CHAPTER 93 Odontogenesis, Odontogenic Cysts, and Odontogenic Tumors
Background
Root Development
The periodontal ligament is actually a joint known as a gomphosis joint. Though the cementum cannot be distinguished except by location, the body, for instance, during orthodontic movement will resorb the bone on the lamina dura side of the periodontal ligament and not the cementum on the tooth side of the ligament. The tooth with the periodontal ligament will retain a certain amount of mobility within the joint. The reader is again referred to various texts for a more in-depth coverage of odontogenesis.1–5,5a
Summary
There are a number of odontogenic epithelial stages and each may provide a basis for odontogenic cysts or tumors. The four main stages considered are (1) dental lamina, (2) enamel organ, (3) reduced enamel epithelium, and (4) Hertwig’s epithelial root sheath. The rests of Serres and Malassez are considered along with their respective progenitors of the dental lamina and Hertwig’s root sheath. In addition, the stomodeal epithelium gives rise to Rathke’s pouch, which retains odontogenic potential. Shafer and colleagues6 suggest the original basal epithelium of the stomodeum also retains potential. In the adult this basal epithelium is represented by the gingival and alveolar mucosal surfaces.6 This concept seems to be supported when peripheral ameloblastomas appear to develop directly from overlying gingival epithelium. With that said, most clinicians would consider the adult basal epithelium to be a rare source of odontogenic neoplasia. The rests of Malassez are common sources of inflammatory odontogenic cysts but retain little neoplastic potential. The rests of Serres, the enamel organ, and reduced enamel epithelium are generally considered the stages most likely to become neoplastic. All stages have the potential to form cysts but to variable degrees. Dentigerous cysts with their origin from the reduced enamel epithelium and radicular cysts from rests of Malassez make up the overwhelming majority of odontogenic cysts.
Odontogenic Cysts
Classification
When reading various sources it becomes quickly clear that what a cyst “is” varies by author and that the classification schemata are in disarray as well. The modified classification scheme seen in Box 93-1 is my attempt at organization. The odontogenic cyst of undetermined origin is a new, admittedly descriptive “diagnosis” used by some oral pathologists. Unfortunately the descriptive nature of that term will not be available to look up in other texts or journals. However, it replaces the diagnosis of primordial cyst. The need to use a descriptive term instead of primordial cyst is due to the ambiguous use of primordial cyst both as an odontogenic keratocyst (OKC) and a simple nonkeratinizing cyst that cannot be classified in relation to the tooth. The descriptive role is to provide a pigeonhole in which to place lesions that are histologically ambiguous, not directly associated with a tooth but located in the alveolus and thus presumably odontogenic in origin.
Box 93-1 Classification of Odontogenic with Other Selected Maxillofacial Cysts
HIV, human immunodeficiency virus; WHO, World Health Organization.
Several classification schemata for odontogenic cysts and oral and maxillofacial cysts exist.7–9 The classification scheme seen here is modified from that of the World Health Organization (WHO). In 1992 WHO published the second edition of Histological Typing of Odontogenic Tumors.10 Unfortunately the most recent WHO treatise on odontogenic tumors is contained within the Pathology and Genetics of Head and Neck Tumors.11 In this change odontogenic cysts are no longer covered within the text. Box 93-1 also contains selected nonodontogenic cysts for completeness and comparison.
By definition a cyst is considered a “pathologic cavity at least partially lined by epithelium.” To be an odontogenic cyst the epithelial lining must be derived from odontogenic epithelium. The best advice to the reader is that all classification schemata are artificial to some extent. The key is to organize them the way that is most useful to you. This modification in Box 93-1 is an attempt at self-clarification and will hopefully be useful for others in either its pure or modified form.
Pathogenesis
Cyst expansion occurs because of numerous factors including accumulation of inflammatory cells, fibrin, serum, and desquamated epithelial cells. As these products enter the cystic cavity, it is the accumulation of the intraluminal products that spurs the cystic expansion of the wall.12–14 Alternatively, cyst expansion may be spurred on by the inherent mitotic activity of the cyst wall itself. If this mitotic activity is the major component of the cyst expansion, it may be better to consider the lesion a cystic neoplasm rather than a simple cyst.14–20 This debate lies at the center of how to classify the OKC, as well as the calcifying odontogenic cyst.21 In the case of the OKC, WHO has renamed it the keratocystic odontogenic tumor. Discussion of this entity is in the odontogenic cyst section. In the case of the calcifying odontogenic cyst (the tumor version), the reader is referred to more detailed articles on the epithelial odontogenic ghost cell tumor.
Multilocularity may in itself be a signal that the lesional growth is mitotically or multifocally driven rather than hydraulically driven.22–25 For this reason the potentially multilocular odontogenic cysts, such as the botryoid odontogenic and glandular odontogenic cyst, may arguably have a neoplastic potential as well.22,26–30 However, in the case of the botryoid cyst the possibility of multifocality cannot be ignored. Cell regulation protein studies to determine cell inhibition and division activities may be helpful in future classifications.31,32 In addition, the ability of epithelia to break down elements of the connective tissue wall could be important.33–37
However, even simple cysts like the periradicular cyst derived from rests of Malassez must possess some mitotic activation, or growth would be impossible. Activation is thought to occur as a result of inflammatory production within the periodontal membrane.13,38 In the skin and gingiva it has been shown that inflammation leads to release of inhibitors, which then allow the renewal of mitotic activity.15 Once a solid epithelial sphere has been formed, it is thought that it eventually outgrows its vascular nourishment and the central area degenerates to form a lumen.15,16 Following the formation of the central lumen, transepithelial flow of fluid is sustained by osmotic forces. Thus hydrostatic pressure plays a role in the development of the classic unilocular appearance of most cysts. How the pressure results to produce osteoclastic resorption is less clear.17,39–41
Cysts that are derived from the more neoplastic dental lamina, or are in themselves “cystic neoplasms,” probably occur as a result of self-sustained or unregulated mitotic activity.42 Even in neoplastic cysts, luminal expansion may occur through degenerative effects, debris accumulation, and hydraulic and mitotic activity.43–45
Periapical Cyst (Radicular Cyst, Periradicular Cyst)
The periapical cyst must be associated with a nonvital tooth. The tooth may be rendered nonvital by trauma, caries, or periodontal space extension. As such, these cysts may be seen at any age, although permanent teeth are more likely to be involved than deciduous teeth.46 They are thought to be derived from rests of Malassez.
Radiographic Features
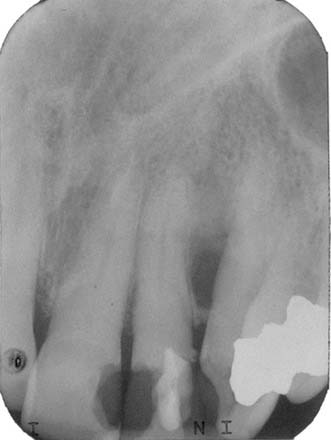
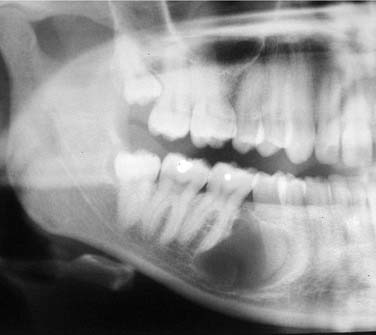
Periapical cysts present as a unilocular radiolucency at the apical portion of the tooth. Though well defined, the border varies from corticated to sclerotic to merely well defined. Variations depend on the amount of inflammation present. Long-standing, neglected lesions can get quite large, though most are less than 1 cm in diameter (Fig. 93-1).
Microscopic Features
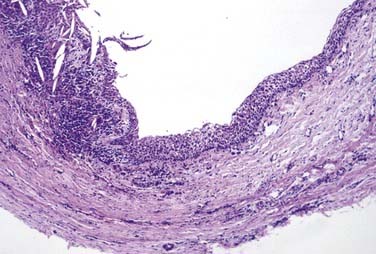
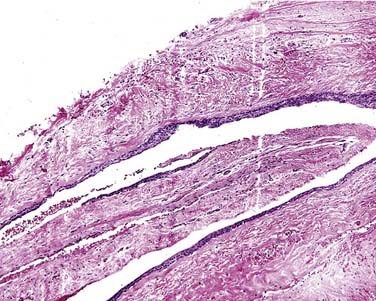
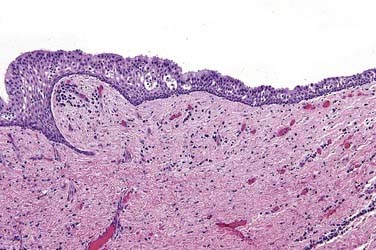
This is the classic inflamed “common odontogenic cyst” and as such the luminal lining will consist of nonkeratinized stratified squamous epithelium. This is an inflammatory cyst and inflammation is invariably present if sufficient sampling is performed (Fig. 93-2). Rests of Malassez are possible in the connective tissue. However, odontogenic rests are rarely seen in the cyst wall even though these rests are thought to be the source of the epithelial proliferation. Cholesterol slits, foreign body giant cells, and hemosiderin deposits are common findings. As in all “common odontogenic cysts,” squamous odontogenic tumor-like” proliferations may be seen in long-standing lesions. These epithelial islands will be cytopathologically benign without evidence of dysplasia. If squamous odontogenic cystlike proliferations are noted, they should essentially be ignored and are of no prognostic significance. In endodontically treated teeth, foreign bodies secondary to endodontic therapy are common.47 Bacterial colonies may also be seen in these cysts. Though actinomycetes colonies may portend a tendency for being slow to resolve, their presence should not result in a diagnosis of osteomyelitis. Such colonies are more commonly an incidental rather than a significant finding. Thus, for multiple reasons, the proper diagnosis of periapical cyst requires radiographic or clinical corroboration.
Treatment
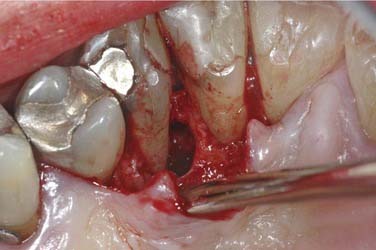
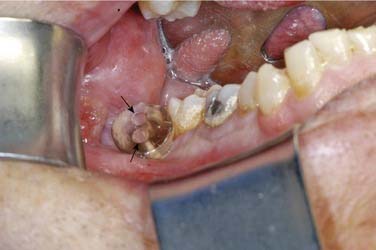
This cyst is treated with simple enucleation (Fig. 93-3). Enucleation is often accomplished at the time of tooth extraction. Uncounted numbers of these cysts are probably adequately resolved with endodontic therapy. If a radiolucency persists longer than 6 months following endodontic therapy, enucleation and histopathologic review are necessary.48–50
Residual Cyst
The majority of these cysts will be the result of leaving a periapical cyst “behind” following tooth extraction. All of these cysts are inflammatory cysts. Occasionally an inflamed dentigerous cyst is incompletely removed and could also be the source of a residual cyst. The clinical, microscopic, radiographic, and histologic features are identical to the periapical cyst.48,49,51,52
Inflammatory Collateral Cyst
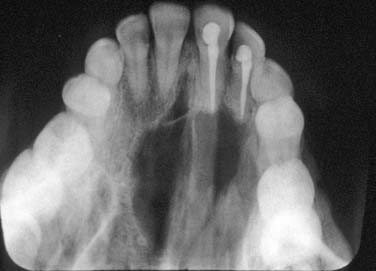
This may or may not be considered by some to be a true cyst. However, because it is an occasionally used diagnosis, a quick summary is included here. This lesion is associated with periodontal disease of a vital tooth. Uncommonly, a deep intrabony periodontal pocket may be sufficiently isolated to allow for hydraulic expansion of the bone (Fig. 93-4). As such, radiographically there will be a radiolucent periradicular lesion (Fig. 93-5). There will also be a periodontal pocket associated with that radiolucency. This diagnosis should be limited to those cases where the clinician indicates the diagnosis as the most likely choice. Otherwise, the clinical, microscopic, radiographic, and histologic features are identical to the periapical cyst.53
Dentigerous Cyst (Follicular Cyst)
The dentigerous cyst by definition must be associated with the crown of an unerupted tooth, developing tooth, or odontoma. The eruption cyst is essentially a subtype of dentigerous cyst that is confined just by the overlying alveolar mucosa. Dentigerous cysts form when fluid accumulates between reduced enamel epithelium and tooth crown.14 As alluded to earlier, the accumulation of fluid may be partially or largely surrounded by connective tissue and epithelium.54,55 Because the third molars and maxillary canines are the teeth most frequently impacted, they are also the most likely to be associated with dentigerous cysts. However, any impacted tooth has an increased risk. There also may be inherent differences in impacted tooth development and how the reduced enamel epithelium is transformed/resorbed.56 They are generally found in the teenage years and early adulthood.57 However, the longer a tooth is impacted, the greater the chance a dentigerous cyst will develop.58
Radiographic Features
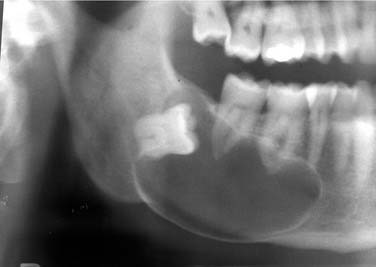
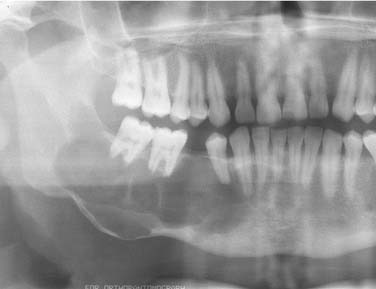
A dentigerous cyst presents as a unilocular radiolucency, which is associated with an unerupted tooth (Fig. 93-6). Dentigerous cysts may also involve odontomas that, by nature, also have “tooth crowns.” The radiolucency is generally well demarcated and well corticated. The border may become sclerotic or display rarifying osteitis if secondary infection is present. Even large cysts that have pushed the associated tooth considerable distances will display evidence of origin from the cementoenamel junction if the film angle is adequate. In large lesions the origin from the cementoenamel junction is best visualized as an area of cortication at the cementoenamel junction. There is considerable overlap between the appearance of small dentigerous cysts and hyperplastic follicles.56
Microscopic Features
The specimen will present primarily as variably dense fibrocollagenous connective tissue with some areas being loose and myxomatous. Odontogenic epithelial rests are usually scattered within the connective tissue and are most common near the epithelial lining. The luminal lining consists of nonkeratinized stratified squamous epithelium. The presence of mucous prosoplasia within the lumen is not uncommon. Care should be taken to not overinterpret the mucus prosoplasia. Cholesterol slits and their associated multinucleated giant cells may be present in inflamed cysts and are generally associated with the connective tissue wall.47 As mentioned earlier, the lumen may be partially or mostly lined by connective tissue.14 If present in the specimen, crevicular epithelium may make microscopic separation of an inflamed dentigerous cyst from pericoronitis impossible. Thus proper diagnosis requires radiographic or clinical corroboration.
Neoplastic Potential
Dentigerous cysts appear to retain the ability to transform into true neoplasms. One study reported that 17% of ameloblastomas were associated with an existing dentigerous cyst.59 This figure varies by study, however.60–62 Both squamous cell carcinomas and mucoepidermoid carcinomas have been reported.63–67
Eruption Cyst
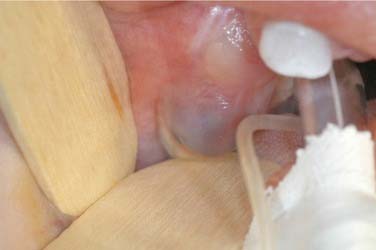
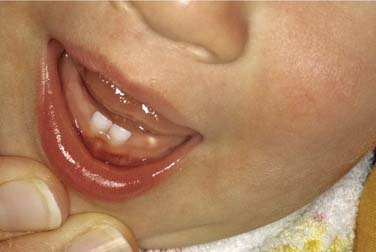
The eruption cyst is a form of dentigerous cyst that is found in the soft tissue overlying an erupting tooth. Because by definition it must be associated with an erupting tooth, eruption cysts occur only during the ages of tooth development.7 They may be seen with erupting deciduous or permanent teeth, but the majority of lesions are seen in the first decade.68 The lesion will present as a soft tissue swelling of the alveolar ridge overlying an area of age-appropriate tooth development. Some eruption cysts will have a slightly blue hue color, though the normal coral pink color of the surrounding mucosa is common (Fig. 93-8). Unlike dentigerous cysts, it is not uncommon for an eruption cyst to be associated with deciduous teeth. Eruption cysts may be seen in newborns with an incidence of 2 per 1000 births reported.69 Clinical follow-up may also serve to confirm the diagnosis because the tooth will erupt within several weeks to months through the cystic expansion.70–74
Paradental Cyst
The paradental cyst is considered by some to be a variant of the dentigerous cyst. This is because the various forms of paradental cyst all originate from the cementoenamel junction just like the dentigerous cyst. However, the paradental cysts are almost uniformly inflamed, so they are generally classified as inflammatory cysts rather than as developmental cysts. Paradental cysts occur on the buccal or distal aspect of an erupted mandibular molar. Though the mesial aspect of a mandibular tooth may rarely be involved, there have been no reported occurrences to the lingual. Craig75 reported the occasional presence of developmental enamel projections near the furcation of some teeth. This is particularly true of the subcomponent of paradental cysts known as infected buccal bifurcation cysts. How big a role these projections play in pathogenesis remains debatable.76–81 In one series the paradental cyst accounted for 3% of all odontogenic cysts.82
Odontogenic Cyst of Undetermined Origin
The problem in diagnosis comes when a cyst is not associated with the crown of a tooth, residually or with the root of a nonvital tooth. Additionally, the cyst is not consistent with a fissural cyst but is at least partially located in the alveolar process. Historically, any cyst that occurred in an area where a tooth should have developed, or where supernumerary teeth could occur, were originally called primordial cysts.83 This term was used to allude to development from the tooth primordium. Unfortunately, this was proposed in 1945 before delineation of the features defining the OKC and a large percentage of these “primordial” cysts had features of what would now be diagnosed as an OKC. When the OKC was defined in the 1950s, some pathologists had already recognized those histologic features in what they termed primordial cysts and thus they began to use the term interchangeably with OKCs. International journals, especially, came to use the term primordial cyst as a synonym of the OKC.45,84 Americans often avoided the term primordial or left the moniker of primordial cyst for those lesions without features of a keratocyst.
Definition of the Odontogenic Cyst of Undetermined Origin
An odontogenic cyst of undetermined origin is a unilocular radiolucent cyst of the jaws with histologic features of common odontogenic cysts but lacking the clinical, histologic, and radiographic features of any defined common odontogenic cyst (Fig. 93-9).
Lateral Periodontal Cyst
These cysts are thought to be derived from the dental lamina and are thus thought to retain some limited neoplastic growth potential. This limited neoplastic potential is best displayed by the associated lesion known as a botryoid odontogenic cyst. Lateral periodontal cysts are located on the lateral surface of a vital tooth.85,86 This assumes that the tooth has not been rendered nonvital by dental caries or trauma unrelated to cyst formation. The most common location is the mandibular premolar/canine area. If present in the maxilla, the lateral incisor area is the most common location. However, the lateral periodontal cyst may be seen in any area of the alveolar processes. This cyst is seen in the interproximal area between tooth roots and is usually an incidental radiographic finding.87,88 Demographically the cyst is most common in males by a 2 : 1 ratio with a peak incidence in the fifth and sixth decades.89 The gingival cyst of the adult and the botryoid odontogenic cysts are essentially subtypes of lateral periodontal cysts. Other cysts, especially the OKC, odontogenic cyst of undetermined origin, and the lateralized periapical cyst, also present interproximally. Histopathologic features and tooth vitality are important diagnostic considerations to separate these lesions. All of these lesions can be separated histologically and the lateralized periradicular cyst will be associated with a nonvital tooth.
Radiographic Features
The lesion presents as a unilocular radiolucency of the alveolus that is usually well corticated. Larger lesions may result in diverged roots.90 Multilocular lesions are a special subset and are classified as botryoid odontogenic cysts (see “Botryoid Odontogenic Cyst” later).27
Microscopic Features
The cyst lining is composed of nonkeratinized simple to stratified squamous epithelium. The lining is most notable for being only a few cells in thickness (Fig. 93-10). Intermixed within this otherwise thin epithelial lining are nodular epithelial thickenings or plaques. The plaques may display somewhat whorled epithelial cell aggregates. The central cells in the aggregate may display cytoplasmic clearing. The clear cells contain glycogen, which can be digested with diastase. Scattered mucous cells may be seen in some lesions but should not be a dominant feature.90,91 The diagnosis of lateral periodontal cyst must be reserved for lesions displaying the thin epithelium described earlier. The plaquelike thickenings are helpful microscopic features but are not always found. The connective tissue wall may contain dental lamina rests, but they are not required for diagnosis.
Botryoid Odontogenic Cyst
The botryoid odontogenic cyst will usually present as a multilocular lesion and is a special variant of the lateral periodontal cyst. Botryoid refers to the fact that these lesions may appear like grapelike clusters, both histologically and usually radiographically as well.23,25,27
Radiographic Features
Radiographically the botryoid odontogenic cyst presents in the same preferred alveolar process locations as the lateral periodontal cyst. Small locules may not be seen on radiographs.23
Gingival Cyst of the Adult
Clinical Features
This lesion is essentially the soft tissue equivalent of the lateral periodontal cyst. As with the lateral periodontal cyst, it derives from dental lamina or rests of Serres.92–94 These lesions generally present as an asymptomatic bluish nodule on the facial aspect of the gingiva. The sessile elevation will be centered in the attached gingiva, though extension below the mucogingival line is possible. Unlike the lateral periodontal cyst, there may be a slightly increased incidence in women.95 The mandibular gingiva is most commonly involved and the peak incidence occurs in the fifth and sixth decades.9,96 Other odontogenic cysts may also occur in the gingiva. Histologically distinctive odontogenic cysts that are appropriately diagnosed histopathologically should not be descriptively called gingival cysts. The possibility of some cysts of the gingivae arising from traumatically implanted surface epithelium does exist. Whether it is best to call these histologically different cysts epithelial inclusion cysts is debatable.95 The term gingival cyst of the adult must be reserved for those lesions with histologically distinct microscopic criteria.
Gingival Cyst of the Newborn (Dental Lamina Cyst, Alveolar Cyst of the Newborn)
These cysts occur on the alveolar ridge of newborns. They are generally only a few millimeters in diameter and seen only in the first few months of life. Though studies are appropriately devoid of histologic sampling data, some clinical reports estimate these cysts occur in up to 50% of all newborns. The cysts are sessile and vary from normal in color to yellow or white. Similar-appearing inclusion cysts of the palatal midline (Epstein’s pearls) or at the junction of the hard and soft palates (Bohn’s nodules) are differentiated by location. The gingival cyst of the newborn is a soft tissue cyst and does not have an intrabony component (Fig. 93-11).97
Keratinizing Odontogenic Cyst (Orthokeratinizing Odontogenic Cyst, Orthokeratinizing Odontogenic Keratocyst)
Recognition of the keratinizing odontogenic cyst is a rather recent phenomenon. When the OKC was first defined, the definition did not separate these lesions. Over time several reviewers noted and separated keratinizing cysts that did not seem to otherwise conform with the microscopic criteria of the OKC.98–100 The most notable difference in these cysts was the presence of orthokeratin rather than parakeratin, though other features such as lack of tombstoning, corrugation, and hyperchromatism are more important criteria. Wright101 was the first to formally separate this group of cysts and proposed the name of OKC, orthokeratinizing variant. He noted the different histopathologic features and ably reported the lack of recurrences associated with these orthokeratinized cysts. However, the nomenclature of including the term keratocyst has created quite a bit of confusion, at least anecdotally. Historically, older texts and articles described keratinization of various odontogenic cysts and recognized the difference of these cysts from OKCs.47,100
The designation as orthokeratinized OKC (OKC, orthokeratinized variant) has evolved since first being described. Current nomenclature has evolved to avoid confusion with the “standard” OKC. It is also worth noting that some OKCs can contain orthokeratin, and the original series of “orthokeratinizing OKCs” described by Wright included 7 of 60 cysts with parakeratin.101 Thus classification based solely on the type of keratin alone is unwise.
The separation of the keratinizing odontogenic cyst is clinically important because the reported recurrence rate of the keratinizing odontogenic cyst is only 2%. The peak incidence of keratinizing odontogenic cysts is in the third, fourth, and fifth decades, with about three fourths of them being in a dentigerous cyst relationship. Other locations are possible, in various relationships to the teeth. Most cysts are asymptomatic, though pain and swelling was reported respectively in 22% and 13% of cases.98,99 The keratinizing odontogenic cyst is not associated with basal cell nevus syndrome (Gorlin-Goltz syndrome).
Radiographic Features
The cyst presents as a well-defined unilocular radiolucency with the border generally being well corticated. If it is in a dentigerous cyst position, a cyst may be large (Fig. 93-12).
Microscopic Features
The epithelial lining will most often be thin with keratinization. However, the thickness will be somewhat variable. Keratin production varies and may only occur on a portion of the cyst wall. Orthokeratin is characteristic but not pathognomonic.102 Most notable are the features that are not present. The features defining an OKC are absent.
Glandular Odontogenic Cyst (Sialo-odontogenic Cyst)
Clinical Features
Controversy encompasses this lesion and undoubtedly further delineation of features will evolve. The glandular odontogenic cyst is usually asymptomatic and involves the mandible more often than the maxilla.29,103 Large cysts may be destructive and expansile.104,105 The glandular odontogenic cyst involves adults and its pathogenesis is not yet understood.106 This cyst may be either multilocular or unilocular. The cyst is generally well defined and often well corticated.
Radiographic Features
The lesion is radiolucent and may be unilocular, but it is more commonly multilocular. The radiographic margins are well defined and usually display a sclerotic rim.107
Microscopic Features
This cyst is lined by cuboidal epithelium of varying thickness, which may display cilia. Unfortunately, separating this particular microscopic feature from “normal” (and insignificant) mucus prosoplasia is sometimes difficult. The following features are more important in establishing the diagnosis. The cyst lining will be mucicarmine positive. The mucin detected by mucicarmine collects in small pools. Characteristically there are cuboidal cells near the surface, which give a slightly papillary appearance to the lumen.103 The epithelium will also contain spheroid aggregates of cells.108,109 The presence of mucous cells in other odontogenic cysts is not diagnostic for this lesion. This lesion, in this author’s opinion, is “overcalled” and second opinion is often necessary. Standard “common odontogenic cysts” with mucus prosoplasia are often “upgraded” to glandular odontogenic cysts (Fig. 93-13). Glandular odontogenic cysts may be “upgraded” to central mucoepidermoid carcinoma. Great care to properly designate these lesions is necessary to ensure proper patient care.
Treatment
Recurrence after enucleation has been reported.103 As in any multilocular lesion, nucleation and curettage of bone is recommended. Few cases are available to draw further treatment conclusions, but the overall prognosis is good.30 Suggestions to treat this cyst aggressively appear to be misguided.
Odontogenic Keratocyst (Parakeratinizing Odontogenic Keratocyst, Keratocystic Odontogenic Tumor)
The OKC remains an enigma for the clinician and researcher, although knowledge gains in recent years have allowed for an improved understanding of this interesting lesion. The lesion was first described by Philipsen in 1956, but even 5 decades later the debate continues over the pathogenesis, behavior, treatment, and classification of this cystic neoplasm.18–20 One major dilemma is whether to classify it as a cyst or a neoplasm. The current World Health Organization (WHO) nomenclature stresses the neoplastic nature and uses the term keratocystic odontogenic tumor.
Soft tissue extension, extension into adjacent bones, and expansion with associated bony destruction have been reported. These reports have prompted clinicians to question what the most appropriate method of treatment really is. Scharfferter and colleagues110 documented increased mitotic activity within the epithelial lining of the OKC, which seems to support its neoplastic nature.
Of particular interest to the clinician is the biologic behavior of the OKC. Recurrence rates of up to 62.5% have been reported with enucleation alone. However, modern reports have a much lower recurrence rate when enucleation and careful curettage are performed. Most modern recurrence rates are less than 10%.111,112 Reasons to explain the recurrence rate include daughter or satellite cyst formation113; incomplete removal of the epithelial lining, leaving satellite cysts left behind; collagenase activity of the cyst114; dental lamina rests left in the cyst wall or overlying mucosa; prostaglandin-induced bone resorption115; and increased mitotic activity. A handful of articles have demonstrated that the OKC can proliferate within muscle, and death from intracranial extension of mandibular OKCs has been reported by Jackson and colleagues.84,116,117 Though these case reports should be remembered, the need to aggressively treat all OKCs should be resisted.
Radiographic Features
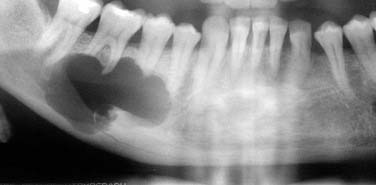
OKCs may be unilocular or multilocular, multiple, or single. OKCs with calcifications within a cyst wall have been reported, but calcification is rare and OKCs are considered radiolucent lesions. OKCs are highly variable in size (Fig. 93-14). They can appear pericoronally, periradicularly, interradicularly, apically, and even peripherally. In summary, OKCs may occur in all areas of odontogenesis and they may extend significant distances.97 The borders are well defined and often corticated.98,118 Patients involved with Gorlin syndrome are more likely to present with metachronous or synchronous cysts. CT scans may be helpful in assessing large lesions and CTs are often essential in assessing maxillary lesions.119–121
Treatment
A handful of reports with significant complications have led some clinicians to perform aggressive surgical procedures on all lesions. Debate persists about how to best manage OKC lesions in general. Treatment should center on decreasing the recurrence rate to a rate expected of the modern era and one that reduces patient morbidity. The reader is referred to other articles. After reading many treatises, the following is my opinion.111,112,122–130
If the patient has more than one cyst, the possibility of basal cell nevus syndrome is elevated. All cysts should still be examined histopathologically to confirm diagnosis. Clinical work-up to assess for syndrome is always prudent when multiple concurrent or sequential OKCs are diagnosed. Even when OKCs are in teenagers or younger patients are diagnosed with a single lesion, at least a cursory family history and gross clinical assessment for the syndrome should be performed. Some investigators have claimed good success with decompression and subsequent enucleation, while others advocate enucleation, excision of overlying mucosa, peripheral osteotomy, and chemical curettage.125,129
The use of Carnoy’s solution is controversial. Carnoy first reported use of his fixative for the study of nematodes in 1887.131 His goal was to fix the tissue and preserve nuclear detail for microscopic examination. However, fixation of the tissue intraoperatively for future microscopic exam is not really the effect that surgeons desire. Surgeons desire the chemical cauterization effect that the solution produces.
Basal Cell Nevus Syndrome (Gorlin Syndrome, Gorlin-Goltz Syndrome, Nevoid Basal Cell Carcinoma Syndrome)
This complex syndrome is called by many names, and the nomenclature often depends on what portions of the syndrome (e.g., bifid ribs, OKC) are present. In the Gorlin and colleagues132–134 description, no fewer than 37 anomalies have been associated with this syndrome. The reader is referred to the original sources.
OKCs are often the first diagnosis of the syndrome to present in childhood and often aid in establishing a diagnosis. This is especially true in cases in which there are new mutations with no prior familial history.135 Up to 40% of cases may be new mutations, though genetic testing is advancing and there may be large variations in expression in this autosomal dominant disease.134 One patient’s first cyst occurred in the seventh decade. For this reason the syndrome should not be ruled out even in a middle-aged patient, and family history and phenotype may help in all age groups.