CHAPTER 192 Microtia Reconstruction
Microtia occurs with an overall frequency of approximately 1 to 3 cases per 10,000 population,1 although certain ethnic groups, such as Navaho Indians in Mexico and the Japanese, may have higher incidence of microtia.2 Microtia deformities appear to be more common on the right side and affect boys more often than girls at roughly a 2.5 : 1 ratio; unilateral cases outnumber bilateral cases by 4 : 1. Surgical management of microtia remains one of the greatest reconstructive challenges for an otolaryngologist.3–6 Auricular reconstruction of the microtia deformity is a complex and labor-intensive process that requires careful preoperative planning, surgical skill, and artistry. This chapter provides a basic overview of clinical issues in children with microtia and reconstruction with autologus rib cartilage. Discussion of aural atresia is presented elsewhere.
Embryology
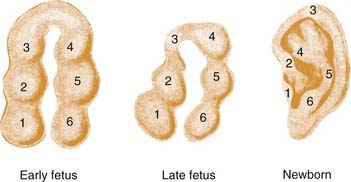
The inner ear begins as the otic placode at approximately the third week of gestation, and it then forms into the otic pit and otic cyst. The external ear begins to form in the fifth week of gestation. The auricle begins as six small buds of mesenchyme, classically known as the hillocks of His, surrounding the dorsal end of the first branchial cleft.7–10 On either side of the first branchial cleft are the first (mandibular) and second (hyoid) branchial arches. Hillocks 1 through 3 arise from the first branchial arch, and hillocks 4 through 6 arise from the second branchial arch. Conventional teaching holds that the first hillock forms the tragus, the second and third form the helix, the fourth and fifth form the antihelix, and the sixth forms the antitragus. The majority of the central ear is derived from hillocks 4 and 5, and the lobule appears to be one of the last parts of the ear to develop (Fig. 192-1). It has been estimated that the hyoid arch forms about 85% of the auricle.5 The exact embryology of each of these hillocks remains unclear. The position of the auricular complex begins at the anterior neck region. As the mandible develops during gestational weeks 8 through 12, the auricular complex moves in dorsal and cephalic directions. The etiology of microtia remains unknown in most cases, although the inciting event that arrests development of the ear must occur during the first trimester, and may be related to abnormal neural crest cell migration.
Known teratogens, such as the retinoic acid derivative isotretinoin (Accutane; Roche, Nutley, New Jersey), can produce forms of microtia in conjunction with craniofacial, cardiac, thymic, and CNS anomalies, a condition commonly known as retinoic embryopathy.11 Thalidomide and mycophenolate mofetil also have been reported to be associated with microtia.12,13 Microtia is associated with other anomalies about 50% of the time. Microtia is most commonly associated with other regional anomalies related to the first and second branchial arches, i.e., facioauriculovertebral syndrome. Within the facioauriculovertebral spectrum are malformations and syndromes that affect the first and second branchial arch derivatives, such as Goldenhar’s syndrome (preauricular nodes, epibulbar dermoids, mandibular hypoplasia), hemifacial microsomia,14 and oculoauricular vertebral dysplasia (microtia, cervical spine anomalies, and epibulbar dermoids).15,16 It may be that Goldenhar’s syndrome, hemifacial microsomia, oculoauriculovertebral dysplasia, and microtia are simply variants of the same entity.4 Microtia also may be seen in syndromes such as Treacher Collins syndrome, which has an autosomal dominant inheritance pattern.
Ear Anatomy
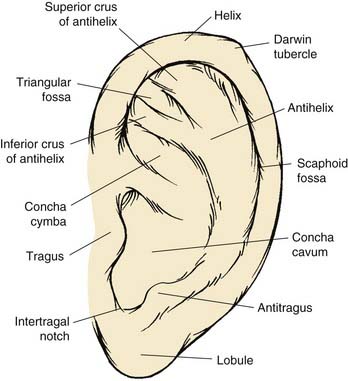
The anatomic contour of the normal ear must first be reviewed in order to understand the nature and extent of microtic ear deformities (Fig. 192-2). The auricle is an aesthetic sculpture of complex convexities, concavities, and curved lines such as the helix and the antihelix, which are smooth and uninterrupted. The elastic cartilage framework of the auricle is usually pliable yet structurally strong and extraordinarily resistant to trauma. The skin of the ear is fibrofatty and loose over the lobule and the margin of the helix but thin and fixed over the remaining cartilage framework.
Normal adult ear height ranges from 5.5 to 6.5 cm, which is typically attained by 13 years of age in boys and 12 years of age in girls. The ear goes through rapid growth between 2 and 3 years of age and reaches 95% of its adult size by about age 5. Thereafter it grows at a modest rate until it reaches its adult size during early teenage years. The horizontal width of the ear is achieved at a much earlier age. Protrusion of the ear from the surface of the mastoid should be 1.5 to 2.0 cm, thereby creating an angle between 15 and 20 degrees.17 The superior margin of the helix is at the level of the tail of the eyebrow in 85% of people. Because the tail of the eyebrow is also quite variable, the level of the upper eyelid also may be used as a landmark if the patient has bilateral microtia.18 The ear inclination is the angle formed by the vertical axis of the face and the longitudinal axis of the ear with the patient in the Frankfort horizontal position. This is approximately 24.8 degrees, with a standard deviation of 6.2 degrees.19 Therefore, the longitudinal axis of the ear is not quite parallel with the dorsum of the nose (as is commonly thought), because the ear is in a slightly more vertical orientation.
Microtia
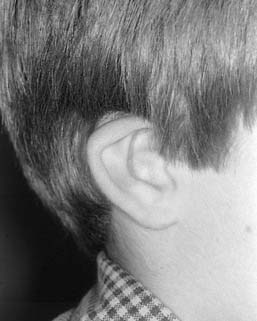
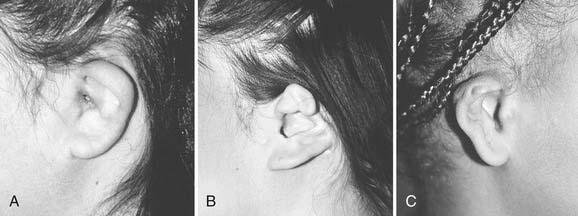
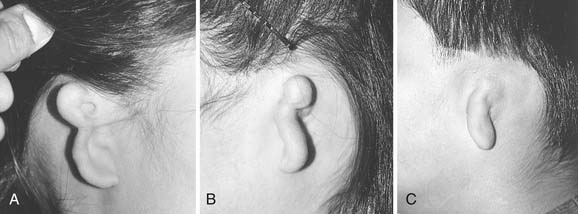
Microtia deformities come in various shapes and sizes, and unfortunately, there is currently no universal, precise, and accurate classification scheme that is clinically useful. Classification schemes are further complicated if the status of the external auditory canal (EAC) and middle ear contents and the presence or absence of an associated syndrome are included.5,20 Classification usually is simplified by evaluating the external auricular deformities alone. Perhaps dysplasia is a better term than microtia to use in describing ear deformities. Dysplasia deformities typically are divided into three broad and simple categories that have a great deal of overlap.21 Type I (Fig. 192-3) is used to designate ears with mild deformity. All major structures are present to some degree, and reconstruction does not require additional tissue. Type II (atypical microtia) dysplasia (Fig. 192-4A to C) includes ears that have all major structures present to some degree. There is a deficiency of tissue, and surgical correction requires the addition of cartilage and skin. Mini-ear and severe cup-ear deformities are included in this category. The external auditory meatus is present, but it may demonstrate some degree of stenosis. Type III (classic microtia) dysplasia (Fig. 192-5A to C) deformities have few or no recognizable landmarks of the auricle, although the lobule usually is present and positioned anteriorly (see Fig. 192-16). Anotia is the complete absence of auricle and lobule. Reconstructive surgeons may find it more useful to simply note the status of the helix and antihelix, the conchal bowl, the lobule, the tragus, and the EAC, because these landmarks dictate the stages of ear reconstruction and the degree of surgical difficulty.
Planning
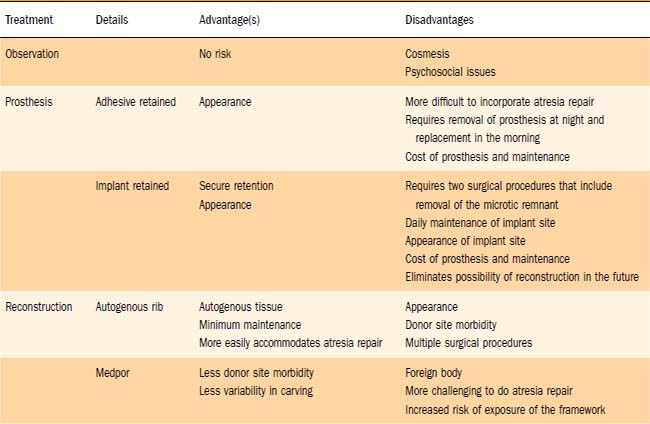
With regard to the pinna, the patient and family are presented with three options: observation; prosthetic management, either adhesive- or implant-retained; and reconstructive options including staged reconstruction with autologous rib graft, and reconstruction with linear high-density polyethylene (Medpor) surgical implant material and temporoparietal fascial flap. Generally, observation is recommended until the child is at least 5 to 8 years old. This allows both growth of the donor rib cartilage and development of the contralateral normal ear, which will act as a template for the microtic side. Waiting until the child is older may reduce the risk of the development of a thoracic deformity as a result of harvesting of cartilaginous rib graft, although we feel this risk is minimal.22 Finally, waiting until the child is school-aged allows the child to participate in these important decisions. We feel that it is important for the child to understand the options and participate in the decision making. The general health of the child is assessed, with particular attention paid to growth, maturation, and lung function. It is important to assess lung function, because the risk of pneumothorax and postoperative atelectasis and pneumonia is significant. This assessment usually is performed by the child’s pediatrician. It is important to question the child about his or her personal concerns and to inquire about the effects of the microtia on social interaction with friends and classmates. Each child and parent will have different concerns that should be explored preoperatively.
During the planning stage of reconstruction, the surgical benefits, risks, and alternatives are discussed with the patient and the parents (Table 192-1). Complications such as infection, bleeding, scarring, graft loss, displacement or resorption, asymmetry, and pneumothorax should be discussed in detail. The possibility of a less-than-optimal aesthetic result also is discussed, and the expectations of the patient, parents, and surgeon must be realistic and clearly defined. Providing postoperative photographs of previous patients often is helpful to patients if the pictures depict a realistic result of a similar deformity.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree