Chapter 121 Retinal Pigment Epithelium and Choroid Translocation in Patients with Age-Related Macular Degeneration
Epidemiology
In indtrialized countries the end-stage of age-related maculopathy (ARM) is the principal cause of legal blindness in persons older than 60 years.1–3 Age-related macular degeneration (AMD) occurs in an atrophic (one-third) and an exudative form (two-thirds). While we currently have an effective treatment to manage exudative AMD, there is no equally effective treatment in sight for geographic atrophy (GA). This situation is likely to increase the relative proportion of blindness through GA compared with exudative AMD in the future. Increasing life expectancy will account for a rise in the absolute number of GA. (Epidemiology is discussed further in Chapter 63, Epidemiology and risk factors for age-related macular degeneration.)
Alternative treatments for AMD
Exudative AMD
The current 2011 standard treatment for many patients with exudative AMD is anti-VEGF injections. (This is discussed further in Chapter 66, Neovascular (exudative or “wet”) age-related macular degeneration.)
However, for patients with a retinal pigment epithelium (RPE) tear4,5 or a large submacular hemorrhage, anti-VEGF therapy is generally less effective in restoring or improving visual acuity (VA), because the underlying anatomy of the outer retina is disrupted. In addition, even with the established anti-VEGF treatments, a small percentage of patients fail to respond. In the original MARINA study, 10% of patients receiving ranibizumab had lost ≥15 letters and overall, 15% of patients on ranibizumab were 20/200 or worse by year 2.6 A recent retrospective analysis of those patients who responded poorly to ranibizumab in both the MARINA and ANCHOR studies by year 2 has highlighted that these patients may have had disruption of retinal anatomy rather than highly active choroidal neovascularization (CNV).7 This highlights the need to consider alternative strategies, such as surgery, to improve the anatomy of the retina–RPE interface in selected patients.
Dry AMD
• Growth factors (ciliary neurotrophic factor (see http://www.clinicaltrials.gov, NCT00447954, for details)
• Modulation of gene expression (DICER protein)8
• Modulation of the visual cycle to decrease toxic by-products (see http://www.clinicaltrials.gov, NCT01002950, for details)
• Modulation of inflammation (see http://www.clinicaltrials.gov, NCT00766649, for details)
• Plasma filtration (see http://www.clinicaltrials.gov, NCT00460967, for details).
Nevertheless, to date, there is no approved treatment regimen for GA.
Surgical treatment
1. Removal of the submacular choroidal membrane and/or hemorrhage. This was shown to be of little benefit in the Submacular Surgery Trials (SST), although the percentage of patients with more than 6 lines VA loss was statistically smaller in the subgroup of patients with large submacular hemorrhage (group B) when compared to natural history.9
2. Macular translocation after 360° retinectomy. This method was first reported by Machemer et al. in 1993.10 A tilted image in successful cases, complex and time-consuming surgery and a high percentage of vision-threatening complications such as proliferative vitreoretinopathy (PVR), particularly in the earlier studies, remain drawbacks of this technique11 (described in more detail in Chapter 120, Macular translocation). In a RCT in Tübingen in 50 patients however, with PDT treatment in the control arm, vision gain was significantly greater in the surgical arm.12 The limit to the extent of retinal rotation possible makes it most effective for smaller lesions, particularly classic CNV, but this clinical presentation is also particularly well suited to anti-VEGF treatments, making surgery less easy to justify in the modern era.
3. Minimal macular translocation by scleral infolding and the induction of retinal detachment was described by de Juan in 1998.13 The macular translocation achieved, however, was less than one disk diameter.14 This has indeed limited its use since the advent of treatments that were particularly effective in small lesions, such as PDT and anti-VEGF (see Chapter 120, Macular translocation).
4. The displacement of an acute hemorrhage by gas and recombinant tissue plasminogen activator, with or without vitrectomy, with subsequent treatment with PDT or anti-VEGF.15,16 Several case series suggest a clear benefit over natural history.17,18
5. Transplantation of an autologous graft of RPE, Bruch’s membrane, choriocapillaris, and choroid. This method was first described in a case report by Peyman et al. in 1991.19 Initially, the graft was harvested from the edge of the macular RPE defect.20 Later studies utilized mid-peripheral tissue.21–26 The most frequent complications of this transplantation technique were recurrent CNV, RD, PVR, nonrevascularization of the graft, macular pucker, and postoperative hemorrhages.
Both macular translocation surgery and the transplantation of a free autologous graft of RPE and choroid will probably achieve a better VA than no intervention. Though macular translocation surgery might produce better outcomes,27 it has a greater risk of complications and there is a physical limit to the amount of rotation possible (even after 360° retinectomy), which makes it less suited to large CNV.
Rationale for reconstitution of retinal pigment epithelium
The spectacular functional restoration achieved in some patients with exudative age-related macular degeneration after macular translocation by rotation proved the principle of reopposing a reversibly compromised fovea to a fresh undersurface of functioning RPE cells.10,28 Other key discoveries in the concept of restoring the underlayer of the macula have included:
• Showing that functioning RPE cells are essential for the preservation of Bruch’s membrane and the underlying choriocapillaris in rabbits29
• Demonstrating that human RPE cells secrete VEGF on their basal side and that the adjoining underlying choriocapillaris has VEGF-receptors30
• Proving the principle that healthy RPE cells can postpone photoreceptor death after subretinal transplantation in a rat model of RPE dysfunction.31
Consequently, many different surgical approaches to recreate a functioning RPE underlayer of the macula have been tried. These approaches may broadly be defined by several complementary techniques: autografts versus allografts; cells in suspension versus cell sheets or patches; RPE versus iris pigment epithelium (IPE) cells (Table 121.1).
Table 121.1 Reconstitution of RPE: cell suspension or cell sheets; allograft or autograft; RPE or IPE
| Author, year of publication (number of patients) | |
|---|---|
| Autograft IPE suspension | Thumann et al. 2000 (12),32 Lappas et al. 2000 (12)33 |
| Autograft IPE patch graft | Navea Tejerina 199834 |
| Autograft RPE suspension | Binder 2002 (14),35 2004 (53),36 van Meurs et al. 2004 (8),37 Falkner-Radler et al. 2011 (7)38 |
| Autograft RPE patch graft | Peyman et al. 1991 (1),19 Stanga et al. 2002 (9),20 van Meurs and Van Den Biesen 2003 (6),21 Bindewald et al. 2004,39 MacLaren 2005 (9),40 Joussen et al. 2006 (45),22 Maaijwee et al. 2007 (84),25 Treumer et al. 2007 (10),24 MacLaren et al. 2007 (12),23 Joussen et al. 2007 (12)‡,41 Chen et al. 2009 (12),27 Chen et al. 2009 (24),42 Gibran et al. 2009 (4)*,43 Ma et al. 2009 (21)*,†,40 Caramoy et al. 2010 (10)‡,44 Cereda 2010 (13)*,45 Falkner-Radler et al. 2011 (7),38 van Zeeburg et al. 2012 (134)26 |
| Allograft RPE suspension | Valtink et al. 1999,46 Valtink et al. 199947 |
| Allograft RPE patch graft | Peyman 1991 (1),19 Algvere 1994 (5),48 1997 (5),49 Del Priore 2001 (12)50 |
*Flapover technique; †Partial thickness graft; ‡Geographic atrophy.
RPE, retinal pigment epithelium; IPE, iris pigment epithelium.
