CHAPTER 120 Radiation Therapy and Management of the Cervical Lymph Nodes and Malignant Skull Base Tumors
Radiation Therapy and Management of Cervical Lymph Nodes
Lymphatic System of the Neck
The head and neck region has a rich network of lymphatic vessels draining from the base of the skull through the jugular nodes, the spinal accessory nodes, and the transverse cervical nodes to the venous jugulo-subclavian confluent or the thoracic duct on the left side and the lymphatic duct on the right side.1,2 The whole lymphatic system of the neck is contained in the celluloadipose tissue delineated by aponeurosis enveloping the muscles, vessels, and nerves. The lymphatic drainage is mainly ipsilateral, but structures like the soft palate, tonsils, base of the tongue, posterior pharyngeal wall, and especially the nasopharynx have bilateral drainage. On the other hand, sites such as the true vocal cord, paranasal sinuses, and middle ear have few or no lymphatic vessels at all.
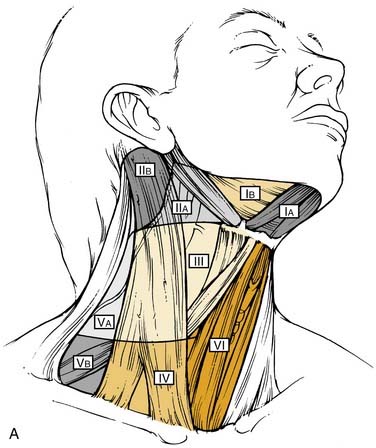
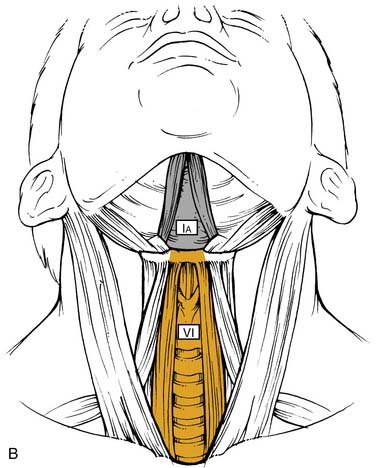
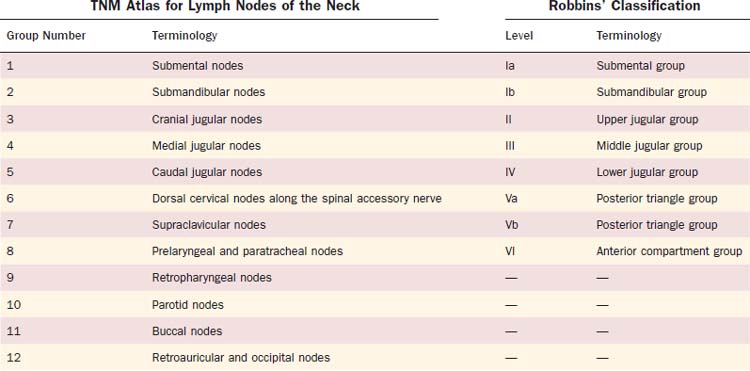
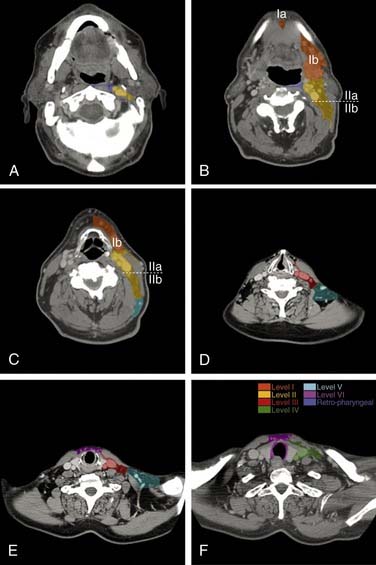
The nomenclature of head and neck lymph nodes has been complicated by various confusing synonyms that are still in use in major textbooks and articles. More recently, several expert bodies have proposed the adoption of systematic classifications aimed at standardizing the terminology. Following the description by Rouvière, the Tumor Node Metastasis atlas proposed a terminology, dividing the head and neck lymph nodes into 12 groups.3 In parallel to this classification, the Committee for Head and Neck Surgery and Oncology of the American Academy for Otolaryngology–Head and Neck Surgery has been working on a classification (the so-called Robbins classification), dividing the neck into six levels including eight node groups (Fig. 120-1).4 This classification is based on the description of a level system, which has been used for a long time by the Head and Neck Service at the Memorial Sloan-Kettering Cancer Center.5 Because one of the objectives of the Robbins classification was to develop a standardized system of terminology for neck dissection procedures, only the lymph node groups routinely removed during neck dissection were considered. The terminology proposed by Robbins was recommended by the Union Internationale Contre Cancer (UICC).6 A comparison between the TNM and the Robbins terminology is shown in Table 120-1. The major advantage of the Robbins classification over the TNM terminology is the definition of the boundaries of the node levels. The delineation of these boundaries is based on anatomic structures such as major blood vessels, muscles, nerves, bones, and cartilage that are easily identifiable by the surgeon during neck dissection procedures. It is beyond the scope of this chapter to go into a detailed description of the node levels. The reader is referred to a recent review article outlining the anatomic basis of the neck node classification.7
Staging of Neck Node Metastasis
The sixth edition (2002) of the American Joint Committee on Cancer (AJCC) staging for neck node metastasis is presented in Table 120-2.8 This classification does not apply to nasopharyngeal carcinoma, thyroid, or skin cancers. In comparison with the fourth (1987) or fifth (1998) edition, the only modification is the possibility to use a descriptor for nodal metastasis located below (L) or above (U) the lower border of the cricoid cartilage. The use of this descriptor will not influence the nodal staging. The classification for nodal staging does apply irrespective of the modality used for the neck assessment (i.e., clinical examination or imaging). However, the routine use of computed tomography (CT) or magnetic resonance imaging (MRI) and, in expert hands, ultrasonography, is recommended, especially to assess nodes not clinically identifiable (e.g., retropharyngeal, intraparotid, or superior mediastinal nodes) or in patients for whom clinical palpation of the neck is less sensitive (e.g., thick or small neck).9 Last, it should be emphasized that the Nx classification only applies when the neck was not assessed or could not be assessed.
Table 120-2 Sixth Edition (2002) of the American Joint Committee on Cancer (AJCC) Staging for Neck Node Metastasis
| Stage | Definition |
|---|---|
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Metastasis in a single ipsilateral node, ≤3 cm in greatest dimension |
| N2a | Metastasis in a single ipsilateral node, >3 cm but ≤6 cm in greatest dimension |
| N2b | Metastasis in multiple ipsilateral nodes, ≤6 cm in greatest dimension |
| N2c | Metastasis in bilateral or contralateral nodes, ≤6 cm in greatest dimension |
| N3 | Metastasis in a lymph node >6 cm in greatest dimension |
Incidence and Distribution of Neck Node Metastasis
Incidence and Distribution of Lymph Node Metastasis from Clinical and Radiologic Assessment
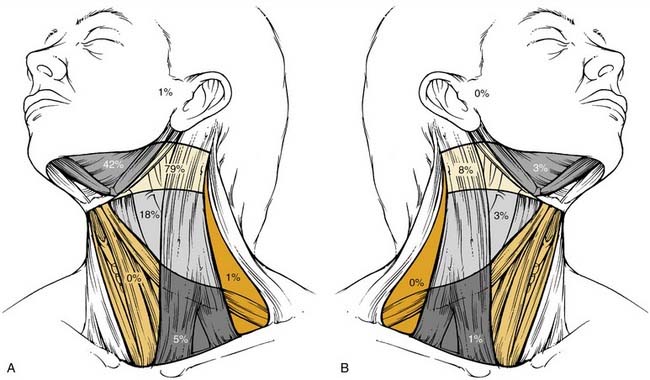
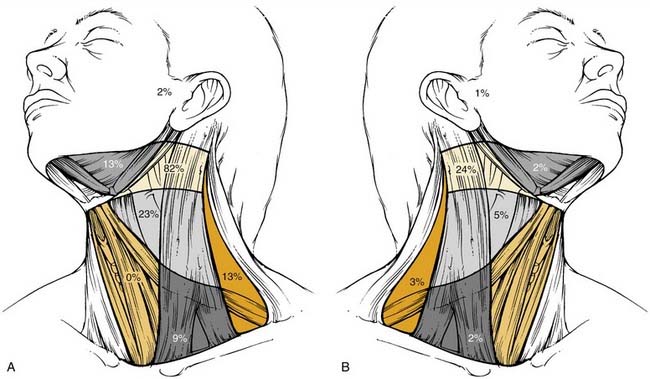
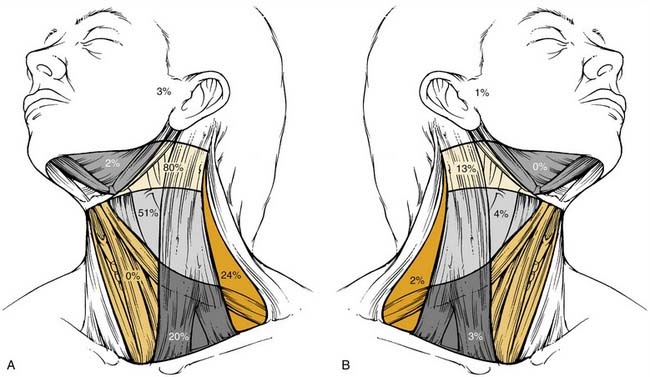
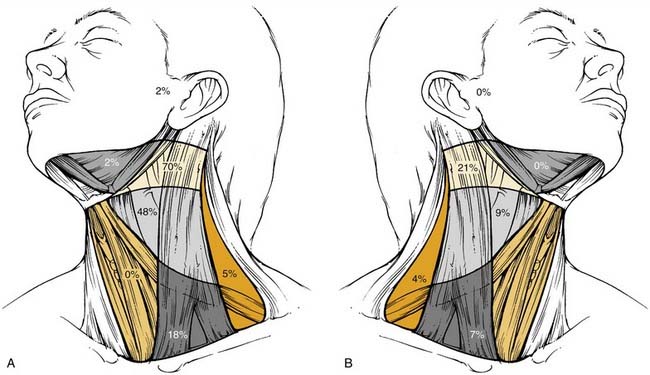
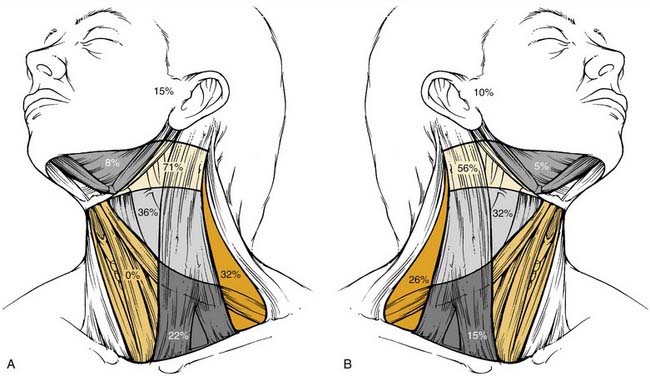
The metastatic spread of head and neck tumors into the cervical lymph nodes is rather consistent and follows predictable pathways, at least in the neck, which has not been violated by previous surgery or radiotherapy. In Figures 120-2 to 120-6, the frequency of metastatic lymph nodes is expressed as a percentage of node-positive patients.10,11
Metastatic lymph node involvement in the neck depends on the size of the primary tumor, increasing with the T stage. In the series reported by Bataini,10 44% of patients with a T1 tumor had clinical lymph node involvement; this increased to 70% for patients with T4 lesions. There are, however, no data suggesting that the relative distribution of involved neck levels varies with the T stage.
Incidence and Distribution of Pathologic Lymph Node Metastasis
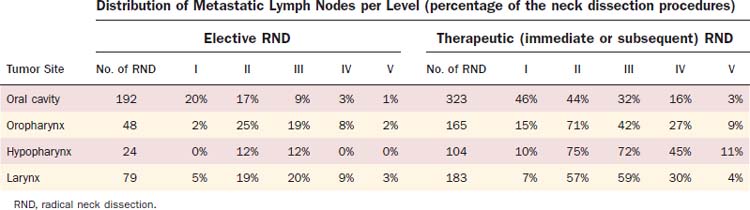
The distribution of pathologic lymph node metastasis in patients with primaries of the oral cavity, oropharynx, hypopharynx, and larynx can be derived from retrospective series in which a systematic radical neck node dissection was proposed as part of the initial treatment procedures.12–15 In essence, retrospective series are biased regarding patient and treatment selection, but these series from the Head and Neck Department at Memorial Sloan-Kettering Cancer Center are the largest and most consistent data ever published on that matter. The results of these retrospective studies are shown in Table 120-3. The data are presented in terms of the number of neck dissections with positive lymph nodes over the total neck dissection procedures and expressed as a percentage. The vast majority of patients (>99% for the N0 neck patients and 95% for the N+ neck patients) only had unilateral treatment, and no distinction between the ipsilateral and contralateral neck was made.
Table 120-3 Incidence of Pathologic Lymph Node Metastasis in Squamous Cell Carcinomas of the Oral Cavity, Oropharynx, Hypopharynx, and Larynx
Incidence and Pattern of Node Distribution in the Contralateral Neck
Few data are available on the pattern of pathologic node distribution in the contralateral neck. Bilateral neck dissection was only performed when the surgeon considered that there was a high risk of contralateral node involvement (e.g., tumors of the oral cavity or the oropharynx reaching or extending beyond the midline, hypopharyngeal and supraglottic tumors). Obviously, in such cases bilateral radical neck dissection was never performed, so an accurate estimate of the pattern of node involvement in levels I to V of the contralateral neck is not possible. Furthermore, in almost every study, data on both sides of the neck were pooled for presentation. Kowalski presented data on 90 patients who underwent bilateral supraomohyoid neck dissection, and in whom the pattern of node distribution in each side of the neck was reported separately.16
The majority of these patients had SCC of the lip or oral cavity. In the ipsilateral neck, pathologic infiltration in levels I, II, and III reached 20%, 15%, and 15%, respectively. In the contralateral neck, corresponding values reached 13%, 11%, and 0%, respectively. These figures are in good agreement with data on clinical node distribution showing that both sides of the neck exhibited a similar pattern of node distribution, but with a lower incidence in the contralateral neck. Foote17 reported the rate of contralateral neck failure in a limited series of 46 clinically N0 patients with base of the tongue tumors treated by some form of glossectomy and ipsilateral neck dissection. None of these patients received postoperative radiotherapy. Ten patients (22%) had contralateral neck recurrence, and the most common sites were in levels II, III, or IV. It appears that in two of these patients, recurrence was also observed at the primary site. The development of delayed contralateral neck metastases was not related to the clinical or pathologic extent of the base of the tongue tumor. O’Sullivan18 reported a retrospective series of 228 patients with tonsillar carcinoma who were treated on the primary tumor and the ipsilateral neck only with radiotherapy. The vast majority of these patients had T1-T2 and N0-N1 disease. Contralateral recurrence in the neck was only observed in eight patients (2%) including five patients with local recurrence as well. No contralateral neck recurrence was observed in the 133 N0 patients. Although not significant due to the small number of events, involvement of midline structure (i.e., soft palate and base of tongue) appeared to be a prognostic factor for contralateral neck recurrence. Similar results were reported in a series of 101 node-negative tonsil carcinoma (mainly T1-T3) treated unilaterally.19 Only two neck recurrences were observed in the contralateral neck.
Recommendations for the Selection of the Target Volumes in the Neck
The data presented in the previous sections indicate that metastatic lymph node involvement of primary SCC of the oral cavity, pharynx, and larynx typically follows a predictive pattern. Both data on clinical and pathological neck node distribution and on neck recurrence after selective dissection procedures support the concept that not all the neck node levels should be treated as part of the initial management strategy of head and neck primaries of squamous cell origin.20,21 One should bear in mind, however, that the data on which such a concept is based have come from retrospective series, and thus may include possible bias (e.g., patient selection, series from pre-imaging area) that could limit its validity.
Tables 120-4 to 120-7 present recommendations for the selection of the target volumes in the neck for pharyngolaryngeal SCCs. These guidelines can be applied irrespective of the treatment modality (i.e., surgery or radiotherapy). The discussion of the choice between these two modalities is beyond the objective of this chapter but should be considered relative to the neck stage, the treatment option for the primary tumor, the performance status of the patient, and the institutional policy agreed on by a multidisciplinary head and neck tumor board.
Table 120-4 Recommendations for the Selection of the Target Volume in the Neck for Oral Cavity Tumors
| Nodal Stage (AJCC 2002) | Selected Levels | |
|---|---|---|
| Ipsilateral Neck | Contralateral Neck | |
| N0-N1 (in levels I, II, or III) | I, II*, III + IV† | I, II*, III + IV† |
| N2a-N2b | I, II, III, IV, V‡ | I, II*, III + IV for anterior tongue tumor |
| N2c | According to N stage on each side of the neck | According to N stage on each side of the neck |
| N3 | I, II, III, IV, V ± adjacent structures according to clinical and radiologic data | I, II*, III + IV for anterior tongue tumor |
AJCC, American Joint Committee on Cancer.
* Level IIb could be omitted for N0 patients.
† For anterior tongue tumor and for any tumor with extension to the oropharynx (e.g., anterior tonsillar pillar, tonsillar fossae, base of tongue).
‡ Level V could be omitted if only levels I through III are involved.
Table 120-5 Recommendations for the Selection of the Target Volume in the Neck for Oropharyngeal Tumors
| Nodal Stage (AJCC 2002) | Selected Levels | |
|---|---|---|
| Ipsilateral Neck | Contralateral Neck | |
| N0-N1 (in levels II, III, or IV) | (Ib)*-II-III-IV + RP for posterior pharyngeal wall tumor | II-III-IV + RP for posterior pharyngeal wall tumor |
| N2a-N2b | Ib, II, III, IV, V + RP | II-III-IV + RP for posterior pharyngeal wall tumor |
| N2c | According to N stage on each side of the neck | According to N stage on each side of the neck |
| N3 | I, II, III, IV, V + RP ± adjacent structures according to clinical and radiologic data | II-III-IV + RP for posterior pharyngeal wall tumor |
AJCC, American Joint Committee on Cancer; RP, retropharyngeal nodes.
* Any tumor with extension to the oral cavity (e.g., retromolar trigone, mobile tongue, inferior gum, oral side of anterior tonsillar pillar).
Table 120-6 Recommendations for the Selection of the Target Volume in the Neck for Hypopharyngeal Tumors
| Nodal Stage (AJCC 2002) | Selected Levels | |
|---|---|---|
| Ipsilateral Neck | Contralateral Neck | |
| N0 | II*-III-IV + RP for posterior pharyngeal wall tumor + VI for apex of piriform sinus or esophageal extension | II*-III-IV + RP for posterior pharyngeal wall tumor + VI for esophageal extension |
| N1-N2a-N2b | Ib, II, III, IV, V + RP + VI for piriform sinus or esophageal extension | II*-III-IV + RP for posterior pharyngeal wall tumor + VI for esophageal extension |
| N2c | According to N stage on each side of the neck | According to N stage on each side of the neck |
| N3 | I, II, III, IV, V + RP + VI piriform sinus or for esophageal extension ± adjacent structures according to clinical and radiologic data | II*-III-IV + RP for posterior pharyngeal wall tumor + VI for esophageal extension |
AJCC, American Joint Committee on Cancer; RP, retropharyngeal nodes.
* Level IIb could be omitted for N0 patients.
Table 120-7 Recommendations for the Selection of the Target Volume in the Neck for Laryngeal Tumors (T1N0 Glottic Carcinoma Excluded)
| Nodal Stage (AJCC 2002) | Selected Levels | |
|---|---|---|
| Ipsilateral Neck | Contralateral Neck | |
| N0-N1 (in levels II, III, or IV) | II*-III-IV + VI for transglottis or subglottis extension | II*-III-IV + VI for transglottis or subglottis extension |
| N2a-N2b | II, III, IV, V + VI for transglottis or subglottis extension | II*-III-IV + VI for transglottis or subglottis extension |
| N2c | According to N stage on each side of the neck | According to N stage on each side of the neck |
| N3 | Ib, II, III, IV, V + VI for transglottis or subglottis extension ± adjacent structures according to clinical and radiologic data | II*-III-IV + VI for transglottis or subglottis extension |
AJCC, American Joint Committee on Cancer.
* Level IIb could be omitted for N0 patients.
For clinically N0 patients with head and neck SCC of the oral cavity, oropharynx, hypopharynx and larynx, selective treatment of the neck is appropriate.21–23 Typically, levels I to III should be treated for oral cavity tumors, and levels II to IV for oropharyngeal, hypopharyngeal, and laryngeal tumors. Robbins has suggested that elective treatment of level IIb is probably not necessary for N0 patients with a primary tumor of the oral cavity, larynx, or hypopharynx.24 On the other hand, Byers suggested that level IV be included in the treatment of the mobile tongue due to the high incidence (10%) of skip metastases.25 Retropharyngeal nodes should be treated in tumors of the posterior pharyngeal wall. For subglottic tumors, tumors with subglottic or transglottic extension, or hypopharyngeal tumors with esophageal extension, level VI nodes should also be included in the treatment volume.
As proposed by Byers, similar guidelines could also be recommended for N1 patients without radiologic evidence of extracapsular infiltration.22 However, when an involved lymph node is located at the boundary with a level, which has not been selected in the target volume, it has been recently recommended to extend the selection to include the adjacent level.26 Typically, this will only apply for oropharyngeal tumors with a single lymph node in level II at the boundary with level Ib or for an oral cavity tumor with a N1 node in level III at the boundary with level IV.
For patients with multiple nodes (N2b), the available data suggest that adequate treatment should include levels I to V. Level I could, however, be omitted for laryngeal tumors, and level V for oral cavity tumors with neck involvement limited to levels I to III. Elective treatment of the retropharyngeal nodes should be systematically performed for oropharyngeal and hypopharyngeal tumors. As for N0 patients, level VI nodes should also be treated for subglottic tumors, tumors with subglottic or transglottic extension, or hypopharyngeal tumors with esophageal extension. Recently, for patients with nodes in the upper neck (i.e., upper level II) it has been proposed to extend the upper limit of the target volume to include the retrostyloid space.26 Similarly, the subclavicular fossae should also be included in the target volume in case of lower neck involvement (i.e., level IV or Vb nodes).26
Elective treatment of the contralateral N0 neck is still in the gray zone and is likely to be based on clinical judgment rather than on strong scientific evidence. Typically, patients with midline tumors or tumors originating from or extending to a site that has bilateral lymphatic drainage (e.g., base of the tongue, vallecula, posterior pharyngeal wall) are thought to benefit from bilateral neck treatment, whereas well-lateralized tumors (e.g., the lateral border of the tongue, retromolar trigone, tonsillar fossa) can be spared contralateral treatment. It has also been reported in tumors of the pharynx and larynx that the risk of contralateral neck metastases increased with involvement of the ipsilateral neck.27 Putting all these data together, one could recommend restricting the treatment to the ipsilateral neck for tumors of the lower gum (not approaching the midline); lateral floor of the mouth; lateral border of the mobile tongue; upper gum; cheek; retromolar trigone; tonsillar fossa (without extension to the base of the tongue, soft palate, or posterior pillar); and lateral wall of the piriform sinus. In the other situations, where elective contralateral neck treatment is recommended, the selection of the node levels to be treated should follow similar rules to those for the ipsilateral neck.
Because of the lymphophilicity of nasopharyngeal carcinoma tumors, bilateral treatment of levels II to V and retropharyngeal nodes is recommended even for patients with a node-negative neck (Table 120-8). For patients with node-positive necks, additional treatment of level Ib might be considered.
Table 120-8 Recommendations for the Selection of the Target Volume in the Neck for Nasopharyngeal Carcinoma
| Nodal Stage (AJCC 1997) | Selected Levels | |
|---|---|---|
| Ipsilateral Neck* | Contralateral Neck* | |
| N0-N2 | (Ib)†-II-III-IV-V + RP† | (Ib)†-II-III-IV-V + RP |
| N3 | (Ib)†-II-III-IV-V + RP ± adjacent structures according to clinical and radiologic data | (Ib)†-II-III-IV-V + RP |
AJCC, American Joint Committee on Cancer; RP, retropharyngeal nodes.
* In case of lymph node involvement in levels IV and Vb, inclusion of supraclavicular nodes may be required,
† Level Ib may be considered for node-positive patients.
In principle, a similar approach should apply for the definition of the node levels to be irradiated postoperatively. However, if one agrees on the selection criteria for postoperative radiotherapy (i.e., capsular rupture, patients with a metastatic node >3 cm in diameter or with more than one metastatic node), irradiation of levels I to V are typically performed. As for primary radiotherapy, the retrostyloid space and the subclavicular fossae should be included in the target volume depending on the location of the metastatic nodes.26 For laryngeal tumors, level I could be omitted. For oral cavity tumors, postoperative irradiation of level V could be omitted in the case of metastatic nodes located in level I and/or II only. Retropharyngeal and paratracheal nodes should be treated as mentioned earlier.
Neck Node Delineation and Irradiation Techniques
Delineation of the Clinical Target Volume
Since the late 1990s, several authors have proposed recommendations for the delineation of the neck node levels.28–32 In the radiation oncology community, however, the so-called Brussels and Rotterdam guidelines have emerged as the most widely used.28–30 A critical review of the two proposals was undertaken in 2003 in collaboration with representatives of the major European and North American clinical cooperative groups to generate an international set of guidelines for the delineation of the neck node levels in the node-negative neck.33 The correspondence between these guidelines and neck dissection procedures was further validated by a recent clip study.34 Last, few amendments were recently proposed to take into account the specific situation of the node-positive and postoperative neck.26,35 In short, this later proposal recommended the delineation of extranodal structures in case of muscular infiltration, the inclusion of the retrostyloid space in case of infiltration of level II, and the inclusion of the subclavicular fossae in case of infiltration of level IV and/or Vb.
It is beyond the scope of this chapter to present an in-depth discussion of the boundaries of the various levels. Interested readers are invited to refer to the original publications or to visit public Web sites, where atlases of neck node delineation have been posted (http://groups.eortc.be/radio/N0HEADNECKCTV.htm; http://www.rtog.org/atlases/hnatlas/main.html).26,33 Figures 120-7 to 120-9 present several CT cuts of the neck with selected levels.
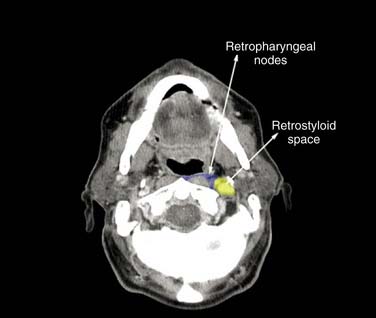
Figure 120-8. Computed tomography section at the level of the base of skull. The image was acquired as explained in Figure 120-7. The delineated areas correspond to the clinical target volume and thus do not include any security margin for organ motion or setup inaccuracy.
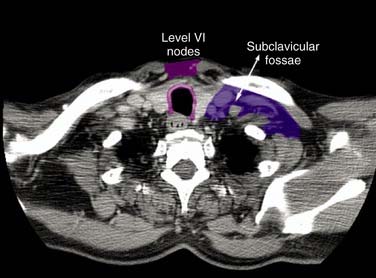
Figure 120-9. Computed tomography section at the level of the subclavicular area, 18 mm above the sterno-clavicular joint. The image was acquired as explained in Figure 120-7. The delineated areas correspond to the clinical target volume and thus do not include any security margin for organ motion or setup inaccuracy.
Irradiation Techniques
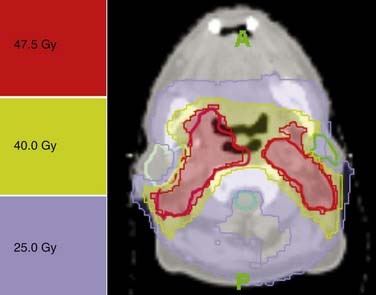
The dose prescription depends on various factors (e.g., elective vs. therapeutic irradiation, the use of combined modality treatment, planned neck node dissection, postoperative irradiation), which are beyond the scope of this section for comprehensive review. Typically, for primary radiotherapy, an elective dose on the order of 50 Gy in about 2 Gy per faction over 5 weeks and a therapeutic dose on the order of 70 Gy in about 2 Gy per fraction over 7 weeks will be prescribed. For postoperative irradiation, depending on the risk factors, doses will range from 60 to 64 to 66 Gy, in 2 Gy fraction over 6 to 6.5 weeks. A typical example of neck irradiation is presented in Figure 120-10.
Control of the N0 Neck
It is generally recommended to perform elective treatment of the neck in patients with primary head and neck SCC clinically staged N0 but having 20% or greater probability of occult lymph node metastases.36 Elective neck dissection and elective neck irradiation are equally effective in controlling the N0 neck. The choice between these two procedures thus generally depends on the treatment modality chosen for the primary tumor, which in turn mainly depends on the institutional policy. However, the basic rule that should govern the choice between surgery and radiotherapy is to favor the use of a single-modality treatment if possible and thus avoid overtreatment. For example, for a T1 or a T2-N0 supraglottic larynx, typically, a supraglottic laryngectomy with a selective neck node dissection or a primary radiotherapy on the larynx and the neck are equally effective therapeutic options. For such stage disease, the need for postoperative radiotherapy is indeed quite low. Conversely, for a T3-N0 supraglottic larynx, a conservative treatment approach with primary radiotherapy or concomitant chemoradiotherapy should be favored because of the necessity of postlaryngectomy radiotherapy and the nonsuperiority of the surgical approach.
Table 120-9 presents the percentage of neck recurrences in large retrospective series of pharyngolaryngeal SCC treated with conventional fractionated radiotherapy.37–39
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree