CHAPTER 18 Sleep Apnea and Sleep Disorders
Over the past decade, interest in sleep and sleep disorders has increased substantially. The majority of this renewed interest within the otolaryngology community has been focused on the sleep-related breathing disorder OSA. As the waistline of the average American has grown, so too has the incidence of OSA. Population-based studies suggest that 2% of women and 4% of men older than age 50 years have symptomatic OSA.1 Advances have also been made in the understanding of the pathophysiology of OSA, diagnostic methods, and medical and surgical treatments. The recognition that sleep disorders require a multidisciplinary approach has led to the creation of a new medical discipline—sleep medicine—with teams made up of internists, pulmonologists, otolaryngologists, neurologists, pediatricians, psychiatrists, oral/maxillofacial surgeons, dentists, behavioral psychologists, and nutritionists who work together to take care of patients with sleep disorders. Considerable advances have been made in the diagnosis and management of sleep disorders, and otolaryngologists are at the forefront of this new field, providing a host of both traditional and novel operative techniques to facilitate treatment.
Historical Perspectives
The idea of obesity being intimately associated with daytime somnolence seems to have been first written about by Charles Dickens in The Posthumous Papers of the Pickwick Club, originally published in 1837. Dickens gives a vibrantly descriptive picture of Joe, a boy so obese that he has difficulty breathing, sounds as if he is snoring even when awake, and frequently falls asleep while standing. In addition, there is evidence to suggest that some of our most noted leaders and dictators suffered from this problem. The 20th president of the United States, William Howard Taft, had a body mass index of 42 kg/m2 while in office and was reported to snore, to fall asleep during the day frequently, and to have hypertension.2 Chouard and colleagues describe several reasons to suspect Napoleon I (1769-1821) had OSA in the last decade of his life: he was obese and retrognathic, his neck was short and thick, he had nasal obstruction, he frequently slept during the day, he complained of declining energy and intellect, and he was quite tired looking and somewhat disheveled.3 In 1956, Drs. A. G. Bicklemann, C. S. Burwell, and colleagues drew on Dickens’ description in their case report titled “Extreme Obesity Associated with Alveolar Hypoventilation: a Pickwickian Syndrome.” This frequently cited paper served as the first description of the Pickwickian syndrome in an academic journal.4 Sleep apnea was first described as a clinical entity in 1965, but it was not until the 1970s that Elio Lugaresi’s group provided a complete description of the OSA syndrome with its potential adverse cardiovascular effects.
In the infancy of this new diagnosis, treatment options were limited to tracheostomy or weight loss. This changed in the early 1980s with the introduction of the uvulopalatopharyngoplasty (UPPP) as described by Fujita and colleagues5 and Simmons and colleagues.6 It was not long after the introduction of UPPP that it was recognized that although this new surgical intervention was helpful in many cases, it left half of the patients with persistent sleep apnea and could be a painful procedure. It was during this same time period that Colin Sullivan, a young Australian medical researcher, developed nasal continuous positive airway pressure (CPAP), which today remains the first-line treatment of adult OSA.
Classification of Obstructive Sleep-Related Breathing Disorders
Sleep-related breathing disorders range from partial airway collapse and increased upper airway resistance to episodes of hypopnea or complete airway collapse with sleep apnea (Box 18-1). In addition, a number of indices are used to describe sleep-disordered breathing (Box 18-2).
Box 18-1 Respiratory Event Definitions and Types
From Kushida CA, Littner MR, Morgenthaler T, et al. Practice parameters for the indications for polysomnography and related procedures: an update for 2005. Sleep. 2005;28:499-521.
| Respiratory Event | Definition |
|---|---|
| Apnea | A cessation of airflow for at least 10 seconds |
| Hypopnea | A reduction in airflow (≥30%) at least 10 seconds with ≥4% oxyhemoglobin desaturation |
| OR a reduction in airflow (≥50%) at least 10 seconds with ≥3% oxyhemoglobin desaturation or an electroencephalogram (EEG) arousal | |
| Respiratory effort-related arousal (RERA) | Sequence of breaths for at least 10 seconds with increasing respiratory effort or flattening of the nasal pressure waveform leading to an arousal from sleep when the sequence of breaths does not meet the criteria for an apnea or a hypopnea |
| Obstructive | Continued thoracoabdominal effort in the setting of partial or complete airflow cessation |
| Central | The lack of thoracoabdominal effort in the setting of partial or complete airflow cessation |
| Mixed | A respiratory event with both obstructive and central features. Mixed events generally begin as central events and end with thoracoabdominal effort without airflow |
Box 18-2 Indices of Sleep-Disordered Breathing
| Indices | Definition |
|---|---|
| Apnea index | Number of apneas per hour of total sleep time |
| Hypopnea index | Number of hypopneas per hour of total sleep time |
| Apnea-hypopnea index | Number of apneas and hypopneas per hour of total sleep time |
| Respiratory effort–related arousals (RERAs) index | Number of RERAs per hour of total sleep time |
| Respiratory disturbance index (RDI) | Number of apneas, hypopneas, and RERAs per hour of total sleep time |
| Central apnea index | Number of central apneas per hour of total sleep time |
| Mixed apnea index | Number of mixed apneas per hour of total sleep time |
Snoring
Snoring is sound generated by the vibration of the pharyngeal soft tissues. It is often louder during inspiration than expiration. It affects at least 40% of men and 20% of women and often accompanies sleep-disordered breathing (SDB).7 However, it can occur in isolation and by definition is not associated with symptoms of excessive daytime sleepiness or insomnia. Snoring, in the absence of OSA, is documented when habitual audible snoring occurs with an apnea hypopnea index (AHI) of less than five events per hour without daytime symptoms. Polysomnography (PSG) is not required for diagnosis, but when performed it reveals an audible microphone signal not associated with arousals, desaturations, airflow limitation, or arrhythmias.8
Upper Airway Resistance Syndrome
The term upper airway resistance syndrome (UARS) was first used to describe patients who do not meet the criteria for OSA syndrome but who experience excessive daytime somnolence and other debilitating somatic complaints.9 UARS is characterized by respiratory effort–related arousals (RERAs). A RERA is defined as a sequence of breaths over at least 10 seconds with increasing respiratory effort that terminates with an arousal.10 A RERA is detected using esophageal pressure manometry, which reveals a pattern of progressively increasing negative esophageal pressure followed by an arousal.10,11 PSG reveals frequent arousals associated with snoring, abnormally negative intrathoracic pressure, or increased diaphragmatic electromyogram activity.9 The current International Classification of Sleep Disorders does not consider UARS to be a separate sleep disorder, but recommends that it be included under the definition of OSA because the pathophysiology is similar.8
Obstructive Sleep Apnea Syndrome
OSA is defined by five or more respiratory events (apneas, hypopneas, or RERAs) in association with excessive daytime somnolence, waking with gasping, choking, or breath-holding, or witnessed reports of apneas, loud snoring, or both. Each episode of apnea or hypopnea must last a minimum of 10 seconds, is commonly accompanied by reductions in blood oxygen saturation of at least 3% to 4%, and is usually terminated by brief, unconscious arousals from sleep. Snoring between apneas is a frequent complaint of bed partners and is often the symptom that prompts these patients to seek medical attention; although, excessive daytime somnolence is a common presenting complaint. Automobile accidents and increased cardiovascular morbidity and mortality are frequent complications if OSA is left untreated. Many OSA sufferers complain of awakening from sleep with morning headache, sore throat, and fatigue or a feeling of being unrefreshed regardless of the duration of sleep (Box 18-3). OSA is exacerbated by ingestion of alcohol, sedative use, and weight gain.
Pathophysiology
Fujita classified the patterns of obstruction by anatomic location in the following manner: type I, collapse in the retropalatal region only; type II, collapse in both retropalatal and retrolingual regions; type III, collapse in the retrolingual region only.12 Subsequent studies have evaluated the prevalence of retropalatal versus retrolingual obstruction in patients with OSA and snoring and found that both retrolingual (77% versus 40%) and retropalatal (100% versus 70%) obstruction are increased in OSA patients, confirmed by PSG, compared with snorers.13
Nasal obstruction contributes to increased airway resistance and may worsen OSA, but it is rarely the sole cause. Nasal obstruction may contribute to open-mouth breathing during sleep, which increases upper airway collapsibility and may decrease the efficacy of dilator muscles.14 Snoring is often a presenting symptom of OSA and can be caused by nasal obstruction. Surgical procedures aimed at improving nasal breathing have demonstrated subjective improvement in snoring following correction of nasal obstruction.15 Even though isolated treatment of the nasal airway rarely leads to cure of OSA, it may allow for decreased CPAP levels to be used for subsequent treatment.16 Nasal obstruction can be caused by bony and cartilaginous deformation or from soft tissue changes. Because there are many causes of nasal obstruction, they all warrant investigation when evaluating a patient with OSA.
Obesity is a major risk factor for OSA. The increased fat deposition around the neck and parapharyngeal spaces is postulated to narrow and compress the upper airway and may offset the effects of dilator muscles that maintain airway patency.17 Obesity is also thought to contribute to OSA through its deleterious effects on metabolism, ventilation, and lung volume, resulting in a mismatch between alveolar ventilation (V) and pulmonary perfusion (Q). Obesity can significantly reduce lung volume, which results in a reduction of functional residual capacity. It has been observed that changes in lung volume significantly reduce pharyngeal upper airway size through the mechanical effect of tracheal and thoracic traction, referred to as “tracheal tug,” increasing the risk for airway collapse.18
Adenotonsillar hypertrophy is the major cause of obstructive sleep apnea in children. In adults there are multiple structural characteristics associated with OSA. Craniofacial variations that have been associated with OSA include increased distance of the hyoid bone from the mandibular plane, decreased mandibular and maxillary projection, a downward and posterior rotation of mandibular and maxillary growth, increased vertical facial length, increased vertical length of the posterior airway, and increased cervical angulation.19
Neuromuscular tone contributes to the patency of the upper airway. During sleep this tone decreases and the airway tends toward collapse. The genioglossus muscle is considered to be the most important muscle in maintaining airway patency in OSA. Elevated genioglossal and tensor palatini muscle activity has been observed in awake OSA patients compared with normal awake subjects who have lower levels of activity. This suggests that even while the patient is awake, airway dilator muscle activity compensates for a more anatomically compromised upper airway in the OSA patient.17 Studies are ongoing to determine the extent and importance of neuromuscular tone in maintaining airway patency during sleep.
Consequences of Untreated Obstructive Sleep Apnea
A number of negative health effects have been attributed to untreated OSA including increased mortality, an increase in cardiovascular disease, and neurocognitive difficulties. In a retrospective study, He and colleagues found that untreated OSA patients with an apnea index (AI) greater than 20 had a statistically significant increase in mortality compared with patients with an apnea index less than 20.20 He also found that untreated patients with an AI greater than 20 had a 63% probability of surviving 8 years compared with 96% in those with an AI less than 20.20 Additionally, untreated OSA is reported to increase the risk of fatal and nonfatal motor vehicle accidents by 2.5-fold.21 A significant proportion of the mortality and morbidity related to OSA occurs through its effect on the cardiovascular system, including hypertension, coronary heart disease, congestive heart failure, arrhythmias, sudden death, pulmonary hypertension, and stroke. Untreated moderate and severe OSA has been reported to result in a threefold increase in fatal and nonfatal cardiovascular events, when compared with both healthy men without OSA and men with CPAP-treated OSA.22 Treatment of OSA with CPAP has also been reported to lower blood pressure by 10 mm Hg.23
Untreated OSA has been demonstrated to be an independent risk factor for insulin resistance.24 Recently, it has been suggested that OSA may contribute to the development of diabetes and metabolic syndrome, the term used to describe the commonly occurring conditions of obesity, insulin resistance, hypertension, and dyslipidemia. However, further research is needed to determine whether there is an independent link between OSA and metabolic abnormalities.
The prevalence of gastroesophageal reflux disease (GERD) in OSA patients is significantly higher than in the general population.25–27 Even though these disorders commonly occur together, no temporal or causal relationship has ever been demonstrated between the two. This may reflect the fact that they share similar risk factors. Treatment of OSA with CPAP has been demonstrated to decrease the occurrence of GERD.28
Aside from the obvious physical effects of OSA, such as excessive daytime sleepiness and impaired mood, neurocognitive deficits have also been associated with OSA. Untreated OSA has been documented to cause problems with attention, working memory, and executive function, all of which are improved with CPAP treatment.29 Bed partner dissatisfaction is also a common complaint among OSA patients and treatment has been shown to improve the quality of life in both the treated individuals and their bed partners.30 Thus the benefits of treating OSA are substantial and well documented.
Diagnosis
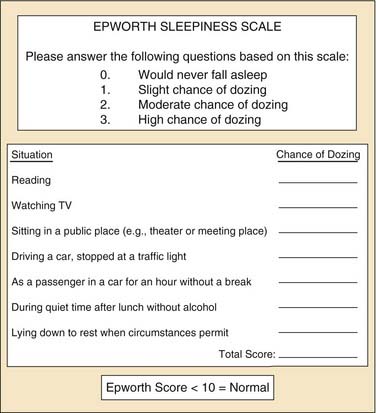
The most common symptoms of OSA include loud snoring, restless sleep, and daytime hypersomnolence. However, a broad range of signs and symptoms have been reported to occur in OSA (see Box 18-3). Obesity is a common finding in patients with OSA with reports that 70% of adult patients with OSA are obese.31 Screening, including a detailed sleep history and physical examination, is recommended for all obese patients. The Epworth Sleepiness Scale (ESS) is a widely used tool that assesses daytime sleepiness (Fig. 18-1). OSA may be suspected in patients with an ESS greater than 10.32 Sleepiness or fatigue may also be precipitated by a number of other medical conditions that should be considered when evaluating patients for possible OSA (Box 18-4). In addition, the presence of additional sleep disorders should be considered.
Due to the increased prevalence of OSA in patients with hypertension, coronary artery disease, congestive heart failure, cerebrovascular accident, and diabetes mellitus, these populations must be carefully screened for the signs and symptoms of OSA.33 It has also recently been recognized that OSA may be underdiagnosed in women due to a lower index of suspicion among physicians or underreporting of the classic symptoms by female patients. Women with OSA are more likely to report symptoms of insomnia, heart palpitations, and ankle edema.34
Although the presence of daytime somnolence and loud snoring are frequently the signs which prompt OSA suffers to seek medical attention, it is the physical examination findings that strengthen the likelihood of the diagnosis. Body mass index calculation, blood pressure measurement, and neck circumference are important general assessment parameters. In addition, body habitus, mandible and maxilla position and size, and facial characteristics should be noted. Assessment of the nose should include evaluation of any external deformity, adequacy of the nasal valve, septal position, turbinate size, nasal mucosa swelling, and the presence or absence of polyps, purulence, and rhinorrhea. In the oral cavity, appraisal of the tongue size and position, elongation of the palate and uvula, tonsil size, modified Mallampati score, dentition, and crowding of oropharynx should follow. In the neck, neck size, hyoid position, and jaw position, including retrognathia, should be evaluated (Box 18-5).
Box 18-5 Physical Examination Findings
Fiberoptic nasopharyngoscopy is an important technique for evaluation of the airway. This examination can be performed in multiple positions in awake and asleep patients and is an important tool to identify level of obstruction: nasal, retropalatal, or retrolingual (Figs. 18-2 and 18-3). Numerous studies have described the benefits and limitations of the addition of the Müller maneuver to this examination for preoperative prediction of the effectiveness of surgical interventions.35,36 The Müller maneuver is performed in an awake patient who generates negative pressure by inhaling against a closed glottis with the nose and mouth closed, which triggers airway collapse (Figs. 18-4 and 18-5). Sher and associates performed both seated and supine examinations using the Müller maneuver in OSA patients, selected those with palatal collapse, and found that 73% of patients had at least a 50% reduction in respiratory disturbance index (RDI) after UPPP.36 Aboussouan found that using the Müller maneuver to guide the decision on UPPP resulted in an AHI reduction by 50% in 78% of patients who had velopalatal collapse, compared with 36% for multilevel obstruction.35 Thus, even though the Müller maneuver can help guide surgical decision making when the area of collapse is purely retropalatal, it appears to be less useful in patients who have multilevel obstruction.
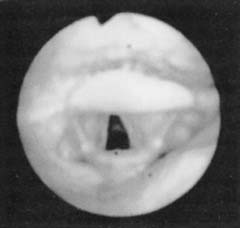
Figure 18-2. Fiberoptic view of the normal hypopharyngeal airway.
(From Troell RJ, Riley RW, Powell NB, Li K. Surgical management of the hypopharyngeal airway in sleep disordered breathing. Otolaryngol Clin North Am. 1998;13:983.)
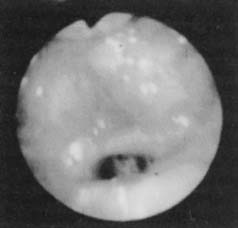
Figure 18-3. Fiberoptic view of base of tongue obstruction.
(From Troell RJ, Riley RW, Powell NB, Li K. Surgical management of the hypopharyngeal airway in sleep disordered breathing. Otolaryngol Clin North Am. 1998;13:983.)
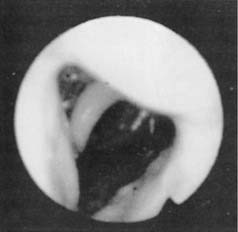
Figure 18-4. Fiberoptic view of the hypopharyngeal airway before the Müller maneuver.
(From Troell RJ, Riley RW, Powell NB, Li K. Surgical management of the hypopharyngeal airway in sleep disordered breathing. Otolaryngol Clin North Am. 1998;13:983.)
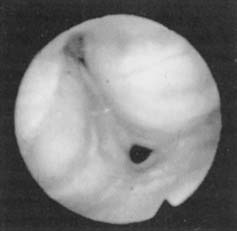
Figure 18-5. Fiberoptic view of hypopharyngeal collapse during the Müller maneuver.
(From Troell RJ, Riley RW, Powell NB, Li K. Surgical management of the hypopharyngeal airway in sleep disordered breathing. Otolaryngol Clin North Am. 1998;13:983.)
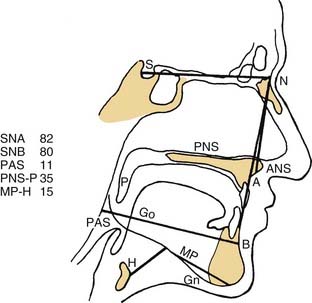
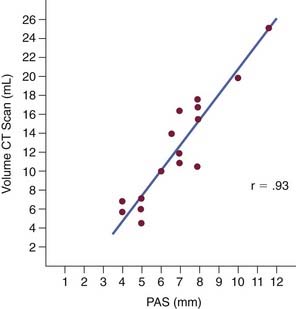
A number of radiologic techniques have been used to aid in the identification of the site and severity of upper airway obstruction or collapse in OSA. Because most of the radiologic techniques for evaluation of OSA are performed in awake patients, they provide limited information about obstruction during sleep. The most frequently used imaging modality has been the cephalometric radiograph. The cephalogram is a two dimensional representation of the airway, which is a standardized evaluation system, with broad availability and relatively low cost (Fig. 18-6).37 These films provide information on both the bony skeleton and the overlying soft tissues. Multiple studies using cephalometry have confirmed that OSA patients have inferior displacement of the hyoid, smaller posterior airway space, and longer soft palates than do non-OSA patients (Fig. 18-7).38–41 Even so, the differences between OSA and non-OSA patients noted on cephalometry have not been significant enough to allow for the use of lateral cephalograms as a sole diagnostic tool.42
Awake computed tomography (CT) provides good anatomic detail of the bone and soft tissue. Like the cephalogram, its has poor sensitivity for diagnosing OSA; however, multiple studies have demonstrated that it is a better modality to illustrate postoperative anatomic changes that correlate with improved PSG parameters.42
Magnetic resonance imaging (MRI) provides excellent soft tissue differentiation and does not require radiation exposure. However, it can be hard to tolerate, is expensive, has limited availability, and is noisy, which may preclude asleep evaluations. Like CT and cephalometry, MRI has not been useful in distinguishing OSA from non-OSA patients.43
Lastly, fluoroscopy has been used as a dynamic airway examination to directly evaluate the sites of obstruction. Somnoflouroscopy, studies done while asleep, have been shown to improve uvulopalatopharyngoplasty success rates when the site of initial obstruction is identified (67% versus 42%); however, these studies are time intensive and expose the patients to increased radiation, which limits their utility.44
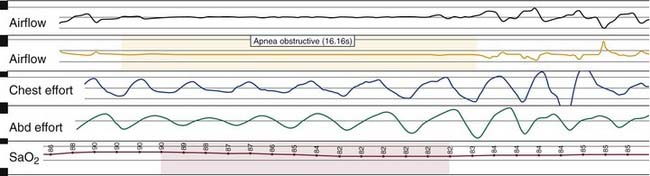
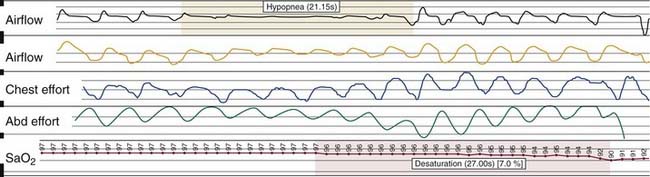
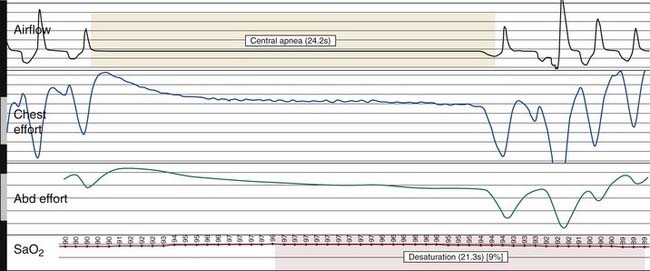
Nocturnal PSG is the gold standard for diagnosis of OSA. A full night diagnostic study is considered to be the most accurate instrument for measuring the presence and severity of OSA. The parameters monitored during PSG include electroencephalogram, electro-oculogram, submental electromyogram, electrocardiogram, airflow, thoracoabdominal effort, and oximetry (Box 18-6). The information obtained from the study is analyzed by a trained polysomnogram technologist and interpreted by a sleep physician. Classification of disordered breathing events recorded during PSG have been standardized to guide practice (Figs. 18-8 through 18-10).33
Box 18-6 Polysomnography Monitors
Treatment
Medical Treatment
Treatment is generally approached in a stepwise manner and begins with conservative medical measures. Weight loss should be recommended for all overweight patients with OSA. However, sustained weight reduction is difficult and patients often regain the lost weight. Therefore other interventions are often also recommended. Consultation with a bariatric surgeon can be considered when treating patients who are morbidly obese. Recent evidence reveals that surgically induced weight loss significantly improves obesity-related OSA and parameters of sleep quality,45 and this improvement can occur as early as 1 month after surgery.46
Continuous positive airway pressure (CPAP) is considered the gold standard treatment for moderate to severe OSA. Studies have demonstrated CPAP effectiveness in reducing the apnea-hypopnea index and in subjectively improving sleep quality; however, patient adherence remains a significant obstacle.11,47,48 CPAP acts as a pneumatic splint and prevents upper airway collapse by providing constant positive intraluminal pressure during inspiration and expiration. Numerous effects of CPAP treatment have been described, chief among these being the reduction in AHI, improvement in objective and subjective sleepiness, improvement in overall quality of life, reduction in risk of cardiovascular events, and reduction of the risk of motor vehicle accidents.20,49,50 Recently, there has also been interest surrounding the beneficial effects of CPAP to cardiovascular health due to decreased inflammation as measured by a decrease in the inflammatory markers C-reactive protein and interleukin-6, improved endothelial function, and reduced diurnal sympathetic activity.51–53 Bilevel positive airway pressure (BiPAP) and autoadjusting positive airway pressure (APAP) were developed to improve pressure titration and treat patients with neuromuscular disorders and ventilatory disease. BiPAP delivers a separately adjustable lower expiratory positive airway pressure and higher inspiratory positive airway pressure. Although BiPAP has not been shown to improve adherence in an unselected patient population, a change in the method of delivery of positive airway pressure may significantly improve adherence in individual patients.54 APAP devices autotitrate positive airway pressure to select an effective level of CPAP, which prevents upper airway collapse. The pressure changes in response to variations in airflow magnitude, airflow limitation, snoring, or airway impedance.55–57
Oral appliances have been used with success in patients with mild, moderate, and some severe OSA (Fig. 18-11), increasing the posterior oropharyngeal airway. Ferguson and colleagues conducted a crossover study comparing oral appliance therapy and nasal-CPAP therapy and concluded that oral appliances are effective in some patients with mild to moderate OSA (AHI 14-50) and are associated with greater satisfaction than CPAP.58 Adherence rates of oral appliance therapy have been reported to be as high as 77%.59 The most often reported complications from oral appliance therapy are tooth and jaw muscle pain, difficulty chewing in the morning, and excessive salivation. Although oral appliance therapy is cost effective and has higher rates of patient adherence, CPAP has proven to be more effective in reducing AHI.60 Therefore this intervention should be recommended cautiously as first-line therapy for moderate OSA, but may serve as an effective treatment option for patients with mild, symptomatic OSA.
Pharmacologic therapy has been proposed as a treatment alternative in patients intolerant of CPAP; however, there is currently insufficient evidence to recommend the use of drug therapy as primary therapy for the treatment of OSA.61 The many proposed mechanisms by which drugs may reduce the severity of OSA include increasing upper airway dilator muscle tone, increasing ventilatory drive, and increasing cholinergic tone during sleep versus reducing the proportion of REM sleep, reducing airway resistance, and reducing surface tension in the upper airway.61
Medications have been investigated as treatment for OSA. Fluticasone has shown some success in treating OSA patients with coexistent rhinitis. Kiely and colleagues reported that the AHI was lower in 24 patients given intranasal fluticasone compared with those given placebo (23.3 versus 30.3).62
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree