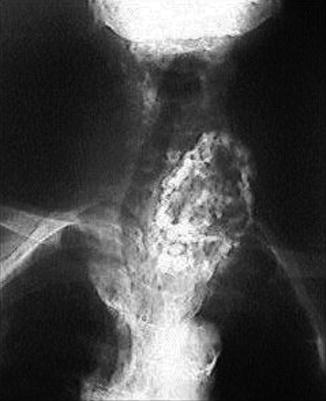

Fig. 11.7
Chest X-ray showing left tracheal deviation (arrow) by a retrosternal goiter (RSG)

Fig. 11.8
Plain X-ray showing calcification in a retrosternal goiter with right tracheal deviation
11.7.5.2 CT Scan and MRI
Both CT scan (Fig. 11.9) and MRI can provide more precise information about the relationship between the various intrathoracic organs and the goiter This information helps the surgeon to plan the operative approach.
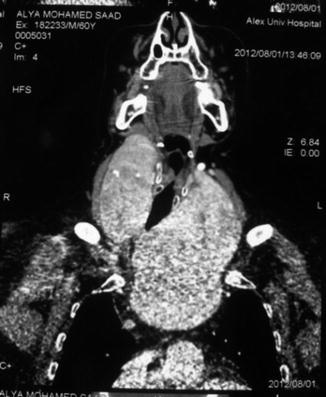

Fig. 11.9
Computed tomography (CT) scan showing secondary retrosternal goiter (RSG) extending from the left thyroid lobe into the mediastinum
11.7.5.3 Scintigraphy
A radionuclide thyroid scan may be useful in differentiating goiter from other mediastinal masses. Nevertheless, a solitary, large cyst may appear as a “cold” nodule on thyroid scan and thus provide a false-negative result.
11.7.5.4 US and FNA
Ultrasonography is generally not necessary but can be helpful in selected patients, and FNAC is not recommended as it is not easily accessible and may cause bleeding or pneumothorax.
11.7.6 Differential Diagnosis
A mediastinal mass in the anterosuperior compartment should be differentiated from a dermoid cyst, aneurysm, lymphoma, teratoma, pleural cyst, and secondary carcinoma. Differential diagnosis of masses in the posterior mediastinum should also include tumors of neurogenic origin.
11.7.7 Treatment
11.7.7.1 Pharmacotherapy
A patient with elevation of TSH or defects in thyroxin synthesis is a candidate for suppressive therapy. However, RSGs, especially those that have cystic change and hemorrhage, do not respond to thyroxin therapy. Overall, only about 20–30 % of patients respond to such treatment after 1 year, and cessation of therapy is often followed by recurrence. Presence of cardiac disease and osteoporosis in elderly patients poses an additional hazard to suppressive thyroxin therapy. Failure of suppressive therapy is probably based on autonomicity. Once stimulated for a long time, autonomous growth occurs, and the hyperplasia is no longer a fully reversible process.
11.7.7.2 Radioactive Iodine (RAI) Therapy
The use of radioactive iodine (RAI) for the treatment of nontoxic goiter was first reported by Keiderling in 1964, but it was not until 1994 that its use in RSGs was evaluated by Huysmans et al. [92]. In their prospective study of 19 patients with large compressive goiters, 11 had intrathoracic extensions for >2 cm. They reported a 40 % reduction in the volume of the goiter using MRI and a 10 % decrease in tracheal narrowing and deviation in 75 % of their patients. However, one-third of their patients did not have any improvement in dyspnea.
Radiotherapy is not without risk of complications, which may include radiation-induced thyroiditis, stridor from a transient increase in volume, neck pain, occasional hyperthyroidism, sore throat, mild dysphagia, and dryness of mouth.
11.7.7.3 Surgical Therapy
Surgical treatment is the most effective therapy for RSG, and the presence of RSG is itself an indication for operation [87]. It is currently believed that TT is the procedure of choice, particularly that about 95 % of cases can be performed via a cervical incision. Ligation of the ITA branches close to the thyroid capsule, preservation of the blood supply to the parathyroid gland, and minimal dissection of the RLN are the hallmarks of a safe operation.
Approach to RSG
Most RSGs can be resected through the standard cervical Kocher’s incision [70, 93, 94]. The head is reclined and the patient positioned in anti-Trendelenburg of about 15–20° to reduce the venous pressure. In order to gain good access, the incision should be placed 1–2 cm higher than usual [93]. The skin/platysma flap is elevated, the cervical fascia is separated at the midline, and the muscles are held aside or incised laterally in the case of a very large goiter.
First, the upper pole is mobilized under ligation of the superior thyroid vessels with preservation of the external branch of the ELN. This is important in the subsequent upward movement of the thyroid gland from the retrosternal to a cervical position. The RLN and superior parathyroid glands are routinely identified [95] particularly that the inferior parathyroids may be more difficult to locate in RSG.
The next step is the delivery of the thyroid gland by blunt dissection with the finger inferiorly, completed by sharp dissection under vision. The inferior vascular structures are then ligated as near as possible to the gland, selectively ligating the branches of the ITA at the level of the thyroid capsule [70]. The inferior thyroid artery (ITA) should not be ligated at the main stem [45, 77]. If the thyroid lobe cannot be brought to the neck, more room is provided by removing the opposite thyroid lobe in its cervical position.
In cases of very large intrathoracic goiters, invasive tumors, dense adhesions in recurrent cases, uncontrollable hemorrhage, or truly ectopic intrathoracic gland with its major blood supply from intrathoracic vessels, a mediastinal approach using sternotomy is required [70, 93]. Indications for sternotomy are listed in Table 11.9. As an alternative to complete sternotomy, a partial upper sternal split (manubriotomy) is possible in most cases [94]. Division of the manubrium to below the manubriosternal junction is performed. The innominate vein and the pleura freed from the back of the manubrium. The manubrium and the upper sternum are divided in the middle and gently spread with a right-angled retractor. Sternotomy is closed using sternal wires. If complete sternotomy is performed, the skin incision is extended to just above the xiphoid process, and the pericardial and diaphragmatic attachments are freed from the back of the sternum before its division. For resection of a crossed RSG with extension from a left-sided gland to the right mediastinum, right anterolateral thoracotomy can be helpful [93].
Table 11.9
Indication for sternotomy for RSG
Goiter size significantly larger than the thoracic inlet |
Primary intrathoracic goiter with intrathoracic blood supply |
Goiters in the posterior mediastinum displacing or compressing the aortic arch |
Goiters associated with SVC syndrome |
Recurrent RSG |
Malignant RSG with LN metastasis |
Complications of RSG Surgery
As with cervical goiter, the main complications of RSG surgery are hemorrhage, RLN injury, and hypoparathyroidism. An intrathoracic goiter was found to be an independent risk factor for postoperative complications [68]. In a prospective study of 2,235 thyroid resections, 312 were performed for RSG [96] in which the complication rate was significantly elevated, including hemorrhage, wound infections, transient hypocalcemia, and transient RLN paresis.
In addition, mediastinal injuries may occur during RSG surgery. If mediastinal hemorrhage occurs, immediate surgical revision via a complete sternotomy is indicated for adequate control. Pneumothorax after pleural injury is treated with insertion of a chest tube. More rare complications are infections (mostly due to an infected hematoma), injury of the pharynx and trachea, or sympathetic chain with resultant Horner’s syndrome. Sternal infection may manifest late and is treated with a surgical debridement.
11.8 Solitary Thyroid Nodule (STN)
The most important distinction in the workup of a solitary thyroid nodule (STN) is whether or not it represents a malignant lesion. Thus, the primary goal is to distinguish those nodules that require surgical excision from those that can be safely observed.
11.8.1 Pathological Classification
Many thyroid diseases can present clinically as an STN such as colloid cysts, adenomas, Graves’ disease, thyroiditis, infections, and malignancies (Table 11.10).
Table 11.10
Pathology of thyroid nodule
Benign |
Adenomatous nodules or colloid nodules |
Follicular adenoma |
Hürthle cell adenoma |
Thyroid cysts |
Inflammatory lesions (thyroiditis) |
Developmental abnormalities (cystic hygroma, dermoid, teratoma) |
Malignant |
Papillary carcinoma |
Follicular carcinoma |
Hürthle cell carcinoma |
Mixed papillary/follicular carcinoma |
Medullary carcinoma |
Anaplastic carcinoma |
Lymphoma |
Metastatic disease |
11.8.2 Clinical Considerations
11.8.2.1 History Taking
Several aspects of the history and physical examination can increase the suspicion of malignancy (Table 11.11). Any nodule developing prior to puberty should be viewed with suspicion. It has been reported that more than 50 % of all thyroid nodules in children are malignant [97]. The incidence of malignancy is also higher in nodules that develop after the age of 65 years. Benign nodules are more common than malignant nodules in both males and females; however, the proportion of malignant nodules in males is twice that of females.
Table 11.11
Important clinical factors in the diagnosis of thyroid cancer
History taking | Physical examination |
|---|---|
Family history Gender Age <20 or >60 years History of head and neck irradiation Rapidity of growth Associated symptoms (pain, dysphagia, dysphonia, dyspnea) Growth on thyroid hormone suppression | Solitary versus multiple nodules A hard nodule Fixation to adjacent structures Diameter 4 cm or more Cervical lymphadenopathy |
One of the most important aspects of the past medical history is whether the patient has received. If a patient with an STN has a history of head or neck irradiation in childhood, the prevalence of cancer is 30–50 % [85]. Other factors to consider in the past medical history include symptoms of pheochromocytoma or hyperparathyroidism (HPT), long-standing constipation and/or diarrhea, hypertension, and/or episodes of nervousness. These should alert the clinician to the possibility of medullary thyroid carcinoma (MTC) in association with a familial MEN syndrome.
The time course for development of an STN is important to note. A nodule that has been stable in size for years is almost always benign. Thyroid malignancies usually develop over weeks or months. Entrapment of the RLN, invasion of thyroid capsule, or spread into adjacent tissues can lead to local pain in the neck or radiating to the jaw and ear. Dysphagia, dysphonia, dyspnea, hoarseness, and hemoptysis may all reflect esophageal or tracheal involvement by a thyroid cancer. Nodules associated with hyperthyroidism are usually benign functioning adenomas, whereas a nodule in a patient with hypothyroidism is often caused by autoimmune thyroiditis.
11.8.2.2 Physical Examination
Although it is almost impossible to distinguish a benign nodule from a malignant nodule by palpation, a thorough physical examination should be performed. A relatively rapidly growing, hard nodule is associated with a higher risk of malignancy. Hard nodules may be due to calcifications in benign adenomas, however.
A nodule fixed to surrounding tissues such as the trachea or strap muscles is most likely malignant. However, fixation of the thyroid can also occur with severe chronic thyroiditis. Vocal cord paralysis strongly suggests an invasive cancer, but again, benign conditions such as Hashimoto’s thyroiditis or MNG can rarely affect vocal cord function. The most significant physical findings suggestive of malignancy are the unilateral, firm, mobile, non-tender, discrete LNs resulting from metastatic thyroid cancer, most commonly papillary thyroid carcinoma (PTC) (Table 11.11).
11.8.3 Laboratory Tests
Thyroid function tests may also be useful in the evaluation of an STN, since a suppressed TSH level is suggestive of benign pathology, as it is uncommon for thyroid cancer to cause thyrotoxicosis or thyroiditis [98]. Abnormalities in the thyroid gland function are most often associated with a benign nodule. Malignant thyroid nodules generally have normal thyroid function tests.
11.8.4 Imaging Studies
11.8.4.1 Ultrasonography (US)
Thyroid US has been used preoperatively to evaluate thyroid nodules (Table 11.12); however, alone, the sensitivity, specificity, and positive predictive value for US are quite low.
Table 11.12
Ultrasound features of thyroid nodule suggestive of malignancy
Highly suggestive |
Extracapsular extension |
Suspicious cervical LNs |
Taller than wider nodule |
Microcalcification |
Irregular ill-defined margins |
Markedly hypoechoic |
Moderately suggestive |
Elastography |
Texture (>50 % solid) |
Increased intranodular vascularity |
Absence of halo |
Minimally suggestive |
Hypoechoic |
Macrocalcification |
Isoechoic, hyperechoic |
Complex nodules |
Peripheral calcification (eggshell) |
Spongiform nodules |
The use of US-guided FNA can improve the diagnostic accuracy and should be considered whenever confronted with a patient whose thyroid nodule is difficult to palpate on physical examination or in whom the initial FNAB was nondiagnostic [5].
Still however, interpretation of the aspirate for definitive diagnosis may still not be possible. In cases of follicular neoplasia, FNA may not be able to distinguish malignant from benign disease, since the diagnosis of follicular carcinoma is histological, specifically by identification of capsular or vascular invasion. This scenario is similar when dealing with Hürthle cell neoplasms [99, 100]. In such cases, pathology reports may be suspicious for malignancy, of which nearly 20 % will actually be malignant Hürthle or follicular neoplasms.
11.8.4.2 Scintiscan
Radioisotopes of iodine or technetium are based on the assumption that malignant thyroid tissue neither traps nor incorporates iodine and therefore should appear nonfunctioning or “cold” on uptake scan. Normally functioning nodules are “warm,” and hyperfunctioning nodules appear as “hot” on the scan. The incidence of malignancy is higher in cold nodules as compared with warm or hot nodules. Thyroid scans have generally been replaced as a first-line test by FNAB [101].
11.8.5 Fine-Needle Aspiration Biopsy (FNAB)
Fine-needle aspiration biopsy (FNAB) has become the diagnostic procedure of choice for STNs. It is a simple outpatient procedure, and complications such as hematoma or infections are rare. It has been shown repeatedly to be a better predictor of malignancy than other preoperative tests and has substantially decreased the number of patients requiring surgery for benign disease [102]. The cytodiagnostic categories of Bethesda Classification [33] with the corresponding estimated risk of malignancy are listed in Table 11.13.
Category | Description | Risk of malignancy (%) |
|---|---|---|
I | Nondiagnostic | 1–4 |
II | Benign | 0–3 |
III | Atypia of undetermined significance/follicular lesion of undetermined significance | 5–15 |
IV | Follicular neoplasm/suspicious for follicular neoplasm | 15–30 |
V | Suspicious of malignancy | 60–75 |
VI | Malignant | 97–99 |
11.8.6 Management
The management of an STN depends on several factors as shown below and demonstrated in the suggested management algorithm (Fig. 11.10).
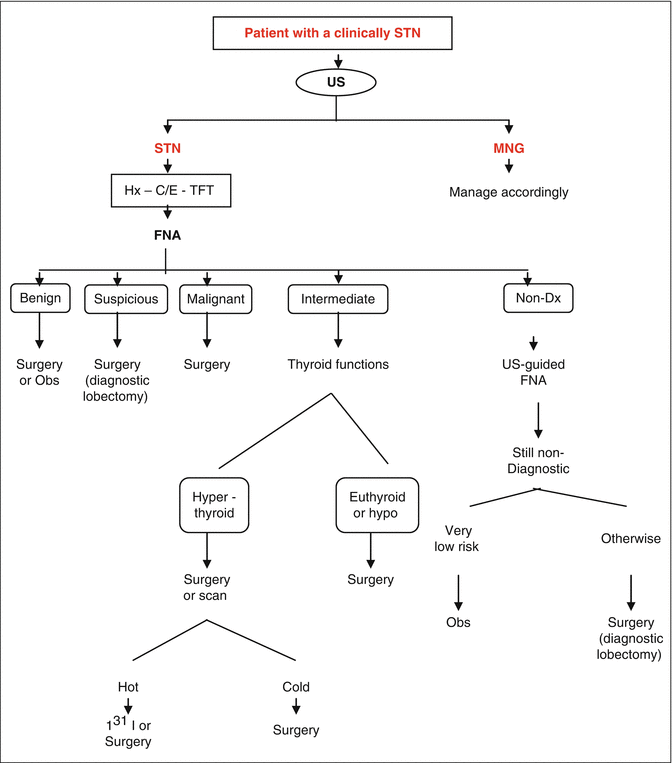

Fig. 11.10
Algorithm for the management of an STN. Hx history, Dx diagnostic, C/E clinical examination, TFT thyroid function tests, Obs observation, US ultrasound, FNA fine-needle aspiration
11.8.6.1 Clinically Non-palpable Incidental Nodule <1 cm (Incidentaloma)
Either non-palpable nodules <1 cm are noted during surgery or imaging performed for another purpose should be observed. These nodules have a very low risk of cancer [32] in patients with low risk (as per history taking, clinical examination, and US findings). In addition, there is no evidence to show that treatment of sub-centimeter micro-carcinomas improves outcome [74, 103]. The exception to the above is an incidentaloma identified by FDG-PET scan. These carry a 50 % chance of malignancy and should be managed as STN or “incidentaloma” >1 cm [74].
11.8.6.2 Benign Nodule
If FNAC indicates a benign nodule, there are three options for treatment: surgery (hemithyroidectomy), observation, and hormone suppression. Surgery may be considered if the nodule is causing symptoms, or is esthetically unpleasant to the patient, and also in those patients who are at increased risk for thyroid cancer despite a benign FNA.
If the patient does not require surgery, the nodule may either be observed or suppressed with L-thyroxin. This should either reduce the size of the nodule or prevent its further growth; otherwise, malignancy should be suspected and surgery considered. Several studies have failed to demonstrate the efficacy of thyroxin therapy for solitary nodules [104]. There has also been some concern that the use of thyroxin, especially in postmenopausal women, may lead to osteoporosis [105].
11.8.6.3 Indeterminate Lesion
When FNAC demonstrates follicular or Hürthle cell neoplasm, surgery is indicated to reach a definite diagnosis. If the nodule is hyperfunctioning, a thyroid scintiscan may be appropriate. A “hot” nodule may be observed, treated with 131I or surgically excised. If the plan is to treat a hyperfunctioning nodule with surgery, the thyroid scan may be omitted.
11.8.6.4 Suspicious Nodule
Suspicious STNs should be treated with surgical resection to avoid missing a thyroid cancer. The principal surgical approach to solitary, undetermined nodule is ipsilateral lobectomy (hemithyroidectomy).
11.8.6.5 Malignant Nodule
Treatment of Differentiated Thyroid Cancer
Principles and Strategies
Differentiated carcinoma of the thyroid includes both papillary and follicular variants. The primary treatment for these cancers is resection, albeit with controversy about its extent. Three primary surgical strategies exist for the management of differentiated thyroid cancer, namely, total thyroidectomy (TT), near-total thyroidectomy, and hemithyroidectomy (lobectomy with isthmusectomy). Whereas TT involves removal of the entire thyroid gland and its capsule, near-total thyroidectomy preserves the posterior capsule of the thyroid contralateral to the tumor. Both procedures are considered as completely ablative approaches. However, they expose the patient to the risk of bilateral dissection and necessitate lifelong thyroid hormone supplementation. Alternatively, hemithyroidectomy allows preservation of normal thyroid tissue, thereby obviating the need for lifelong supplementation and eliminating the risk for hypoparathyroidism and bilateral vocal cord paralysis. Subtotal thyroidectomy (preservation of several grams of thyroid tissue) is not a recommended treatment option for patients with differentiated thyroid cancer due to the higher complication rates encountered when subsequent surgery is indicated [106].
Treatment: Papillary Thyroid Cancer (PTC)
Total Thyroidectomy
In patients with high-risk lesions or extrathyroidal extension, it is generally accepted that TT is the appropriate treatment as it results in excision of cancer that was occult on the preoperative assessment and permits a more sensitive postoperative follow-up surveillance for recurrent disease. After a TT (or near-total thyroidectomy), postoperative RAI can be used to identify and ablate any residual thyroid cancer, and serum thyroglobulin is a more accurate marker of recurrent or persistent PTC following TT when compared with more conservative thyroid resections. It is thus conceivable that TT is a safe and effective primary treatment for patients with PTC, particularly with the low complication rate (<2 %) reported by experienced surgeons [107, 108]. Pacini et al. [109] reported that approximately 44 % of 182 patients studied harbored histologically confirmed PTC at completion thyroidectomy and suggested TT in order to eradicate all neoplastic tissue in patients with PTC. The authors also recommended completion thyroidectomy for patients with a history of PTC that were initially treated with hemithyroidectomy. Moreover, Hay et al. [110] found that patients who underwent lobectomy for PTC had a higher RR (14 %) and nodal metastases (19 %) as compared to TT%, though with a similar survival rate and distant metastases. Finally, elimination of all PTC via TT may prevent progression to anaplastic thyroid cancer from residual tissue left behind during thyroid hemithyroidectomy.
Hemithyroidectomy
The use of hemithyroidectomy for the treatment of PTC is supported by some authors as it avoids the risk of some potential complications of bilateral procedures such as RLN and parathyroid injuries and also provides similar survival benefit to TT [111–113]. In patients with excellent prognosis (tumor size <1 cm, confined to the thyroid without evidence of any metastatic disease, in an otherwise healthy female below 45 years of age), hemithyroidectomy may be used.
Lymph Node Dissection
Careful stratification of patients with PTC into those who require lymph node (LN) dissection and those who do not is important for minimizing surgical morbidity. Gross nodal disease occurs in 20–30 % of adult patients with PTC [114]. Nodal metastases confirmed by preoperative US or CT or by intraoperative exploration should be treated with node dissection [106], specifically removal of ipsilateral central neck LNs (level 6) and lateral compartment nodes (levels 2–5). On the other hand, microscopic regional LN metastases occur in approximately 80 % of patients with PTC [110] and may often be ablated by adjuvant RAI therapy, but better be removed at the initial operation as they may be the site of persistent disease [115–117]. Removal of central neck LNs has been reported in retrospective studies to be associated with an improvement in survival rate and regional RR [118–120]. The current American Thyroid Association (ATA) Guidelines advise that staging/prophylactic level 6 LN dissection for all patients undergoing thyroidectomy for well-differentiated thyroid carcinoma should be considered [121]. Prophylactic lateral neck node dissection is not recommended because it is not associated with improved overall survival of patients and involves violation of additional planes by a more extensive operation.
Treatment: Follicular Thyroid Cancer (FTC)
Surgical resection remains the primary method of therapy for follicular thyroid cancer (FTC). If the tumor is confined to the thyroid (T1–2 N0 M0), TT and hemithyroidectomy with extirpation of central LNs are both adequate. For larger tumors (T3–T4), TT along with postoperative RAI therapy is indicated. Morbidity is minimal in the hands of experienced surgeons. Since FTC spreads primarily via hematogenous routes, cervical LN metastases are not as common as with PTC (35 % versus 67 %, respectively). Still however, therapeutic modified neck dissection is appropriate for patients with clinically apparent disease. Postoperative RAI scans and ablation are also essential to help detect any residual disease and eliminate it.
11.8.6.6 Treatment of Oncocytic (Hürthle Cell) Thyroid Cancer
Treatment strategies of oncocytic (oxyphilic or Hürthle cell) carcinoma, a variant of FTC, are similar to those of FTC because of similarities in the natural history and prognosis. However, it is important to distinguish the two entities. Radioactive iodine uptake in oncocytic carcinoma is much less than that of FTC; hence, postoperative diagnosis and ablation of residual disease with RAI are more difficult [122].
11.8.6.7 Treatment of Medullary Thyroid Carcinoma (MTC)
Prior to surgery, patients with MTC should have baseline calcitonin levels and assessment of catecholamine secretion to rule out a pheochromocytoma. Medullary carcinoma must be treated with a TT since it may be unknown if the patient is part of a familial syndrome and therefore would be expected to have multicentric disease. A central LN dissection is indicated for occult cancers detected after family screening, whereas palpable lesions require an ipsilateral modified radical neck dissection.
11.8.6.8 Treatment of Anaplastic Carcinoma
Though rare, anaplastic carcinomas are very aggressive neoplasms with a very poor prognosis. Two-thirds of patients die within 1 year of diagnosis. In the rare case that all gross disease can be excised, resection may be appropriate. Chemotherapy and radiotherapy may be used for palliative treatment.
11.8.6.9 Nondiagnostic Biopsies
The results of FNAB, even with US guidance, may be repeatedly nondiagnostic. This particularly occurs with cystic nodules. The rate of malignancy in these nodules is extremely low, and observation may be appropriate. However, diagnostic lobectomy should be performed after a repeat nondiagnostic FNAB. The routine use of frozen section is excessively costly, and false-positive results could lead to unnecessary thyroidectomies. The decision to do a more extensive resection should be based on the patient’s history or characteristics of the nodule [123].
11.9 Thyrotoxicosis
11.9.1 Introduction
Thyrotoxicosis refers to a syndrome characterized by signs and symptoms of sympathetic overactivity and hypermetabolism due to excessive amounts of thyroid hormone. Hyperthyroidism is defined as excess synthesis or secretion of thyroid hormone by the thyroid gland. Some patients with thyrotoxicosis, such as those with subacute thyroiditis or excess thyroxin intake, do not have hyperthyroidism (Table 11.14).
Table 11.14
Causes of thyrotoxicosis
Associated with hyperthyroidism | Not associated with hyperthyroidism |
|---|---|
Graves’ disease Toxic multinodular goiter Solitary toxic adenoma Iodine-induced Hashimoto’s thyroiditis Thyrotropin-producing pituitary tumor Thyroid hormone resistance syndromes | Subacute thyroiditis Radiation thyroiditis Excess thyroid hormone ingestion (iatrogenic, thyrotoxicosis factitia) Struma ovarii Functioning metastatic thyroid cancer |
11.9.2 Clinical Manifestations
The clinical manifestations of thyrotoxicosis are diverse and result from increased thyroid hormone levels that sensitize nerve cells to catecholamines and cause the symptoms of increased sympathetic nervous system activity. Older patients usually have fewer and more subtle symptoms (apathetic hyperthyroidism). They also more frequently present with cardiovascular manifestations such as atrial fibrillation, angina, or congestive heart failure (CHF).
11.9.2.1 Symptoms
Patients complain of a neck swelling of variable size in addition to symptoms of toxicity, which include nervousness, anxiety, irritability, weight loss despite increased appetite, palpitations, heat intolerance, excessive sweating, insomnia or sleep disturbances, fatigue, weakness, hair loss, brittle nails, dyspnea on effort, increased frequency of bowel movements, reduced libido, impaired fertility, and menstrual disturbances.
11.9.2.2 Local Examination
The thyroid gland becomes diffusely enlarged with a smooth surface and fleshy consistency. In moderate and severe cases, pulsations due to increased vascularity could be seen (inspection), a thrill may be felt (palpation), and a bruit may be heard (auscultation). The skin overlying may show dilated veins.
11.9.2.3 General Examination
Findings of general examination depend on the severity of the condition and age of the patient. They are summarized in Table 11.15.
Table 11.15
General examination findings in thyrotoxicosis
Systems/organs | Manifestations |
|---|---|
Body metabolism | Increased metabolism leads to excessive sweating, loss of weight in spite of good appetite, and intolerance to hot weather |
Nails | The junction of the nail with its bed becomes straight or concave (thyroid acropachy) and onycholysis |
Digestive system | Diarrhea, nausea and vomiting (thyrotoxic crisis), abdominal distention, increased glucose intolerance, and glycosuria (polyphagia and polydipsia) |
Respiratory system | Dyspnea on effort |
Nervous system | Increased irritability, nervousness, easy excitability, fine tremors, choreiform movement of the hands and arms, and frank psychosis (in severe cases) |
Cardiovascular system | Attacks of palpitation on exertion or on rest, tachycardia, cardiac arrhythmias superimposed on a sinus tachycardia (as the disease progresses) in the form of multiple extrasystoles, paroxysmal atrial tachycardia, paroxysmal atrial fibrillation, persistent atrial flutter not responsive to digoxin, and congestive heart failure (CHF) |
Musculoskeletal system | Myopathy (weakness), osteoporosis (vertebral collapse or fractures), and pretibial myxedema (localized, bilateral leathery thickening of the skin of the fronts of the legs) |
Eye manifestations | |
Lid retraction (Dalrymple’s sign): Spasm of the involuntary part of the levator palpebrae superioris muscle (Muller’s muscles) causes widening of the palpebral fissure, so a band of white sclera appears between the margin of the upper eyelid and the cornea (no proptosis) | |
Lid lag (Von Graefe’s sign): Lack of harmony between movement of the upper eyelid and the eyeball. The eyelid lags behind the eyeball as the patient looks down following the examiner’s finger | |
Stellwag’s sign: Staring look due to infrequent blinking, a mild degree of lid retraction, or exophthalmos | |
Joffroy’s sign: No wrinkling of the forehead on looking upward due to weakness of the frontalis muscle | |
Moebius sign: Lack of convergence on looking at a near object for a long time due to easy fatigability of skeletal muscles of the eye | |
Gifford’s sign: Difficulty in passive eversion of the eye | |
Rosenbach’s sign: Involuntary spasm of the eyelids when closed | |
Backer’s sign: Abnormal pulsation of the retinal vessels | |
Thyrotoxic exophthalmos: Abnormal protrusion of the eye ball, usually bilateral but may be unilateral | |
Malignant exophthalmos: Edema and pigmentation of eyelids, diplopia and squint from ophthalmoplegia, edema and chemosis of conjunctiva, and corneal, which may end in panophthalmitis and blindness | |
11.9.3 Diagnosis
The TSH level is low in all patients with thyrotoxicosis except in rare cases of TSH-secreting pituitary tumors or pituitary resistance to thyroid hormone. When TSH level is low, T3 and T4 levels should be obtained. In patients with a suppressed TSH and a normal FT4 level, T3 levels are important to make a diagnosis of T3 toxicosis. Measurement of thyroid-stimulating immunoglobulins and antithyroid antibodies is useful for diagnosing Graves’ disease or thyroiditis in selected patients. A thyroid scintiscan can differentiate a hypofunctioning nodule in a patient with Graves’ disease from a solitary toxic nodule.
11.9.4 Differential Diagnosis
Thyrotoxicosis should be considered in children with a growth spurt, behavioral problems, or myopathy and in elderly patients with tachycardia or arrhythmias, unexplained diarrhea, and loss of weight. It should be differentiated from anxiety neurosis; organic diseases, which cause heart diseases, anemia, or gastrointestinal diseases; myasthenia gravis or other muscular disorders; menopausal syndrome; pheochromocytoma; and other causes of exophthalmos and primary ophthalmopathy.
11.10 Graves’ Disease
Graves’ disease (Basedow’s or Parry’s disease) is an autoimmune disorder with a genetic predisposition that typically affects young individuals between 20 and 40 years of age, with a female to male ratio that varies from 4:1 to 10:1. It is the most common cause of spontaneously occurring thyrotoxicosis, accounting for 60–90 % of all cases, and frequently occurs in association with other autoimmune diseases.
11.10.1 Pathogenesis
Reduced activation of suppressor T lymphocytes by specific antigen that occurs due to an inherited abnormality in antigen presentation encoded for by histocompatibility genes has been postulated to be the main defect behind the development of thyrotoxicosis [124]. The autoimmune dysfunction may be precipitated by environmental factors such as stress, infection, or trauma. The defect in suppressor T-cell function allows for thyroid-directed B lymphocytes, which are normally suppressed, to produce thyroid antibodies directed against the TSH receptor which stimulates the follicular cells in a manner similar to TSH. Graves’ disease is also characterized by thyroid autoantibodies to other antigens including thyroglobulin and thyroid peroxidase (TPO).
11.10.2 Pathology
Macroscopically, the gland is smooth and diffusely enlarged, reddish brown, and friable. The cut surface shows a varying picture depending on the colloid content, vascularity, and the amount of fibrous stroma. Microscopically, the gland shows marked vascularity, epithelial proliferation of the cells lining the follicles, and lymphocytic infiltration of the stroma.
11.10.3 Clinical Presentation
Patients with Graves’ disease typically have a diffuse symmetric goiter (Fig. 11.11) often with an audible bruit in addition to the variable presence of ophthalmopathy, dermopathy, and acropachy.
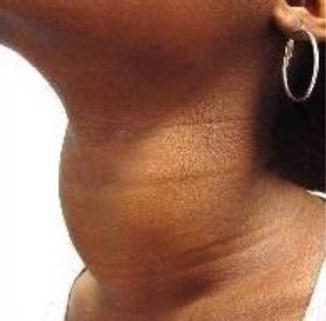

Fig. 11.11
Diffuse goiter with no nodules (smooth)
The extrathyroidal manifestations of Graves’ disease result from tissue deposition of glycosaminoglycans in response to the immune reaction against tissue antigens shared with the thyroid gland or antigens that cross-react with the TSH receptor.
Ophthalmopathy occurs more in patients with higher levels of thyroid receptor antibodies [125]. Eyelid retraction, lid lag, and stare look may occur with thyrotoxicosis regardless of its cause; however, periorbital edema, chemosis, exophthalmos (Fig. 11.12a–b), diplopia, and decreased visual acuity are more specific for Graves’ disease and occur as a result of edema, glycosaminoglycan deposition, leukocyte infiltration, and fibrosis of the orbit and extraocular muscles.
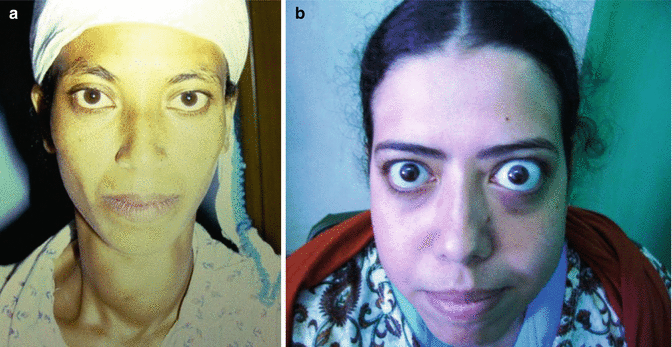

Fig. 11.12
(a) A 33-year-old patient with exophthalmos. (b) A 24-year-old lady with severe exophthalmos. Note white sclera all around
Dermopathy, in the form of pretibial myxedema, occurs in 0.5–4 % of patients. It consists of violaceous, plaque-like thickening or induration of the skin of the lower legs and feet and may be associated with pain and pruritus. Acropachy is rare (<1 %) and is manifested by thickening or clubbing of the fingers or toes, nail changes, and periosteal new bone formation.
11.10.4 Diagnosis
The diagnosis of Graves’ disease is usually established by the presence of hyperthyroidism, diffuse symmetric goiter, low TSH levels, and increased thyroidal RAI uptake (typically diffuse and symmetric). Measurement of thyroid receptor antibodies is not routinely necessary. Documentation of high titers of thyroid-stimulating immunoglobulins during pregnancy in women with a history of Graves’ disease may be important in predicting the risk of fetal and neonatal thyrotoxicosis [124]. Scintigraphy is used selectively to help differentiate thyrotoxicosis caused by Graves’ disease from toxic MNG, a solitary toxic thyroid nodule, or Graves’ disease with a concomitant dominant thyroid nodule.
11.10.5 Management
11.10.5.1 Radioactive Iodine (RAI)
In the United States, the majority of patients with Graves’ disease are treated with RAI, which emits beta particles that locally destroy the follicular cells of the thyroid gland. Nordyke and Gilbert reported that 90 % of their patients treated with a 10 mCi of RAI were cured. A higher dose may be required in patients with large thyroid glands (>50 g). However, they emphasized that delayed hypothyroidism develops in most patients with Graves’ disease treated with RAI regardless of the dose of 131I used. Accordingly, definitive treatment of hyperthyroidism is the most important consideration [126].
Symptomatic improvement usually occurs 6–8 weeks after receiving RAI treatment, and complications are rare. Serum TSH levels are monitored in all patients, and thyroid replacement with L-thyroxin is begun when TSH levels are elevated. A pregnancy test should be obtained prior to RAI administration in all women of the child-bearing age as it is contraindicated during pregnancy (and lactation).
11.10.5.2 Antithyroid Drugs (ATDs)
The thioamide drugs propylthiouracil (PTU) and methimazole are used for the treatment of Graves’ disease in children, pregnant or breast-feeding women, elderly patients with mild to moderate symptoms without a goiter, and in preparation of patients for RAI or surgery. Both drugs decrease thyroid hormone synthesis by a dose-dependent inhibition of the thyroid peroxidase enzyme. In addition, PTU blocks the peripheral conversion of T4–T3. When compared to methimazole, PTU has a greater protein binding which results in less passage across the placenta and the mammary epithelium, and that is why it is preferable in women who are pregnant or breastfeeding. The half-life of PTU is 2 h and is given 2–3 times/day, whereas methimazole has a half-life of 6 h and is given 1–2 times/day.
A high thioamide dose is given initially (PTU 100–200 mg or methimazole 10–30 mg). Once the free T4 and T3 levels have normalized, the thioamide dose is tapered to the lowest dose that will maintain a euthyroid state. Patients are kept on a maintenance dose usually for 1–2 years. Remissions are variable and most often last for <6 months. Hedley et al. reported that 40–80 % of patients develop recurrent thyrotoxicosis after discontinuation of ATDs [127].
Minor side effects of thioamides may be dose related or agent related and include skin rash, pruritus, urticaria, nausea, vomiting, myalgias or arthralgias, fever, and transient leukopenia. In such cases, drug dosage is reduced or the other thioamide drug substituted, though cross-sensitivity may occur. Major side effects are idiopathic and include agranulocytosis, hepatitis, aplastic anemia, and vasculitis.
11.10.5.3 Surgical Treatment
Surgery for treatment of Graves’ disease results in immediate symptomatic improvement and is indicated in pregnant patients intolerant to ATDs, large goiters with compressive symptoms, concomitant solitary cold nodule, patients who fail to respond to multiple doses of RAI, and those who prefer surgery.
The standard operation had been a bilateral subtotal thyroidectomy in an attempt at maintaining a euthyroid state postoperatively and reducing the risk of RLN injury and hypoparathyroidism while minimizing the risk of recurrent hyperthyroidism. However, it has been reported that 10–15 % of patients suffer from recurrent hyperthyroidism after bilateral subtotal thyroidectomy and the majority of patients become hypothyroid within 10 years. Consequently, total thyroidectomy (TT) has become the procedure of choice provided it is performed by experienced hands. In addition, TT has also been recommended for patients with severe or progressive ophthalmopathy and high TSH receptor antibody titers [125]. Total removal of the thyroid gland is advocated to decrease TSH receptor antibodies and other antibodies directed against the extraocular muscles, orbit, and optic nerve [125].
Prior to elective surgery, patients are rendered biochemically euthyroid using ATDs. A β- adrenergic-blocking agent (propranolol) is also used for symptomatic treatment and maintaining the resting heart rate between 60 and 80 beats/min. Preparing the patient for surgery is important in order to eliminate the risk of perioperative thyroid storm.
11.11 Toxic Multinodular Goiter (Plummer’s Disease)
Toxic MNG accounts for 5–15 % of cases of thyrotoxicosis. It more commonly affects women and typically occurs in elderly patients with a long-standing MNG. It is thought to occur due to progressive generation of autonomously functioning thyroid follicles overtime that have a greater capacity to synthesize T4 and T3, eventually resulting in toxic MNG.
11.11.1 Diagnosis
Thyrotoxicosis is generally mild in comparison to Graves’ disease, and infiltrative ophthalmopathy does not occur; however, patients often have large goiters with compressive symptoms, and cardiovascular manifestations occur more commonly because the patients are older. Laboratory evaluation reveals a low serum TSH level with or without elevated serum FT4 and/or T3 levels. Routine scintiscanning is not necessary.
11.11.2 Treatment
The goal in treatment is to eradicate all autonomously functioning thyroid follicles by surgical resection or 131I therapy. Because of the marked thyroid enlargement and frequent associated compressive symptoms, surgical resection, in the form of total thyroidectomy, is the usual treatment. A subtotal thyroidectomy is performed only when it can be accomplished without leaving abnormal thyroid tissue behind. Patients are also pretreated with ATD preoperatively to normalize their free T4 or T3 levels before proceeding with thyroidectomy.
Radioiodine may also be used for treatment. However, toxic MNGs can be resistant to RAI therapy, which is also usually not effective in alleviating compressive symptoms related to thyroid enlargement. Treatment with 131I is usually reserved for elderly patients with multiple concurrent medical problems that place them at high risk for surgery. Treatment with ATDs should be considered prior to RAI administration especially in patients with underlying heart disease. It must be discontinued 3–5 days prior to treatment to optimize RAI uptake and then resumed 1 week after treatment.
11.11.2.1 Solitary Toxic Nodule
A solitary toxic nodule is a discrete, autonomous, hyperfunctioning nodule that occurs in an otherwise normal thyroid gland and causes hyperthyroidism. It accounts for nearly 3–10 % of all cases of spontaneous thyrotoxicosis. The term hyperfunctioning nodule means that it takes up greater radioiodine than the normal adjacent thyroid tissue. Only 25 % of all hyperfunctioning nodules are toxic nodules. The term autonomous means it functions independently of the hypothalamic-pituitary-thyroid feedback mechanism and secretes thyroid hormone despite suppressed TSH levels.
11.11.3 Diagnosis
The clinical thyrotoxic manifestations of a solitary toxic nodule are generally milder than in patients with Graves’ disease. It usually occurs more commonly in women and in patients <50 years of age. Physical examination reveals a single, discrete nodule in the thyroid gland.
The initial diagnostic test is serum TSH, free T4, and free T3 levels. Hyperfunctioning nodules preferentially secrete T3, and so serum T3 levels are more likely to be elevated in patients with an autonomous nodule. A thyroid scan using 131I confirms the presence of a hyperfunctioning nodule. The pathology of a toxic solitary nodule is almost uniformly either a follicular adenoma or an adenomatous nodule. Carcinoma occurs in only about 1 % of cases [128].
11.11.4 Treatment
Patients with a hyperfunctioning thyroid nodule who are asymptomatic can be observed. Treatment is recommended in the presence of subclinical hyperthyroidism for patients who are at high risk of cardiac side effects, for postmenopausal women with decreased bone mineral density, and those who have a hyperfunctioning nodule >3 cm in diameter.
Both 131I and surgery (hemithyroidectomy) have been reported to be effective in the treatment of solitary toxic nodules. Surgical treatment has the advantages of immediate symptomatic relief, avoidance of radiation exposure to the normal thyroid tissue, and the low risk of complications. Both postoperative hypothyroidism and recurrence of hyperthyroidism are uncommon. Radioiodine treatment usually requires higher doses of 131I than are normally used for treatment of Graves’ disease. It has the disadvantages of delay in symptomatic relief; exposure of normal thyroid tissue to radiation, which may result in hypothyroidism in up to 35 % of patients; and concerns related to persistence of the nodule [129].
Other less attractive therapeutic options include ATDs and percutaneous ethanol injection. The ATDs are not curative and must be given lifelong because to avoid recurrence of hyperthyroidism. Their use is limited to preparing patients for surgical or RAI treatment, but may also be considered in elderly patients with medical problems that preclude surgery or RAI therapy. Ultrasound-guided ethanol injection is effective in reversing hyperthyroidism, but it requires multiple painful injections and can be complicated by transient RLN paresis.
11.12 Thyroiditis
Thyrotoxicosis secondary to thyroiditis is uncommon. It is typically transient and self-limited. It may occur as a result of chronic lymphocytic or Hashimoto’s thyroiditis (hashtoxicosis), silent (painless) thyroiditis, subacute (de Quervain’s) thyroiditis, and RAI-induced thyroiditis. In contrast to hashtoxicosis in which RAI uptake is increased, silent, subacute, and RAI-induced thyroiditis are all characterized by the inability to trap iodine, follicular cell destruction, and release of preformed thyroid hormone resulting in thyrotoxicosis with a low RAI uptake.
11.12.1 Hashimoto’s Thyroiditis
Thyrotoxicosis in patients with Hashimoto’s thyroiditis typically occurs in the early course of the disease and is transient in nature. It is thought to be the result of lymphocyte production of stimulatory anti-TSH receptor antibodies, which are present in 10–25 % of all patients with chronic lymphocytic thyroiditis. Those patients have marked elevation of antithyroglobulin and antithyroid peroxidase (antimicrosomal) antibody titers and focal or diffuse lymphocytic infiltration of the thyroid gland. Most patients are women between the ages of 30 and 50 years. They may have a firm goiter and rarely ophthalmopathy. As the disease progresses, thyrotoxicosis resolves and hypothyroidism develops. If symptoms of thyrotoxicosis become problematic, a beta-adrenergic blocking agent or ATD may be used. Patients are followed up clinically, and their serum TSH levels are monitored for the inevitable development of hypothyroidism, which will require hormone replacement therapy.
11.12.2 Silent (Painless) Thyroiditis
Silent thyroiditis is the major cause of thyrotoxicosis in patients with low RAI uptake. It is an autoimmune disorder which accounts for <5 % of all cases of thyrotoxicosis. It is a form of lymphocytic thyroiditis characterized by single or recurrent episodes of acute inflammation of the thyroid gland resulting in release of stored thyroid hormone. Patients are usually women between the age of 30 and 40 years. Symptoms of thyrotoxicosis are usually acute, mild, and self- limited and may be followed by transient hypothyroidism. Clinically, patients may have a firm, non-tender goiter. Antithyroid peroxidase and antithyroglobulin antibodies may be elevated, and serum thyroglobulin level is markedly elevated. In general, the condition requires no therapy, unless symptoms become problematic. In such cases, a beta-adrenergic antagonist and anti-inflammatory therapy with prednisone can be used. Because increased thyroid hormone synthesis is not the cause of thyrotoxicosis, ATDs are not effective. Surgical or RAI treatment may be beneficial in the rare patient with recurrent disabling episodes of silent thyroiditis with thyrotoxicosis.
11.12.3 Subacute Thyroiditis (de Quervain’s, Granulomatous, or Giant Cell Thyroiditis)
Subacute thyroiditis is a subacute, self-limited inflammatory condition of the thyroid gland, characterized by neck pain, fever, myalgias, malaise, mild to moderate thyroid enlargement, exquisite neck tenderness, and symptoms of thyrotoxicosis, which occur during the initial phase of inflammation. Etiology is multifactorial. A viral infection may trigger an abnormal cell-mediated immune response directed at the thyroid follicular cells causing follicular cell destruction and release of preformed thyroid hormone. A genetic predisposition may also be involved as suggested by the association of the HLA BW35 haplotype with subacute thyroiditis in certain patients [130]. Diagnosis is supported by the presence of a markedly elevated erythrocyte sedimentation rate (ESR), an increased serum thyroglobulin level, and a suppressed RAI uptake. Treatment is primarily supportive using nonsteroidal anti-inflammatory agents, or prednisone. Thyrotoxicosis usually requires no treatment and resolves within 3–6 weeks. If symptoms become problematic, a beta-adrenergic blocking agent, but not ATDs, may be given. If follicular cell destruction is extensive, hypothyroidism may develop during the recovery phase. Nevertheless, nearly 95 % of patients become euthyroid within 6 months of onset.
11.13 Iodine-Induced Thyrotoxicosis
Iodine-induced thyrotoxicosis usually occurs in elderly patients with a preexisting MNG who are given a large iodine (I2) load (e.g., oral expectorants, intravenous contrast material, etc.). It is the only cause of hyperthyroidism with a low RAI uptake and accounts for <1 % of all causes of thyrotoxicosis. Pathogenesis is not fully understood. In normal individuals, large doses of iodine lead to an inhibition of I2 transport and a rapid decrease in thyroid hormone synthesis and release (Wolff-Chaikoff effect). It may also occur as a result of supplying excess I2 to areas of autonomous function in the thyroid gland (Jod-Basedow effect) or due to increase in the I2 set point of the thyroid gland which leads to increased thyroid hormone synthesis. Diagnosis is suspected by a history of a recent exogenous I2 load in a patient with a goiter and is supported by a serum iodide concentration >1.5 mg/dl and a 24-h urinary iodide excretion >1000 mg. Treatment most often consists of discontinuation of the iodide source, although this may be problematic in patients with refractory arrhythmias on amiodarone [131]. Thioamide drugs may also be used either alone or in combination with a beta-adrenergic antagonist and/or potassium perchlorate, which competitively inhibits I2 uptake by the thyroid gland. Radioiodine therapy is not an option because the high I2 load suppresses RAI uptake by the thyroid gland. Total thyroidectomy may be indicated in patients with amiodarone-induced thyrotoxicosis that is refractory to medical therapy or as an initial therapy for patients who present with resurgence of life-threatening cardiac arrhythmias [131].
11.14 Thyroiditis
11.14.1 Introduction
Thyroiditis represents about 20 % of all thyroid diseases [132] and is caused by several factors, most commonly autoimmune diseases (Table 11.16). Thyroid autoantibodies are mainly directed against thyroid peroxidase (TPO) or thyroglobulin (Tg) and have an association with defined HLA haplotypes implying a genetic predisposition [133]. Iodine therapy, viral infections, pregnancy, menopause, stress [134], and immune-modulating drugs such as interferon-α have also been linked to autoimmune thyroiditis. Except for Graves’ disease, most cases of autoimmune thyroiditis present initially with hyperthyroidism that returns to euthyroidism or falls to permanent hypothyroidism (subclinical or overt).
Table 11.16
Etiology of thyroiditis
Autoimmune thyroiditis | Nonimmune thyroiditis |
|---|---|
Chronic lymphocytic thyroiditis (Hashimoto’s) | Acute infectious thyroiditis |
Fibrotic variant of Hashimoto’s thyroiditis | Radiation-induced thyroiditis |
Atrophic thyroiditis (primary myxedema) | Palpation/trauma-induced thyroiditis |
Variants of autoimmune thyroiditis | Sarcoidosis |
Postpartum thyroiditis | Vasculitis-associated thyroiditis |
Silent or painless thyroiditis | Postoperative necrotizing thyroiditis |
Subacute de Quervain’s thyroiditis | Drug-induced thyroiditis |
Fibrotic Riedel’s thyroiditis | Carcinoma-associated thyroiditis |
Clinically, thyroiditis is divided into acute, subacute, and chronic forms [135], and patients may present either with severe thyroid pain (e.g., acute suppurative thyroiditis, subacute de Quervain’s thyroiditis, radiation thyroiditis, traumatic thyroiditis) or without evident inflammation but with goiter or thyroid dysfunction (e.g., silent thyroiditis, Hashimoto’s or Riedel’s thyroiditis).
11.15 Autoimmune Thyroiditis
11.15.1 Hashimoto’s Thyroiditis (Chronic Lymphocytic Thyroiditis, Struma Lymphomatosa)
Hashimoto’s thyroiditis is the most frequent autoimmune thyroiditis and the most common cause of hypothyroidism [132]. Women are 10–20 times more affected than men, with a peak incidence in the fifth decade of life. A genetic association with the haplotypes HLADR3, HLADR4, and HLADR5 has been reported. Many other autoimmune diseases are associated with Hashimoto’s thyroiditis such as Graves’ disease, juvenile diabetes, Addison’s disease, pernicious anemia, rheumatoid arthritis, Sjogren’s syndrome, and systemic lupus erythematosus (SLE).
Hashimoto’s thyroiditis is caused by a breakdown in self-tolerance to thyroid autoantigens. This is exemplified by the presence of circulating autoantibodies against Tg and thyroid TPO in the vast majority of cases. The inciting events have not been elucidated, but possibilities include abnormalities of regulatory T cells or exposure of normally sequestered thyroid antigen. Induction of thyroid autoimmunity is accompanied by a progressive depletion of thyroid epithelial cells by apoptosis and replacement of thyroid parenchyma by mononuclear cell infiltration and fibrosis. Multiple immunological mechanisms may contribute to thyroid cell death, including CD8+ cytotoxic T-cell-mediated cell death, cytokine-mediated cell death, and, less likely, binding of antithyroid antibodies followed by antibody-dependent cell-mediated toxicity.
Patients with Hashimoto’s thyroiditis mostly present with a painless homogeneous goiter and manifestations of hypothyroidism. Low levels of thyroid hormones with high TSH and circulating thyroid autoantibodies against TPO (in 70–90 % of cases) and Tg (in 40–70 % of cases) confirm the diagnosis [132]. Occasionally, the patient presents initially with hyperthyroidism (hashtoxicosis) associated with the presence of anti-TSH receptor antibodies. The FNAC may frequently show Hürthle cells, and it may be difficult to distinguish Hashimoto’s thyroiditis at times from a follicular neoplasm, PTC, or low-grade MALT lymphoma. Immunohistochemistry studies may help to reach the diagnosis.
Being an autoimmune disease, the clinical course of Hashimoto’s thyroiditis is one of relapsing episodes, with up to 25 % of the patients showing a spontaneous recovery. The binding of autoantibodies to the thyrocytes accounts for complement and T-lymphocyte-mediated lysis of the thyrocytes and non-regulated release of T3 and T4, resulting in the transient hyperthyroidism occasionally noted. Later on, destruction of the thyroid parenchyma may lead to permanent hypothyroidism. Replacement thyroid hormone therapy is indicated when overt hypothyroidism is identified, and most patients will require lifelong therapy.
Fibrotic Hashimoto’s Thyroiditis
A fibrotic variant of Hashimoto’s thyroiditis accounts for up to 10 % of cases, mainly in elderly patients with a preexisting goiter. It is characterized by a rapid increase of goiter size, which may lead to the suspicion of malignancy, Riedel’s fibrosing thyroiditis. However, the extensive fibrotic changes and metaplasia noted on biopsies are always limited to the gland.
Atrophic Hashimoto’s (Autoimmune) Thyroiditis (Primary or Idiopathic Myxedema)
The atrophic autoimmune thyroiditis is the cause of primary myxedema and should not be confused with end-stage fibrotic Hashimoto’s thyroiditis. Most of the patients do not show signs or symptoms of hypothyroidism until the fourth to sixth decade of life, and women are five times more affected than men [136].
11.15.2 Focal Lymphocytic Thyroiditis (Focal Autoimmune Thyroiditis, Chronic Nonspecific Thyroiditis)
This low-grade autoimmune thyroiditis is characterized by focal lymphocytic infiltrates of <5 % of the thyroid gland and is found coincidentally in 50 % of women’s and 25 % of men’s autopsies, without clinical relevance.
11.15.3 Postpartum Thyroiditis
A postpartum thyroiditis occurs in 2–16 % of women within 1 year after delivery (or abortion) [137, 138]. The disease represents an exacerbation of a preceding (undiagnosed) autoimmune thyroiditis and is classically linked to the haplotypes HLADR3, HLADR4, and HLADR5. Most patients (85 %) develop autoantibodies against TPO and Tg, which may disappear with time. Women with a known autoimmune thyroiditis prior to pregnancy and an elevated titer of autoantibodies against Tg during pregnancy nearly always suffer from a postpartum exacerbation of their autoimmune thyroiditis. Clinically, patients may show a transient hyperthyroidism state, which rapidly converts to hypothyroidism, and then to euthyroidism within 12 months. Treatment consists of thyroid hormone replacement when required.
11.15.4 Subacute de Quervain’s Thyroiditis (Granulomatous, Pseudotuberculous, or Giant Cell Thyroiditis)
Subacute de Quervain’s thyroiditis is a self-limiting disease accounting for 0.5–3 % of all thyroid diseases and lasts for up to 2 months [139]. A post-viral cytokine-mediated inflammation of the thyroid is suspected because of the seasonal frequency and association with upper respiratory tract infection. In 50 % of patients, antibodies against mumps, measles, influenza, adenovirus, coxsackievirus, or echovirus are found. Furthermore, a genetic predisposition exists with the haplotype HLA-Bw35.
Women are 3–6 times more affected than men, with a peak incidence between the second and fifth decades of life. Symptoms include pain in the neck that irradiates to the jaw, ear, face, and down to the chest, fever, lassitude, and weakness. Physical examination reveals an exquisitely tender and enlarged gland. The erythrocyte sedimentation rate (ESR) is markedly elevated. Initially, there is transient hyperthyroidism from destruction of the thyroid follicles by the local inflammation process. Later on, hypothyroidism emerges due to inability to cope with the body’s demand for thyroid hormones, and finally, as healing occurs, euthyroidism is restored. However, permanent hypothyroidism requiring replacement therapy may occur in 15 % of patients, and recurrence may be noted, at a low rate of 4 % [140].
Subacute should be differentiated from acute suppurative thyroiditis. In de Quervain’s thyroiditis, the gland sonographically reveals irregular hypoperfused areas instead of hyperperfused tissue seen with acute suppurative thyroiditis. On FNA, the differential diagnosis further includes palpation thyroiditis, in addition to other granulomatous diseases such as sarcoidosis, tuberculosis (TB), and rheumatoid diseases.
Treatment is supportive with nonsteroidal anti-inflammatory drugs (NSAIDs) and β-blockers in severe cases with hyperthyroidism. Corticosteroids are useful when the NSAID medication is not successful, and symptoms usually improve within 2–3 days after the initiation of treatment. However, it may take about 4 weeks for the disappearance of the thyroid mass.
11.15.5 Painless Thyroiditis (Subacute Lymphocytic Thyroiditis)
Patients with painless thyroiditis present with a diffuse but mild enlargement of the thyroid gland. Thyroid function tests reveal a transient hyperthyroidism, followed by hypothyroidism. Autoantibodies against TPO and Tg are found as well as an association with HLADR3 and HLADR5 haplotypes. Histological examination reveals lymphocytic infiltration with destruction of follicles (in contrast to Hashimoto’s thyroiditis) and absence of giant cell granulomas (in contrast to subacute thyroiditis). Women are more often affected than men with a peak of incidence in middle life and in the postpartum period. It is a self-limited disease and rarely necessitates thyroid replacement therapy.
11.15.6 Riedel’s Fibrosing Thyroiditis
Riedel’s thyroiditis is a rare chronic thyroiditis in which the thyroid gland is replaced by fibrous tissue. The underlying etiologic mechanisms are unclear, though an autoimmune relation is suspected, due to elevated titers of thyroid autoantibodies. It is generally considered as a part of a multifocal fibro-inflammatory process also involving other tissues such as the mediastinum, liver, lung, retroperitoneum, and orbit.
Women in middle to advanced ages are more affected than men. The clinical picture often resembles malignancy due to the hard consistency of the gland. Patients usually complain of a rapid indolent enlargement of the thyroid that becomes very hard and may suffer from neck discomfort, dysphagia, hoarseness of voice, and hypoparathyroidism (due to involvement of the RLN and parathyroid glands). Approximately, 30–40 % of patients develop overt hypothyroidism. Physical examination, laboratory analysis, cytology, and imaging features are not useful for differentiating between Riedel’s thyroiditis and neoplastic diseases or the fibrotic variant of Hashimoto’s thyroiditis [141].
Histological examination is necessary to establish the final diagnosis, and so surgical biopsy is mandatory. The differential diagnosis further includes anaplastic carcinoma and sarcoma of the thyroid. In contrast to the fibrotic variant of Hashimoto’s thyroiditis where fibrosis is strictly limited to the gland, Riedel’s thyroiditis displays a dense fibrotic replacement of thyroid parenchyma that penetrates the capsule and extends into contiguous neck structures. Once the diagnosis is confirmed, treatment is supportive with thyroid hormone replacement, when required.
11.16 Non-autoimmune Thyroiditis
Nonimmune thyroiditis consists of a heterogeneous and rare group of thyroid inflammatory diseases. Some of them are infectious while others are clearly iatrogenic, such as drug-induced thyroiditis and postoperative necrotizing thyroiditis.
11.16.1 Acute Infectious Thyroiditis (Acute Suppurative Thyroiditis)
Infectious thyroiditis is a rare disease of the thyroid caused by bacterial or fungal infection, though mycobacterial, parasitic, and viral forms of thyroiditis have also been described, particularly in immunosuppressed hosts. The thyroid gland appears to be relatively resistant to infection. The protective mechanisms include a rich vascular supply, extended lymphatic drainage, a fibrous capsule, fascial planes that anatomically separate the thyroid from other cervical structures, and the high iodine content of the gland, which may have bactericidal effect. Infection of the gland occurs, either through hematogenous spread from a primary focus or by direct extension from adjacent neck structures, especially in children [142]. Other less common sources of infection include neck trauma or lymphatic spread, or rarely, surgical site infections [143]. The most common predisposing factor for suppurative thyroiditis is immunosuppression associated with HIV, TB, old age, or debilitating diseases. Other predisposing factors include preexisting thyroid diseases, such as MNG, autoimmune thyroiditis, and cancer [143–165].
Patients usually present with fever and a painful, mostly unilateral, enlargement of the thyroid and local inflammatory signs. The thyroid function tests are usually normal, but a slight hyper- or hypothyroidism may occur. Laboratory tests show leukocytosis and elevated ESR. Neck US shows patchy hyperperfused areas in the thyroid with liquid content (pus) when an abscess is present. An FNAC and cultures can identify the pathogen identification and guide the antimicrobial treatment. Immunosuppressed patients tend to present with more chronic thyroid infections, bilateral disease, and less prominent signs and symptoms. Diagnosis may be reached with a high index of suspicion and aspiration biopsy.
The differential diagnoses include de Quervain’s thyroiditis, hemorrhage into a thyroid nodule, infected thyroglossal or branchial cyst, infected cystic hygroma, and cervical lymphadenitis. In addition to FNA, US helps to establish the diagnosis, which may further be refined by CT scan.
When an abscess is identified, surgical drainage is essential. Occasionally, complete resection of an affected gland is required. In patients with recurrent acute thyroiditis, an undetected fistula must be postulated. Complete removal of the infected fistula is therefore required to prevent recurrence. Injection of 0.5 % methylene blue solution through a Nelaton’s catheter into the fistula usually enables the complete resection of the tract. When the origin of the fistula is difficult to identify, transection of the inferior pharyngeal constrictor muscle makes intervention easier.
11.16.2 Drug-Induced Thyroiditis
Certain drugs have been reported to cause thyroiditis, such as chronic I2 therapy, long-term lithium therapy, and anticonvulsants (e.g., phenytoin, carbamazepine) [132]. Moreover, patients with chronic hepatitis or cancer treated with interferon-α will develop a painless thyroiditis in about 1–5 % of cases [146]. Elevated antithyroid antibodies are noted in a higher percentage in these patients, and permanent hypothyroidism or Graves’ disease may appear. Thus, TSH should be measured prior to initiation of interferon-α therapy and periodically during treatment [147].
Interleukin-2 (IL-2) is also used for immune-modulation and in malignant melanoma, renal cell carcinoma, and leukemia, alone or in combination with chemotherapy. A painless thyroiditis has been reported in about 2 % of the patients treated with IL-2. Finally, the antiarrhythmic drug amiodarone contains 35 % I2 and may cause a thyrotoxic crisis or, conversely, cause hypothyroidism. Amiodarone decreases the conversion of T4 to the biologically active T3. It is worth mentioning that if the decision is taken to stop amiodarone therapy, the drug is not eliminated for months due to its very long half-life [148].
11.16.3 Postoperative Necrotizing Thyroiditis
Postoperative necrotizing thyroiditis is a rare surgical complication owing to the very rich vascular supply of the thyroid [149, 150]. It is related to a trauma of the gland by vigorous manipulation at surgery or through repeated FNA [151]. Such manipulation could induce an acute thyroiditis, which in turn may lead to thyrotoxicosis or to a necrotizing thyroiditis. No predictive marker or factor has been identified. Histologically, the specimen typically shows postoperative granulomas, as found in other organs (bladder, prostate) following surgery.
11.16.4 Radiation Thyroiditis
Radiation thyroiditis occurs in a dose-related fashion after RAI or external beam radiation therapy (EBRT), which causes follicle destruction resulting in a transient hyperthyroidism, followed eventually by hypothyroidism. Neck pain and tenderness usually develop 5–10 days following treatment. Symptoms are mild and subside spontaneously in a week.
11.16.5 Other Causes of Nonimmune Thyroiditis
Other causes of thyroiditis are related to a local process, such as an acute hemorrhage into a thyroid cyst or nodule. Palpation thyroiditis refers to a mild, self-limited thyroiditis occurring after physical examination, surgery, or trauma to the thyroid. It is not associated with any thyroid disease. Finally, thyroiditis may be caused by systemic diseases, such as a vasculitis-associated thyroiditis (phenytoin therapy), sarcoidosis, [152], metastatic cancer, or a globus hystericus.
11.16.6 Indications of Surgery in Thyroiditis
Surgical interventions are exceptionally indicated for the management of a thyroiditis, accounting for <1 % of all thyroid procedures [153]. Fortunately, patients with autoimmune thyroiditis are effectively managed with thyroid hormone replacement therapy, and surgical treatment is the exception. Technical difficulties are encountered by the surgeon as the glands are firm, rigid, and highly vascular and the surrounding tissues are inflamed with lymphadenopathy, rendering the parathyroids and RLNs at risk. In the rare instance where a large Hashimoto’s goiter may develop and become symptomatic, total thyroidectomy (TT) is an option [135, 153, 154]. Moreover, as thyroiditis patients bear a higher risk of developing thyroid carcinoma, a cold nodule suspicious on FNA may indicate a thyroid lobectomy. Similarly, the rapid growth of a chronic lymphocytic thyroid gland is suggestive of non-Hodgkin lymphoma (NHL). While TT may surgically cure a stage I lymphoma (confined to the thyroid), most thyroid lymphomas involve regional LNs and distant sites and require multimodal systemic therapy. Open biopsy or thyroid lobectomy is sufficient in these cases to establish the definitive diagnosis.
A subacute de Quervain’s thyroiditis exceptionally deserves thyroidectomy. This indication is given when intractable neck pain is present in spite of a consequent analgesic and L-thyroxin replacement therapy over a period of 6 months [153]. The fibrotic variant of Hashimoto’s thyroiditis is characterized by a rapid enlargement of a preexisting goiter which causes suspicion of a thyroid cancer and consequently surgery. Riedel’s fibrosing thyroiditis often requires an open biopsy to confirm the diagnosis and rule out an anaplastic carcinoma or isthmectomy and/or lobectomy to decompress the trachea and esophagus. However, thyroidectomy can be highly demanding because of the dense fibrotic reaction extending beyond the thyroid, which puts the surrounding structures at risk of injury. Amiodarone-induced thyrotoxicosis in the setting of a rare patient with otherwise intractable arrhythmia is an indication for thyroidectomy. Finally, the acute suppurative thyroiditis is a classic indication for surgical drainage followed by antibiotic therapy and rarely for lobectomy when the suppurative process is necrotizing.
11.17 Malignant Thyroid Disease: Introduction
Thyroid cancer is the most common endocrine malignancy, but only represents 1 % of all newly diagnosed malignancies [32]. The wide spectrum of aggressiveness of thyroid cancer is extraordinary, ranging from differentiated malignancies in which most patients live out close to their normal lifespan to anaplastic varieties that are almost universally lethal. It is crucial to ensure that patients presenting with thyroid nodules are not over- or undertreated.
11.17.1 Classification
11.17.1.1 World Health Organization (WHO) Classification
A classification of thyroid tumors, as suggested by WHO, is shown in Table 11.17 [155].
Table 11.17
WHO classification of thyroid neoplasms
I. Primary tumors |
1. Epithelial tumors: |
(a) Tumors of follicular cells: |
Benign (follicular adenoma) |
Conventional |
Variants |
Malignant (carcinoma) |
*Differentiated: |
Follicular carcinoma. |
Papillary carcinoma: |
Conventional |
Variants |
*Poorly differentiated: |
Insular |
Others |
*Undifferentiated (anaplastic) |
(b) Tumors of C (and related neuroendocrine) cells: |
Medullary carcinoma |
Others |
(c) Tumors of follicular and C cells |
2. Sarcoma |
3. Lymphoma (and related hematopoietic neoplasms) |
4. Miscellaneous neoplasms |
II. Secondary tumors |
III. Tumorlike lesions |
1. Tumors with oncocytic (Hürthle cell) features: |
Oncocytic adenoma (Hürthle cell adenoma) |
Oncocytic carcinoma (Hürthle cell carcinoma) |
Papillary oncocytic (Hürthle cell tumors) |
2. Tumors with clear cell features |
3. Tumors with squamous features |
4. Tumors with mucinous features |
11.17.1.2 Pathological Classification
Most thyroid tumors arise from the follicular cells, and most are well differentiated. Poorly differentiated and undifferentiated types are rare (Table 11.18). Well-differentiated carcinomas include papillary and follicular carcinomas.
Table 11.18
Pathological classification of thyroid neoplasms
I. Well-differentiated (low-grade malignancy) | 1. Usual papillary thyroid carcinoma (PTC) |
2. Micro-carcinoma (<1 cm) | |
3. Follicular variant of PTC | |
4. Usual follicular thyroid carcinoma (FTC) | |
5. Hürthle cell carcinoma | |
II. Intermediate differentiation | 1. Medullary thyroid carcinoma (MTC) |
2. Diffuse sclerosing variant of PTC | |
3. Columnar cell variant of PTC | |
4. Insular carcinoma | |
5. Tall cell variant of papillary carcinoma | |
III. Poorly differentiated (high-grade malignancy) | Anaplastic (undifferentiated) carcinoma |
11.17.2 Screening
At present there is no screening program to detect thyroid cancer for the general population. Screening is possible for familial MTCs associated with specific oncogene mutations. The genetic basis of papillary, follicular, and anaplastic thyroid cancer has been investigated, and the roles and potential prognostic value of several genes, e.g., RET, TRK, ras, BRAF, and p53, have been identified. Testing for these genes is not routinely available in clinical practice [156].
While screening generally is not possible, a family history for thyroid cancer should be taken in each case, and if there is a strong familial incidence of thyroid cancer or association with other cancers, genetic advice should be considered in appropriate cases from the regional genetics service.
11.17.3 Risk Factors for Thyroid Carcinoma
11.17.3.1 History Taking
It is important to focus on gender; age; duration of the swelling and, more importantly, its rate of growth; history of neck irradiation; and family history of thyroid cancer. Other associated symptoms such as difficulty swallowing or breathing would suggest compressive effect. A hoarse voice is a strong indication of RLN palsy and malignancy [73]. Other associated diseases should also be noted. These are summarized in Table 11.19 [103].
Risk factors | |
|---|---|
History taking | Physical examination |
Male gender Age <20 or >70 years Low-iodine diet (endemic goiter) Radiation exposure (during childhood) Family history of thyroid cancer Hashimoto’s thyroiditis (risk of lymphoma) Family or personal history of thyroid adenoma Multiple endocrine neoplasia (MEN II a, b) Gardner syndrome Familial adenomatous polyposis Cowden disease Nonpolyposis colon cancer syndrome (NPCC) | True vocal cord fixation Very firm or hard nodules Fixation to skin, surrounding musculature, and extracapsular spread Associated cervical lymph nodes (LNs) Recent rapid growth Large nodules (>4 cm) Persistent diarrhea (MTC) |
Male gender carries 2–3 times the risk of thyroid cancer as compared to women [74, 157]. At an age below 20 years, the risk of malignancy is doubled, while above 70 years, the risk of malignancy is quadrupled [74, 157].
There is a 40 % absolute risk of malignancy for a thyroid nodule in a patient with previous exposure to irradiation, particularly during childhood. Low dose carries a 100 times increased risk of malignancy (lifetime risk), while high dose carries a 300 times increased risk (lifetime risk). The latency period is 10–15 years, and cancer mostly occurs 20–30 years after radiation exposure [73, 158]. History of rapid growth in a few weeks [73, 158, 159] and the presence of hoarseness of voice or vocal cord palsy indicate a higher risk of malignancy [159].
11.17.3.2 Clinical Examination
The larger tumor size, especially when >4 cm, and the presence of obstructive symptoms indicate higher risk of malignancy [157, 159]. Firm/hard consistency or fixed swelling indicates high risk, while a soft, mobile, or cystic swelling indicates a low risk of malignancy [159]. Presence of cervical lymphadenopathy indicates high risk of malignancy [159] (Table 11.19).
Flexible laryngoscopy is important to assess vocal cord movements. The so-called “classic” red flag physical finding is true vocal cord paralysis. However, this finding by itself can be of limited help in assessing malignant potential as the three main causes of unilateral vocal cord paralysis can be broadly categorized as iatrogenic, malignant, and idiopathic. Reviews of unilateral cord paralysis quote figures for malignancy ranging from 7 to 25 % with the vast majority due to lung cancer. If lung cancer is excluded, all other malignancies, including thyroid cancer, represent well under 10 % of unilateral vocal cord paralysis. In contrast, idiopathic causes account for 30–40 % of vocal cord paralysis cases.
11.18 Well-Differentiated Thyroid Cancer
Differentiated thyroid carcinoma (DTC) refers to both papillary and follicular carcinomas, which arise from the thyroid follicular cell. Among many unique features of DTC, two require special mention. First, age is the most important prognostic factor. It is interesting to note that the mortality in patients with thyroid cancer in the younger age group is extremely low, while that in the elderly patient is quite high. There is no other human cancer that parallels this biological behavior. This is the only cancer where age is included in the staging system. There is no stage III and IV cancer in patients below the age of 45 years [160–162]. Second, the presence of nodal metastasis has almost no prognostic implication. This clinical behavior is not seen in any other malignancy. In the majority of cancers, the presence of nodal metastasis decreases the survival by almost 50 %, while in well-differentiated thyroid cancer (WDTC), there is no apparent effect on outcome [163].
The mortality of DTC remains low; most deaths are directly related to the high-risk group, generally elderly patients with poorly differentiated histology or locally aggressive tumors. There is considerable debate and controversy about the management of the disease [164]. There are vigorous proponents of routine total thyroidectomy (TT), whereas other authors recommend less than TT, depending on the prognostic factors and risk groups [165].
11.18.1 Staging (Tables 11.20a, 11.20b, and 11.20c)
Primary tumor (pT) | |
pT1 | Intrathyroidal tumor, ≤1 cm in greatest dimension |
pT2 | Intrathyroidal tumor, >1–4 cm in greatest dimension |
PT3 | Intrathyroidal tumor, >4 cm in greatest dimension |
pT4 | Tumor of any size, extending beyond thyroid capsule |
pTX | Primary tumor cannot be assessed |
Regional LNs (cervical or upper mediastinal) | |
N0 | No nodes involved |
N1 N1a N1b | Regional nodes involved Ipsilateral cervical nodes Bilateral, midline, or contralateral cervical nodes or mediastinal nodes |
NX | Nodes cannot be assessed |
Distant metastases | |
M0 | No distant metastases |
M1 | Distant metastases |
MX | Distant metastases cannot be assessed |
Table 11.20b
Papillary or follicular carcinoma staging
Stage | Under 45 years | 45 years and older |
|---|---|---|
Stage I | Any T, any N, M0 | pT1, N0, M0 |
Stage II | Any T, any N, M1 | pT2, N0, M0 pT3, N0, M0 |
Stage III | pT4, N0, M0 Any pT, N1, M0 | |
Stage IV | Any pT, any N, M1 |
Stage | 10-year cancer-specific mortality (%) |
|---|---|
I | 1.7 |
II | 15.8 |
III | 30 |
IV | 6 |
11.18.2 Management of DTC
11.18.2.1 Surgical Treatment
Fine-needle aspiration cytology (FNAC) should be used in the planning of surgery. Patients with a PTC >1 cm or with high-risk FTC should undergo near-TT or TT, while those with PTC ≤1 cm or low-risk FTC may be treated with thyroid lobectomy alone. Serum thyroglobulin (Tg) should be checked in all postoperative patients with DTC, but not sooner than 6 weeks after surgery. Patients will normally start on L-thyroxin 100 μg daily after the operation. This should be stopped 2 weeks before 131I ablation or therapy.
Most patients with a tumor >1 cm, who have undergone a near-TT/TT, should have 131I ablation. Pregnancy and breastfeeding should always be excluded before administering 131I. Breastfeeding should be stopped 4 weeks and preferably 8 weeks before 131I ablation or treatment and should not be resumed. A post-ablation scan (3–10 days after 131I ablation) should be performed.
Patients treated with 131I will require L-thyroxin therapy in a dose sufficient to suppress the serum TSH to <0.1 mIU/L. L-thyroxin can be started 3 days after 131I in a dose sufficient to suppress TSH to <0.1 mIU/L. In low-risk patients, TSH <0.5 mIU/L is acceptable.
Reassessment with a whole-body scan (WBS) after stopping L-thyroxin for 4 weeks and stimulated serum Tg is indicated no earlier than 6 months after 131I ablation. If abnormal uptake of the tracer is detectable, a 131I treatment dose should be given and a posttreatment scan (3–10 days after 131I treatment) performed. The patient should then restart L-thyroxin.
11.18.2.2 Radioactive Iodine (RAI) Ablation and Treatment for DTC
Following a TT or near-TT, some radioiodine uptake is usually demonstrable in the thyroid bed. 131I-induced destruction of this residual thyroid tissue is known as radioiodine remnant ablation. Radioiodine therapy refers to administration of 131 I with the intention to treat recurrent or metastatic disease. The principles and procedures are similar for the administration of 131I for ablation or treatment.
Preparation for 131I Ablation or Therapy
Patients should adopt a low I2 diet for 2 weeks prior to 131I, and other sources of excess I2 should be eliminated (e.g., recent CT with contrast) [168]. If 131 I can be administered within 3–4 weeks of thyroidectomy, no thyroid hormone replacement is required in the interim period. This would usually allow TSH to rise to >30 mIU/L at the time of ablation. For most centers, however, the interval between thyroidectomy and 131I ablation will be longer. In such cases, patients should start T3 20 mg tds after surgery; this should be stopped 2 weeks before planned ablation to allow serum TSH to increase to >30 mIU/L.
If there is doubt about completeness of surgery, a pre-ablation scan can be performed to assess remnant size [169]. Demonstration of large thyroid remnants should lead to consideration of further surgery before 131I ablation.
Pregnancy must be excluded before 131I ablation. Breastfeeding must be discontinued for 4 weeks, preferably 8 weeks before 131I ablation or treatment, and should not be resumed. Pretreatment sperm banking should be considered in male patients likely to have >2 high-dose 131I therapies [170]. Adequate hydration at the time of treatment and for several days afterward, regular emptying of the urinary bladder, and avoidance of constipation help to prevent a reduction in sperm count.
Postoperative 131I Ablation
Patients >45 years with tumors >1.5 cm should receive 131I ablation to reduce local and distant recurrence and cancer mortality [171, 172]. The benefit of 131I ablation for low-risk patients may however be questionable. Other factors such as invasion, metastases, completeness of excision, and associated disease should be considered. Benefits of 131I ablation include:
1.
Eradication of all thyroid cells including residual postoperative microscopic disease and thus possible reduction of risk of local and distant tumor recurrence
2.
Reassurance to patients imparted by the knowledge that serum Tg is undetectable and iodine scan negative, implying that all thyroid tissue has been destroyed
4.
Increased sensitivity of monitoring by serum Tg measurements and possibly earlier detection of recurrent or metastatic disease [174]
Indications of Remnant Ablation with 131I
Patients should satisfy all the following criteria for 131I ablation to be omitted: complete surgery, favorable histology, tumor unifocal, ≤1 cm, N0, M0, or minimally invasive FTCs, without vascular invasion, <2 cm [177], and no extension beyond the thyroid capsule.
Definite Indications
The presence of any of the following is a definite indication for 131 I ablation: distant metastases, incomplete tumor resection, or complete tumor resection but high risk of recurrence or mortality (tumor extension beyond the thyroid capsule, or >10 involved LNs and >3 LNs with extracapsular spread) [178].
Probable Indications
Any one of the following categories is a “probable” indication for 131I ablation: less than TT, status of LNs not assessed at surgery, tumor size >1 cm and <4 cm, tumors <1 cm with unfavorable histology (tall cell, columnar cell, or diffuse sclerosing papillary cancers, widely invasive or poorly differentiated follicular cancers), and multifocal tumors <1 cm [178].
Short-Term and Long-Term Side Effects of 131I Ablation Treatment
The main side effect is transient hypothyroidism, unless rh TSH is used [179–183]. The possible early effects include abnormality of taste and sialadenitis (can be minimized by good hydration), nausea (can be minimized by antiemetics), neck discomfort and swelling within a few days of RAI (rare, simple analgesics should be tried initially, but a short course of steroids may be necessary), radiation cystitis, radiation gastritis, bleeding into secondary deposits, and edema in cerebral secondary deposits (extremely rare) [178].
The possible late effects include dry mouth, abnormal taste, sialadenitis, lacrimal gland dysfunction, lifetime risk of leukemia and secondary cancers (0.5 %) [184], radiation fibrosis [185], increased risk of miscarriage (may persist for 1 year after 131I therapy) [186], and infertility in men [170].
11.18.2.3 External Beam Radiotherapy (EBRT) for the Treatment of DTC
External beam radiotherapy (EBRT) is only occasionally used in the treatment of DTC. Postoperative adjuvant EBRT may be indicated to reduce local recurrence in patients at high risk due to residual disease where further surgery is not appropriate [158, 187]. High-dose EBRT as part of primary treatment is indicated for unresectable tumors that do not concentrate RAI and unresectable bulky tumors in addition to RAI treatment.
11.18.3 Follow-Up of WDTC
Follow-up should be lifelong because (1) the disease has a long natural history, (2) late recurrences are not rare and can be treated successfully, (3) regular follow-up is also necessary for monitoring of treatment (TSH suppression, the consequences of supraphysiological L-thyroxin replacement, treatment of hypocalcemia, (4) lifelong suppression of serum TSH level below normal (<0.1 mIU/L) is one of the main components of treatment in high-risk cases, and (5) patients should be monitored for late side effects of 131I treatment.
Surveillance for recurrence is based on (1) annual clinical examination, (2) annual measurement of serum Tg and TSH, and (3) diagnostic imaging and FNAC when indicated.
11.18.3.1 Voice Dysfunction
Voice dysfunction may result if there is ELN and/or RLN injury. It must be investigated if symptoms persist beyond 2 weeks after surgery. The patient should be referred to a specialist for direct and/or indirect laryngoscopy [188].
11.18.3.2 Management of Hypocalcemia
Serum calcium (Ca) should be checked on the day after surgery and daily until the hypocalcemia improves [189]. A decline in serum Ca in the first 24 h after surgery indicates the need for Ca supplementation [190]. If hypocalcemia develops, Ca supplement should be started at an initial dose of 500-mg elemental calcium three times daily. The dose is adjusted as indicated by the response. Occasionally IV Ca gluconate may be required. Mild asymptomatic hypocalcemia usually does not require treatment, although monitoring is indicated. If hypocalcemia does not improve or worsens, alfacalcidol should be added.
Hypoparathyroidism is often transient, and a predictor of this is increase in serum PTH at the time of occurrence of hypocalcemia [191]. Thus, most patients on calcitriol/alfacalcidol/Ca supplements can have this treatment withdrawn. Supplements should be slowly and gradually reduced and serum Ca monitored every few months until withdrawn and eucalcemia restored. Calcitriol/alfacalcidol/calcium supplement withdrawal should take place during euthyroidism.
If hypoparathyroidism is permanent, the lowest dose of supplements should be given to maintain the serum Ca at the lower end of the normal range, while avoiding hypercalciuria. In stable cases annual measurement of serum Ca is recommended. Close monitoring of serum Ca is needed to prevent hypercalcemia. After TT, 30 % of patients will need Ca supplement ± alfacalcidol. By 3 months, <10 % of patients will still need Ca [191].
11.18.3.3 Long-Term Suppression of Serum Thyrotropin
L-thyroxin should be used in preference to T3 for long-term suppression. The dose should suppress the TSH to <0.1 mIU/L [192] and should be adjusted by 25 μg (every 6 weeks) until serum TSH is <0.1 mIU/L). To achieve this, most patients may require 175–200 μg daily.
11.18.3.4 Measurement of Serum in Long-Term Follow-Up
Thyroglobulin (Tg) is secreted by both normal and cancerous thyroid cells. In patients who have not had a TT and 131I ablation, the interpretation of serum Tg measurements is limited by the inability to differentiate between tumor and thyroid remnants [193]. Detectable serum Tg is highly suggestive of thyroid remnant, residual, or recurrent tumor. A serum Tg rising with time while on suppressive thyroxin treatment highly suggests tumor recurrence or progression.
TSH-Stimulated Serum Tg Measurement
The diagnostic sensitivity of serum Tg measurements increases by elevated TSH concentration [194]. Tumor recurrence or progression can be diagnosed earlier by detecting increased Tg after TSH stimulation than by measuring Tg on suppressive thyroxin therapy; Tg should be measured when serum TSH is >30 mIU/L.
11.18.3.5 Role of US and Whole-Body 131I Scan (WBS) in Routine Follow-Up
After TT and postoperative 131I ablation, diagnostic WBSs have relatively low sensitivity in detecting residual or recurrent disease compared with measurement of serum Tg [195]. Ultrasound is sensitive for the detection of residual disease in the thyroid bed and metastatic disease in LNs. It may also have a particular role when serum Tg measurements are unreliable because of the presence of assay interference.
11.18.4 Recurrent/Persistent DTC
Early detection of recurrent disease (Fig. 11.13) can lead to cure or long-term survival, particularly if disease is operable or takes up RAI [173, 178]. Distant metastases develop in 5–23 % of patients with DTC, mainly in the lungs and bones.

Fig. 11.13
A 47-year-old lady with recurrent thyroid cancer. Note the swelling on the right side and the scar of previous surgery
11.18.4.1 Recurrence in the Thyroid Bed or Cervical LNs
Surgical re-exploration is the preferred method of treatment, usually followed by 131 I therapy [195]. Recurrent neck disease uncontrolled by surgery and 131I treatment is best treated by high-dose palliative EBRT. As patients are likely to survive for a significant period, radical EBRT (doses 50–66 Gy) is often necessary with a daily fractionation.
11.18.4.2 Metastases in the Lungs and Other Soft Tissue Areas
These sites are usually not amenable to surgery and should be treated with 131I therapy [185]. If the tumor takes up radioiodine, long-term survival is possible in such cases [196]. There is no maximum limit to the cumulative 131I dose that can be given to patients with persistent disease. A normal CBC must be confirmed prior to each 131I treatment, and impairment of renal function demands a lower dose [197].
11.18.4.3 Cerebral Metastases
EBRT has a palliative role in the treatment of cerebral metastases along with surgery if appropriate.
11.18.4.4 Bone Metastases
Extensive bony metastases are generally not curable by 131I treatment alone. For solitary or limited number of bony metastases that are not cured by 131I therapy, EBRT with or without resection and/or embolization should be considered in selected cases. The EBRT also has a very important role in the treatment of spinal cord compression for vertebral metastases [198].
11.18.4.5 Other Metastatic Sites
Metastasectomy or radiofrequency ablation may be helpful in cases with a limited number of metastases.
11.18.4.6 Palliative Care
Palliative care is not necessary in most patients with DTC because they are cured. High-dose palliative EBRT may be appropriate in good performance status patients with anticipated survival of >6 months. It also has a role in palliation of symptoms from fungating LNs, bleeding tumor, stridor, SVC obstruction, and dysphagia [199]. Stridor can also be alleviated by palliative surgery. Palliative chemotherapy may have a role in end-stage disease uncontrolled by surgery, 131I therapy, or EBRT.
11.19 Papillary Thyroid Cancer (PTC)
Papillary thyroid carcinoma (PTC), which is a differentiated type of thyroid cancer derived from follicular epithelial cells, is the most common histological type, constituting 80 % of all thyroid carcinomas [195]. Women are more affected than men in a ratio of 2:1–4:1. It has become the sixth most common cancer in women. It can present in any age group, the mean age at the time of initial diagnosis being approximately 40 years. Papillary carcinoma accounts for more than 90 % of thyroid malignancies in children [200].
11.19.1 Risk Factors
Both genetic and environmental factors may increase the risk of developing PTC. About 3 % of cases of PTC are familial [201]. Some familial syndromes known to be associated with PTC include familial adenomatous polyposis (FAP) and its variant, Gardner syndrome (both caused by a mutation in the APC gene), Cowden syndrome (also known as multiple hamartoma syndrome, caused by a mutation in the PTEN gene), and Carney complex (caused by a mutation in the PRKAR1A gene) [202, 203]. A family history of PTC in two first-degree relatives increases the risk of PTC three- to ninefold, and these families are likely part of a familial non-medullary thyroid cancer (FNMTC) kindred, whose specific genetic defect has not yet been determined [204].
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


