CHAPTER 103 Neoplasms of the Hypopharynx and Cervical Esophagus
Anatomy of the Hypopharynx and Cervical Esophagus
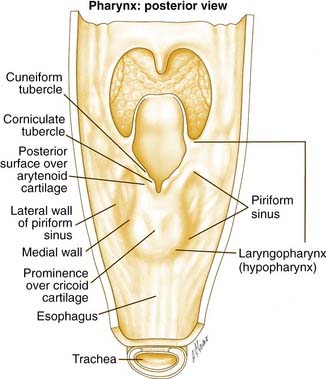
The hypopharynx is the region of the pharynx that extends from the oropharynx superiorly to the cervical esophagus inferiorly. The cervical esophagus is that portion of the esophagus that extends to the thoracic inlet. The superior extent of the hypopharynx is approximately at the level of the hyoid bone or at the level of the pharyngoepiglottic folds. Inferiorly, the hypopharynx tapers to the esophageal introitus at the cricopharyngeus muscle. It is bordered anteriorly by the larynx and posteriorly by the retropharyngeal space. It is subdivided into three regions: the pyriform sinuses, the postcricoid region, and the posterior pharyngeal wall (Fig. 103-1).
The pyriform sinuses (one on each side) are composed of anterior, medial, and lateral walls that form an inverted pyramid with the base of the pyramid at the level of the pharyngoepiglottic fold and the apex extending to just below the cricoid cartilage. This lower region is lateral to the aryepiglottic folds and medial to the thyroid lamina. Because of this close relationship, tumors of the hypopharynx often extend to invade the larynx.1 The medial pyriform mucosa forms the posterior wall of the paraglottic space and is separated from the endolarynx by the aryepiglottic folds and the lateral cricoarytenoid muscles. Hypopharyngeal tumors extending medially from the medial wall of the pyriform sinus can, therefore, invade into the larynx.
The aryepiglottic folds separate the endolarynx from the medial wall of the pyriform sinus bilaterally and form what has been termed the “marginal area.” Although the aryepiglottic folds are actually part of the supraglottic larynx, tumors arising at this site behave aggressively like hypopharyngeal cancers and not supraglottic carcinomas.2
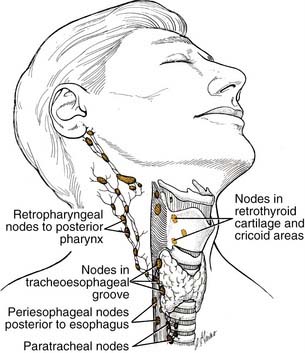
Lymphatic drainage from the pyriform sinuses passes through the thyrohyoid membrane primarily to the jugulodigastric lymph node and to the midjugular and spinal accessory chains (Fig. 103-2). Lymphatic vessels from the inferior portion of the hypopharynx and from the postcricoid region also drain to the paratracheal and paraesophageal nodes and to nodes in the supraclavicular fossa. Lymphatic drainage from the posterior hypopharyngeal wall is to the retropharyngeal nodes and to the midjugular chain. The retropharyngeal nodes are divided into a medial and lateral group. Lateral retropharyngeal nodes, also known as the nodes of Rouvière, are present at the level of the skull base.
Incidence
The incidence of cancer of the hypopharynx varies depending on worldwide geographic location. In addition, the incidence of cancer arising from specific subsites within the hypopharynx may also vary by geographic location. In the United States, three large database reviews shed light on the epidemiology of hypopharyngeal carcinoma. Neoplasms specific to the cervical esophagus are not mentioned in the first two of these three studies. Canto and Devesa3 analyzed data from 1975 to 1988 in nine registries of the Surveillance, Epidemiology, and End Results (SEER) database. Although these data are not a random sample because each registry is from a specific geographic location, they represent approximately 10% of the U.S. population. The rates of cancer per 100,000 person-years vary with race and sex. It is also not clear from this study why the sites are listed separately as hypopharynx and pyriform. Whether there is overlap in these numbers is not stated in this study. For white males the rate is 0.4 and 0.9 for the hypopharynx and pyriform sinus, respectively. For African American males the rate is 0.8 and 2.3 for the hypopharynx and pyriform sinus, respectively. For white females the rate is 0.2 for both the hypopharynx and pyriform sinus. For African American females the rate is 0.2 and 0.5 for the hypopharynx and pyriform sinus, respectively.
Hoffman and colleagues4 conducted a second major database review of the National Cancer Data Base (NCDB) report, which is a comprehensive hospital-based registry of all cancer cases encountered. The data cover the years 1985 to 1994 and reveal that hypopharyngeal malignancy comprised 4.3% of all head and neck cancers, which included salivary gland and thyroid tumors in this study. Ninety-five percent of the hypopharyngeal tumors were squamous cell carcinomas with the remainder being adenocarcinomas, lymphoma, and other pathologies. Stage III and IV tumors comprised 77.3% of cancers, demonstrating the advanced stage of hypopharyngeal tumors at presentation. The survival data in this study were noteworthy in that hypopharyngeal cancers had the worst 5-year survival compared with any other primary site in the head and neck. The 5-year disease-specific survival was 31.4%.
Finally, Davies and Welch5 analyzed the updated SEER database for the years 1999 to 2001 and found an average of 75,000 new cases of head and neck cancer. Interestingly, compared with the average 1975 statistics, this study found that incidence and mortality rates for hypopharyngeal and cervical esophageal cancer were decreasing. The one caveat to consider in saying that this trend was a definitive finding is that the rates of these tumors were below 1 per 100,000. Notwithstanding this statistic, this study shows a continued significant incidence of these deadly cancers within all head and neck squamous cell carcinomas.
The statistics outside of the United States reveal similar epidemiologic data. Wahlberg and colleagues6 analyzed the comprehensive Swedish databases, covering 1960 to 1989, and found rates of 1.22/100,000 for men and 0.45/100,000 for women, similar to rates described by Canto and Devesa.3 During this time the rates for men increased, but the rates for women decreased. The authors suggest that this may reflect a decrease in Plummer-Vinson syndrome, the sideropenic anemia common in Northern Sweden that predisposes to development of postcricoid carcinoma.
The data on cervical esophageal carcinoma are much more limited. Esophageal carcinoma causes approximately 12,000 deaths per year in the United States.7,8 In one series, cervical esophageal carcinoma represented only 5.3% (9/168 patients) of all esophageal cancers.9 Given this low frequency, the data are limited on the incidence and optimal treatment of this cancer. Additionally, these tumors are often included in statistics regarding all esophageal cancers.
Clinical Evaluation
PATIENT SYMPTOMS
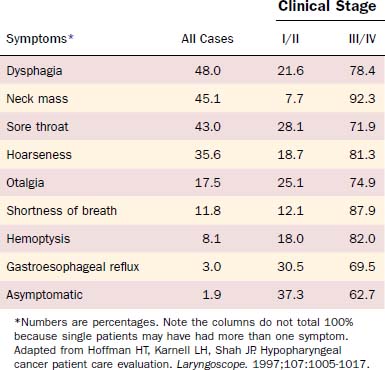
Patients with lesions in the hypopharynx and cervical esophagus usually present at an advanced stage of disease. Lesions in this area can grow unabated to a size larger than most other head and neck sites because the anatomic boundaries of neighboring structures are not as limiting as for other sites (e.g., the larynx). Thus a disturbance of function is not seen until the disease is advanced. Because this area also has a rich lymphatic drainage, a neck mass is not an uncommon finding, especially in the advanced-stage disease. Hoffman and colleagues10 analyzed presenting symptoms in 2939 cases from the Patient Care Evaluation study of the American College of Surgeons Commission on Cancer (Table 103-1). For stage I/II disease, gastroesophageal reflux, a common and nonspecific symptom, was most common (30.5%) followed closely by sore throat (28.1%). Importantly, 37.3% of patients with early-stage disease were asymptomatic at presentation. For stage III/IV disease, the most common symptom was a neck mass (92.3%) followed by shortness of breath (87.9%). Dysphagia occurred in 21.6% of early-stage and 78.4% of advanced-stage lesions. Additionally, referred otalgia was present in 25.1% of early-stage and 74.9% of advanced-stage lesions.
PHYSICAL FINDINGS
The overall appearance of these patients will likely reveal a malnourished individual. A complete examination of the head and neck should be performed with a focus on the mucosa of the upper aerodigestive tract to evaluate the extent of the primary tumor and to assess for second primaries. Synchronous tumors were present in a significant proportion of asymptomatic stage I/II patients in the Hoffman and colleagues’ study.10 In the same study a significant portion of patients presented with a neck mass; thus clinical staging of the neck to define the nodal stage is also critical. In the clinic, fiberoptic laryngoscopy should be used to evaluate the airway because impending airway distress is a possibility with advanced tumors. Immobility of the vocal cord(s) may reveal whether the larynx has been invaded.
In defining the options for managing a patient with hypopharyngeal and cervical esophageal cancer, the following must be addressed. The extent of tumor and lymph node involvement must be defined by office examination, operative endoscopy, and imaging. For tumors involving the posterior pharyngeal wall, invasion of the prevertebral fascia needs to be assessed clinically and radiographically to ensure an adequate surgical margin. Because distant metastases in certain series are the highest for hypopharyngeal cancers,11 a metastatic workup by imaging and laboratory testing should also be completed. Comorbidities of the patient are also a significant contributing factor in managing these patients. For example, if a conservation surgical procedure is planned, the pulmonary function status of the patient needs to be evaluated because these patients are at significant risk for aspiration and must have adequate pulmonary reserve.
Imaging
In the treatment of hypopharyngeal malignancies, the role of imaging involves the pretreatment evaluation of the extent of the primary tumor and possible metastases, as well as the evaluation of the patient following treatment. Hypopharyngeal tumors have a propensity for submucosal spread that may be undetectable on clinical or radiographic examination.12 Causes for surgical failure include submucosal extension, involvement of the thyroid gland, and metastasis to paratracheal and upper mediastinal lymph nodes.13 Thus imaging is essential to assess tumor extent or recurrence.
Although the barium swallow has been historically used to identify malignancies in this region,14 its use is now more limited. Some centers continue to use the swallow to evaluate for second primaries in the esophagus. In the case of a cervical esophageal cancer where an endoscope cannot be passed distally, the barium swallow can be used for imaging the distal extent of disease. Another use of the barium swallow study is in the postoperative setting to examine the deglutition process or to identify anastomotic problems such as stricture or fistula.
The primary imaging modality for pretreatment evaluation of the hypopharynx is cross-sectional imaging with computed tomography (CT) or magnetic resonance (MRI). In multiple studies examining the impact of cross-sectional imaging on the staging of hypopharyngeal and/or esophageal cancer, the clinical tumor stage was upstaged in up to 90% of patients.15 Accuracy of tumor staging, determined by comparison to pathologic findings, is 58% for clinical examination, 80% for CT, and 85% for MRI.16 CT is often preferred to MRI to assess cartilage invasion; however, an experienced radiologist looking for an increased T2 and decreased T1 signal intensity on MRI, which indicates cartilage involvement, can achieve a high degree of sensitivity (89% to 100%).15 The specificity of MRI for cartilage invasion, however, is inferior to CT (62% vs. 84%).17 A wide variability in specificity reflects the variability in ossification and the degree of inflammation, edema, and/or fibrosis at the tumor site. The specificity for cartilage invasion is lowest for the thyroid cartilage (57%), highest for the arytenoid cartilages (95%), and intermediate for the cricoid cartilage (87%).16 Figure 103-3 demonstrates a T4 left pyriform sinus squamous cell cancer that has invaded the arytenoid cartilage and the thyroid lamina. The incidence of a second neoplasm of the aerodigestive tract, either synchronous or metachronous to an index tumor of the hypopharynx, has been reported to be 16% to 18%11; therefore a thorough radiographic evaluation of the entire aerodigestive tract including the esophagus is necessary.18,19
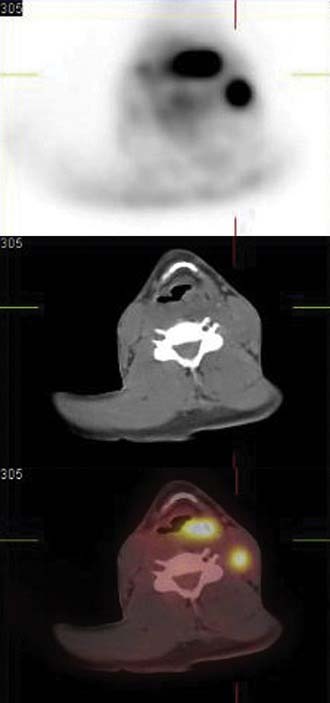
There are no available data supporting the routine use of imaging with CT or MRI in the post-treatment period to follow disease-free status. Typically patient symptoms or clinical endoscopic findings will dictate the utilization of specific imaging. The role of positron emission tomography (PET) has been explored as a modality to assist in the detection of locoregional recurrence and/or persistent disease. Several studies have reported on the improved sensitivity of PET over MRI in the evaluation of response to therapy.20,21 Although this modality appears to have an increased sensitivity (86%) for recurrence over CT/MRI (57%), there is a lower measured specificity with PET (75% vs. 92% for CT/MRI).21 An improvement in specificity with PET may be achieved as newer tracers are studied. For example, Chesnay and colleagues22 described the successful use of 11C-methionine to follow post-chemotherapy tumor response. The combination of PET and CT scans (PET/CT) provides significant advantages due to the ability to correlate fluorodeoxyglucose (FDG) avid lesions with anatomic findings and is increasingly used in all phases of hypopharyngeal tumor management.23 Figure 103-4 illustrates the use of PET/CT in a patient with an advanced left pyriform sinus squamous cell carcinoma with cervical lymphadenopathy. The fused scan not only shows the primary FDG avid lesion but also highlights the left cervical lymph node with obvious tumor involvement. Thus the combination of CT, MRI, and PET scanning can be used in all phases of the management of hypopharyngeal and cervical esophageal carcinoma.
Pathology
Although the vast majority (95% in the Hoffman and colleagues’ study10) of neoplasms of the hypopharynx and cervical esophagus are squamous cell carcinomas, other rare pathologic entities have been described (Fig. 103-5). In addition, variants of squamous cell carcinoma have also been described.
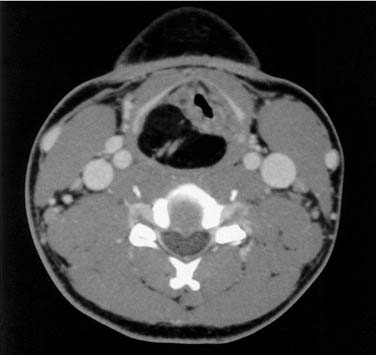
Figure 103-5. Axial computed tomography scan of a patient with a hypopharyngeal lipoma displacing the larynx.
Unusual malignant lesions include lymphomas, which can present in the hypopharynx as the primary site or can secondarily involve the hypopharynx and cervical esophagus after systemic presentation. Some of the subtypes include angiocentric T cell lymphomas, non-Hodgkin’s extranodal lymphomas, and mucosa-associated lymphoid tissue (MALT) lymphomas.24 A high index of suspicion for lymphoma should be maintained in patients with AIDS and a hypopharyngeal mass.25 Lymphomas need to be extensively staged and, depending on subtype, are treated with a combination of radiation and/or chemotherapy. The neuroendocrine tumors of the larynx and hypopharynx are a diverse subset of small cell tumors.26 Depending on the histologic subtype, treatment varies from chemotherapy and radiation to surgical therapy.
Adenocarcinomas are another rare malignant entity seen in the hypopharynx and cervical esophagus. These lesions may initiate in the minor salivary glands within the hypopharynx or ectopic gastric mucosa in the cervical esophagus. The outgrowth of adenocarcinomas from ectopic gastric mucosa is a rare event.27 Thyroid malignancies may secondarily involve the hypopharynx or cervical esophagus via direct invasion.28 Other extremely rare malignancies include sarcomas29 such as liposarcomas,30 angiosarcomas,31 and synovial sarcomas.32
Of the squamous cell cancers, there are three histologic subtypes that deserve special attention. First is basaloid squamous cell carcinoma,33 which is a bimorphic variant of squamous cell carcinoma that has distinct pathologic features. Clinically, these lesions appear primarily in the supraglottis, pyriform sinus, and tongue base and have a distinctly more aggressive clinical course.34 Second, lymphoepitheliomas are the hypopharyngeal counterpart of nasopharyngeal cancers.35,36 These tumors are treated with radiation and chemotherapy, similar to nasopharyngeal carcinoma. Finally, the adenosquamous carcinoma is another rare variant, which also behaves clinically in an aggressive manner.37
Location of Tumors and Patterns of Spread
An understanding of the site of initiation and patterns of spread of hypopharyngeal carcinoma is critical in the management of these tumors. Substantial data exist regarding the origin of the carcinoma within specific subsites of the hypopharynx. Kirchner1 described experience at the Yale University hospitals in managing hypopharyngeal carcinoma. In their patient population, 152 (86%) carcinomas arose in the pyriform sinus, 17 (10%) were in the posterior pharyngeal wall, and 8 (4%) were located in the postcricoid region. In the series of Carpenter and colleagues,38 117 (72%) carcinomas arose in the pyriform sinus, 37 (23%) in the posterior pharyngeal wall, and 8 (5%) were located in the postcricoid region. Saleh and colleagues39 described their series of patients from Egypt and found that postcricoid cancers were the majority of presenting lesions (50.1%) followed by pyriform sinus (26.5%) and finally posterior pharyngeal wall tumors (23.4%). A European cancer registry review40 demonstrated highest rates of pyriform sinus cancer in France (78%) and lowest rates in Sweden (5%). Although difficult to identify the precise reason for these subsite differences, one explanation is the possibility that an assignment of the exact origin of these tumors may be difficult except in the earliest stages.
Regarding the histologic spread of tumor, Kirchner1 performed whole organ serial sections on 51 surgically resected pyriform sinus carcinoma specimens and described several interesting findings. Tumors were able to infiltrate the larynx and behave as transglottic cancers. They may invade the lateral and posterior pharyngeal walls and they may spread into the supraglottis and base of tongue. Invasion inferiorly into the cervical esophagus was not a common finding in these specimens. Twenty-two specimens invaded the thyroid cartilage at its posterior border, and all were found to have lateral pharyngeal wall involvement. Kirchner demonstrated in this study that the exclusion of tumors with pyriform apex involvement from conservation surgery was indeed appropriate because all of these tumors had laryngeal framework invasion.
A distinct feature of hypopharyngeal carcinoma that was noted early in the surgical treatment of this disease was its tendency to spread in a submucosal fashion.13,41 This is an often-cited feature of hypopharyngeal cancer, especially in discussing its poor prognosis. Ho and colleagues12 performed a detailed analysis of submucosal extension by serial sectioning of 57 specimens of hypopharyngeal cancer. Three classes of submucosal extension were identified: type 1 was a direct submucosal extension visible on gross inspection as elevated mucosa, type 2 was direct submucosal extension that was only visible on histologic examination, and type 3 was a true “skip” lesion with no connection to the primary. Only 1 of 57 specimens had type 3 extension; however, 33 (58%) patients did have submucosal extension of some type. In contradiction to the findings of Harrison,13 significant inferior extension toward the cervical esophagus was observed. Patients treated with preoperative radiotherapy had significantly increased amounts of type 2 extension. Interestingly, in comparing overall 5-year survival, no differences were found between the group that had submucosal extension and the one that did not. Additionally, submucosal extension was not associated with increased locoregional recurrences. Thus although it is true that submucosal extension occurs in a significant number of patients with hypopharyngeal cancer, most of it is detectable clinically and a poorer prognosis is not seen in patients with submucosal extension of tumor.
Included in the data from serial sectioning by Ho and colleagues and Wei12,42 was the finding that submucosal extension was greatest in the inferior direction, followed by lateral extension, and finally superior extension. On the basis of these data, they recommended that resection margins should be 3 cm inferiorly, 2 cm laterally, and 1.5 cm superiorly in patients who have not received previous radiation. For those with previous radiation, these margins were recommended to be 4 cm, 3 cm, and 2 cm, respectively. The deep margin was recommended to be greater than 1 mm in all patients.43
Etiology and Biology
The causal relationships among alcohol and tobacco intake, genetic predisposition, diet, and socioeconomic conditions in the development of squamous cell cancers of the head and neck apply as well to hypopharyngeal cancer.44–46 In examining these factors, two specific components appear to be more closely associated with hypopharyngeal cancer. The first is alcohol intake, which appears to be more common in patients with hypopharyngeal cancer, at least when compared with laryngeal cancer.10,47–49 What the specific effect of alcohol may be in hypopharyngeal cancer development is unclear. Alcohol may have direct carcinogenic effects, may be more of a promoter of tobacco’s carcinogenic effects, or may have both functions. Two recent studies found that specific chewing tobacco products or exposure to wood smoke were associated with hypopharyngeal cancers in India.50,51
A condition specifically associated with postcricoid carcinoma is the Plummer-Vinson or Paterson-Brown-Kelly syndrome, which primarily affects women (85% of the cases).52 This syndrome represents the combination of dysphagia, iron deficiency anemia, and hypopharyngeal and esophageal webs. It is hypothesized that chronic irritation results in the hypopharyngeal webs that then progress to carcinoma. The syndrome is also geographically biased in that patients are predominantly located in the United States, Wales, and Sweden. The etiology is believed to be due to nutritional deficiency and since these issues have been addressed, the incidence of postcricoid carcinomas has declined in Sweden.6
Staging
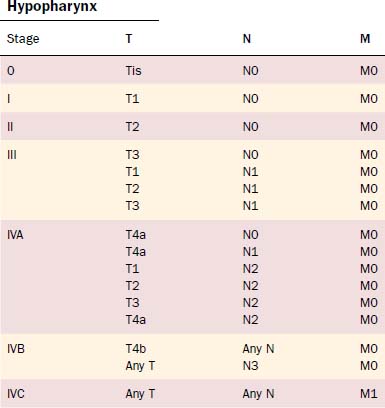
The TNM system continues to be the standard by which the morphologic extent of the tumor is described. The American Joint Commission on Cancer updated the TNM staging in 2002, and the relevant hypopharyngeal and cervical esophageal system is listed in Tables 103-2 and 103-3.53 The TNM system’s major advantages are that it allows for comparing end results, communicating about patients, determining prognosis, and selecting treatments.54 However, it is also acknowledged to have deficiencies due to inconsistencies; inaccuracies; observer variability; problems with various T, N, and M classification criteria; and exclusion of host factors. Piccirillo and colleagues from our institution have examined the role of host factors including patient symptom severity and comorbidities on outcomes of treatment. Piccirillo and colleagues55,56 demonstrated that comorbidities are an independent prognostic factor in outcomes of patients with head and neck cancer. The most recent AJCC Cancer Staging Manual53 recommends comorbidity assessment but does not yet incorporate these factors into staging.
Table 103-2 Tumor and Lymph Node Staging
| Primary Tumor (T) | |
| Hypopharynx | |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Tumor limited to one subsite of hypopharynx and ≤ 2 cm in greatest dimension |
| T2 | Tumor invades more than one subsite of hypopharynx or an adjacent site, or measures > 2 cm but not > 4 cm in greatest dimension without fixation of hemilarynx |
| T3 | Tumor > 4 cm in greatest dimension or with fixation of hemilarynx |
| T4a | Tumor invades thyroid/cricoid cartilage, hyoid bone, thyroid gland, esophagus, or central compartment soft tissue (including prelaryngeal strap muscles and subcutaneous fat) |
| T4b | Tumor invades prevertebral fascia, encases carotid artery, or involves mediastinal structures |
| Cervical Esophagus | |
| TX | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ |
| T1 | Tumor invades lamina propria or submucosa |
| T2 | Tumor invades muscularis propria |
| T3 | Tumor invades adventitia |
| T4 | Tumor invades adjacent structures |
| Regional Lymph Nodes (N) | |
| Hypopharynx | |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Metastasis in a single ipsilateral lymph node, ≤ 3 cm in greatest dimension |
| N2a | Metastasis in a single ipsilateral lymph node > 3 cm but not > 6 cm in greatest dimension |
| N2b | Metastasis in multiple ipsilateral lymph nodes, none > 6 cm in greatest dimension |
| N2c | Metastasis in bilateral or contralateral lymph nodes, none > 6 cm in greatest dimension |
| N3 | Metastasis in a lymph node > 6 cm in greatest dimension |
| Cervical Esophagus | |
| NX | Regional lymph nodes cannot be assessed |
| N1 | No regional lymph node metastasis |
| N2 | Regional lymph node metastasis |
| Distant Metastasis (M) | |
| MX | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis present |
Therapeutic Modalities—Surgical Options Including Techniques
Cheever57 first described the lateral pharyngotomy in combination with a mandibulotomy approach in 1878. Resection of a large tonsil tumor and a node plucking were performed; however, the patient recurred with local and regional metastases. With no anesthesia, attempts at control of the local recurrence were made with 18 applications of white-hot irons. Not surprisingly, the patient’s tumor continued to grow. Other highlights in the development of hypopharyngeal cancer treatment include Sebileau’s (1904) description of the lateral retrothyroid pharyngectomy; Trotter’s (1913) development of the lateral pharyngectomy; the supracricoid hemilaryngopharyngectomy (SCHLP) described by Andre, Pinel, and Laccourreye (1962); and Ogura’s (1965) development of the extended supraglottic laryngectomy. Many other surgeons contributed to the development of other techniques including Orton, Alonso, and Harrison.
The available surgical options can be divided among the organ-preservation procedures and more radical operations (Table 103-4). The conservation surgical procedures are limited to T1, T2, or highly selected T3 lesions, except for the endoscopic approach used by Steiner and colleagues.58 The more radical operations typically require extensive reconstruction of the alimentary tract. The options and the algorithm for deciding on a specific reconstruction are discussed in a separate section.
| Procedure | T Stage | Reconstruction |
|---|---|---|
| Partial pharyngectomy | T1, T2 | Primary closure |
| Partial laryngopharyngectomy | T1, T2, T3 | Regional or free flap |
| Supracricoid hemilaryngectomy | T1, T2, T3 | Primary closure |
| Endoscopic CO2 laser resection | T1, T2 (possible T3, T4) | Secondary intention |
| Total laryngectomy with partial/total pharyngectomy | T3, T4 | Primary closure vs. regional or free flap |
| Total pharyngo-laryngo-esophagectomy | T4 | Gastric pull-up |
PARTIAL PHARYNGECTOMY
A tumor that more extensively involves the lateral hypopharyngeal wall needs to be addressed by wider margin. This can be accomplished with a lateral transthyroid pharyngotomy (Fig. 103-6) by a wider resection that involves the posterior portions of the thyroid cartilage and the hyoid bone.59 In this variation, instead of approaching the tumor through the pyriform, an entry through the vallecula is chosen. Once the neck dissection is completed, a medial and inferiorly based perichondrial flap is elevated off the thyroid cartilage. The hyoid bone is exposed from lesser cornu extending laterally. The hyoid bone is then cut through the lesser cornu, and cartilage cuts are made in a vertical fashion at the junction of the anterior two thirds and posterior one third of the thyroid cartilage. The hypopharynx is then visualized by entering through the vallecula. A blunt retractor placed into the vallecula assists entry. The incision is then carried laterally and inferiorly through the cartilage cuts. As these cuts are made, visualization through the initial incision ensures a safe margin around the tumor. The lesion is reflected laterally and the other required circumferential cuts are completed. Frozen sections for control of the margins are obtained. Reapproximating the mucosa and suturing the perichondrial flap as a second layer closes the defect.
With both these approaches, complications include pharyngocutaneous fistula with wound breakdown and dysphagia. Fistula problems are addressed by attempting to decrease or divert salivary contamination, meticulous wound care, and the possibility of secondary flap closure. A cricopharyngeal myotomy performed intraoperatively is one prophylactic measure to address possible dysphagia, although one study has shown no benefit of this maneuver in head and neck cancer patients.60
The anterior transhyoid pharyngotomy (Fig. 103-7) is another approach for lesions that primarily involve the posterior pharyngeal wall. Thus T1 or T2 lesions are good candidates for this approach. A tracheotomy is first performed. Again, a collar incision is used to identify the hyoid bone. The hyoid may be resected or the approach may be above or below the hyoid. The vallecula is identified, and sharp dissection through this mucosa with subsequent tongue base retraction provides access to the posterior hypopharyngeal wall. The tumor is removed with the prevertebral fascia as the deep margin. After all margins are assessed by frozen section, a split-thickness skin graft can be used to cover the defect. The wound is closed by reapproximating the vallecular mucosa. The primary limitation to this procedure is that there is limited visualization of the full hypopharynx. Thus choosing the appropriate tumor for this approach is critical. Complications of this approach include pharyngocutaneous fistula and dysphagia.
PARTIAL LARYNGOPHARYNGECTOMY
This operation combines the classic hemilaryngectomy operation with a partial pharyngectomy.61 In certain cases the laryngeal remnant is still able to maintain speech, swallowing, and airway protection. The tumor must fit strict criteria including involvement of the medial pyriform sinus. The lesion may be a marginal aryepiglottic fold tumor. Extension into the base of tongue, lateral pyriform sinus, and vallecula can potentially be included in the resected specimen. Contraindications include (1) involvement of the pyriform apex, (2) extension to the postcricoid region, (3) ipsilateral true vocal cord paralysis, or (4) encroachment into the cricopharyngeus.
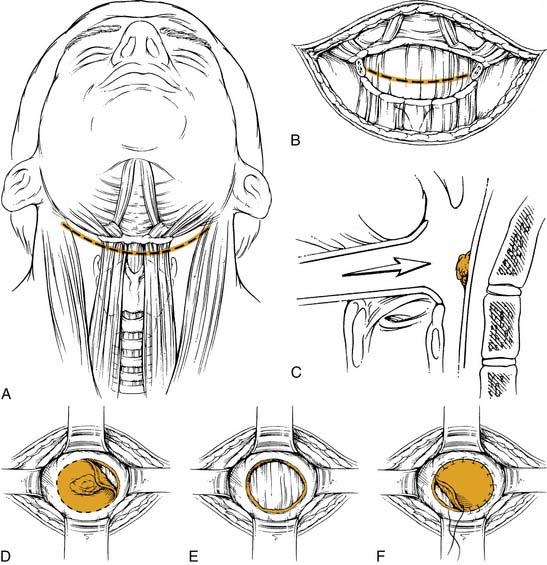
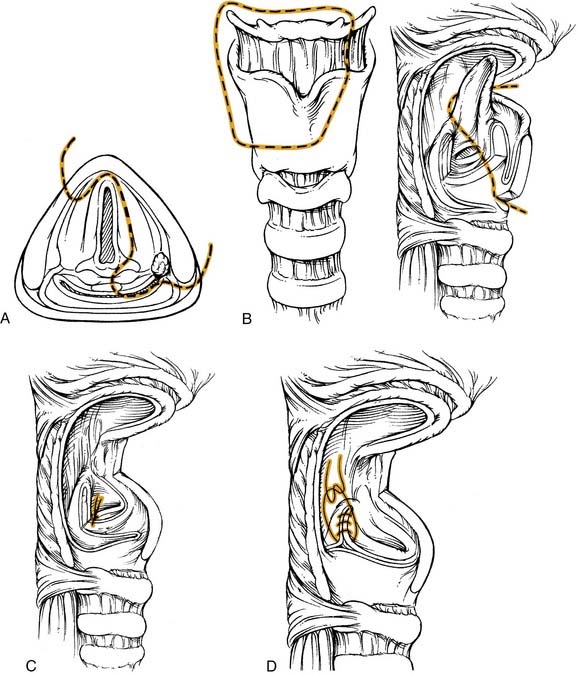
The procedure (Fig. 103-8) is begun after the completion of the tracheotomy and the neck dissection(s). For a lesion involving the medial wall of the pyriform sinus, minimal extension onto the epiglottis, and encroachment onto the arytenoid, the final resected specimen will contain the tumor on the medial wall, a portion of the epiglottis, the ipsilateral arytenoid, the ipsilateral half of the hyoid bone and the superior two thirds of the ipsilateral thyroid cartilage. After transection of the suprahyoid musculature, a superiorly based perichondrial flap is elevated off the thyroid cartilage. The thyroid cartilage is cut in the midline in an incision extending approximately two thirds of its height. This cut is then extended horizontally to the posterior border of the cartilage. The ipsilateral hyoid is freed and the vallecula is entered. Extension of this incision along with visualization of the epiglottis allows better exposure. At this point the epiglottis is grasped and retracted, providing visualization of the tumor. The epiglottis is then divided with a safe margin around the tumor and this cut is brought to, but does not include, the anterior commissure. One blade of the scissors is placed into the ventricle, and the other blade approximates the horizontal thyroid cartilage cut. This incision is made to the arytenoid, which is spared only if there is no involvement; however, it has to be sacrificed with tumor involvement. The vallecular incision is extended inferiorly with a good margin around the tumor. This is where a portion of the lateral pyriform/pharyngeal wall can be included in the resection if involved with tumor. The medial and lateral cuts meet at the arytenoid. If tumor involves more of the supraglottic area, a standard supraglottic laryngectomy can be performed to resect the entire epiglottis, pre-epiglottic space, and/or a portion of the base of tongue.
SUPRACRICOID HEMILARYNGOPHARYNGECTOMY
The major proponent of this technique is the French group of Laccourreye.62–65 This procedure extends the partial laryngopharyngectomy procedure to include the entire ipsilateral supracricoid hemilarynx along with the pyriform sinus. Thus contraindications to this approach include involvement of the pyriform apex, ipsilateral cord fixation, postcricoid involvement, or posterior pharyngeal wall invasion.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree