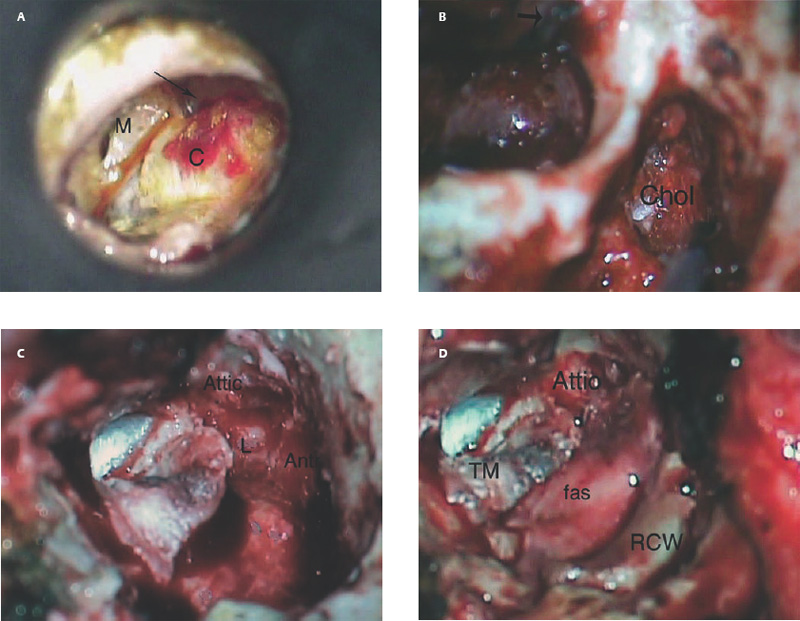
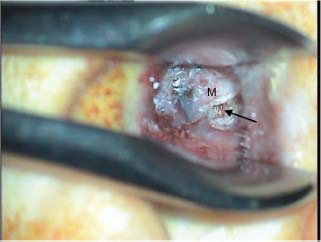
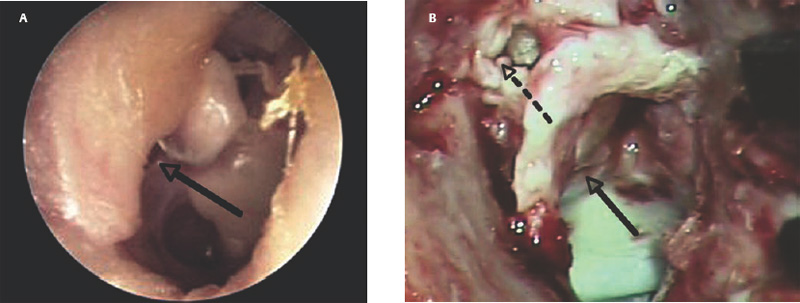
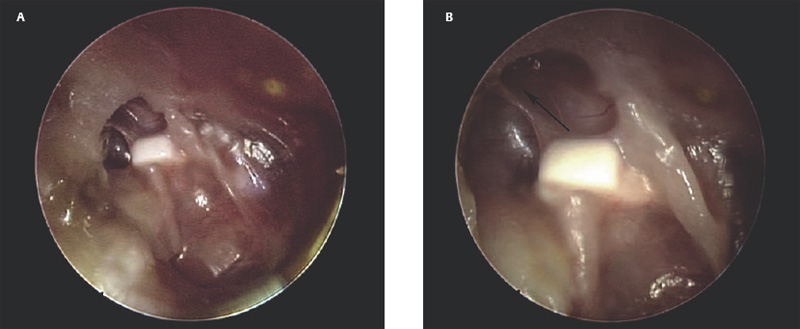
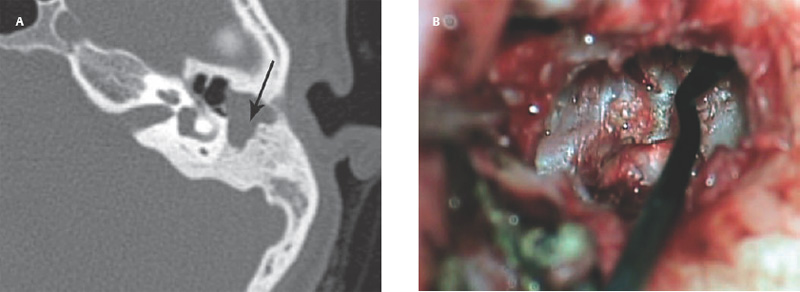
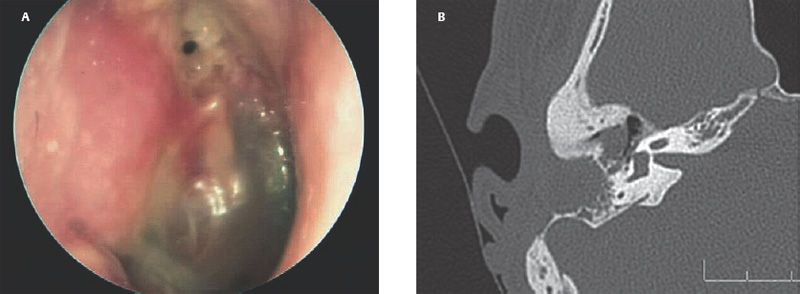
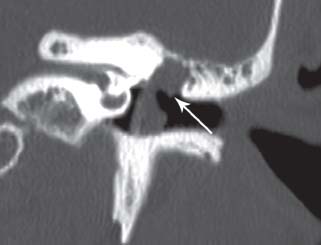
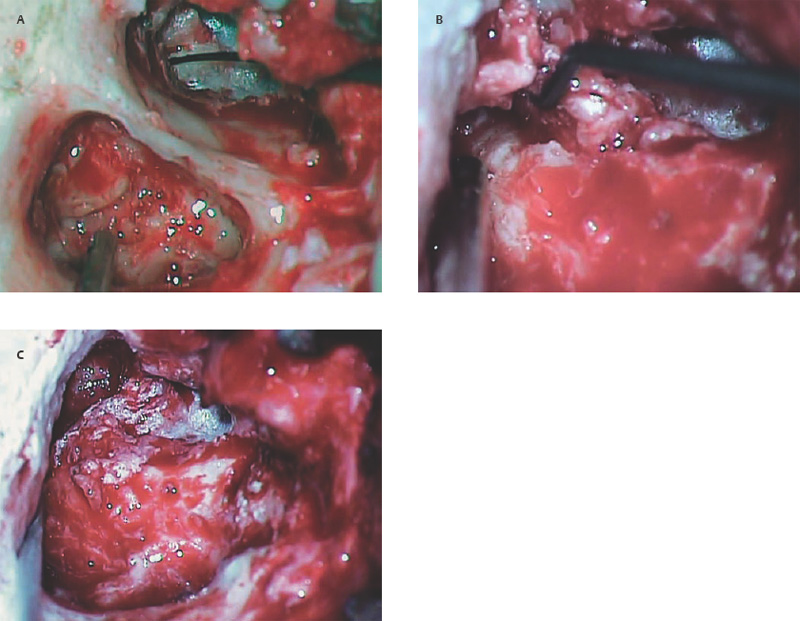
10 Recidivism The possibility of disease recurrence makes the surgical manage ment of cholesteatoma challenging. Recurrent cholesteatoma is defined as cholesteatoma that re-forms after complete surgical excision. Residual cholesteatoma is disease left behind by the surgeon. Recidivism refers to the combination of recurrent and residual disease. The rate of recurrent disease after canal wall up surgery is high, ∼20%, and may be even higher in children.1,2 Recurrent disease may form after canal wall up mastoidectomy and occasionally after canal wall down procedures. In contrast to residual cholesteatoma, recurrent disease occurs when a retraction pocket re-forms from the tympanic membrane, extending upward to the attic or backward toward the sinus tympani. Failure to reconstruct the scutum after intact canal wall mastoidectomy can provide a space for recurrence to form, but disease may recur even when the scutum is rebuilt with cartilage at the initial operation.3 This may begin by invagination of tympanic membrane epithelium into the middle ear space, which can then extend back to the attic and later the mastoid. In other cases, the recurrence may erode the scutum or the posterior bony canal wall and extend superiorly. Recognition of recurrent disease is not always easy. In cases in which the scutum was originally reconstructed with cartilage, the recurrent cholesteatoma may begin in the space between the cartilage graft and the bony scutum, and the opening of the sac may not be apparent. Examination under the microscope is the single best method of detecting recurrent cholesteatoma. A retraction pocket can be gently probed with a blunt hook to determine its depth and whether it contains squamous material. The case shown in Fig. 10.1A is a patient with a canal wall up operation for attic cholesteatoma with cartilage graft reconstruction of the scutum, who complained only of fullness, without hearing loss or discharge. Gentle probing revealed the scutum defect to be part of a deep sac. The operative findings are shown in the Fig. 10.1B, a recurrent cholesteatoma extending from attic to antrum. This was managed by canal wall down mastoidectomy with bony obliteration of the mastoid cavity (Fig. 10.1C,D). She has remained free of recurrence for 4 years. Recurrent cholesteatoma can also form in the middle ear. Retraction pockets that redevelop in the pars tensa typically grow either upward, toward the attic, or medially and backward, toward the sinus tympani (Fig. 10.2). The endoscope can be a useful tool for detecting a recurrence in the office. By allowing illumination deep in the cavity, and by putting the viewer’s eye close to the area of interest, the endoscope may allow the physician to look into crevices and around bony obstructions and see more than can be appreciated with the microscope. The case shown in Fig. 10.3 is an endoscopic view of the right ear of a young girl who had undergone a canal wall up mastoidectomy with staged middle ear reconstruction by another surgeon. She complained of hearing decline after initial hearing gain. On otomicroscopic exam, the meatus was constricted and there was middle ear collapse, but the attic could not be adequately assessed. With endoscopy, squamous material could be seen in the attic, under the ledge of bone, signifying regrowth of cholesteatoma (Fig. 10.3A). At surgery, there was a very large recurrent cholesteatoma filling the attic, and a canal wall down procedure was performed (Fig. 10.3B). The presence of recurring discharge and polyp, especially in the absence of a tympanic membrane perforation, should suggest the presence of recurrence even when a retraction pocket and/or squamous debris are not evident. Declining hearing in ears with good initial postoperative hearing is also suggestive of recurrence. The case illustrated in Fig. 10.4 is a 6-year-old child who underwent canal wall up mastoidectomy with total ossicular replacement prosthesis (TORP) reconstruction of the middle ear, with an excellent hearing result. After 9 months the hearing declined. Microscopic evaluation revealed pars tensa collapse (Fig. 10.4A) but endoscopic exam showed a large retraction pocket extending to the attic (Fig. 10.4B). A canal wall down mastoidectomy was performed. Fig. 10.1 (A) The patient had undergone left canal wall up mastoidectomy with cartilage graft (C) reconstruction of the scutum and had declining hearing and ear fullness. The patient had a stable attic retraction pocket (arrow) without drainage or squamous debris. (M = malleus) (B) At surgery, a moderately large cholesteatoma (Chol) is filling the posterior epitympanum and extending to the antrum. (C) Canal wall down mastoidectomy, with disease completely excised. The canal wall was removed en bloc. L, lateral semicircular canal; Antr, antrum. (D) The middle ear was reconstructed with a type 3 tympanoplasty (tympanic membrane on stapes head). The posterior canal bone was replaced to create a reconstructed canal wall (RCW). The attic was left open to prevent further recurrence, and fascia (fas) was used to reline the base of the cavity. Computed tomographic (CT) scan can be helpful, particularly when the original procedure was done by another surgeon. In the case of prior intact wall mastoidectomy, the scan demonstrates the size of the mastoid and the extent of bone removal. It also might show the presence of neocortical bone formation, and anatomical areas that might have been left unexplored during the original procedure. CT also demonstrates the presence or absence of ossicles. CT is useful in predicting a complication, such as a lateral semicircular canal fistula or dehiscent tegmen, and may prevent problems during revision surgery. In the case of prior canal wall down mastoidectomy, CT shows the remaining bony anatomy, and possible shortcomings of prior surgery, such as a high facial ridge and retained mastoid. For example, Fig. 10.5 shows a CT image of a patient with a recurrent cholesteatoma that had grown through a tight attic defect in a canal wall down mastoid cavity. CT, however, might not always prove the diagnosis of recurrent cholesteatoma. Cholesteatoma appears as a soft tissue density on CT scan but may be indistinguishable from inflamed mucosa, granulation tissue, or retained fluid. Residual disease may appear as a spherical density in a well pneumatized mastoid cavity (Fig. 10.6). In intact canal wall mastoidectomy, fibrous tissue often fills the mastoid cavity and also appears as a soft tissue density. This perfectly benign finding may be differentiated from recurrent cholesteatoma or other forms of inflammatory soft tissue by its concave margins, although this is not routinely true (Fig. 10.7). The use of magnetic resonance imaging (MRI) in conjunction with CT has been advocated by some as the means of establishing or disproving recurrence.4 Diffusion-weighted MRI in particular appears to have a high positive and negative predictive value.5 However, MRI is not routinely utilized in the clinical setting because scanning is expensive and subject to motion artifact, and surgery will probably be recommended in highly suspicious cases regardless of the radiologic findings. The patient is prepped and draped in the same manner as for primary surgery. Lidocaine (1%) with epinephrine 1:100,000 is injected into the canal and postauricular skin for hemostasis. First the drum is examined through the meatus, debriding any pus and polyp from the ear canal. Usually the diagnosis will be evident at this point if it could not be confirmed during office microscopy. Gentle probing may reveal bony defect in the scutum or posterior bony canal (Fig. 10.8A), and pressure on the soft tissue may express squamous debris, thereby confirming the diagnosis. Canal incisions should be made just lateral to the bony defect so as to maintain as much viable canal skin as possible for later reconstruction. A postauricular incision is created, and the mastoid periosteum is incised posterior and superior to the bony mastoidectomy defect if possible. The fibrous tissue in the mastoid can then either be excavated directly from the depth of the cavity or incised at the plane of the mastoid cortex and left in place until after the canal skin is separated from the bone (the latter method allows for more control when dissecting the tissue out of the mastoid). The superior periosteal incision should proceed along the temporal line and far enough anterior so that the anterior epitympanum can be accessed. Bony exposure is obtained starting from the mastoid and working forward. In most revision mastoidectomy cases, if the disease fills the mastoid extensively, the posterior bony canal wall will almost certainly have to be removed. In cases in which the recurrent disease is localized to the attic, however, it may be possible to maintain the posterior canal wall. As in primary cases, it pays to begin the bony dissection posteriorly, preserving the canal wall at least initially. First the neocortical bony edges are removed and the tegmen mastoideum is identified. Posteriorly, the sigmoid sinus is identified and the retrosigmoid bone is removed. The tegmen is followed anteriorly until the attic is opened. The superior portion of the posterior bony canal wall can be removed at this point. Cortical bone should be taken down as far as possible to the zygomatic root, and the tegmen is followed medially until the remaining portion of the scutum is identified. If the attic is filled with cholesteatoma and the scutum is partially eroded, the remaining scutum can be removed with a drill. If there is concern about injuring an intact ossicular chain, however, the scutum can be thinned down and removed with a curette. The facial recess can be opened at this point by following the fossa incudus downward with a 2 mm diamond burr using constant suction-irrigation. Once the facial recess is opened widely, the remainder of the posterior bony canal wall can be taken down with a rongeur. The contents of the sac are debulked, and the matrix is dissected beginning posteriorly and working forward toward the lateral semicircular canal and stapes. The lateral semicircular canal can usually be identified before removing the matrix (Fig. 10.8B). If fistula is suspected, the matrix should be left over the lateral canal until later in the case. If the field is infected with purulent material, it may be preferable not to remove the matrix over the lateral canal. The incus and malleus head, if present, should be removed. The matrix is peeled away from the medial wall of the attic, working toward the tympanic segment of the facial nerve. It is wise to assume that the bony fallopian canal is dehiscent in every case. The matrix should be peeled away from the facial nerve working from above downward, avoiding sharp instruments on the nerve. The backside of a curved Rosen needle is helpful, using the tip to elevate the free edge of matrix, and sweeping the blunt surface of the instrument gently along the nerve while peeling the matrix away. The stapes should be palpated through the matrix. If the disease is adherent to the stapes, the matrix should be cut just lateral to the capitulum with a Bellucci microalligator scissors and the remainder of the disease dissected later, after better exposure is obtained. Disease in the middle ear is dealt with as in primary surgery. The drum should be elevated first. If the middle ear space is free of disease, there may be no need to interrupt any previously made ossicular reconstruction. If on the other hand there is invagination of epithelium into the middle ear, the stapes capitulum should be separated from its attachment and the middle ear explored and all disease removed widely as in primary surgery. If the middle ear disease is extensive, it may be safer to avoid reconstruction and simply create a radical mastoidectomy cavity. Any remaining disease must be cleared from the mastoid cavity. Once the matrix is elevated, all the remaining bony crypts need to be carefully inspected to be sure that there is no plunging disease. All remaining air cells should be exenterated with a polishing burr. Fig. 10.8 (A) Recurrent cholesteatoma arising from the tympanic membrane. Gentle probing reveals extension of the disease to the attic. (B) After the canal wall has been taken down, the posterior limit of the sac is identified, and the matrix is elevated away from the lateral semicircular canal. (C) Canal wall down cavity after complete removal of recurrent disease. The bony edges have been beveled, and the facial ridge lowered to the level of the mastoid segment of the facial nerve. The cavity can be made functionally smaller by using a Palva (conchomeatal soft tissue) flap. Cavity management is addressed after all cholesteatoma is removed. The goal is to create a compact cavity. This involves beveling all the bony edges widely, including the retrosigmoid bone (removing more bone paradoxically leads to a smaller cavity, as the soft tissues collapse medially to fill the space). The mastoid tip should be amputated down to the level of the digastric muscle. The bone anterior to the mastoid tip should be taken down so that the most dependent portion of the mastoid cavity is confluent with the inferior canal wall. In a sclerotic mastoid, the cavity can be sculpted to a round shape. In a well-pneumatized mastoid, as much cellular bone as possible should be removed. The facial ridge should be removed completely so that the facial nerve is skeletonized to a thin layer of bone (Fig. 10.8C), and the bone anterior to the facial nerve should be removed as well. This results in a tympanic cavity that is shallower than normal. A meatoplasty should be performed as previously described. Obliteration of the cavity with fibrous tissue flap (Palva flap) is optional in small cavities but should be performed in larger cavities, except when there is a real concern of burying residual disease. Ossicular reconstruction can be undertaken in revision cases, often with good results.6 Options include the following: • Leaving the original reconstruction intact (feasible in cases in which recurrence begins in the attic and does not involve the middle ear) • Repairing the tympanic membrane and conducting a simultaneous ossicular repair • Repairing the tympanic membrane and postponing the ossicular repair • Performing a type 4 reconstruction (cavum minor) • Excising the drum completely and creating a radical mastoidectomy cavity
♦ Recurrent Cholesteatoma
Revision Surgery for Recurrent Cholesteatoma in Canal Wall Up Cavity
Middle Ear Reconstruction
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


