CHAPTER 174 Surgery of the Anterior and Middle Cranial Base
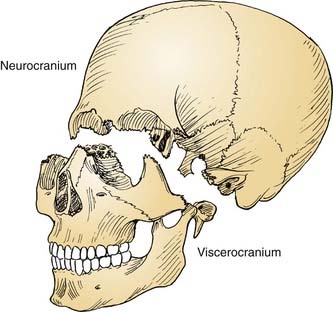
The cranial base often is conceptually divided into three anatomic regions that are named for their intracranial relations to the overlying cranial fossae: anterior, middle, and posterior (Fig. 174-1). From a diagnostic and therapeutic point of view, it is useful to consider the anterior and middle cranial base regions together, for several reasons. They contribute to what is commonly referred to as the craniofacial junction, where the neurocranium and the viscerocranium meet (Fig. 174-2). Both share anatomic relationships with the orbits, the nasal airway, and the paranasal sinuses, and they are therefore affected by similar pathologic processes. The anterior and middle cranial base regions have traditionally been approached using craniofacial disassembly techniques, however, endoscopic approaches to the skull base have assumed a growing role in selected cases while minimizing surgical morbidity. Additionally, robotic-assisted surgery has been investigated and holds promise for the future of anterior and middle cranial base surgery. The posterior and lateral cranial base are clinically regarded as a separate region, often approached with combined neurotologic and neurosurgical approaches (Chapters 127, 173, 176, and 177). This chapter focuses on the surgical management of lesions that affect the anterior and middle cranial base.
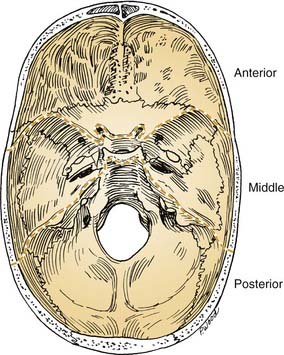
Figure 174-1. Intracranial view of cranial base showing boundaries of anterior, middle, and posterior divisions.
Surgical Anatomy
Anterior Cranial Base
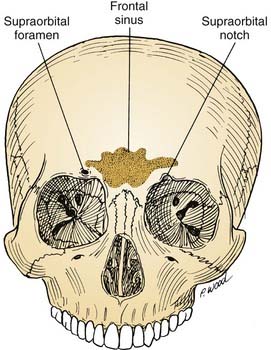
The anterior cranial base can be defined as that portion of the skull base adjacent to the anterior cranial fossa. It is bounded anteriorly by the frontal bone, which contains two surgically important structures: the frontal sinus and the supraorbital foramina (Fig. 174-3). The anatomy of the anterior cranial base is described in detail in Chapter 175, and briefly reviewed here. The supraorbital foramina, which may be incomplete (and therefore referred to as supraorbital notches), transmit the supraorbital nerves and vessels. These vessels supply the galea and the pericranium of the frontal region, and their preservation facilitates subsequent use in reconstructing anterior cranial base defects.
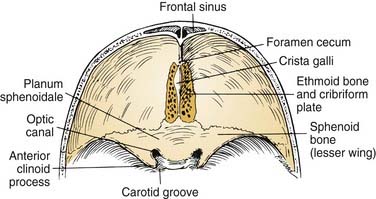
Superiorly, the anterior cranial base is formed by the frontal, ethmoid, and sphenoid bones (Fig. 174-4). An important visible landmark is the foramen cecum, which is the site of a communication between veins of the nasal cavity and the origin of the superior sagittal sinus. The next landmark, the crista galli, protrudes upward from the midline to provide attachment for the falx cerebri. On either side of the crista are the openings of the cribriform plate, through which olfactory nerves are transmitted. Just posterior to the last of these olfactory foramina is a smooth-surfaced area known as the planum sphenoidale; it forms the roof of the sphenoid sinus when the sinus is well pneumatized. The anterior clinoid processes and lesser sphenoid wings delineate the most posterior limit of the anterior cranial base and delineate its boundary with the middle cranial base. Between and slightly below the clinoids are the optic canals and the internal carotid arteries (ICAs).
Extracranially, the anterior cranial base is related to the nasal cavity, the ethmoid and sphenoid sinuses, and the orbits (Fig. 174-5). The floor of the anterior cranial fossa is uneven, with the relatively flat orbital roofs sloping downward as they join the roof of the ethmoid sinuses medially. Thus, the ethmoid roof is lower than the orbital roof. The downward slope becomes even more exaggerated as the ethmoid roof joins the cribriform area (the roof of the nasal cavity), near the midline, which usually is the lowest point of the anterior skull base. The relative relationship of the height of the cribriform plate to the roof of the ethmoid is variable and has been classified by Keros into three categories: class 1, 1 to 3 mm; class 2, 4 to 7 mm; and class 3, 8 to 16 mm.1
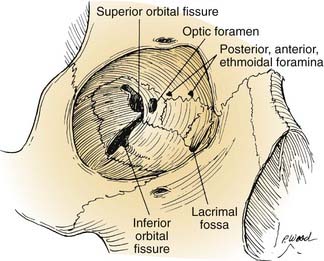
The orbits contain several landmarks that can help surgical orientation during cranial base operations (Fig. 174-6). The superior orbital fissure transmits the oculomotor, trochlear, ophthalmic, and abducens nerves (cranial nerves III, IV, V1, and VI) and the ophthalmic vein, and it communicates with the middle cranial fossa. The inferior orbital fissure contains the maxillary nerve (V2) and communicates primarily with the pterygopalatine fossa; the lateral end of this fissure is an important landmark for the placement of lateral orbital osteotomies. The optic canal transmits the optic nerve and the ophthalmic artery. The anterior and posterior arteries transmitted through ethmoid foramina should be controlled to reduce intraoperative bleeding in the nasal vault region. More important, these ethmoid foramina mark the position of the frontoethmoid suture line, a valuable guide to the level of the ethmoid roof and the anterior fossa floor. The posterior ethmoid foramen is of additional significance because of its consistent relationship with the optic canal, found 4 to 7 mm posteriorly.
Middle Cranial Base
The middle cranial base forms the floor of the middle cranial fossa. From the intracranial perspective (Fig. 174-7), the middle cranial base begins anteriorly at the posterior edge of the lesser sphenoid wing; posteriorly it ends at the posterosuperior edge of the petrous part of the temporal bone. The intracranial surface of the middle cranial base is formed by the greater wing and body of the sphenoid bone and the petrous and squamous portions of the temporal bone. As such, it forms the roof of the infratemporal fossa, middle ear, mastoid, and condylar fossa and the lateral wall of the sphenoid sinus.
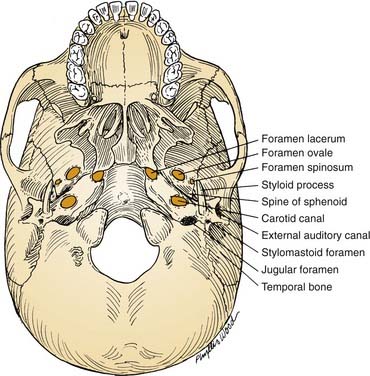
From an extracranial viewpoint, the middle cranial base extends from the posterolateral walls of the maxillary sinuses anteriorly to the petro-occipital sutures posteriorly (Fig. 174-8). It is formed by the greater wing and body of the sphenoid bone and by the temporal bone, including the condylar fossa. As on its intracranial surface, this region also contains numerous openings for major nerves and blood vessels, including the foramina ovale, spinosum, and lacerum and the stylomastoid foramen (cranial nerve VII), the jugular foramen (internal jugular vein, inferior petrosal sinus, cranial nerves IX, X, and XI), and the carotid canal (entrance of ICA into the temporal bone).
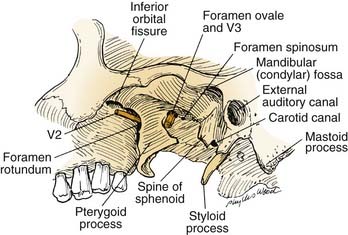
Operative approaches in this region often traverse the infratemporal fossa. Here, the muscles of mastication—the temporalis, masseter, and medial and lateral pterygoids—receive their blood supply from branches of the internal maxillary artery, which should be preserved if, for example, the versatile temporalis muscle is to be used as a flap for reconstruction. Both the lateral and medial pterygoid muscles originate in part from the lateral pterygoid plate (of the sphenoid bone), which serves as an excellent landmark owing to the following features: (1) it is a palpable guide for surgical orientation; (2) it can be easily exposed by dissection carried out medially along the greater sphenoid wing; and (3) it is easily identified on radiographic imaging studies, especially computed tomography (CT). The root of this lateral pterygoid plate is situated immediately posterior to the foramen rotundum and anterior to the foramen ovale. It can be used as an index to the positions of the maxillary (V2) and mandibular (V3) divisions of the trigeminal nerve (Fig. 174-9). After the foramen ovale is identified, the foramen spinosum (transmitting the middle meningeal artery) will be found just posterior to it. This leads to the next critical landmark: the spine of the sphenoid.
The sphenoid spine, which is situated just medial to the condylar fossa, serves as a palpable, radiographically identifiable landmark that is important because of its location immediately lateral to the carotid canal (see Fig. 174-8). It helps the surgeon locate the highest portion of the cervical ICA, which can be followed distally to expose and mobilize the petrous ICA. Because the sphenoid spine is just medial to the condylar fossa, the mandibular condyle often is displaced anteroinferiorly or resected to enhance exposure of the ICA as it enters the cranial base.
Preoperative Considerations
The principles of the clinical evaluation of presenting symptoms and physical findings (Chapter 175) and diagnostic imaging2 (Chapter 135) are considered in detail elsewhere in this book.
Angiography
Angiography also gives anatomic information about the integrity of the circle of Willis, existing collateral circulation to the cerebral hemispheres, and other possible anomalous blood supply patterns that may dictate the approach or prudent use of embolization.3
Angiography alone, however, does not give sufficient physiologic information about the adequacy of the circle of Willis or other collaterals with regard to supplying circulation to the brain.4 To obtain detailed physiologic information, cerebral blood flow studies are performed.
Cerebral Blood Flow Evaluation
If a cranial base lesion involves or impinges on the ICA or if it will be necessary to manipulate the ICA during surgery, cerebral blood flow studies should be considered. These studies give a physiologic index of the adequacy of the circulation to the brain in a quantitative way and are useful for predicting whether a patient can tolerate occlusion of the ICA without major neurologic consequence.5–8
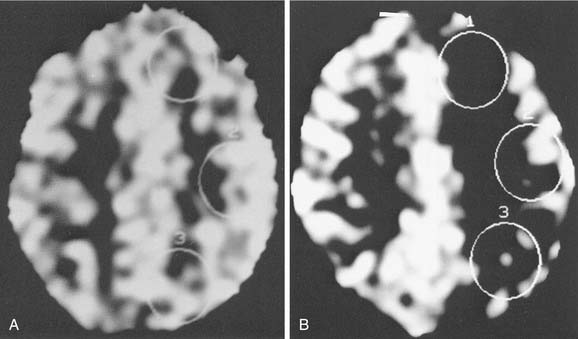
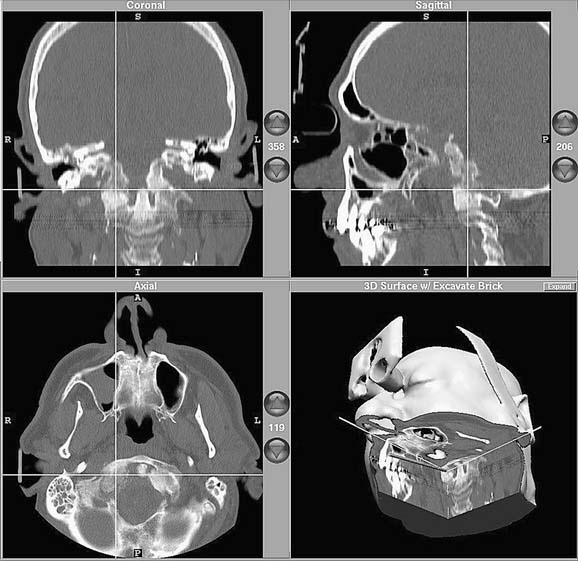
The test begins with a temporary occlusion of the ICA by means of a balloon-tipped catheter inflated within the vessel. The catheter is placed percutaneously through the femoral artery and guided into the cervical portion of the ICA under fluoroscopic control, just as is done for routine cerebral angiography. This is performed with the patient awake, and serial neurologic assessments are done during the 15-minute ICA occlusion. If a neurologic deficit develops during this part of the test, occlusion is immediately discontinued. A patient demonstrating such a deficit is considered to have failed the test and is presumed to be highly dependent on the flow in that ICA; thus, the risk for stroke is increased if the carotid is compromised at surgery. If the patient tolerates 15 minutes of ICA occlusion without the development of neurologic deficit, further study uses a quantitative test in which stable xenon gas is inhaled. The inhaled xenon is distributed throughout the circulation and into the brain, where it is visible on CT scanning; this gives a picture of cerebral blood flow distribution (Fig. 174-10). This xenon-enhanced CT scan is performed with the balloon inflated and deflated in the ICA. The uptake of xenon within both cerebral hemispheres is quantitated using the digitized data from the CT scan (when xenon CT is not available, single-photon emission computed tomography [SPECT] imaging can give similar blood flow imaging, although it is not quantitative).
Preoperative Embolization
Another advantage of preoperative angiography is the possibility for elective embolization of vascular tumor beds to help reduce intraoperative bleeding. In addition, the ICA may be electively embolized using detachable intra-arterial permanent balloons if the decision is made preoperatively that the ICA will have to be sacrificed. This is sometimes done to permit the resection of malignant tumors. These procedures are considered in more detail in Chapter 136.
Other Evaluations
Pathology and Management Planning
Benign tumors and related lesions affecting the skull base have been reviewed by several authors and are summarized in Box 174-1.9–11 In general, benign cranial base neoplasms are managed surgically. The surgical technique often involves “piecemeal” removal, with careful progression from one surgical landmark to the next, to permit the maximal preservation of functionally important structures. For larger tumors or those for which removal carries a high risk of damage to neighboring structures, incomplete resection with postoperative irradiation may be indicated.12
Box 174-1 Benign Lesions of the Skull Base
Modified with permission from Dickins JRE. Approach to diagnosis of skull base lesions. Am J Otol. 1981;3:35.
Malignant skull base lesions are listed in Box 174-2.10 With some notable exceptions (i.e., leukemia, lymphoma, myeloma, metastases), malignant neoplasms are managed surgically, although in most cases surgery will not be used as the sole modality. Adjuvant management with radiation (by external beam, implantation, or brachytherapy) or chemotherapy is usually included in the therapeutic plan. Single-modality radiation therapy, including sterotactic radiotherapy, has been recommended by some investigators for certain smaller lesions.13 Malignant lesions are surgically removed en bloc, with margins of uninvolved tissue after broad circumferential exposure whenever possible.14
Box 174-2 Malignant Lesions of the Skull Base
Modified with permission from Dickins JRE. Approach to diagnosis of skull base lesions. Am J Otol. 1981;3:35.
Anesthesia
Anesthetic management is crucial for determining the outcome of cranial base operations. Techniques of neuroanesthesia are used, with the primary goals of maximal neuronal preservation and simultaneous facilitation of a controlled surgical environment.15 Maintenance of hemodynamic factors is key in this scheme, because cerebral blood flow cannot be allowed to drop below critical levels for any significant length of time. Thus, close monitoring of arterial pressure, central venous pressure, cardiac function, and urine output is of paramount importance.
Operative Techniques
General Considerations
As the field of skull base surgery has evolved, more and more lesions previously thought to be unreachable or unresectable are now able to be addressed surgically. The contraindications to skull base surgery for malignancy as outlined by Donald16 include anatomic, tumor, and patient factors. Absolute anatomic contraindications include involvement of the brain stem, portions of the cerebrum, superior sagittal sinus, both internal carotid arteries, both cavernous sinuses and certain vital bridging veins. Tumor factors representing contraindications to resection usually include distant metastic disease; although some authors advocate significantly prolonged survival in those with isolated distant metastases after resection of the primary tumor, this has not been well demonstrated in skull base tumors. Certain malignancies typically display aggressive behavior despite treatment, which must be considered before extensive resection. Patients should be well informed and motivated and without medical contraindications to the operation as well.16
Overview of Approaches
Surgical treatment of lesions involving the anterior skull base, nasal cavity, and paranasal sinuses has evolved into a single category of craniofacial approaches. A myriad of surgical approaches have been developed for exposure of the anterior and middle cranial base regions; these range from purely intracranial to purely extracranial. However, most approaches for dealing with lesions of the skull base use combined intracranial and extracranial methods. For anterior cranial base lesions, the most commonly used approaches combine frontal craniotomy with some form of transfacial (transnasal, transmaxillary, or transorbital) exposure. Most commonly, a team of neurosurgeons and otolaryngologists performs this procedure resulting in a bifrontal craniotomy from above and a transfacial approach from below. The transfacial approach often involves midfacial degloving, and facial disassembly, requiring facial incisions and facial osteotomies. Lateral rhinotomy, removal of the frontonasal unit, Le Fort I and II osteotomies, and splitting of the maxilla have been described as means of accessing lesions of the anterior cranial base. Janecka and coworkers17 described the facial translocation approach, which also involves an extensive facial incision and facial disassembly for access to tumors in the anterior cranial base, cavernous sinus, clivus, and infratemporal fossa.
Since the 1990s, endoscopic sinus surgery has virtually replaced the conventional open techniques used by otolaryngologists in treating sinonasal disorders and is discussed in detail in Chapter 175. Use of the endoscope as a supplement to the surgical approaches can alleviate the need for some facial incisions by allowing the surgeon to observe areas hidden from the field of view of the microscope. For those tumors that invade the anterior cranial base, the endoscope may be utilized as an adjunct to the standard bifrontal craniotomy by allowing visualization of paranasal extension into the sphenoid, ethmoid, frontal, and maxillary sinuses. From nasal or maxillary portals, the endoscope allows visualization of all of the paranasal sinuses. As the technology of endoscope optics and video systems improves, the role of endoscopic cranial base surgery will augment and supplement current microsurgical techniques. Intraorbital lesions may be approached by frontal craniotomy or subcranial approaches, often combined with transfacial approaches.18
For middle cranial base lesions, access is most often provided by combining temporal or frontotemporal craniotomy with infratemporal fossa dissection, transfacial exposure, or transtemporal techniques. In addition, endoscope-assisted and image-guided navigational approaches are increasingly being applied (see Chapter 177). In anterior and middle cranial base approaches, craniofacial disassembly techniques have been widely used.
Implicit in the term craniofacial disassembly is the concept of the systematic, stepwise dissection of cranial and facial soft tissues on the basis of the knowledge of regional vascular territories and functional anatomy, followed by osteotomies and dismantling of the craniofacial skeleton. Some of these techniques, which were developed originally by plastic and reconstructive surgeons for the correction of congenital craniofacial deformities, are of major importance in cranial base surgery, because they allow wide exposure of the skull base through temporary displacement of the viscerocranium.19,20 The enhanced exposure of the skull base from below the neuraxis significantly reduces the need for brain retraction and therefore helps minimize postoperative neurologic dysfunction. It also allows the surgeon greater oncologic precision during the extirpative phase, with preservation of the functional and aesthetic units of the face for reconstruction.58
Planning the Operative Approach
Whether the surgery is being performed for a benign tumor, a malignancy, or other indications (e.g., inflammatory or vascular lesions, CSF fistula), the approach should be planned and executed to accomplish the four specific goals that are applicable to all cranial base operations, summarized in Box 174-3.
GOAL 2
Image-guided (stereotactic) computer-assisted surgery has become a very useful alternative—or adjunct—to wide surgical exposure in selected cases.21–26 Such “navigational surgery” employs one of a variety of referencing systems that can assimilate preoperative imaging studies into a computer-based algorithm that allows image reformatting in three dimensions (The preoperative reference images may be CT- or magnetic resonance imaging [MRI]-based, or they may be a “fusion study” that takes advantage of both types of studies). The reformatted images then serve as the basis for intraoperative navigation, which involves the use of an interactive probe that transmits spatial-orientation information back to the computer via transmitters; these can be either electromagnetic or optical. The computer screen then displays the appropriate preoperative images with the location of the probe superimposed, thus “targeting” the area of interest (Figure 174-11). With navigational systems, structures that are anatomically fixed (i.e., orbital walls, carotid canal, optic canal) can be precisely located, and the risk of injury can be minimized.
Reported uses of image-guided surgery include primary and revision endoscopic sinus surgery, osteoplastic frontal sinusotomy, transsphenoidal hypophysectomy, endoscopic cerebrospinal fluid leak and encephalocele or meningocele repair, endoscopic skull base tumor resection, orbital surgery, and endoscopic pterygomaxillary fossa biopsy.27,28
The American Academy of Otolaryngology–Head and Neck Surgery endorses use of computer-aided surgery for the following specific procedures or conditions29:
Intraoperative MRI or CT also has been proposed as a useful adjunct in skull base surgery. Proponents advocate the improved ability to detect residual tumor and evaluate for intraoperative complications, however, limitations of time and cost have prevented its widespread use outside of certain academic centers.30
Although large, multi-institutional prospective studies are yet to be accomplished, smaller studies and anecdotal reports demonstrate the efficacy of endoscope-assisted approaches for the treatment of selected skull base problems, most notably sinonasal neoplasms, pituitary tumors, and CSF leaks.31–37
Stamm38 classifies endoscopic skull base approaches as follows:
The location and suspected pathology dictate the selection of approach.
Endoscopy-based approaches to the nasal cavity, olfactory groove, nasopharynx, ethmoid roof (anterior skull base), orbit and orbital apex, sphenoid sinus and sellar and parasellar areas, clivus, cavernous sinus, optic chiasm, pterygopalatine and infratemporal fossae, parapharyngeal space, craniocervical junction, petrous apex, and jugular foramen all have been described. In short, access to the central skull base from the frontal sinus to C2 and from the sella to the jugular foramen is now possible using the expanded endonasal approach.39–43
The endoscopic approach to the orbital apex and optic canal for orbital decompression in fibrous dysplasia, Graves’ orbitopathy, compressive or traumatic optic neuropathy has been described. Of note, decompression is indicated only in patients with fibrous dysplasia with continuous deterioration of vision or undergoing removal of neighboring bone, but not as a prophylactic procedure.44,45 This approach also is used in the management of clival lesions, anterior skull base tumors, and vascular malformations of the skull base.46,47
Kassam and associates described an approach to identify the petrous portion of the carotid artery following identification and retrograde dissection of the vidian artery. They found this to be a consistent landmark radiographically and intraoperatively.48
Endoscopic approaches to pituitary surgery offer benefits of improved cosmesis, decreased recovery time, and fewer complications. Three approaches exist: transnasal-transethmoid, transnasal-transseptal, and direct transnasal. The direct transnasal approach has been touted because of the advantages of avoiding the ethmoid complex, decreased operative time, and improved visualization.2 Although endoscopic skull base surgery has been more extensively described for the anterior cranial base, endoscopic-assisted approaches to the middle cranial base also have been described.49
The protection of critical structures at the cranial base also is enhanced by the appropriate use of electrophysiologic monitoring, including intraoperative somatosensory evoked potentials, auditory brainstem evoked response, and EMG.14 The injection of low-dose intrathecal fluorescein has been shown to be a useful adjunct intraoperatively to decrease the CSF leak rate and, when appropriate medication is provided, has been proven safe and without significant adverse effects.50
GOAL 3
Such vascularized local flaps are preferable to free flaps or nonvascularized grafts when available, however others have displayed excellent closure rates with materials ranging from acellular dermis to free microvascular transfers.51,52 In general, smaller defects (less than 2 cm) are less likely to require vascularized tissue to achieve closure.53,54
Robotic endoscopic skull base surgery and transoral robotic surgery have been advocated by some researchers for the advantages of improved visualization, access, precise technique, and improved watertight closure following endoscopic skull base surgery. Although promising, more clinical investigation is needed before widespread acceptance of these techniques.55,56
GOAL 4
The choice of operative approach should reflect consideration for functional and aesthetic reconstruction, and it should include the placement of incisions within natural skin lines that respect aesthetic units of the face. The soft tissue closure should follow plastic surgery principles. Excellent skull base exposure usually can be achieved by elective osteotomies and the temporary removal of craniofacial bone segments, which are subsequently replaced, thereby preserving facial contour.17,51,57–59 Even in cases in which aesthetically important segments should be removed for oncologic reasons, acceptable cosmesis can usually be accomplished through the judicious use of bone grafts and soft tissue flaps or through surgical closures designed to support alloplastic or prosthetic materials.
Anterior Cranial Base Approaches
Surgical approaches to the anterior cranial base include methods that are purely extracranial and those that use combined extra- and intracranial exposures. The extracranial techniques—external ethmoidectomy, frontal sinusotomy, and intranasal ethmoidectomy—are suitable only for the management of discrete, well-localized lesions, such as CSF fistulas and some very limited benign anterior cranial base tumors. These procedures are well described elsewhere.60,61
Technical Note: The Bicoronal Incision
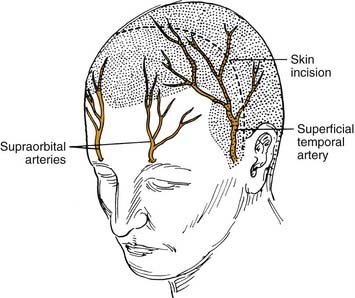
The bicoronal incision should be in the true coronal plane at the level of the top of the helix of the ear or slightly anterior to it (Fig. 174-12). A short, forward-directed, preauricular extension can be made on both sides to enhance scalp flap rotation. This coronal placement of the incision preserves the anterior branches of the superficial temporal artery, enhancing viability of the scalp flap. In addition, it substantially increases the length of vascularized galea and pericranium available for reconstruction as compared with incisions along the anterior hairline, the midforehead crease, or the brow. Additional length can be gained by back-elevating the scalp flap before division and elevation of the pericranial flap.
The central portion of the anterior scalp flap (i.e., the portion between the two superior temporal lines) is elevated in the subgaleal plane if a pericranial flap may be used and in the subperiosteal (subpericranial) plane if not. Lateral to the two superior temporal lines, it is elevated in the plane just above the deep temporal fascia. Therefore, at the temporal lines, the pericranium should be sharply incised to separate it from the origin of the deep temporal fascia (see Fig. 174-15). The pericranium is divided far enough posteriorly to provide sufficient length to the flap; then the dissection is carried forward in the subperiosteal plane as described earlier.
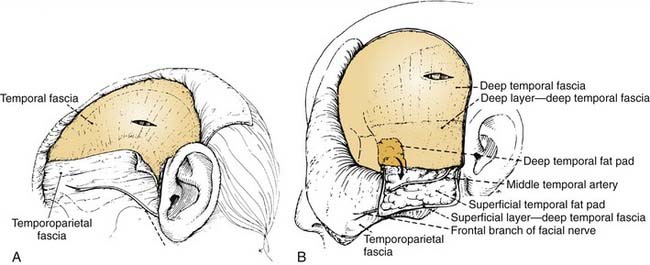
This deep temporal fascia begins to split into superficial and deep layers, beginning at approximately the level of the superior orbital rim.62 These fascial layers diverge to envelop the lateral and medial surfaces of the zygomatic arch inferiorly; between the layers is the temporal fat pad. The frontal (temporal) branches of the facial nerve course just superficial to the zygoma along the superficial temporal fascia (temporoparietal fascia) and are prone to injury if the dissection is done at that level (Fig. 174-13A). Often these injuries can be avoided by maintaining the plane of dissection at the level of the deep layer of the deep temporal fascia (i.e., at the surface of the temporalis muscle itself). This deep plane of exposure essentially elevates the fat pad along with the superficial fascia and the superficial layer of the deep temporal fascia, thereby protecting the facial nerve branches (see Fig. 174-13B).63,64 After the dissection reaches the level of the zygomatic arch, the arch is palpated, and its superior surface is directly exposed by sharply incising the fat pad and periosteum.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree