CHAPTER 91 Oral Mucosal Lesions
Red/White Lesions
Leukoedema
Etiology and Pathogenesis
No clear cause of this condition has yet been established. Factors that may have a role in the manifestation of this condition include smoking, alcohol ingestion, bacterial infections, electrochemical interactions, and possibly inflammatory salivary disorders, although no specific, reproducible, or definitive cause has been proven to date. Many authorities indicate that this hardly represents a disease entity but rather may be considered a variation of what is normal. Similar edematous alterations of mucosa may also be seen in the vagina and larynx, further supporting the notion that this is indeed a normal variant of mucosal surface appearance. The incidence of leukoedema increases with age up to 40 to 49 years, followed by a decline.1
Clinical Features
Leukoedema is asymptomatic in nature and is usually discovered incidentally during routine examination or by the patient. The distribution tends to be symmetric and involves the buccal mucosa and, to a lesser extent, the labial mucosa. The appearance may be that of a diffuse filmy grayish surface with white streaks, wrinkles, or milky alteration (Fig. 91-1). At rest the tissue may appear opaque, whereas when stretched, the opaque alteration dissipates to the point where it is barely visible.
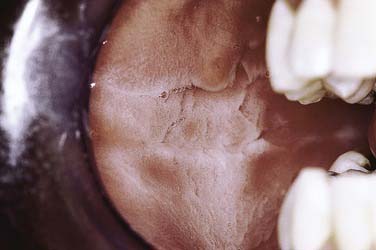
Figure 91-1. Leukoedema is characterized by a milky, translucent, and diffuse alteration of the buccal mucosa.
Histopathologic Features
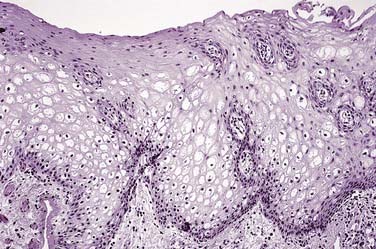
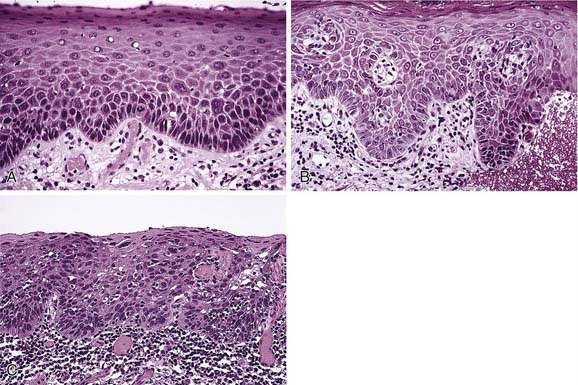
The surface of the epithelium is usually parakeratinized, whereas the epithelial ridges are broad and elongated with occasional spike-type morphology. Dominating the histomorphology is marked intracellular edema within the prickle cell or spinous cell layer, where individual cells will appear enlarged and optically clear or vacuolated, with peripherally displaced pyknotic nuclei and an overall bland appearance in association with a volumetric increase in the thickness of the epithelial layer (Fig. 91-2).
Oral Leukoplakia
Definition
Experts commonly agree that leukoplakia represents the most common premalignant oral mucosal lesion; however, most cases are benign and remain so over time. The concept of malignant transformation is essentially related to the development of cancer or preinvasive cancer that is further characterized by variable degrees of epithelial dysplasia. The possibility of carcinoma and dysplasia must be carefully considered, with the percentages of leukoplakia showing microscopic evidence of dysplasia ranging from 3.7%2 to 28.7%.3 A more recent review4 pointed out that the malignant transformation potential of leukoplakia was noted by Sir James Paget in 1851, when he provided the essential description of what we currently consider to be representative of leukoplakia. The entity of oral leukoplakia was initially and specifically described in the latter half of the 19th century by the Hungarian dermatologist Schwimmer. Since the late 18th century and into the 19th century, oral leukoplakia has been recognized and fully established as a definitive precancerous lesion, often serving as a harbinger for the development of oral cancer. The major clinical factor of importance in this lesion resides in its relationship to the general morbidity and relatively high overall mortality associated with invasive squamous cell carcinoma.
Conceptually, therefore, the definition of a premalignant lesion may be that of a morphologically altered tissue within which cancer is more likely to develop when compared with its apparently normal counterpart. In a more contemporary sense, the work of Waldron and Schafer (1975)5 and Bouquot and Gorlin (1986)6 established the clinical and microscopic correlation of leukoplakia in a large number of persons within the United States. In those studies the presence of dysplasia, carcinoma in situ, and invasive carcinoma in relation to oral leukoplakia from all sites ranged from 17% to 25%.
Etiology
Along the lower lip vermilion surface, patches of leukoplakia may develop in response to chronic sun exposure (actinic cheilitis), demonstrating close correlation with cumulative exposure and the level of cutaneous pigmentation (Fig. 91-3).
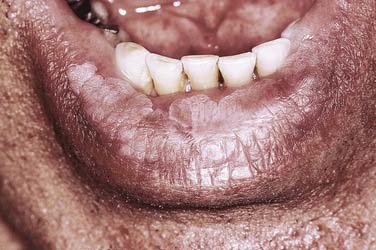
Figure 91-3. Lower lip vermilion surface affected with leukoplakia interspersed with patchy melanotic pigmentation.
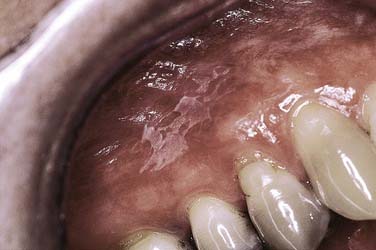
A recently described form of keratosis has been attributed to the use of mouthwash preparations and toothpaste containing an herbal root extract, sanguinaria (Viadent). Characteristically, the lesion develops along the labial alveolar mucosa of the maxilla as an often translucent to slightly opaque white patch with well-defined margins and generally smooth surface features (Fig. 91-4). The relationship to development of dysplasia or malignant transformation in association with this entity remains controversial, with reversal of lesions often noted on withdrawal of sanguinaria-containing products.7,8
Clinical Features
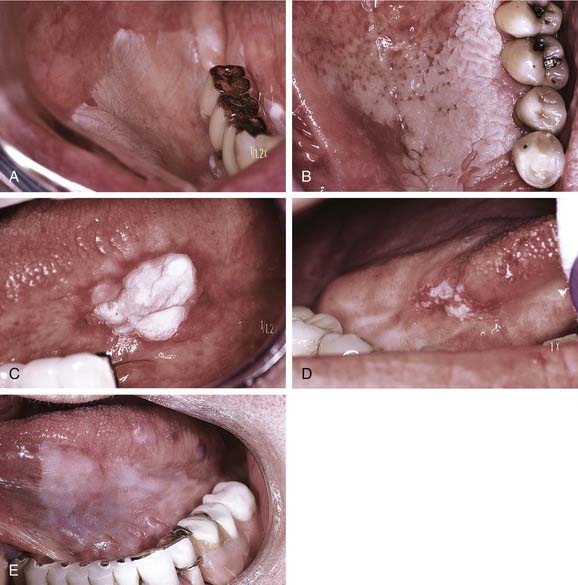
A widely variable clinical pattern characterizes leukoplakia. In general the appearance can range from a thin grayish surface alteration demonstrating a white translucent quality with ill-defined margins (Fig. 91-5A) and generally smooth surface qualities to discrete, sharply marginated, thick and opaque plaques. In general terms such lesions may be homogeneous and smooth, focal or diffuse, or heterogeneous and multifocal with variable textures (see Fig. 91-5B and C). Surface textural alterations can range from a fine granularity to a slightly papillary outline. In addition, leukoplakia can appear ulcerative, erosive, speckled, nodular, or verrucous to the naked eye. Importantly, the various forms of leukoplakia often transition from one to another; however, this change of clinical appearance may not necessarily correspond to a shift in biologic behavior.9,10 In cases where there is a clinical shift in appearance from homogeneous leukoplakia to a heterogeneous, speckled, or nodular form, a rebiopsy is mandatory (see Fig. 91-5D) because of the well-established correlation between increasing levels and frequencies of dysplasia and an increase in the level of regional heterogeneity or speckled quality.11
The term thin leukoplakia (see Fig. 91-5E) has been used to describe early leukoplakia formation. Such lesions generally progress over time from a macular to a slightly elevated translucent plaque with blending margins into normal-appearing mucosa, to later developing a more thickened appearance lending a more opaque white color to the lesion. Likewise, textural features may develop with increasing duration, which may be furrowed, leathery, or wrinkled in appearance. Similarly, lesions can become exophytic or verruciform in quality and might project above the surface. Although many lesions may remain for extended periods as homogeneous or more textured, thickened alterations, some lesions may disappear over time.
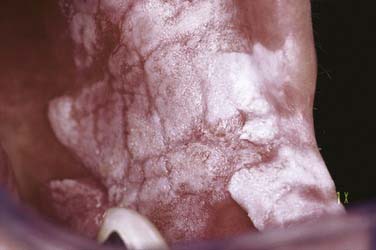
A recently described, uncommon variant of leukoplakia, proliferative verrucous leukoplakia, requires separate designation in that it is often noted in individuals without the usual risk factors for leukoplakia development, with lesion development noted in areas less commonly affected by oral squamous cell carcinoma. Behavior of this variant of leukoplakia is characteristically aggressive with changing histopathologic features, a more relentless behavior pattern, and greater tendency to develop into squamous cell carcinoma.12 No linkage to human papillomavirus has been demonstrated in comparison with other forms of oral leukoplakia.13 It is characteristically multifocal and persistent and tends to occur more often in women. An evolution from a thin, flat, white patch to a leathery, thickened, and ultimately papillary to verrucous stage characterizes this lesion over time (Fig. 91-6). When the lesion is microscopically evaluated at the papillary or verrucous stage, the diagnosis can vary from that of a benign or atypical verrucous hyperplasia to verrucous carcinoma or squamous cell carcinoma with papillary features. Likewise, characterizing this entity is persistence, a high recurrence rate, and development of squamous cell carcinoma in up to 74% of cases.14,15 Gaining a better understanding of the behavioral qualities and variation across the spectrum of oral leukoplakia involves several factors including clinical appearance, location, and the presence and degree of dysplasia. Recent studies have demonstrated an association between progression of oral mucosal premalignancy and a loss of heterozygosity (LOH).16 An essential step in the initiation of escape from normal epithelial proliferation and differentiation involves LOH at 3p and 9p where a modest 3.8-fold relative risk for oral cancer development has been noted. However, in cases where additional losses (4p, 8p, 11q, or 17p) existed, there was a 33-fold increase in relative risk for cancer development.17 Lending a risk-targeting or prognostic strategy to this concept has been controversial, with earlier work concerning ploidy analysis being discredited and more recent studies refuting this claim.18 No single or specific molecular pathway has been identified as the primary factor in the progression of epithelial dysplasia to squamous cell carcinoma. Continuing studies have revealed a range of molecular and genetic changes associated with oral squamous cell carcinoma tumorigenesis including mitochondrial alteration and several epigenetic changes, with continued ongoing investigation including those of altered methylation patterns and early carcinogenesis.19 Finally, the relationship between the site of leukoplakia and the presence and risk of dysplasia or carcinoma must be considered. Earlier and more recent studies have underscored this relationship with the floor of the mouth, ventrolateral tongue, and retromolar trigone/soft palate complex carrying risk levels greater than other oral sites.5,20
Histopathologic Features
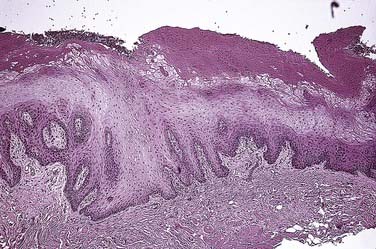
A spectrum of histologic alterations characterizes idiopathic or true leukoplakia that ranges from hyperkeratosis and acanthosis to variable degrees of dysplasia or carcinoma in situ and ultimately invasive squamous cell carcinoma (Fig. 91-7). Cellular atypia, a component of dysplasia, refers to abnormal cellular features, whereas dysplasia refers to disordered cell growth and architectural distortion. With dysplasia, a grading system of mild, moderate, or severe is the convention; the level is determined subjectively in an attempt to convey tissue alteration to the level of intraepithelial localization or non-neoplastic or noninvasive quality.
Treatment
The role of chemoprevention strategies in cases of mild dysplasia or treated dysplasia remains an active and important area of research, though there is no proven and effective prevention strategy regarding malignant transformation. Retinoids, antioxidants, and cyclooxygenase (COX)-2 inhibitors hold promise as management alternatives within the area of chemoprevention, though no effective strategy has yet been proven by randomized clinical trials. In general there is no effective treatment in preventing malignant transformation of leukoplakia.21 On balance and regardless of location, the dynamic nature of oral leukoplakia and its cancer-related potential should be appreciated and understood. The key to limiting the likelihood of late-stage disease evolving from such lesions concerns itself with an appreciation of the potential of this group of mucosal alterations coupled with the establishment of early diagnosis, intervention, and follow-up.
Oral Hairy Leukoplakia
Definition
A distinctive asymptomatic white lesion of the oral mucosa, oral hairy leukoplakia has been defined both clinically and microscopically with a distinct relationship in the vast majority of cases with systemic immunosuppression. The Epstein-Barr virus is considered the etiologic agent of this lesion that characteristically arises along the lateral tongue margins bilaterally, ranging from subtle white keratotic vertical streaks to thick corrugated and then to shaggy surface alterations (Fig. 91-8).22 Early lesions may be more smoothly textured and macular in presentation. Less common sites include the dorsum of the tongue, buccal mucosa, and floor of the mouth.
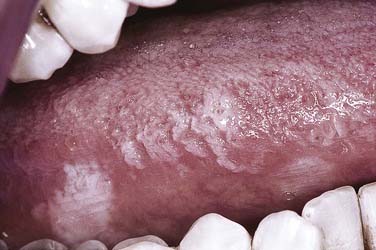
Figure 91-8. Oral hairy leukoplakia along the lateral tongue margin is characterized by vertically corrugated keratotic ridges.
Diagnosis by routine microscopy and in situ hybridization to demonstrate the presence of the Epstein-Barr virus is essential in that the confirmed diagnosis almost always correlates with systemic immunosuppression. The microscopic features suggesting this diagnosis include hyperkeratosis and irregular surface projections and irregularities (Fig. 91-9). Ballooning degeneration of the subsurface layers with nuclear alterations consisting of clearing and chromatin displacement by viral replication occurs along the inner aspect of the nuclear membrane in the form of “beading.” Confirmation of the impression of a viral process can be achieved by in situ hybridization, Southern blot procedure, polymerase chain reaction, or ultrastructural demonstration of virus.
Oral Lichen Planus
Although the general etiology of lichen planus is unknown, it is generally thought to represent a T-cell-mediated or immunologically driven chronic inflammatory process in the absence of any consistent serologic changes. Immunologic studies have demonstrated that the lymphocyte subpopulations involve cells of mixed CD4+ and CD8+ types that express α-1 integrin molecules. The majority of the T cells present are activated CD8+ cells.23 Existing data suggest a complex nonspecific mechanism might be involved in pathogenesis including major histocompatibility complex (MHC) class 1- and MHC class 2-restricted antigen presentation by lesional keratinocytes, activation of antigen-specific CD4+-positive helper T cells and CD8+-positive cytotoxic T cells, clonal expansion of antigen-specific T cells, and keratinocyte apoptosis triggered by antigen-specific CD8+-positive cytotoxic T cells. More recent studies, however, have discussed the monoclonal nature of lesional T cells in oral lichen planus.24 Antigen-specific mechanisms therefore include antigen processing by resident dendritic cells and antigen-specific keratinocyte killing by CD8+-positive cytotoxic T cells. A host of nonspecific mechanisms also seem to be involved in pathogenesis including activation of matrix metalloproteinases, mast cell degranulation, alterations in the epithelial basement membrane region, and elaboration of an array of proinflammatory cytokines including RANTES, which play a vital role in inflammatory cell recruitment into the area of the affected skin or mucosa.
As a result of lymphocyte adhesion along the inner aspect of superficial vessels within the lamina propria of the mucosa, they soon exit the vasculature to traverse the basement membrane and migrate into the overlying epithelium secondary to the up-regulation of chemoattractants by keratinocytes.25
Clinical Features
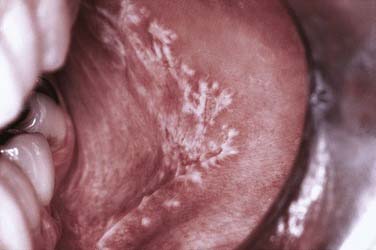
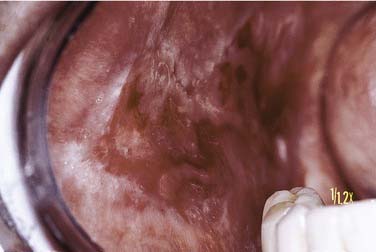
Oral mucosal lesions of reticular lichen planus are most commonly seen in the middle-aged population with a characteristic distribution over the buccal mucosa in a symmetric bilateral manner. The surface of the mucosa is characterized by delicate white keratotic striae that intersect and arborize over a mucosal surface that may be diffusely erythematous to normal in quality (Fig. 91-10). Striae may be preceded by keratotic papules, which fuse and ultimately form the striae. The pattern is generally lacy to annular in configuration with extension over the buccal mucosa and often inferiorly into the mucobuccal folds. Striae may also appear over the dorsum and lateral portion of the tongue and less frequently over the attached gingiva and vermilion portion of the lip. Of note is the general freedom from symptoms related to this form or variant of oral lichen planus.
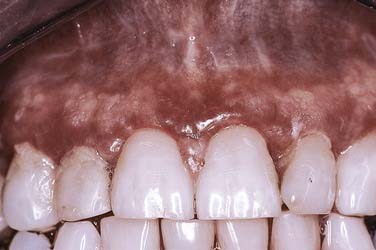
Atrophic or erythematous lichen planus demonstrates only faint white striae, being dominated by thinned, reddened mucosa, often noted concomitantly with reticular or erosive forms of this disease. The attached gingiva is most likely to be involved by this type of lichen planus where there is a loss of the usual pink, stippled quality of the tissue surface (Fig. 91-11). In its place is a glossy, edematous, and erythematous alteration that is tender and bleeds easily, leading indirectly to the accumulation of large amounts of dental bacterial plaque. This usually results from the inability of patients to maintain their usual levels of dental hygiene given the friability and tenderness of the gingiva, which bleeds with the slightest provocation. Bacterial antigens and toxins in turn elicit the formation of inflammatory cytokines locally that further enhance the immunologically driven process within the lichen planus.26
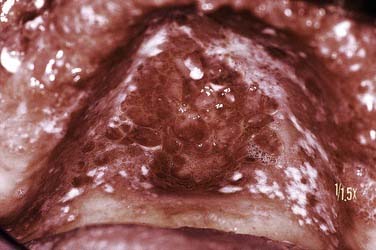
Erosive lichen planus is characterized by central ulceration that can be deep and painful but is usually superficial in nature (Fig. 91-12). The ulcer is surfaced by a pseudomembrane or firmly adherent fibrinous plaque, which is usually sharply demarcated. Often it is possible to identify peripherally localized white striae, which gradually transition to an erythematous, nonstriaform region as the ulcerated area is approached. As the erosions heal, new areas will break down into ulcerations with variable patterns of change noted over time.
Association with Systemic Diseases
Oral lichen planus has been reported in conjunction with primary sclerosing cholangitis, lupus erythematosus, primary biliary cirrhosis, and Sjögren’s syndrome.27,28 Although controversial in certain geographic areas, within the United States there appears to be a lesser association between oral lichen planus and many systemic diseases, in particular hepatitis B and C, in comparison with patients with oral lichen planus in Italy and Japan, thus suggesting genetic susceptibility or predisposition.
Histopathologic Features
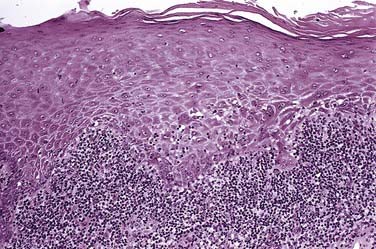
At the free surface of the epithelium, hyperkeratosis/parahyperkeratosis will be present. A slightly thinned to atrophic spinous layer overlies the basal and parabasal layers, which are often degenerate and vacuolated (Fig. 91-13). Over time, the epithelium becomes thin and may assume a sawtooth pattern on occasion. Scattered throughout the epithelium are increased numbers of dendritic cells that have been reported to function as antigen processing units, delivering prepared antigen to the subjacent lymphocyte population, which morphologically forms a dense bandlike pattern at the epithelial connective tissue junction.29 At this junctional zone, occasional ovoid, anucleate eosinophilic droplets with hyaline features are noted. These represent apoptotic keratinocytes termed colloid or Civatte bodies.30 Although nonspecific and nondiagnostic of lichen planus, they are highly suggestive of such when correlating other pathologic and clinical findings.
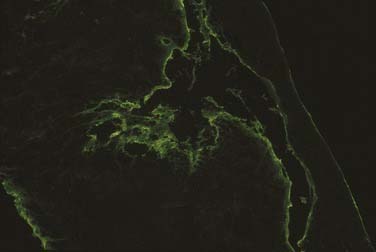
Reflective of the altered basement membrane region is the nonspecific deposition of fibrin that can be demonstrated by direct immunofluorescence studies as a uniform linear deposition along the basement membrane (Fig. 91-14). This pattern can be noted in up to 90% to 100% of cases.
Treatment
Important in communicating the therapeutic end point to the patient is the notion that the goal of management is relief of symptoms because immediate cure is not to be achieved. Topically applied agents, usually corticosteroids, are the mainstay of management in most forms of oral lichen planus that are symptomatic in nature. The specific form or choice of corticosteroid will relate to the potency of such, with moderate to ultra-potent agents considered the modality of choice. More recently, the nonsteroidal topical agents tacrolimus and pimecrolimus have been shown to be effective in the management of symptomatic or lichen planus without the usual adverse effects related to topical steroid use, most commonly oral candidiasis.31
Relationship to Oral Squamous Cell Carcinoma
The notion of lichenoid dysplasia versus dysplasia developing in lichen planus may not be readily settled. However, studies suggest a significant degree of allelic loss in dysplastic oral lichenoid lesions. Reports stating a degree of dysplasia in association with lichenoid features should alert the clinician to either remove the balance of the altered mucosa or follow the patient carefully.32 Mattsson and colleagues33 support the relationship between oral lichen planus and oral squamous cell carcinoma, yet they dispute, on an economic and practical basis, the need for continuous recall of these patients in specialists’ offices and clinics.
Finally, the issue of an “oral lichenoid lesion” versus oral lichen planus has been discussed within the context of malignant transformation. Stated is the possibility of an increased risk of transformation within the context of lichenoid lesions, whereas true lichen planus may be less of a transformation risk.34
Submucous Fibrosis
Definition
Submucous fibrosis represents a multifactorial disorder with the considered chief etiologic factor being the consistent and habitual use of areca (betel) nut, either in the form of chewing or simply placing a quid of material (paan masala) in the buccal or labial sulcus several time per day, or in a packaged powdered form with other components (guthka), over many years. Submucous fibrosis is considered a premalignant condition, with transformation rates as high as 7.6% reported from India over a 17-year period.35 Other researchers have reported 2.54% of cases demonstrating epithelial dysplasia.36
Etiology and Pathogenesis
Some experts believe the basic process is a failure of collagen remodeling and that altered epithelial-mesenchymal interactions result in formation of collagenous bands and aggregates within the submucosa and lamina propria. A diminished level of functional collagenase levels has been postulated as another mechanism to explain increased collagen accumulation.37
Clinical Features
Initial mucosal alterations characteristic of oral submucous fibrosis include erythema with or without vesiculation. The former reaction (erythema) is believed to be related to increased levels of inducible nitric oxide synthetase.38 This is followed by slow diminishment of erythema and a progressive decrease in the degree of oral opening and tongue mobility reflective of the altered collagen metabolism. Pallor of the normally pink mucosa becomes evident as the underlying chronic inflammation recedes and fibrosis and hyalinization progress. Scar bands may become evident deep within the buccal soft tissues, further limiting jaw opening and function.
Histologic/Diagnostic Features
Dominating the histopathology is a juxtaposition of atrophic epithelium surfacing a subjacent fibrosis. Early connective tissue alterations are characterized by delicate and loosely arranged collagen fibers with progressive degrees of hyalinization until, in the late stages, there is complete hyalinization of the supportive connective tissue. Variable degrees of chronic inflammation occur in the form of lymphocytes and plasma cells. Variable levels of dysplasia have been noted. In one study, mild dysplasia was present in 46% of cases, moderate dysplasia in 52%, and severe dysplasia in 2%.39
Treatment
Management of oral submucous fibrosis is problematic, particularly in advanced cases and when the use of areca-containing products remains in place. Surgical release procedures of scar bands have been only modestly successful. More recently collagenase and pentoxifylline administration in separate studies has been proposed.37,40
Verruciform Xanthoma
Etiology and Pathogenesis
The cause is unknown. The cellular lineage of the proliferative xanthoma cells is that of monocyte/macrophage type. They are noted chiefly in the fifth decade and have been classified into three types according to their microscopic surface qualities. These include the verrucous, papillary, and flat types.41
Histopathologic/Diagnostic Features
Variable levels of surface hyperkeratosis cover a papillary or verrucous configuration with deeply invaginated crypts alternating with papillary extensions. Epithelial ridges composed of normal keratinocytes are elongated and extend uniformly into the lamina propria, which is engorged with numerous xanthoma cells containing characteristic foamy to flocculent cytoplasm. PAS-positive and diastase-resistant granules are present within the cytoplasm of these cells, as well as lipoid droplets, and are macrophages in nature.42
Candidiasis
Etiology and Pathogenesis
Candida albicans represents the most common candidal species, whereas Candida tropicalis, Candida krusei, and Candida glabrata among others are less commonly involved. In immunosuppressed and neutropenic patients, however, these latter species are more often present. Of note is the essential shift from a commensal state to that of a “pathologic” state, in which former asymptomatic carriers of the organism now suffer from its overgrowth. Risk factors for development of candidiasis may include local and systemic factors (Tables 91-1 and 91-2). Commonly, systemic conditions such as poorly controlled diabetes mellitus and immunosuppression are associated with oropharyngeal candidiasis. Local effects or altered ecology of the region by topically delivered drugs (corticosteroids), xerostomia (with loss or diminution of saliva’s protective function), heavy smoking, and denture appliances favor development of candidal overgrowth.43–46
Table 91-1 Oropharyngeal Candidiasis: Risk Factors
| Local factors (mucosal barrier function) | Heavy smoking |
| Foreign bodies (dentures, nasogastric tubes) | |
| Radiation-induced mucositis | |
| Inhalational and topical corticosteroid use | |
| Xerostomia | |
| Mucosal tumors | |
| Systemic factors | Immunosuppression, age, viral-retroviral, chemotherapy, corticosteroid use |
| Diabetes mellitus | |
| Intrinsic immunodeficiency | |
| Myelodysplasia/leukemia | |
| Antibiotic administration |
Table 91-2 Clinical Forms of Oral/Oropharyngeal Candidiasis
| Acute candidiasis | Pseudomembranous (thrush) |
| Erythematous/atrophic | |
| Chronic candidiasis | Hyperplastic (candidal leukoplakia) |
| Denture-related (chronic erythematous/atrophic) | |
| Median rhomboid glossitis | |
| Angular cheilitis |
From Akban A, Morgan R. Oral candidiasis. Postgrad Med J. 2002;78:455-459.
Crucial to the establishment of infection, which implies a shift from commensalism, is fungal adhesion to mucosal surfaces and subsequent replication. Yeast adherence to several ligands is complex. It is aided by intrinsic candidal adhesion that in turn binds to salivary proteins that are absorbed onto cell surfaces.47,48 Such binding, one measure of virulence, is a covalently mediated bonding in part related to fungal synthesis of this hyphae-associated proline-rich surface protein.
Clinical Features
Of the clinical forms, the erythematous, atrophic, and pseudomembranous types are most likely to produce symptoms (Fig. 91-15). So-called denture sore mouth (chronic erythematous/atrophic candidiasis) is generally minimally to completely asymptomatic, whereas the acute forms are uniformly problematic. Deeply invasive candidal infection in immunosuppressed persons may present as an ulcerative lesion. Of note is the tendency of the various clinical forms to cycle from a bland, asymptomatic state to a symptomatic one in relation to general host factors or degree of medical compromise. Acute pseudomembranous candidiasis (thrush) with characteristic soft, white, and superficial colonies of organisms may be widespread and often asymptomatic (Fig. 91-16).
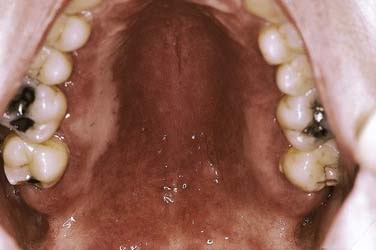
Figure 91-15. Acute erythematous candidiasis over the hard palate mucosa developed subsequent to a course of broad-spectrum antibiotics.