Fig. 3.1
Embryological development of the salivary glands
The third stage is marked by maturation of acini and intercalated ducts and reduced prominence of interstitial connective tissue.
The primordial parotid gland is the first to appear, during the sixth gestational week, when oral ectodermal outpouchings extend into the adjacent mesoderm and serve as the site of origin for growth of glands. After a short journey of dorsal and lateral migration, the parotid gland resides in the preauricular region. The facial nerve arbitrarily divides the gland into superficial and deep lobes by the 10th week of gestation. A fully developed capsule from the nearby mesenchyme surrounds the gland by the 12th week [1].
During the sixth week of embryonic life, small buds appear in the floor of the mouth lateral to the tongue and extend posteriorly around the mylohyoid muscle into the submandibular triangle. These buds eventually develop into the submandibular glands. A capsule from the surrounding mesenchyme is fully formed around the gland by the third-gestational month [2].
During the ninth embryonic month, the sublingual gland anlage is formed from multiple endodermal epithelial buds in the paralingual sulcus of the floor of the mouth. Absence of a capsule is due to infiltration of the glands by sublingual connective tissue. Intraglandular lymph nodes (LNs) and major ducts also do not generally develop within sublingual glands. Upper respiratory ectoderm gives rise to simple tubuloacinar units. They develop into the minor salivary glands during the 12th intrauterine week [4].
Aside from heterotopic salivary glands, developmental disorders of these glands are rare. Reported congenital anomalies include aplasia (absence) [5–8], gland duplication [9], as well as congenitally atretic, imperforate, ectatic, and duplicated ducts [10, 11]. Agenesis may be associated with abnormalities of the lacrimal apparatus and may also be genetically transmitted [7]. Agenesis may be partial or total or unilateral or bilateral and may involve more than one salivary gland. Association with specific syndromes such as Levy-Hollister syndrome, Down syndrome, or Klinefelter syndrome has been reported. The true incidence of parotid agenesis is unknown since most of the cases are asymptomatic. Symptoms include dental caries, thirst, xerostomia, and gingival infections. Oral examination reveals absence of the papilla of Stensen’s duct. When suspected, magnetic resonance imaging (MRI) is the imaging tool of choice to demonstrate glandular bed being replaced by fat. Congenital parotid fistula is another developmental disorder.
Tumorlike presentations of maldevelopment include rare hamartomas of the hard palate [12] and unusual cystic choristomas of the submandibular gland [13], which appear to be a mixture of epithelium of both ectodermal and endodermal derivation. The congenital salivary gland anlage tumor or congenital pleomorphic adenoma (PA) is thought to represent a hamartoma of minor salivary gland origin [14].
3.3 Histology
All glands in general are derived from epithelial cells and consist of parenchyma (secretory unit and associated ducts) and stroma (surrounding connective tissue that penetrates and divides the gland into lobules). Salivary glands are exocrine glands that secrete saliva through ducts from a flask-like, blind-ended secretory structure called the salivary acinus.
The acini of the parotid gland are lined exclusively by serous cells. The acini of the submandibular gland are also mainly serous (90 %) but also contain mucous as well as mixed acini. The sublingual gland is composed primarily of mucous acini. Minor salivary glands are either mucinous or seromucinous except for the serous Ebner’s glands on the posterior aspect of the tongue. This histological description results in parotid thin watery, devoid of mucins, saliva; submandibular mixed saliva; and sublingual more viscous, mucin-rich, saliva [15].
The acini are drained by a series of ducts, the smallest of which are the intralobular intercalated ducts, which are comprised of an irregular myoepithelial cell layer lined with squamous or low cuboidal epithelium and in turn drain into the striated ducts. The striated ducts are also intralobular but lined by a different epithelial arrangement. Striated ducts drain into the interlobular ducts. In the major salivary glands, these ducts drain into major excretory ducts (Stensen’s duct in the parotid gland, Wharton’s duct in the submandibular gland, and Bartholin’s duct in the sublingual gland), and the epithelium of these ducts changes to squamous as they exit through the oral mucosa. The minor salivary gland duct system is simpler than that of the major salivary glands, where the intercalated ducts are longer and the striated ducts are either less developed or not present [15].
3.4 Physiology
The major function of the salivary glands is the production of saliva, which performs many functions including lubrication of the food bolus, maintaining the pH of the oral cavity within 6–7, maintaining the teeth integrity, fighting bacteria, aiding taste and digestion, as well as providing a continuous lavaging biofilm for the oral cavity [4].
The amount of saliva is affected by the total body fluid volume, and so dehydration decreases its amount and so one feels thirsty. Many agents and viruses are actively excreted in saliva. Mercury poisoning can manifest as stomatitis and lead poisoning by the gingival deposition of lead. The rabies and poliomyelitis viruses are excreted into saliva and can be transmitted in this manner. The two main triggers for salivary production are mastication and gustatory stimuli. Acidic foods are the best stimulus and sweet tastes the least. Olfaction is surprisingly a poor secretory stimulus.
3.4.1 Salivary Flow Rates
In normal circumstances the minimal total unstimulated salivary flow rate is defined as 0.1 mL per minute, and the minimal stimulated flow rate is 0.2 mL per minute. Maximal stimulated flow rate is 7 mL per minute. The 24-h volume of salivary secretion has been estimated to be 500–1,500 mL. Salivary flow in the unstimulated glands is produced primarily by the submandibular glands (65 %), with the parotid, sublingual, and minor glands providing 20 % and 7–8 % of the flow, respectively. Once stimulated, the relative contributions of the parotid and submandibular glands are reversed, with the parotid gland supplying greater than 50 % of the flow. The minor salivary glands, independent of stimulation, produce less than 10 % of the total flow.
Bilateral tympanic neurectomies (bilateral parasympathetic denervation) have been used for patients with ptyalism (drooling) with good initial results. Others, however, advocate bilateral parotid duct rerouting with or without bilateral submandibular gland excision for long-term management of drooling. Intraglandular botulinum toxin has been reported to have good results for patients with hyper-sialorrhea. Most resting salivary gland flow arises from the submandibular glands, and surgery should be focused on this gland to control uncontrolled sialorrhea. Salivary flow rates are independent of age. Xerostomia in the elderly is probably the result of systemic disease or medication side effects. Salivary gland hypofunction is defined as an unstimulated flow rate less than 0.1 mL per minute or a 50 % reduction below basal rates if they have been determined. Basal flow rates should be recorded after 15 years of age [4, 16].
3.5 Parotid Glands
The parotid gland is the largest of the major salivary glands and consists of two lobes: superficial and deep with regard to its relation with the facial nerve. It is wrapped around the mandibular ramus and secretes saliva through the parotid (Stensen’s) duct and the oral cavity. The word “parotid” (paraotic) literally means around the ear.
3.5.1 Surgical Anatomy
The parotid gland is a paired organ, weighing about 15–30 g each. Its superficial lobe overlies the lateral surface of the masseter muscle and is bounded superiorly by the zygomatic arch, while its deep lobe is located in the pre-styloid compartment of the parapharyngeal space between the mastoid process posteriorly, the ramus of the mandible anteriorly, and the external auditory meatus (EAM) superiorly. Medially, the gland reaches to the styloid process. Inferiorly, the parotid tail extends down to about the anteromedial margin of the sternocleidomastoid (SCM) muscle.
Several structures run through the parotid gland. These are (1) the terminal segment of the external carotid artery (ECA), which gives the posterior auricular artery just before entering the gland and terminates by dividing into the superficial temporal artery and the maxillary artery; (2) retromandibular vein; (3) facial nerve which soon gives 5 branches inside the gland radiating forward superficial to the vein and artery; and (4) parotid lymph nodes (LNs).
3.5.1.1 Parotid Fascia
The deep cervical fascia continues superiorly to form the parotid fascia which is split into superficial and deep layers to enclose the parotid gland. The thicker superficial fascia extends up to the zygomatic arch, while the deep one extends to the stylomandibular ligament, which separates the superficial and deep parotid lobes. The parotid fascia forms a dense inelastic capsule.
3.5.1.2 Parotid Duct (Stensen’s Duct)
The parotid gland drains its serous secretions through a long duct (Stensen’s duct) that arises from the most anterior superficial portion of the gland and travels parallel and 1 cm below the zygoma, running on the lateral surface of the masseter, just deep to the skin, and then it finally dives at an angle of about 90° at the anterior border of the masseter to pierce the buccal pad of fat and the buccinator muscle and opens in the oral vestibule opposite the 2nd maxillary molar over the summit of a papilla [4, 17].
3.5.1.3 Accessory Parotid Gland
Accessory parotid tissue (lobe or gland) may sometimes exist and come to reside over the masseter muscle between the zygomatic arch and Stensen’s duct. As proved histologically, this accessory glandular tissue secretes mucous in addition to serous secretions drained through a single short duct that joins the main duct [18].
3.5.1.4 Surface Anatomy of the Parotid Gland
The anterior border of the gland corresponds to a line that extends from the ear tragus to the posterior border of the masseter opposite the angle of the mouth, the inferior border from there to below and behind the angle of the mandible, the posterior border from there to the mastoid process, and the superior border from the mastoid process to the ear tragus. Surface anatomy of the parotid duct corresponds to the middle third of an imaginary line that extends from the tragus of the ear to the midportion of the upper lip.
3.5.1.5 Nerve Supply
The facial nerve (CN VII) exits the skull base through the stylomastoid foramen, which is slightly posterolateral to the styloid process and anteromedial to the mastoid process. The nerve immediately gives off three motor branches for the posterior belly of the digastric, the stylohyoid, and the postauricular muscles. Its main trunk then enters the posterior portion of the gland. Inside the gland, at the pesanserinus (Latin: goose’s foot), it divides into upper temporofacial and lower cervicofacial division approximately 1.3 cm from the stylomastoid foramen (Fig. 3.2). The temporofacial division gives off the temporal, zygomatic, and buccal branches, while the cervicofacial gives off the marginal mandibular and cervical branches.
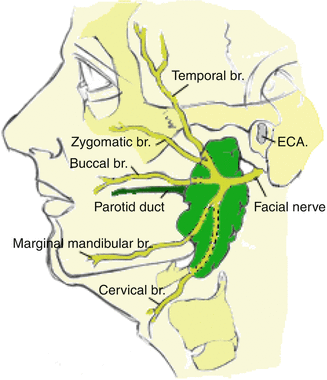

Fig. 3.2
Anatomy of the facial nerve. It divides first into two divisions: upper temporofacial and lower cervicofacial. The upper division gives off the temporal, zygomatic, and buccal branches, while the lower gives off the marginal mandibular and cervical branches
The temporal branch passes with the superficial temporal vessels over the zygoma to supply the frontal belly of occipitofrontalis, orbicularis oculi, corrugator supercilii, and anterior and superior auricular muscles. The zygomatic branch passes over the periosteum of the zygomatic arch to supply the zygomatic, orbital, and infraorbital muscles. The buccal branch travels with the Stensen’s duct to supply the buccinator, upper lip, and nostril muscles. The marginal mandibular branch travels along the inferior border of the parotid gland just deep to the platysma muscle but superficial to the posterior facial and retromandibular veins, to supply the lower lip and chin muscles. Running in the same plane is the cervical branch, which supplies the platysma muscle. All the muscles supplied by the facial nerve are muscles of facial expression.
The great auricular nerve (C2, C3) is a sensory branch of the cervical plexus that provides general sensation to the parotid gland and the skin of the posterior portion of the ear pinna and the ear lobule. It accompanies the external jugular vein along the lateral surface of the SCM muscle toward the tail of the parotid gland, and there it splits into anterior and posterior branches. It is often injured during parotidectomy leaving the ear lobule senseless. Its harvesting for facial nerve grafting may be needed when the latter is injured or sacrificed.
The auriculotemporal nerve (ATN) is a branch of the mandibular division of the trigeminal nerve (CN V). After exiting the foramen ovale, it runs parallel to the superficial temporal vessels and anterior to the external auditory canal to innervate the skin and scalp immediately anterior to the ear.
The glossopharyngeal nerve (CN IX) provides visceral secretory innervation to the parotid gland. The nerve carries preganglionic parasympathetic fibers from the inferior salivatory nucleus in the medulla through the jugular foramen. Then, a small branch of the CN IX (Jacobsen’s nerve) reenters the skull through the inferior tympanic canaliculus and into the middle ear to form the tympanic plexus.
The preganglionic fibers then travel as the lesser petrosal nerve into the middle cranial fossa and out through the foramen ovale to synapse in the otic ganglion. Postganglionic parasympathetic fibers exit the otic ganglion to join the ATN in the infratemporal fossa. The ATN enters the substance of the gland from its deep aspect along the neck of the mandible and emerges from the gland just inferior to the root of the zygomatic arch. The ATN carries fibers that innervate the parotid gland for secretion of saliva (Fig. 3.3).
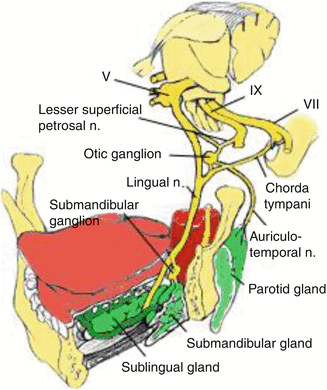

Fig. 3.3
Parasympathetic innervations of the salivary glands
Postganglionic sympathetic fibers innervate the salivary glands, sweat glands, and cutaneous blood vessels, mediating vasoconstriction, through the external carotid plexus from the superior cervical ganglion. Acetylcholine serves as the neurotransmitter for both postganglionic sympathetic and parasympathetic fibers. This physiologic coincidence allows for the occasional development of “gustatory sweating,” also known as Frey’s syndrome, following parotidectomy [19].
3.5.1.6 Arterial Supply
The parotid gland receives its blood supply through branches of the external carotid artery (ECA), mainly through the posterior auricular artery. The ECA travels parallel to the mandible under the posterior belly of digastric muscle. It then travels medial to the gland and splits into its two terminal branches: the superficial temporal artery that leaves the superior portion of the gland and the maxillary artery that leaves the medial portion of the gland. The transverse facial artery is a branch of the superficial temporal artery that runs anteriorly between the zygoma and parotid duct to supply it together with the parotid gland and the masseter muscle.
3.5.1.7 Venous Drainage
The retromandibular vein, formed by the union of the maxillary and the superficial temporal veins, runs through the parotid gland just deep to the facial nerve. It drains into the external jugular vein. There are many variations as regards the surgical anatomy of the retromandibular vein.
3.5.1.8 Lymphatic Drainage
As a result of late encapsulation during embryological development, the parotid gland, unlike the submandibular and sublingual glands, contains intraparenchymal lymph nodes (LNs), with salivary gland structures, usually ducts and less frequency acini, in the intra- and peri-parotid LNs. Ninety percent of the nodes draining the parotid gland are located in the superficial layer between the gland and its capsule (3–20 nodes). These drain the parotid gland, external auditory canal, pinna, scalp, eyelids, and lacrimal glands. Deep LNs, residing on the lateral wall of the pharynx, drain the gland, external auditory canal, middle ear, nasopharynx, and soft palate. Both groups drain into level II of cervical LNs.
3.5.1.9 Parapharyngeal Space (PPS)
Tumors of the deep lobe of the parotid gland often extend medially into the PPS, which is shaped like an inverted pyramid, where the greater cornu of the hyoid bone serves as the apex and the petrous bone of the skull base as the base. The PPS is bound medially by the lateral pharyngeal wall, which consists of the superior constrictor muscles, buccopharyngeal fascia and tensor veli palatine. It is bounded laterally by the ramus of the mandible and the medial pterygoid muscle, anteriorly by the pterygoid fascia and the pterygomandibular raphe, and posteriorly by the carotid sheath and prevertebral fascia.
A line from the styloid process to the medial portion of the medial pterygoid plate divides the PPS into pre- and post-styloid compartments. The pre-styloid one contains the deep parotid lobe, internal maxillary and ascending pharyngeal arteries, and inferior alveolar, lingual, and ATNs. The post-styloid compartment contains the internal jugular vein (IJV), carotid artery, vagus nerve (tenth cranial nerve, CN X) all within the carotid sheath, as well as the cranial nerves IX, XI, and XII and the cervical sympathetic chain. Thus, neurogenic tumors or paragangliomas arising from these nerves lie in this post-styloid compartment [20].
Hints and Tips
The parotid bed is an irregular space located between the ramus of the mandible, the EAM, the mastoid and styloid processes, the digastric, and SCM muscles.
Structures entering the parotid gland exit from its posterior, superior, inferior, and anterior surfaces. The posterior auricular artery exits from the posterior aspect of the gland. The superficial temporal artery and vein, ATN, and temporal branches of the facial nerve are seen at the superior margin of the gland. Inferiorly, the retromandibular vein exits the parotid gland. Emanating from the entire facial margin of the gland are the terminal branches of the facial nerve, grouped into five major branches: the temporal, zygomatic, buccal, mandibular, and cervical branches.
General sensation to the parotid gland is provided by the great auricular nerve of the cervical plexus. Sympathetic innervation is supplied by the carotid plexus, whereas secretomotor innervation is supplied by the glossopharyngeal nerve and delivered to the gland by the ATN.
The gland cannot be moved over the deep structures and becomes more prominent when the patient clenches his teeth by contracting the masseter muscles.
3.5.2 Evaluation of the Parotid Gland
3.5.2.1 Clinical Evaluation
Despite the availability of modern technology in diagnosis of parotid gland disorders, great care should be taken during history taking and thorough physical examination as they still play important roles in the clinical diagnosis of the patient.
History Taking
Patients with parotid gland disorders usually complain of swelling, pain, xerostomia (dry mouth), foul taste, and occasionally sialorrhea (excessive salivation). Swelling and pain during meals followed by reduction in symptoms after meals may indicate partial stenosis of the duct. Demographic data (age and gender) are of considerable importance. The autoimmune disorder known as Sjogren’s syndrome, for example, is common in menopausal women, while mumps usually occurs in children.
The medical profile of the patient may provide helpful clues to diagnosis. Dysfunction of the gland is often associated with certain systemic disorders such as diabetes mellitus (DM), atherosclerosis, hormonal imbalances, and neurologic disorders [21, 22]. A careful dietary and nutrition history should not be neglected, for patients who suffer from chronic dehydration due to bulimia or anorexia or during chemotherapy are at risk for parotitis.
Drug history of the patient should also be considered, for salivary function is often affected by certain drugs such as diuretics and other antihypertensive drugs, which may cause xerostomia [21, 22]. Since xerostomia is also a debilitating consequence of radiation therapy to the head and neck, history of prior irradiation should be sought.
Physical Examination
Initial clinical evaluation involves careful examination of the head and neck regions. Both extraoral and intraoral examinations should be carried out in a systematic way to avoid missing any crucial signs. Bimanual palpation (extraoral with one hand and intraoral with the palmar aspects of the fingertips of the other) must be also performed to properly examine the submandibular glands.
Inspection
Extraoral inspection is performed with the patient facing in front of the examiner, three to four feet away. The examiner should inspect symmetry, color, pulsations, and discharging sinuses on both sides of the patient. Enlargement of the parotid gland may be unilateral or bilateral. Parotitis typically presents as preauricular swelling, but may not be visible if deep in the parotid tail or within the substance of the gland. Neurologic deficits should also be examined. Facial nerve paralysis in conjunction with a parotid mass, for example, should raise the suspicion of a malignant parotid neoplasm. Intraoral inspection is done using a torch or headlight. The orifices of the parotid (Stensen’s) ducts are inspected opposite the second upper molar tooth and the two sides compared. Saliva may be seen pouring from the non-affected side only.
Palpation
With the examiner standing in front of or behind the patient, the patient’s head is inclined forward to maximally expose the parotid gland region. Size, tenderness, consistency, mobility, and other features of the parotid gland and associated masses can be easily evaluated with extraoral and intraoral palpation owing to the superficial anatomical location of the gland.
Auscultation
Rare clinical entities, such as hemangiomas and other vascular anomalies, may be identified by auscultation.
3.5.2.2 Imaging
For patients with no specific symptoms (swelling and pain) and unclear physical signs, imaging studies can play an important role in clarifying the etiology of the gland disorder and assist in treatment selection and planning.
Sialography
Sialography is used to evaluate sialolithiasis (or other obstructive disorders) and inflammatory and neoplastic diseases. In this technique, 0.5 to 2 mL of a suitable radiopaque liquid such as Hypaque (sodium diatrizoate) or Lipiodol is introduced into the duct system through a fine polythene catheter, or a blunt metal cannula, and a plain-film radiograph is taken. Sialography is contraindicated in case of iodine allergy and acute sialadenitis. Any filling defect (e.g., stone), retained sections (e.g., chronic sialadenitis), stricture (e.g., inflammation), extravasation (e.g., Sjogren’s syndrome), or irregular borders (e.g., tumor) are noted. Fistulae and abscesses cavities can also be displayed with this technique.
Computed Tomography (CT)
The parotid gland has low attenuation due to its high fat content and is therefore easily discernible by CT scanning. The advantage of CT imaging is the two-dimensional view of the salivary gland, which can elucidate relationships to adjacent vital structures, differentiate intrinsic from extrinsic disease, and assess the draining cervical LNs. It is also extremely valuable in defining abscess formation versus phlegmon. However, it is limited in evaluating the ductal system unless combined with simultaneous sialography (CT sialography) [23]. Differences between intrinsic and extrinsic parotid gland masses, however, are often difficult to assess especially when present in the parapharyngeal space (PPS) [24].
Magnetic Resonance Imaging (MRI)
Compared with CT, MRI provides better contrast resolution, exposes the patient to less harmful radiation, and yields detailed images on several different planes without patient repositioning. It is more often used for assessment of parapharyngeal space abnormalities especially in discriminating between deep lobe parotid tumors and other lesions, such as schwannoma and/or glomus vagale. However, MRI is inferior to CT scanning for the detection of calcification and early bone erosion. Chronic inflammation of the salivary glands and calculi are not indications for MRI.
3.5.2.3 Endoscopic Examination (Sialendoscopy)
Sialendoscopy is a minimally invasive technique that inspects the salivary glands using narrow-diameter, rigid fiber optic endoscopes, introduced under direct vision through the ductal orifice after its dilatation with a lacrimal probe [25]. It is well tolerated, with minimal complications, and has thus opened up a new frontier for evaluation and treatment of salivary gland disorders [26]. Direct inspection of the glandular duct and hilum is performed during lavage of the duct. Thus, in one setting, at the time of diagnosis, treatment of benign lesions can be performed [27]. Through a CO2-laser papillotomy, sialolithectomy can be easily performed. Pharmacotherapy and laser ablation can also be performed. This relatively new technique has shown much promise in the diagnosis and treatment of chronic obstructive sialadenitis (COS) and sialolithiasis.
3.5.2.4 Biopsy
Fine-needle aspiration cytology (FNAC) is an accurate investigation for the diagnosis of a parotid mass in up to 93 % of cases [25–27]. It allows for improved patient selection for surgery since it can identify disorders such as reactive lymph nodes that might mimic parotid tumors clinically. The information gained by FNAC is thus useful for patient counseling and for surgical timing and planning. Open biopsy of the lip should be considered when the diagnosis of Sjogren’s disease is contemplated.
3.5.3 Parotid Injuries and Fistulae
Successful treatment of parotid injuries depends on early recognition and appropriate early intervention. Sequelae of inadequate diagnosis and treatment include parotid fistula and sialocele formation, which are inconvenient for the patient and more difficult to treat than the initial injury.
A parotid fistula is a communication between the parotid gland (glandular fistula) or duct (ductal fistula) and the skin externally (external fistula) or the oral cavity internally (internal fistula). A sialocele is a collection of saliva beneath the skin that occurs if the duct leaks but no fistula forms or when the glandular substance, but not the duct, is disrupted.
3.5.3.1 Etiology
Causes of the fistula include: (1) penetrating or blunt injury in the region of the parotid gland, (2) improper incision and drainage or spontaneous rupture of a parotid abscess (or sialocele), (3) intraoperative iatrogenic injury, (4) complication of parotid duct cannulation for sialography, and (5) malignant tumors invading the surface [28].
3.5.3.2 Clinical Presentation
Males are twice as likely to experience parotid duct injury as females, and the mean age of patients with parotid duct injury is approximately 30 years.
History
Important aspects of history of the wound include the circumstances surrounding the injury, precipitating cause, exact mechanism and site of injury, time of occurrence, and treatment initiated prior to presentation.
Other important aspects of the history include tobacco, alcohol, or drug use; tetanus immune status; and comorbid conditions that may place the patient at a higher risk for infection such as diabetes mellitus and immunosuppression.
Physical Examination
An internal fistula constitutes no consequences and requires no treatment. However, an external fistula connected with large ducts causes extreme discomfort every time the patient has a meal, smells, or even thinks of food, due to excessive outpouring of saliva on the cheek causing skin excoriation. A sialogram will determine whether the fistula is ductal or glandular.
A thorough clinical examination is necessary for proper evaluation of the overall state of health, comorbidities, nutritional status, and mental status of the patient. Important signs or symptoms related to the wound include pain, fever, edema, discharge, and/or odor. Important aspects of wound assessment include location, shape, size, type (blunt or penetrating), depth, drainage (quality, character, odor), presence of a foreign body (e.g., glass, tooth fragments), loss of tissue, tenderness, asymmetry, surrounding skin (erythema, edema, crepitus), and status of the facial nerve.
An injury classification system that divides the parotid duct into 3 regions has been devised for parotid duct injuries.
Site A: Posterior to the masseter muscle or intraglandular (glandular)
Site B: Overlying the masseter muscle (masseteric)
Site C: Anterior to the masseter muscle (pre-masseteric)
3.5.3.3 Treatment
Various treatment modalities to treat the parotid fistula have been advocated, if conservative measures fail. Tympanic neurectomy involves drilling into the temporal bone and disruption of the tympanic nerve, which carries parasympathetic secretory nerve fibers to the parotid gland [29, 30]. This technique aims at reducing salivary flow and causing spontaneous fistula resolution. Although popular in the past, this method tends to be abandoned due to short-term and poor results due to reinnervation of the gland with time [31].
Radiation has been used in the past to suppress the gland, but use of radiation for benign disease is now avoided. Some authors advocate use of anticholinergics to suppress glandular function during healing, but this is not a frequently used modality. Three operative techniques have been popularized over time. These include repair of the duct over a stent, ligation of the duct, and fistulization of the duct into the oral cavity.
Medical Therapy
Wound care is the cornerstone of therapy. Prophylactic antibiotics should be administered and continued for 5–7 days, but it should be noted that antibiotics cannot avert or cure infections in the setting of poor wound care. In rare cases, human saliva contains and occasionally transmits Clostridium tetani. Accordingly, all patients should be assessed for their tetanus immune status and immunization should be updated as appropriate.
Some authors choose to use anticholinergic agents to suppress glandular function during healing or in an attempt to close a fistula or resolve a sialocele spontaneously. A commonly used agent is propantheline bromide (Pro-Banthine), which inhibits the action of acetylcholine at the postganglionic nerve endings of the parasympathetic nervous system (adult dose 15 mg PO qid half an hour prior to meals).
Sialocele and salivary fistula can frequently be managed nonoperatively with antibiotics, pressure dressings, and serial aspiration. Anticholinergic medications and the injection of botulinum toxin represent additional measures before resorting to surgical therapies such as tympanic neurectomy or parotidectomy [32].
Surgical Therapy
Copious irrigation of the wound with normal saline solution has been shown to decrease the incidence of wound infection. Careful debridement of devitalized tissue, particulate matter, and clot is necessary to reduce the infection risk and to improve the cosmetic result. Clean, surgically created wound margins allow for faster wound healing and better scarring.
Head and neck wounds, less than 12 h old and not obviously infected, being in a cosmetically sensitive area, may be closed directly with a low incidence of infection. The low infection rate is probably related to the excellent regional blood supply and infrequency of edema in these areas. For a good cosmetic result, closure is performed in a simple interrupted fashion, using fine stitches and avoiding layered closure with buried sutures.
The most important initial step for proper surgical repair is the identification of key structures, namely, the buccal branches of the facial nerve (with the aid of a nerve stimulator intraoperatively if available) and the parotid duct itself. If the buccal branch was transected, repair it with fine sutures (8–0 to 10–0 monofilament is appropriate) under microscopic aid. The distal end of the parotid duct is identified by a Silastic tube, placed via cannulation of the intraoral papilla. The proximal parotid duct can usually be identified by the flow of saliva into the wound, with gentle pressure over the gland if necessary. Once all key structures are identified, a decision is made regarding which repair technique to employ.
Distal lacerations, occurring at site C, may be treated in several ways. If the papilla is uninjured, the proximal portion may be dissected free and reimplanted into the papilla. If the papilla is injured or if the proximal duct does not reach the papilla, the duct may be reimplanted (under magnification) into the oral mucosa posterior to the papilla, using fine interrupted absorbable sutures with meticulous approximation of duct epithelium to oral mucosa. If the distal injury is too short to be reimplanted into the oral mucosa without undue tension, then the best decision is to ligate the proximal duct.
Injuries occurring over the masseter muscle, at site B, are the most common injuries to the parotid duct and may be treated by repair or ligation. If enough length remains, primary anastomosis over a Silastic stent, using a single layer of interrupted fine sutures (8–0 to 10–0 monofilament) is performed under magnification. If a portion of the duct is damaged beyond repair or is missing, the proximal and distal duct should be ligated. Reports of attempts to use a vein graft (saphenous) in such cases have generally found such attempts unsuccessful. Sialendoscopy-assisted repair of a transected Stensen’s duct in zone B has been reported [33].
Injuries of the proximal duct near the parotid substance, at site A, are usually best treated by duct ligation as the amount of proximal duct remaining is usually insufficient to result in a useful repair. Laceration of the gland itself without disruption of the parotid duct may be repaired with fine absorbable sutures (5–0 or 6–0 Vicryl).
A drain in the wound bed is recommended to drain any residual salivary leak and prevent early sialocele formation. Drains are removed once drainage is minimal and the skin has become adherent to the operative site. Postoperatively, a compression dressing is placed over the operative field postoperatively for several days. If ductal injury required ligation of the proximal duct, marked temporary swelling of the gland followed by rapid glandular atrophy is expected. If leaking of saliva occurs as in the development of a fistula or sialocele, the pressure dressing should be continued or reinstituted. Intermittent aspiration of sialoceles has led to resolution in many cases. Anticholinergics may be used to temporarily decrease salivary flow in order to effect wound healing. In the case of a chronic parotid duct fistula, an intraoral diversion technique to reestablish salivary flow in case of a nonfunctional parotid duct punctum has been described. The fistula tract and the surrounding ellipse of skin are passed in the oral cavity and sutured to the buccal mucosa with 4–0 chromic sutures without need for stenting [34]. Alternatively, chronic fistula and sialocele have been medically managed with botulinum toxin [35].
3.5.3.4 Complications
Persistent Salivary Fistula
If the fistula occurs in the oral cavity, it is of no consequence and requires no further therapy. If the fistula occurs to the overlying skin, the patient experiences saliva dripping down the cheek. Initial expectant management, with or without anticholinergic medications, has led to resolution in many cases. Other cases have required surgical excision of the fistula tract with repair of the duct or superficial parotidectomy. Sialocele usually resolves with intermittent aspiration and compression and rarely requires drain placement. Anticholinergics may be beneficial in the treatment of sialoceles. Duct ligation may lead to early edema of the gland with accompanying pain from stretching of the capsule. This usually subsides spontaneously within 1–2 weeks as atrophy of the gland occurs. Infection of the remaining glandular substance may occur as a late complication of duct ligation. Sialadenitis may result from manipulation of the intraoral papilla or from sialography and may require drainage and antibiotics. Facial nerve injury and sensory nerve injury may occur when surgery is conducted in the region of the parotid duct, particularly in cases where trauma and blood extravasation have discolored the tissues and disrupted tissue planes.
3.5.4 Parotid Calculus
Parotid stones are much less common than submandibular stones (20 % versus 80 %, respectively). However, recent studies showed some rapprochement between these figures. This is attributed to a number of factors such as the difference in the composition of the saliva produced by each gland and the dependence of the submandibular (Wharton’s) duct, which hinders easy drainage of its viscid secretions. Parotid stones are composed of organic substances such as cellular debris, mucopolysaccharides and glycoproteins, and inorganic substances such as different calcium and magnesium salts. The organic substances are mainly found as the core of the stone, while the inorganic substances are in its periphery. Apatite is the most frequent component present throughout the calculus. The annual growth rate of an established stone is about 1 mm per year. Their shape is either rounded or irregular and the average size is about 3.2 mm. Clinical presentations of a parotid stone vary from being asymptomatic, to intermittent obstructive parotitis, to an acute abscess. Parotid stones are usually imaged through sialography being mostly radiolucent [36].
Stones present in the gland or within the collecting ducts are treated with parotidectomy, while those present near the papilla may be treated with longitudinal incision of the duct releasing the stone. The advent of external lithotripsy in the early 1990s [37] paved the way for conservative treatment of all calculi, with a success rate of 40 % in the submandibular gland and 75 % in the parotid. Unfortunately, results were discouraging, specifically in patients with large calculi [38–41].
Interventional sialendoscopy, first described in the 1990s, offered much less invasive therapeutic options and became popular with technological improvements in the years after 2000 [42–47]. It can be used to retrieve stones from the ducts (Fig. 3.4) as well as dilating strictures.
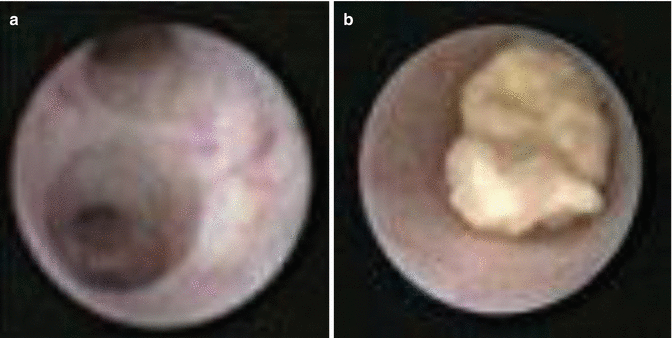

Fig. 3.4
Sialendoscopy showing interior of the parotid (Stensen’s) duct (a) and intraductal stones (b)
Nevertheless, success rates of interventional sialendoscopy with intraductal laser fragmentation and basket extraction of calculi remained possible in only 80 % of patients [42]. Unsuccessful treatment of the remaining 20 % of patients was attributed not only to large-sized calculi (6 mm and larger) but also due to associated ductal stenosis [48]. In such cases, the only solution was to remove the gland, with its associated significant morbidity [38–40].
3.5.5 Inflammatory Disorders (Sialadenitis)
3.5.5.1 Viral Infections
Mumps [49]
Mumps is a specific acute viral infection due to a paramyxovirus, an RNA virus that belongs to the influenza and parainfluenza family. In 85 % of cases, it affects the school-age children under the age of 15 years, with an incubation period of 2–3 weeks. The illness starts by 1–2 days of prodromal influenza-like syndrome (discomfort, loss of appetite, nausea, chills, fever, sore throat, and headache), followed by the appearance of the characteristic face swelling. The patient is infectious from 3 days before the onset of salivary swellings to 7 days after, through airborne droplets of infected saliva. A single episode of infection confers lifelong immunity. Fortunately, this condition has been mostly eradicated as a result of vaccination.
The swelling, which usually starts unilateral and in a few days becomes bilateral, lasts from a couple of days to 1 week (Fig. 3.5). The virus causes fever and is considered the most common cause of acute painful profuse swelling of the parotid gland.
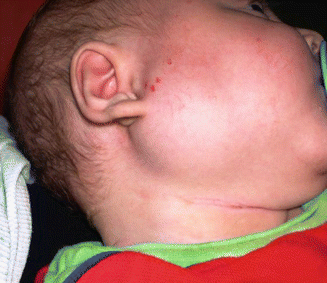

Fig. 3.5
Mumps in a 10-month child with a smooth enlargement of the right parotid gland
The gland is soft and tender, but never suppurates. Impingement on the auriculotemporal and great auricular nerves causes much pain as the gland is pressured during mastication. Symptoms usually resolve within 5–10 days.
Mumps may be complicated by pancreatitis, orchitis, and oophoritis. It might be a cause of abortion during the first trimester of pregnancy because of fetal endocardial fibroelastosis. Sensorineural deafness (1/20,000) and meningoencephalitis are rare but are more likely to occur in adults.
Laboratory findings include leukocytopenia with relative lymphocytosis. Serum amylase peaks in the 1st week and normalizes by the 2nd or 3rd week. Soluble antibodies directed against the nucleoprotein core of the virus appear within the final week of infection and disappear within 8 months. Antibodies directed against the outer surface appear several weeks after soluble antibodies, and persist for 5 years. Mumps is a self-limited disease that requires rest and symptomatic treatment only, which includes antibiotics, sialagogues, and rehydration.
Non-Mumps
Parotitis with the same clinical picture as that described for mumps can be caused by other viral agents, namely, Coxsackie A and B, parainfluenza types 1 and 3, echovirus, and lymphocytic choriomeningitis virus.
Human Immunodeficiency Virus (HIV)-Associated Sialadenitis [50, 51]
The presentation of HIV-associated sialadenitis is in the form of chronic, bilateral enlargement of parotid glands where the glands are painless, hard, and nodular. The disease is more common among children than among adults. The condition may be associated with xerostomia and xerophthalmia, being so similar to the classical Sjogren’s syndrome in adulthood. Although both conditions share similar histological characteristics, HIV-associated sialadenitis is usually associated with a negative autoantibody screen. Multiple glandular cysts causing gross parotid swelling and facial disfigurement are another pattern of presentation. Thirty percent of HIV-infected children have been reported to have enlargement of their parotid glands. In addition, patients with HIV are more susceptible to infection with cytomegalovirus (CMV) and adenoviruses as causative agents for non-mumps parotitis.
Useful imaging tools include ultrasound (US), computed tomography (CT), and MRI. The latter two demonstrate the characteristic “Swiss cheese” appearance of multiple large cystic lesions (Fig. 3.6). They can also guide fine-needle aspiration, which reveals serous fluid with the presence of lymphocytes and macrophages. As the parotid gland contains many LNs at different levels, they might be enlarged as the HIV virus mainly affects the lymphoid tissue. The sole indication of surgery might be to improve the appearance.

Fig. 3.6
MRI showing the characteristic Swiss cheese appearance of multiple large cystic lesions
3.5.5.2 Bacterial Infections
Acute Suppurative Parotitis (Abscess) [52]
The infective organism can be any of the following bacteria: Staphylococcus aureus, Streptococcus viridans, Penicillin-resistant coagulase-positive staphylococcus, Streptococcus pneumoniae, beta-hemolytic streptococcus, methicillin-resistant Staphylococcus aureus (MRSA), or gram-negative germs, such as E. coli. Studies have shown the presence of 30–40 % of anaerobic bacteria: bacteroides, peptostreptococcus, and fusobacteria. In Southeast Asia, Pseudomonas pseudomallei has been reported. In many cases the infection is a mixed one.
Ascending retrograde infection spreads from an infected dry mouth through the duct into the gland, or it may be blood-borne. One or more of the following factors that cause dehydration and/or electrolyte imbalance are usually evident: septicemia, obstruction of Stensen’s duct by a stone, after major surgery, or bad oral hygiene. However, sometimes the condition is totally idiopathic. Parotids are affected more frequently than submandibular glands. One of the possible reasons is that the bacteriostatic activity of the saliva secreted by the parotid gland is inferior to that secreted by the submandibular gland as the former is predominantly serous and thus lacks the protective constituents (IgA, sialic acid, and lysosomes) seen in mucinous secretions of the other salivary glands.
The patient generally complains of anorexia, fever, tachycardia, and malaise. Over several hours, the local condition starts with a swelling which is very tender, hot, and with throbbing pain that is exacerbated by mastication. The gland size may reach 3–4 times larger than that of the normal gland, which is usually diffuse but may localize to the lower pole. The skin overlying the gland is red, edematous, and tethered (Fig. 3.7). Movements of the ipsilateral temporomandibular joint (TMJ) are restricted and painful. The upper deep cervical LNs are enlarged and tender.
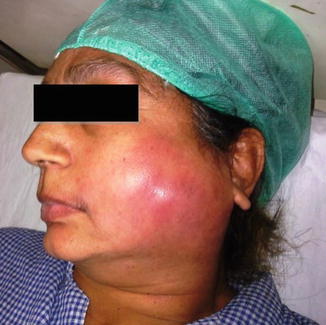

Fig. 3.7
Large left parotid abscess with erythematous, edematous, and tethered overlying skin
The parotid gland, being enclosed in a dense capsule, is liable to fulminating inflammation and necrosis due to increased tension within this tightly closed fascial compartment. Fluctuation is difficult to elicit and is never waited for. Pus may exude from the duct orifice on massage of the gland, which is in spite of being painful at the moment; it relieves the pressure inside the ductal system and thence the pain. Diseases of the parotid gland cause pain to be referred to the ear, the TMJ, and the external auditory meatus. This is due to the overlapping of sensory branches of various nerves serving the regions of the parotid bed, ear, and TMJ. Infections of the parotid gland may be confused with toothache as a result of trigeminal nerve (CN V) involvement. An inflamed parotid papilla (parotid duct orifice in the oral cavity) provides clue to diagnosis.
A parotid abscess may be complicated by local spread in the form of cellulitis, chronicity due to inadequate management, development of a fistula, septicemia, and rupture into the external auditory meatus or along the carotid sheath. The patient shows increased white blood cell count, and culture and sensitivity should be done from the pus. Sialography is contraindicated during acute infection and US may be needed; however, the diagnosis is mostly done on clinical grounds.
Prophylaxis against the development of such suppuration is considered much safer through adopting good oral hygiene, preventing dehydration, and early adequate correction of electrolyte imbalance. As an early treatment of any inflammatory condition affecting the parotid gland, one should use warm local fomentations, intravenous antibiotics, anti-inflammatory agents, and analgesics. When suppuration ensues (known by throbbing pain, hectic fever, aspirated pus, or US-identified abscess formation), formal drainage under general anesthesia becomes necessary.
Decompression of the Parotid (Hilton’s Method)
Do not wait for fluctuation. Under general anesthesia, a vertical incision is done in the skin down to the parotid capsule. The capsule in then incised transversely along the course of the branches of the facial nerve to avoid their injury. Pus is evacuated, a drain is put in the lower part of the incision for 24–72 h, and then closure is achieved. Complications of this procedure include parotid fistula and facial nerve injury. Alternatively, the skin incision may be made low to avoid damage to the lower branch of the facial nerve.
Chronic Pyogenic Parotitis
Chronic bacterial sialadenitis is rare in the parotid gland. It may result from improper treatment of acute parotitis, presence of stones in the duct, or stenosis of the Stensen’s duct [53].
Actinomycosis
Actinomycosis affects LNs adjacent to salivary glands, mimicking a salivary gland infection. The pathogen is Actinomyces israelii, A. propionica, A. viscosus, or A. odontolyticus. Infection may be acute, subacute, or chronic. The acute form is associated with suppuration, the subacute with a slightly tender and tumorlike mass attached to the mandible, and the chronic with marked induration that may be misdiagnosed as a neoplasm. The finding of pollen grain like “sulfur granules” on clinical examination or “sulfur grounds” on pathological evaluation is pathognomonic of this condition. Treatment of the acute phase is surgical, with eventual drainage of the exudates and administration of broad-spectrum antibiotics [54].
Cat-Scratch Disease
A disease caused by infection with a gram-negative bacillus called Bartonella henselae. The disease originally involves LNs adjacent to salivary glands, which may be secondarily involved by continuous spread. The disease is due to contact with cats, and children or young adults are the most often involved. Laboratory tests include specific PCR or serology. Antibiotics do not seem to shorten the course of the disease but may be given as prophylaxis against secondary bacterial infection. The affected LNs disappear spontaneously within a few months [55, 56].
3.5.5.3 Recurrent Parotitis of Childhood
This is a distinct clinical entity of unknown etiology and unsure prognosis. It is the most frequent nonviral disorder of salivary glands in children [57]. Suggested etiologies include congenital malformation of the parotid ducts, primary or secondary infections, and local manifestations of systemic immunological disease.
The clinical picture comprises fever, malaise, rapid swelling of one or both parotid glands, and pain which is made worse by mastication. This usually lasts from 3 to 7 days followed by a quiescent period of weeks to several months. The condition may rarely start as early as 4 months of age, but usually children present between the ages of 3 and 6 years and usually resolve around puberty. The diagnosis is made by obtaining typical history and findings of the clinical examination. Sialography may be confirmatory. It shows a characteristic punctate sialectasis likened to a “snowstorm” (Fig. 3.8) which persists until adult life. Sialendoscopy may show diffuse reduction of the caliber of Stensen’s duct, associated sometimes with multiple localized stenosis and may be salivary calculi. Endoscopy may also show sialectasia (dilated ductules and acini). Sialendoscopy has proven recently to be effective therapeutically through injection of antiseptic solutions in some cases [57].

Fig. 3.8
Sialography showing a characteristic punctate sialectasis (snowstorm appearance)
No specific treatment is available; however, therapeutic options include good oral hygiene, prophylactic low-dose antibiotics for several months or even years especially if recurrence is frequent, anti-inflammatory agents, sialendoscopy, and total conservative parotidectomy [58].
3.5.5.4 Papillary Obstructive Parotitis
Although there are many recognized causes of obstructive parotitis, however, papillary obstruction due to trauma to the parotid papilla through either an overextended upper denture flange or a fractured upper molar tooth is merely the most common of all. Subsequent edema and then fibrosis of the papilla obstruct salivary flow, especially at mealtimes, in intermittent and then chronic forms, respectively. Thus, the patient suffers from an intermittent painful swelling of the parotid, accumulating over minutes after the start of his meal. If stenosis ensues, the symptoms will not resolve except by a papillotomy. Obstructive parotitis is less common than obstructive submandibular sialadenitis.
3.5.5.5 Granulomatous Sialadenitis
Granulomatous sialadenitis can result from tuberculosis, mycosis, sarcoidosis, or duct obstruction from calculi or malignant tumors. In the latter instance, the granulomas result from rupture of ducts and may contain small pools of mucin.
Tuberculosis (TB)
Mycobacterium tuberculosis and atypical mycobacterium both affect LNs adjacent to salivary glands or intraglandular LNs. The patient may suffer from one or more of the systemic manifestations of TB such as low-grade fever, night sweating, and anemia. The gland is firm and nodular but may become cystic with sinuses and little pain. Polymerase chain reaction (PCR) is the best preoperative laboratory diagnostic tool, but diagnosis is only confirmed with histopathology following parotidectomy, if needed. Specific treatment of this condition includes multidrug therapy for an average of 1 year together with exposure to direct sunlight, good aeration, and well-balanced nutrition to enhance general health and immunity [59, 60].
Sarcoidosis
Sarcoidosis is a systemic disease involving multiple organs. Its etiology remains unclear, but several hypotheses have been made, including autoantigens and infectious organisms. It resembles TB but with no caseation. Salivary glands are usually affected and specifically the parotid glands. Symptoms include swellings and xerostomia. Sometimes, the disease my present as a mass, and the diagnosis is only made after surgical excision of the presumed tumor: the sarcoid pseudotumor. Laboratory diagnosis is made through amylase, kallikrein, and the ACE test. Radiological and histological evidences of non-caseous epithelial granulomas are confirmatory. Corticosteroids are the best therapeutic option. Heerfordt’s syndrome is a rare form of sarcoidosis that involves parotid swelling, anterior uveitis, facial palsy, and fever (uveoparotid fever). It affects young patients in their third decade [61].
3.5.5.6 Autoimmune Sialadenitis
Mikulicz Disease (MD)
It is an autoimmune disease, which is characterized by chronic, symmetrical, bilateral enlargement of all salivary and lacrimal glands. The disease almost always occurs in association with another underlying disease such as TB, leukemia, syphilis, or systemic lupus erythematosus (SLE). Sometimes patients may experience recurring fever. Patients with MD are at higher risk of developing lymphomas. Some believe that it should be considered a form of Sjogren’s syndrome (SS). Actually the main clinical difference between MD and SS is the preservation of the lacrimation and salivation in MD due to much less gland cell apoptosis than that found in SS [62].
Sjogren’s Syndrome (SS)
This disease is an autoimmune one involving the parotid glands more frequently than the submandibular glands. Females are affected more than males (10:1). It leads to xerostomia and keratoconjunctivitis sicca due to progressive destruction of both salivary and lacrimal glands. The diseased gland is occasionally enlarged and is occasionally painful. Secondary bacterial ascending infection may occur as a result of dry mouth. Histologically, SS is characterized by massive progressive lymphocytic infiltration, acinar cell destruction, and ductal epithelial cell proliferation affecting all salivary and lacrimal glands. This is the primary SS. Secondary SS differs in that it is associated with a connective tissue disorder, symptoms are less severe, and the incidence of lymphomatous transformation (most commonly monocytoid B-cell lymphoma) is also less [62].
There is no known specific treatment available to stop Sjogren’s syndrome, and thus management is totally symptomatic. Artificial tears and periodic ophthalmological examination is mandatory to avoid corneal ulcerations. Artificial salivary substitutes are available and their importance increases in the dentate patient where fluoride should be added as well. Usually the patient consumes a large amount of water daily carrying a bottle of water with him all the time [63].
3.5.5.7 Benign Lymphoepithelial Lesion (Myoepithelial Sialadenitis, MESA)
This disease mostly affects females over 50 years of age. Patients present with a diffusely enlarged, firm, and often painful parotid gland, which is bilateral in 20 % of cases. Sialography shows sialectasia, which ranges from punctuate to cavitary. Management is usually parotidectomy to establish a final diagnosis. Histopathological examination of the excised gland carries the very same previously described histopathological features of Sjogren’s syndrome, and so the differentiation between both is only done on clinical grounds. Prolonged follow-up of such patients is mandatory as 20 % of them will develop lymphoma at a certain time [64].
3.5.5.8 Other Autoimmune Sialadenitis
Wegener’s granulomatosis is an autoimmune disease characterized by upper and lower respiratory and renal disease. The most common salivary gland to be involved is the parotid gland [61]; however, this occurs in less than 1 % of the cases [65]. Kimura’s disease occurs typically in young Asian males and is characterized clinically by painless lymphadenopathy of the head and neck region, including peri-parotid and intra-parotid LNs [61, 64, 66]. Chronic sclerosing sialadenitis presents as a firm localized swelling of the salivary gland mimicking a neoplasm, most commonly involving the submandibular gland. It may be associated with autoimmune extra-salivary disease such as primary sclerosing cholangitis and idiopathic retroperitoneal fibrosis [64].
3.5.6 Sialadenosis (Sialosis)
Sialadenosis is a noninflammatory disorder causing diffuse enlargement of salivary glands, usually the parotid glands and less commonly the submandibular [64, 67, 68], and minor salivary glands [69].
3.5.6.1 Clinical Presentation
Sialadenosis is frequently bilateral and has an equal sex distribution. Most of the patients are between 40 and 70 years of age. There is a painless, soft, and diffuse enlargement of both parotid glands giving the patient a striking facial feature, known as the “hamster-like appearance”.
3.5.6.2 Etiology
Causes of sialadenosis can be categorized as: (1) nutritional (alcoholism, cirrhosis, bulimia, kwashiorkor, and pellagra), (2) endocrine (DM, thyroid disorders, gonadal dysfunction), and (3) neurochemical (vegetative state, lead and mercury poisoning, iodine, thiouracil, isoproterenol) [64, 68]. However, in many cases no underlying disorder can be detected.
3.5.6.3 Pathogenesis
The proposed pathogeneses include prolonged malnutrition with resultant glandular atrophy and fatty replacement. Sialadenosis is also thought to be a neurosecretory disorder. Diabetic neuropathy may be the clue causing acinar cell atrophy, in some cases. Ultrastructural and animal experimental studies point to a disturbance in the autonomic innervation of salivary glands. This is considered to be the initiating factor for sialadenosis [67, 68, 70–72].
3.5.6.4 Pathology
Grossly, there is only diffuse enlargement of the affected gland. Histologically, the condition is characterized by acinar hypertrophy and fatty infiltration [73]. Zymogen granules are increased in size and number by light and electron microscopy [71]. No inflammation or fibrosis can be detected, which differentiates sialadenosis from sialadenitis. While amyloidosis may also present with diffuse enlargement of the salivary glands, histologically, there will be interstitial fibrosis as well as the characteristic pale amyloid deposition that can be demonstrated with a Congo red stain [74].
3.5.6.5 Treatment
Treatment is in the form of controlling the underlying disorder or withdrawing the incriminated drug. There is usually little morbidity associated with the condition itself, and surgery is resorted to only in case of significant cosmetic complaint.
3.5.7 Cystic Parotid Lesions
Cysts of the parotid gland represent a clinical dilemma. The differential for cystic parotid lesions is summarized in Table 3.1. Apart from the cystic forms of benign and malignant neoplasms, cysts of the parotid gland may be the presentation of a variety of diseases. Such nonneoplastic cysts are uncommon and represent 2–5 % of all salivary gland lesions [75]. A first branchial arch anomaly must be considered when one encounters a case of parotid cyst [76].
Table 3.1
Cystic parotid lesions
Bilateral |
Warthin’s tumor |
Benign lymphoepithelial lesions of HIV |
Sjogren’s syndrome |
Sialocele |
Unilateral |
Warthin’s tumor |
Sialocele |
First branchial cleft cyst: parotid lymphoepithelial cyst |
Necrotic LN(s) especially SCC |
Infected LN(s) |
Disruption of the parotid duct or parenchyma results in extravasation of saliva into the glandular or peri-glandular tissues forming what is known by sialocele. The cause might be either facial trauma or surgery especially when Surgicel is used.
Parotid duct cysts are also known as sialocysts, simple cysts, and retention cysts and result from obstruction due to various causes [74, 77, 78]. They are true cysts lined by epithelium, unlike mucocele, which is lined by granulation tissue [77]. Parotid duct cysts should not be confused with duct ectasia (Fig. 3.9).
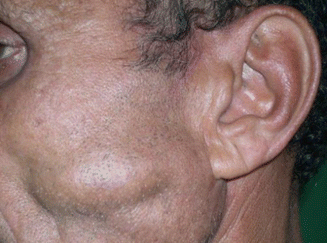

Fig. 3.9
Left parotid duct ectasia
Duct cysts are unilocular and may grossly contain mucoid material or sialoliths in long-standing cases. Histologically, they are lined by a cuboidal, columnar, or squamous epithelium, though oncocytic metaplasia may be seen in older patients. However, these cysts are not associated with lymphoid elements [64]. Prognosis is excellent. Complications are rare and include superimposed infections. Recurrence is also rare and results from incomplete excision [64].
3.5.8 Tumors of the Parotid Glands
3.5.8.1 WHO Histological Classification of Tumors of Salivary Glands
Benign Epithelial Tumors
Pleomorphic adenoma (PA) (mixed salivary tumor)
Myoepithelioma
Basal cell adenoma
Warthin’s tumor (adenolymphoma, papillary cystadenoma lymphomatosum)
Oncocytoma
Canalicular adenoma
Sebaceous adenoma
Lymphadenoma
Sebaceous
Non-sebaceous
Duct papilloma
Inverted duct papilloma
Intraductal papilloma
Sialadenoma papilliferum
Cystadenoma
Malignant Epithelial Tumors
Acinic cell carcinoma
Mucoepidermoid carcinoma (MEP)
Adenoid cystic carcinoma (ACC)
Polymorphous low-grade adenocarcinoma (PLGA)
Epithelial-myoepithelial carcinoma
Clear-cell carcinoma, not otherwise specified
Basal cell adenocarcinoma
Sebaceous carcinoma
Sebaceous lymphadenocarcinoma
Cystadenocarcinoma
Low-grade cribriform cystadenocarcinoma
Mucinous adenocarcinoma
Oncocytic carcinoma
Salivary duct carcinoma
Adenocarcinoma, not otherwise specified
Myoepithelial carcinoma
Carcinoma ex-pleomorphic adenoma (CEPA)
Carcinosarcoma (malignant mixed salivary tumor)
Metastasizing pleomorphic adenoma
Squamous cell carcinoma
Small cell carcinoma (SCC)
Large cell carcinoma
Lymphoepithelial carcinoma
Sialoblastoma
Soft Tissue Tumors
Hemangioma
Hematolymphoid Tumors
Hodgkin’s lymphoma
Diffuse large B-cell lymphoma
Extra-nodal marginal zone B-cell lymphoma
Secondary Tumors
Tumors of the salivary glands are relatively uncommon and represent less than 2 % of all head and neck neoplasms.
3.5.8.2 Benign Tumors (BTs)
Salivary gland BTs occur mostly in the parotid gland, and most of them are pleomorphic adenomas. The BT usually presents as a slowly growing painless mass in front of the tragus of the ear, below the ear lobule, or in upper part of the neck, arising from the superficial lobe of the gland which represents 80 % of the cases. Less commonly, the mass presents as a cheek mass overlying the masseter, arising from the accessory parotid lobe. If the tumor arises from the deep lobe, it presents as a parapharyngeal mass with completely different clinical presentation. In this instance, the patient will complain of difficultly in swallowing and snoring, and physical examination will reveal a soft palate and tonsillar firm, diffuse bulge.
Pleomorphic Adenoma (PA)
This mixed salivary tumor represents 75–80 % of all benign parotid tumors. It may affect as well the submandibular and minor salivary glands. Its incidence is slightly higher in females and affects most commonly patients between 30 and 50 years of age [80–82]. It is uncommon to be bilateral [78, 83]. It is usually a solitary lesion, although synchronous or metachronous involvement of two or more salivary glands can occur. It may also occur in combination with other tumors, mostly Warthin’s tumor.
It is a well-defined tumor, but with small extensions into the adjacent normal tissue through an incomplete capsule, which explains the recurrence after enucleation. It has a pleomorphic matrix, with a non-cystic cut section that may show some islets of cartilage. Microscopically, it is formed of epithelial cells intermingled with a pleomorphic stroma: fibrous, myxomatous, and pseudo-cartilaginous (Fig. 3.10). It is categorized into four types: (1) principally myxoid, (2) mixed myxoid and cellular components in equal proportions, (3) predominantly cellular, and (4) extremely cellular [84].
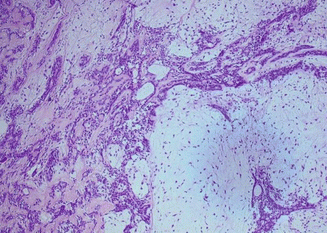

Fig. 3.10
Pleomorphic adenoma: biphasic population of epithelial and mesenchymal cells. Epithelial cells assume tubular profiles, cords, or nests. Stroma is myxoid (H&E, ×100)
It carries a malignant potential of 5–10 %. The presence of hyalinized stroma is the most predictive histological parameter for malignant transformation. The tumor may rarely metastasize without having the histological features of malignancy, but this almost always occurs after inadequate surgical excision, possibly due to altered anatomy secondary to surgery, which gave access to vascular and lymphatic channels.
Roughly 70 % of PAs have cytogenetic alterations that likely play a major role in pathogenesis (tumorigenesis) and can be categorized into 4 groups: those with 8q12 rearrangements, those with 12q13-15 rearrangements, those with miscellaneous clonal changes, and those that are karyotypically normal [85, 86].
The patient presents with a painless, slowly growing, spherical mass, of variable size ranging from pea-size to a large mass, 20–50 cm across [87–89] (Fig. 3.11). The mass usually lies over the angle of the mandible and below the lobule of the ear (Fig. 3.12). However, a swelling of the lower pole of the parotid gland may present in the lateral side of the neck, below and behind the angle of the mandible, giving rise to clinical diagnostic difficulty (Fig. 3.13). The patient’s complaint is usually disfigurement. The mass is of heterogonous consistency and mobile, being neither attached to the skin nor to the deep structures. Bosselation of the tumor is demonstrable if it is bigger than 3 cm.
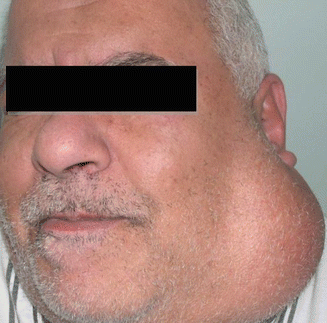
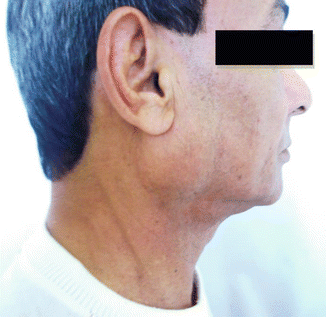
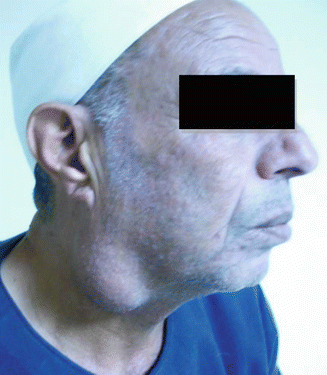

Fig. 3.11
A large left pleomorphic adenoma (PA) of the parotid gland in a 56-year-old male patient

Fig. 3.12
Pleomorphic adenoma in its typical position over the angle of the mandible and below the lobule of the ear

Fig. 3.13
Pleomorphic adenoma arising below and behind the angle of the mandible. Not related to the ear lobule
While most PAs occur in the superficial lobe of the parotid gland (80 %) [90–92], the deep lobe can be involved (20 %) and is clearly demonstrable by CT scan (Fig. 3.14). Extensions of deep lobe PAs are the most common tumors of the parapharyngeal space constituting 40 % of tumors in this region [83, 92]. The draining LNs are not enlarged and the facial nerve is always intact.
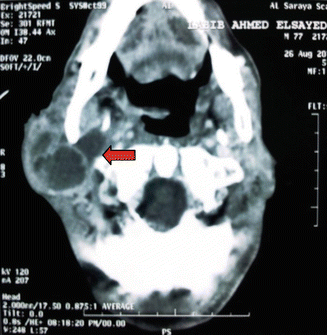

Fig. 3.14
CT scan showing extension of the PA to involve the deep lobe of the parotid gland (red arrow)
As for any parotid mass, US, CT, and MRI (preferably with diffusion-weighted and perfusion sequences) are the best imaging tools. Fine-needle aspiration cytology (FNAC) is a recognized method for obtaining preoperative diagnosis and is advised to be done after imaging for the fear of hemorrhage. Many studies showed that the use of flow cytometry as a biological parameter for the prediction of recurrence in PAs should be considered. Tumors of a larger size showed a higher percentage of cells in the S-phase fraction and probably a greater tendency for recurrence [93].
Treatment is through surgical excision. Superficial or total conservative parotidectomy with preservation of the facial nerve is done. Partial superficial parotidectomy and extracapsular dissection are other recognized options of resection [94–96]. Partial superficial parotidectomy entails dissection of the main trunk of the facial nerve and only one division with its terminal branches (Fig. 3.15). Enucleation is never done for its high recurrence rate. The skin incision can be the classical lazy-S incision, or recently one can resort to the cosmetically better face-lift incision.
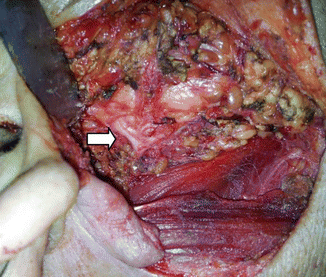

Fig. 3.15
Dissection of the main trunk and only one division, related to the parotid mass (white arrow)
The flaps are raised and the facial nerve trunk is exposed at the stylomastoid foramen, and all its five terminal branches are followed to the muscles to avoid their injury. The incidence of recurrence following surgery varies depending on the surgical technique used, the experience of the surgeon, and the duration of patient’s follow-up [97]. Whatever the series and its duration of patient follow-up, a recurrence rate of less than 1 % is considered acceptable. Recurrent PAs have a higher likelihood for second recurrence of about 6–15 % [98]. Uninodular recurrences have a better outcome than multinodular recurrences [99, 100].
Warthin’s Tumor (Adenolymphoma)
Warthin’s tumor represents about 5 % of all benign parotid tumors and is bilateral in 5–10 % of cases. It occurs almost exclusively in the parotid gland, affecting males more than females. The ratio has decreased drastically during the past 50 years to 2:1 [101, 102]. This gender predilection shift is referred to that it is highly linked to smoking. Smokers have an 8 times higher incidence of developing Warthin’s tumors than nonsmokers [103]. The mean age is 62 years. Patients are rarely below 40 years [104, 105].
Macroscopically, it is a reddish-brown mass with a true capsule. Its cut section has many cystic spaces, and the fluid content is characteristically granular brown resembling “motor oil”. Under the microscope, it is comprised of a papillary proliferation lined by a double layer composed of surface columnar oncocytic epithelium and a smaller basal layer of small cuboidal cells with myoepithelial characteristics. The surrounding stroma contains a highly ordered lymphoid architecture similar to an actual LN (Fig. 3.16). When Warthin’s tumor arises in a cervical LN, it may be mistaken for metastatic carcinoma, particularly from a papillary thyroid carcinoma. Malignant transformation is extremely rare where MEC is the most common histology. The lymphoid component may lead to lymphoma.
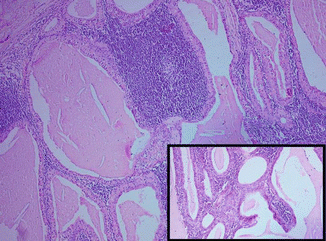

Fig. 3.16
Warthin’s tumor: cystic spaces lined by double layer of epithelial cells resting on dense lymphoid stroma. Inset depicts intracystic polypoid projections with lymphoid stroma (H&E, ×100)
The mass is painless, slowly growing, soft, freely mobile, and usually 1–3 cm in diameter. It does not attain a huge size (Fig. 3.17). It has a smooth surface and homogenous consistency and is sometimes cystic. It is often multifocal and may be fixed to the overlying skin [106]. Rarely, infarcted Warthin’s tumors may cause pain [102]. Adenolymphoma is the only parotid tumor that can give a hot spot in Tc99 scan. If the preoperative diagnosis is certain, then enucleation can be added to the operative options of the surgeon as the tumor is well capsulated and does not recur.
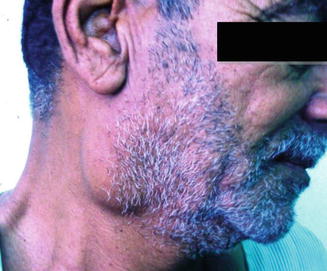

Fig. 3.17
Right adenolymphoma presenting as a small (3-cm), smooth, mass in a 61-year-old gentleman
Basal Cell Adenoma (BCA)
Basal cell adenoma tends to occur over the age of 50 years with a female-to-male ratio of 2:1 [107]. It usually occurs in the parotid gland (75 %), much less commonly in the submandibular gland (5 %) [108], and rarely in the minor salivary glands with the exclusion of canalicular adenomas, which were previously categorized with BCA [109, 110]. It typically presents as a slowly growing solitary painless mass [64]. A special variant of BCA, the membranous type (dermal analog tumor) has a propensity for multifocality and can be associated with multiple trichoepitheliomas and cylindromas (Brooke-Spiegler syndrome) [111]. Prognosis of BCA is generally excellent. Recurrence rate is low, except for the membranous subtype, which may recur in about 25 % of cases. Malignant transformation is rare, again favoring the membranous subtype [101, 112, 113].
Canalicular Adenoma
Canalicular adenoma, previously categorized with BCA, is a rare tumor comprising <1 % of all salivary tumors [101, 112, 113]. The mean age is 65 years with a female-to-male ratio of 1.8:1 [101, 114]. It rarely affects the parotid glands [101]. The minor salivary glands of the upper lip are the ones most commonly involved (80 %) followed by the buccal mucosa [113] and palate [115]. It typically presents as painless, slowly growing submucosal nodule. Rarely, multiple/multifocal canalicular adenomas may occur and present clinically with multiple discrete masses, typically occurring in the upper lip and buccal mucosa [64]. The prognosis is excellent and recurrences are extremely rare. Some of these recurrences may be considered as separate tumors [101, 116].
Myoepithelioma
Myoepitheliomas account for about 1.5 % of all salivary tumors. This tumor primarily affects adults with a peak incidence in the third to fourth decades (range 8–82 years) [117, 118]. The parotid gland is the most common site affected (40–50 %), followed by the minor salivary glands in the palate [64]. It usually presents as a slow-growing painless mass. Prognosis is generally favorable; recurrences are relatively rare and are usually the result of incomplete excision [119]. Malignant transformation is uncommon [120].
Cystadenoma
Cystadenoma is a rare benign cystic salivary tumor that resembles Warthin’s tumor, though with different clinicopathologic features. Unlike Warthin’s tumor, there is a slight female predilection, there is no association with smoking [101, 121], and it is not exclusive for the parotid gland, though it is affected in nearly half of the cases. Other sites include the lip and buccal mucosa [122] and rarely the supraglottic larynx [123]. It affects adults with a mean age of 57 years [124]. Prognosis of cystadenoma is excellent; complete excision is curative.
3.5.8.3 Malignant Tumors
Parotid cancer represents <1 % of all body cancer and about 20 % of all parotid tumors. It is either de novo or on top of PA. There is no sex predilection or males are slightly more affected. Age of developing this cancer is usually above 50 years.
Signs of malignant transformation in a preexisting BT include rapid rate of growth, harder consistency, or fixation to the underlying muscles or the overlying skin, which may be severe enough to show fungation (Fig. 3.18). The patient may develop pain and tenderness which are usually due infiltration of the auriculotemporal nerve with the pain being referred to the ipsilateral ear. Trismus due to invasion of the masseter or pterygoid muscles or due to restricted movements of the temporomandibular joint (TMJ) is another finding.
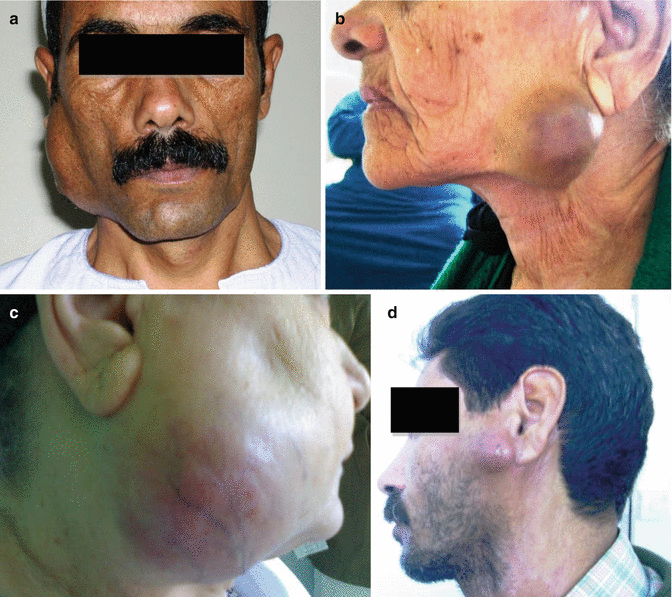

Fig. 3.18
Malignant tumors of the parotid gland present with a hard, rapidly growing mass (a) that may become fixed to the overlying skin (b) and with erythema and increased vascularity (c) miming an abscess and may be severe enough to show fungation (d)
In 10–15 % of the cases, the facial nerve will be paralyzed. Unequal pulsations of superficial temporal artery may as well be noticed. Manifestations of metastases to regional LNs or distant organs such as the lungs or the liver may ensue. On the first suspicion of any change in the behavior of the preexisting tumor, FNAC should be done immediately after MRI preferably with a diffusion-weighted and perfusion sequences.
Spread of parotid malignancy occurs through the well-established routes of metastases, where the first echelon LN is the intra- and peri-glandular nodes. The next echelon is level II LNs. Local spread can affect any of the critical nearby organs. Hematogenous spread occurs very late and is mainly to the lungs and bones particularly the vertebral column. However, adenoid cystic carcinoma (ACC) tends to grow through perineural lymphatics with increased risk of nerve affection and intracranial extension, as well as increased rate of recurrence.
Mucoepidermoid Carcinoma (MEC)
This is the most common malignancy of the salivary glands [125] and that of the parotid gland (80 % of cases) seems to be less aggressive than that of the submandibular gland (8–13 % of cases) and of better prognosis [126]. It mostly occurs around an age of 50 years, but still, it is the most common salivary malignancy of the pediatric age group [126, 127].
Low-grade and high-grade variants are recognized, where the low-grade one very rarely metastasizes [128–130]. It usually presents as a slowly growing painless mass; however, rapid growth, pain, and tenderness may be seen with the high-grade variant [131, 132] (Fig. 3.19). Metastases occur to LNs, lungs, and bones [126]. Treatment is surgical resection. Neck dissection is needed in high-grade variants [128].
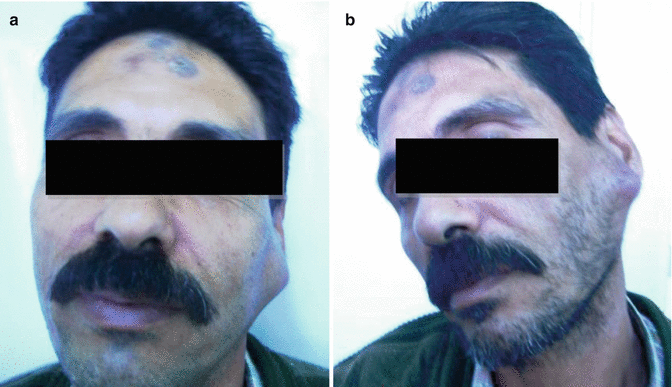

Fig. 3.19
A rapidly growing (high-grade) mucoepidermoid carcinoma (MEC) in a 51-year-old gentleman (a) that became fixed to the overlying skin (b) within 2 weeks of presentation
Prognosis is influenced by the grade and stage of the tumor and patient’s age and gender. Overall 5-year survival rates range from 92 to 100 % for low-grade tumors and 0 to 43 % for high-grade tumors [133].
Adenoid Cystic Carcinoma (ACC)
This tumor has been given many names since it was first described by Robin and colleagues in 1853. From these names, “cylindroma” was particularly discouraged to avoid confusion with the benign cutaneous appendage tumor carrying the same name. This tumor arises not only from major and minor salivary glands but also from seromucous glands throughout the body [134, 135]. It is the second most common salivary malignancy and affects patients between 40 and 60 years of age. It is a slowly growing tumor frequently presenting as a painless mass giving a false sense of security. Lymph node metastases are uncommon, but distant metastases occur in up to 60 % of the cases, mainly to the liver, lung, bones, and brain [135, 136]. It has a high affinity to perineural invasion, which results in paresthesia or paralysis of the nerve affected.
Histologically, low-grade (Fig. 3.20) and high-grade (Fig. 3.21) variants are recognized. Best results of treatment are obtained through radical surgery and postoperative radiotherapy. The tumor is radiosensitive but not radiocurable. Although controversial, neck dissection may be reserved for patients with clinically positive LNs [136–138]. Some studies showed that age over 45 years, advanced clinical stage, paresthesia, as well as increased expression of p53 are associated with poor prognosis [135, 136].
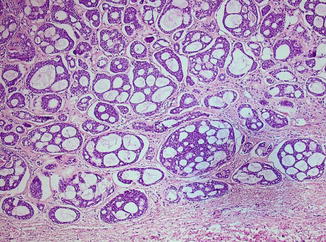
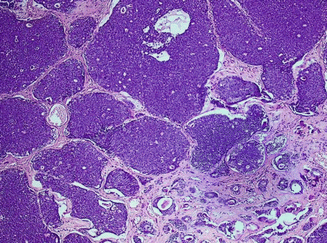

Fig. 3.20
Low-grade adenoid cystic carcinoma (ACC) with a notable cribriform pattern (H&E, ×100)

Fig. 3.21
High-grade adenoid cystic carcinoma (ACC) with a predominant (>70 %) solid pattern. The lower right corner still depicts tubular structures (H&E, ×100)
Carcinoma Ex-pleomorphic Adenoma (CEPA)
This is by definition the malignant transformation of a long-standing PA, which occurs in about 25 % of such cases [138–140]. Clinical features predictive of malignant transformation are age, tumor size, long history, and submandibular location. Histologically, the presence of hyalinized stroma is the most predictive parameter for malignant transformation [64, 108]. The carcinoma is usually a high-grade adenocarcinoma or an undifferentiated carcinoma although numerous other types including SCC, MEC, ACC, myoepithelial carcinoma, clear-cell carcinoma, papillary carcinoma, and terminal duct carcinoma have been reported. Carcinoma ex-pleomorphic adenoma (CEPA) occurs in the major salivary glands in 80 % of the cases, from which again 80 % occurs in the parotid gland. The average age at diagnosis is 50–60 years, which is approximately 10 years older than most individuals with PA. It is extremely uncommon in individuals below the age of 20 years [138, 141]. In 12–55 % of cases, rapid enlargement will be painful and often associated with facial nerve palsy and fixation to the surrounding soft tissues [142].
This carcinoma metastasizes in up to 70 % of the cases to distant sites rather than regionally, with special affinity to the lungs and the vertebral column. Treatment is surgical resection usually with neck dissection. In situ variant is that tumor which did not invade the capsule of a PA and thence was diagnosed postoperatively. In this case, the performed standard resection of a PA is considered curative.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