Transplantation of a full-thickness patch from the midperiphery
With the current lack of demonstrable presence or function of RPE or IPE suspension transplants35,37,38 the authors decided to pursue the use of a sheet of autologous RPE on its own substratum. Peyman reported a case history on the use of a full-thickness flap with a pedicle. Follow-up was 6 months and stabilization of vision at 20/400 was reported.19 Aylward, in nine patients, used a full-thickness patch, cut out from a location adjacent to the removed subfoveal membrane. In four patients, some function on microperimetry could be shown over the patch; but the preoperative vision was too low to assess vision in detail using the techniques available at the time.20 Fibrosis of the patch, however, developed in the second year of follow-up in most patients and previously documented function on the graft had disappeared.51 In Aylward’s patients the grafted paramacular choriocapillary appeared sclerotic and damaged by the surgery and we speculated that it was therefore less likely to be successfully revascularized. Van Meurs and Van Den Biesen21 sought to improve on Aylward’s technique by harvesting a relatively healthy midperipheral full-thickness RPE and choroid patch with the advantage of having easy access to cut out the patch and direct control of bleeding from the donor site. Surgeons in Cologne, Kiel, Liverpool, Vienna, Verona, Beijing, London, and Rotterdam have published further reports, some with a different surgical approach with a flapover technique to expose the subretinal space. Surgeons have also reported the use of a split-thickness graft, removing most of the choroid with an excimer laser39 or with a spatula,40 or using a spontaneously occurring free RPE sheet in patients with a pigment epithelium detachment.52 The last approach is particularly interesting in view of the recent development of free RPE sheets, without choriocapillary, grown from embryonic stem cells (see below).
Surgery
Keyhole approach
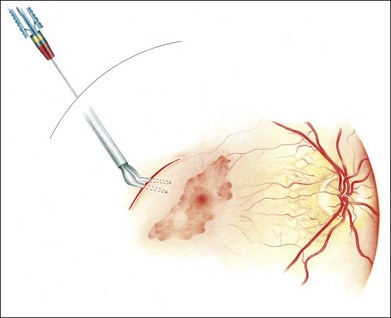
After the induction of a posterior vitreous detachment, a complete vitrectomy is performed (see video under http://www.eyemoviepedia.com/videos/3971697493/153). The macular retina can be separated from the RPE and/or blood and/or CNV membrane by injecting a balanced salt solution into the subretinal space through a 28-gauge subretinal cannula. A paramacular temporal retinotomy is made, through which the CNV membrane and/or subretinal hemorrhage is removed from the subretinal space with Thomas subretinal forceps. Remaining hemorrhage is flushed out by irrigation with a 135° bent 32-gauge needle connected by tubing to a syringe filled with BSS and operated by an assistant.
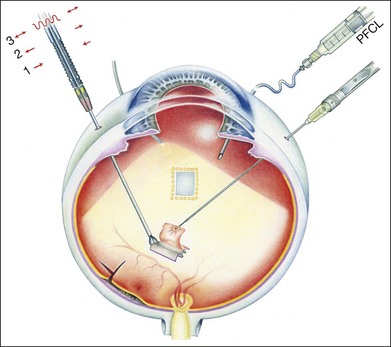
Initially, a small retinotomy was made in the raphe; in later surgeries a radial cut was made along the raphe, from the retinotomy further peripheral. However, a vertical retinotomy temporal to the macula (Fig. 121.1) was found to work best for two reasons: (1) less tendency to enlarge in the direction of the fovea and (2) easier for later introduction of the flat graft (Francois Devin, Marseille, pers. comm.). After circular heavy diathermy or laser in the midperiphery at the 6 or 12 o’clock position, vitreous scissors are used to cut a full-thickness graft of retina, RPE, Bruch’s membrane, and choroid of approximately 2.5–3 × 2.5–4 mm. Initially, the graft was loaded onto an aspiration-reflux spatula. Subsequently the technique was changed so that the graft could be grasped from the choroidal side by fine forceps. The retina is removed from the graft just before it is repositioned under the macula through the existing paramacular retinotomy (Fig 121.2). Perfluorocarbon liquid is injected over the macula to hold the graft in position during retraction of the instrument. A vibration device attached to the forceps facilitates the release.53 The midperipheral donor site is then surrounded with laser photocoagulation, followed by silicone oil tamponade.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree