CHAPTER 112 Radiation Therapy for Cancer of the Larynx and Hypopharynx
Early-Stage Glottic Cancer (T1-T2 Lesions)
Advanced-Stage Glottic Cancer (T3-T4 Lesions, N+)
Early-Stage Hypopharyngeal Cancer
Advanced-Stage Hypopharyngeal Cancer
Overview
Of the millions of species on planet Earth, none has the ability to communicate as effectively as Homo sapiens. Of course, central to this ability to communicate is the larynx. Besides directly assisting the human voice, the larynx is indirectly involved in other critical functions such as swallowing. Hence it is not surprising that cancer of the larynx is a devastating diagnosis for a patient not only because of its potentially life-threatening implications but also due to the numerous implications on the individual’s quality of life. The American Cancer Society estimated that 12,250 cases of laryngeal cancer were diagnosed in 2008 and that 3670 deaths resulted from this disease.1 Cancer of the hypopharynx, though rare with 2500 new cases estimated every year, has similar implications on the individual patient’s well-being and quality of life.
In order to achieve succinctness and clarity, this chapter is organized as follows:
Anatomic Considerations
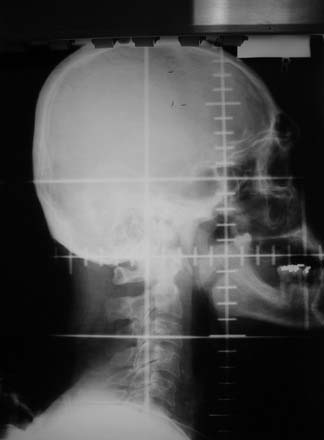
The larynx is intimately related to the hypopharynx; it is situated anteriorly to the hypopharynx, at approximately the same cervical levels. Anatomically, the larynx extends from the tip of the epiglottis at the level of the lower border of the C3 vertebra to the distal extent of the cricoid cartilage at the level of the C6 vertebra. The larynx is subdivided into three anatomic regions: the supraglottis, the glottis, and subglottis regions. The supraglottic region includes (1) the epiglottis, (2) the aryepiglottic folds, (3) the arytenoids, (4) the false vocal cords, and (5) the ventricle. The glottis consists of the true vocal cords and the anterior and posterior commissures. Importantly, the anterior commissure is usually within 1 cm of the skin surface, an important consideration for radiation treatment planning. The lower boundary of the glottis extends 5 mm below the free margin of the vocal cords, or to the horizontal plane 1 cm below the apex of the ventricle. The vocal cords attach to the thyroid cartilage at what is visualized to be the “figure-of-eight” on a lateral plain film. The subglottis extends from the lower boundary of the glottis to the inferior margin of the cricoid cartilage. These anatomic boundaries are readily visualized on plain lateral x-ray films (Fig. 112-1).
Lymphatic Drainage
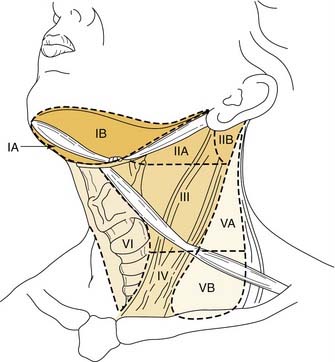
Knowledge of the lymph node levels in the neck in relation to bony landmarks, as well as surgical landmarks, is important for the radiation oncologist when planning radiation portals. Level I nodes are above the hyoid bone, below the mylohyoid muscle and anterior to the back portion of the submandibular gland. This corresponds to the submental and submandibular triangles. Level II nodes are located from the skull base to the lower body of the hyoid bone inferiorly, posterior to the back of the submandibular gland, and anterior to the posterior extent of the sternocleidomastoid muscle. The jugular chain lymph nodes, which run from the mandible down to the carotid bifurcation, are included in this level. Level III nodes extend from the lower body of the hyoid to the inferior margin of the cricoid, also anterior to the posterior margin of the sternocleidomastoid muscle, corresponding to the region from the carotid bifurcation to the omohyoid muscle. Level IV lymph nodes are bordered superiorly by the lower border of the cricoid cartilage and inferiorly by the clavicles and are anterior to the posterior margin of the sternocleidomastoid muscle, which corresponds to the region from the omohyoid muscle to the clavicle. Level V lymph nodes run from the skull base to the level of the clavicle in the region posterior to the sternocleidomastoid muscle; this area is the posterior triangle bounded by the SCM anteriorly, the trapezius posteriorly, and the omohyoid inferiorly. Level VI lymph nodes are between the lower body of the hyoid bone and the suprasternal notch, otherwise known as the anterior compartment between the carotid arteries (Fig. 112-2).
Clinical Presentation and Evaluation
Staging
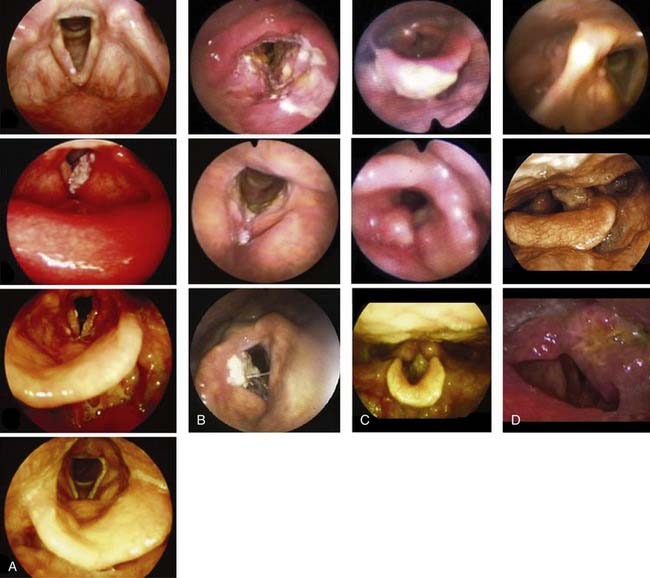
Laryngeal and hypopharyngeal cancers are staged according to the American Joint Commission for Cancer Tumor Node Metastasis system. The sixth and most recent version of the staging system published in 2002 can be found in other chapters. In contrast to staging in other sites, the staging for tumors in the larynx is based on involvement of anatomic structures and not on the size of the lesion. Staging in the hypopharynx is based on size and subsite involvement in early stages. Note that in both the larynx and hypopharynx, fixation of either vocal cord automatically stages the tumor as a T3, regardless of the tumor bulk. T4 disease is the same in all glottic subsites and hypopharynx, reflecting invasion outside of thyroid cartilage or beyond. Nodal staging is the same as that in other head and neck malignancies (with the exception of the nasopharynx). The ability to visualize the stage of the disease from an anatomic framework, rather than memorizing lines of description in text, is useful. Direct laryngoscopy images of early T-stage laryngeal and hypopharyngeal cancers are shown in Figures 112-3A-D.
Treatment Options, Radiotherapy Techniques, Dose-Fractionation Schemes, Outcome, Prognostic Factors, and Complications
Laryngeal Cancers
Early-Stage Glottic Cancer (T1 or T2)
Radiation Techniques and Dose-Fractionation Schemes
Multiple-dose fractionation schemes have been used to treat patients with T1 and T2 glottic carcinoma, with total dose equaling 66 to 70 Gy at 2.0 Gy/fraction (fx) per day, 63 Gy at 2.1 Gy/fx per day, or 60.75 Gy to 65.25 Gy at 2.25 Gy/fx per day, 5 days per week.2–4 At the Department of Radiation Oncology, University of Southern California (USC) Keck School of Medicine (KSOM), patients with Tcis, T1 (including verrucous carcinoma), and small T2 lesions are treated to 66.0 Gy at 2.0 Gy/fx. Patients with T2 lesions are treated to a total dose of 70 Gy at 2.0 Gy/fx. These dose-fractionation schemes are used at USC because of the ease of tolerability (i.e., rarity of treatment related interruptions) in our experience and excellent local control rates. Doses less than 2.0 Gy/fx should not be used to treat early-stage glottic tumors because of the extended interval of RT, which enhances tumor repopulation rates, leading to inferior local control rates.5,6 Although altered-dose fractionation schemes (i.e., hyperfractionated or accelerated radiotherapy) can be used to treat patients with unfavorable early-stage glottic cancers (e.g., bulky T2 lesion), the treating radiation oncologist should only embark on this path if he or she has experience in using these alternative-dose fractionation schemes.
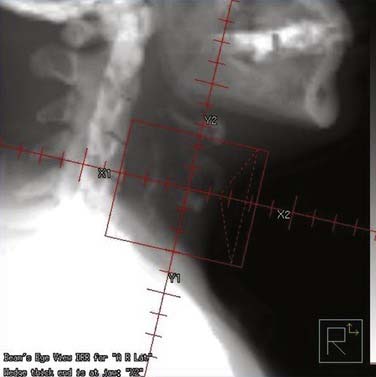
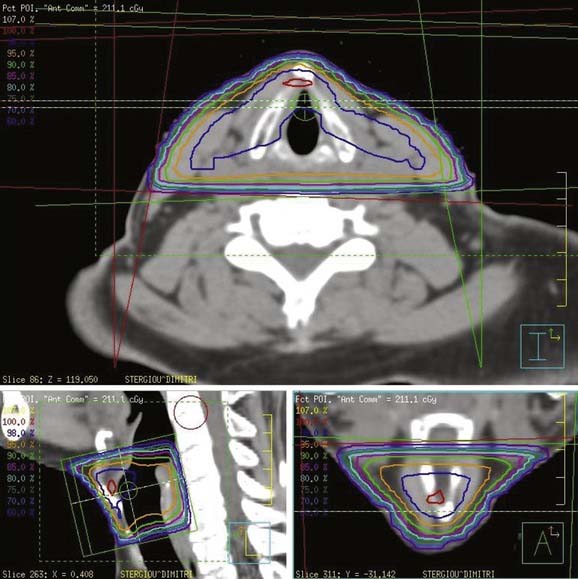
At USC, a three-dimensional conformal RT (3-D CRT) approach is used to treat patients with early-stage laryngeal carcinoma. A planning CT scan is done with the patients in a supine treatment position, with the head hyperextended and immobilized with an individually made face mask. For T1 and very early T2 lesions, two small opposing lateral fields, usually measuring 5 × 5 cm2 to 6 × 6 cm2, are used (Fig. 112-4). The fields typically extend from the upper thyroid notch superiorly to the lower border of the C6 vertebral body inferiorly. The anterior border of the field should flash at least 1 cm in front of the skin surface at the level of the vocal cords (approximately C4). The posterior border of the field should include the anterior portion of the vertebral bodies. During fluoroscopic simulation (if possible) of the fields, the patients should be asked to swallow to monitor the superior extension of the glottis to ensure that it is still within the radiotherapy field because sometimes a patient may actually swallow during the radiation treatment. During treatment planning, radiation dose homogeneity, especially in the anterior commissure region, using wedges should be achieved to prevent overdosing or underdosing (Fig. 112-5). The lymph nodes are not treated in patients with Tcis and T1 disease because of the virtually nonexistent incidence of lymphatic nodal metastases. The incidence of lymphatic nodal involvement varies from 5% to 15% with T2 disease. Generally, for T2 lesions, we recommend treatment of at least the first-echelon lymph nodes (subdigastric and midjugular, or lower levels II and III), especially in a patient with bulky disease.
Outcome and Prognostic Factors
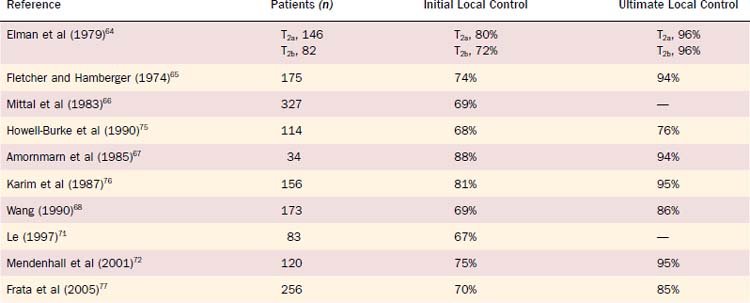
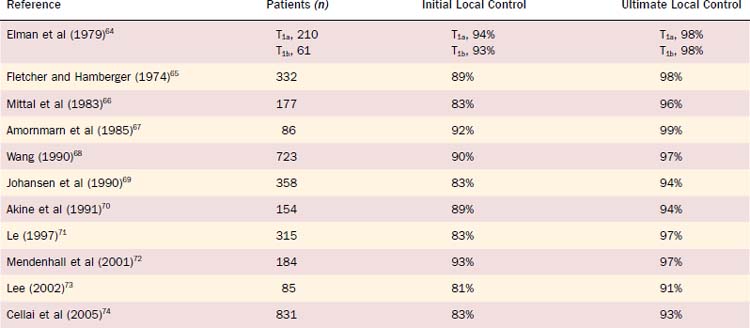
For Tcis and T1 lesions, local tumor control rates vary between approximately 85% and 95% at 5 years (Table 112-1). With surgical salvage, ultimate local control rates between 95% and 99% at 5 years can be achieved. For T1 verrucous carcinoma, similar high local control rates have been reported with definitive RT. For T2 glottic lesions, initial local control rates vary between 70% and 90% at 5 years with RT alone (Table 112-2). Following surgical salvage of radiotherapy failures, ultimate local control rates between 80% and 96% at 5 years can be achieved for T2 lesions.
Table 112-1 Local Control of T1 Carcinoma of the Glottis Treated with Definitive Radiotherapy and Surgical Salvage
Anterior commissure involvement has been shown in older studies to be associated with lower tumor local control rates. However, in the era of 3-D CRT with computer treatment planning, which can detect and thus limit underdosing of the anterior commissure region, this prognostic factor is no longer significant. Fraction sizes of less than 2.0 Gy result in inferior local control rates and therefore should not be used. Studies have also shown that extending treatment times beyond approximately 42 days results in inferior outcomes secondary to accelerated repopulation by tumor cells.8 In a study of 91 patients with T1N0 squamous cell carcinoma of the glottis treated with definitive radiotherapy, both overall treatment time (i.e., ≤42 days vs. > 42 days) and dose per fraction (i.e., <200 cGy vs. ≥200 cGy) predicted for local control. Typically, a loss of approximately 1.4% (0.4% to 2.5%) in local control occurs for every day of a treatment break or overall extension of treatment time. Multiple studies have shown an adverse effect of low pretreatment and/or post-treatment hemoglobin levels (Hgb) on outcome in early- and advanced-stage laryngeal cancer.9 For example, in a study from Fox Chase Cancer Center that analyzed 109 patients with T1 or T2 glottic cancers treated with definitive RT, pretreatment Hgb levels less than or equal to 13 g/dL adversely affected both 2-year local control rates and overall survival (66% vs. 95% and 46% vs. 88%, respectively) on multivariate analysis.
Complications from Radiation Therapy
Because the treatment fields for early-stage glottic cancer are small, patients typically experience few side effects from RT. Common side effects include skin erythema and/or hyperpigmentation, desquamation, hoarseness, sore throat, and resultant dysphagia. These effects are self-limiting, manifesting approximately 2 to 4 weeks into therapy and usually resolving within 6 to 8 weeks after the completion of treatment. Hypothyroidism is also a possible consequence of radiation, and therefore patients should have thyroid function testing before treatment and every 6 to 12 months. Hypothyroidism rates as high as 30% to 40% have been reported in the literature after radiation.13 Hormonal replacement can be given to replete thyroid hormone levels. More serious though rarer late sequelae from RT include laryngeal arytenoid edema and laryngeal arytenoid necrosis. In patients who have been irradiated for carcinoma of the glottis, the incidence of mild to moderate laryngeal edema persisting for more than 3 months after RT is about 15.4% to 25%.14 The incidence of severe laryngeal edema is about 1.5% to 4.6%,15–18 and this increases with greater total dose, field size, dose per fraction, and T stage of the lesion.16,18,19 Initially these patients should be managed conservatively with voice rest, abstinence from alcohol and cigarettes, and careful, close follow-up examinations. Antibiotics and low-dose steroids (i.e., 2 mg po bid) may be used when there is suspicion of infection or when the edema is severe enough to significantly compromise the airway. However, if the edema is progressive and unresponsive to conservative measures and recurrent disease is strongly suspected, biopsies should be carried out to rule out malignancy as the underlying cause. Salvage surgery is performed if biopsies are positive. Late laryngeal arytenoid necrosis after RT is rare, with a reported incidence of about 0.5% to 1.8% for glottic cancer.15,20,21
Advanced-Stage Glottic Cancer (T3 or T4, Stage III or IV)
Overall Considerations: Balancing Cure, Organ Preservation, and Quality of Life
Hence we recommend a primary surgical approach in the following settings:
Primary Surgical Approach: Adjuvant Therapy
At USC, we prefer to use postoperative versus preoperative radiotherapy in the setting of a primary surgical approach based on results of the Radiation Therapy Oncology Group (RTOG) 7303 and the “inherent” advantages of adjuvant treatment.22 RTOG 7303 was a randomized trial comparing preoperative (50 Gy) versus postoperative (60 Gy) RT in advanced-stage, operable supraglottic larynx and hypopharynx cancers. The 10-year locoregional control was significantly improved in the postoperative versus the preoperative group (70% vs. 58%, respectively). Additionally, the rate of complications was much higher in the patients undergoing preoperative RT (9%) versus the postoperative group (5%). On the basis of the results of this RTOG study and the inherent advantage of having pathologic information after surgery, postoperative RT is preferred over preoperative RT. At USC, the role of preoperative RT is limited to shrinking locally advanced disease considered to be unresectable.
Although locoregional tumor control rates with total laryngectomy followed by postoperative RT are relatively high, some patients with high-risk disease features still do poorly with adjuvant radiotherapy alone. Hence several randomized trials have evaluated the role of adding concurrent chemotherapy to adjuvant RT following surgery for locally advanced head and neck cancer including primary laryngeal tumors. These studies include RTOG 9501, European Organization for the Research and Treatment of Cancer (EORTC) 22931, and the German ARO 96-3 study (Table 112-3).23–26 All three of these randomized trials showed that adjuvant concurrent chemoradiation therapy is better than radiotherapy alone for local control and disease-free survival in patients with high-risk disease following surgery. In the RTOG 9501 trial, 459 patients with high-risk locally advanced head and neck cancer defined as any T-stage disease with two or more involved lymph nodes, positive extracapsular extension (ECE), and/or positive mucosal margins of resection were randomized to either RT alone (60 to 66 Gy at 2.0 Gy/fx) or the same RT with concurrent cisplatin (100 mg/m2 on days 1, 22, and 43). At 2 years, local control rates (82% vs. 72%, p = 0.01, respectively) and disease-free survival (35% vs. 25%, p = 0.04, respectively) were significantly better among those who underwent adjuvant chemoradiation therapy versus radiotherapy alone, respectively. However, there was no statistical difference in overall survival between the two arms (45% vs. 35%, p = 0.19, respectively). In the EORTC 22931 trial, high-risk disease was defined as any patient with a pT3 or pT4 tumor with any nodal stage, or a pT1/pT2 tumor with N2 or N3 disease, or pT1/pT2 and N0 or N1 disease with involved mucosal margins, positive ECE, perineural invasion, and/or vascular emboli. In the EORTC trial, 334 patients were randomized to RT alone (66 Gy at 2.0 Gy/fx) or the same RT with cisplatin (100 mg/m2
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree