23 Management of Persistent Maxillary Sinusitis: The View from Japan Buoyed by the sentiment that the Japanese economy could “no longer be termed postwar,” Japan’s gross national product expanded at an average annual rate of 10% between the mid-1950s and the end of the 1960s.1 Until then, chronic sinusitis (CS) was so common in Japan that it was considered a national affliction. It was treated mainly with surgery, although a considerable number of cases were recalcitrant. The pathophysiology of CS has changed and the infiltrating cells in polyps or sinus mucosa have changed dramatically since then. In 1964, Okuda2 reported the dominant infiltrating cells in polyps found in Europeans were eosinophils, which were different from those seen in Japanese patients (lymphocytes and neutrophils). Since then, the rate of eosinophil-dominant polyps has increased and has matched the European level of eosinophil infection. Before 1970, sinusitis developed mainly due to an anatomic abnormality in the lateral nasal wall. Inflammation was located in the maxillary sinus and the dominant infiltrating cells in the sinus mucosa were lymphocytes or neutrophils. However, this type of sinusitis has decreased and is now treated by macrolide treatment and endoscopic sinus surgery (ESS). After 1970, increased numbers of allergic rhinitis patients with a type-1 allergy developed CS. Insufficient ventilation and drainage due to mucosal swelling in the nose were regarded as a cause. This was often seen in severe cases of allergic rhinitis or in pediatric cases. Concomitant usage of drugs for allergic rhinitis such as receptor antagonists or release inhibitors of chemical mediators is effective for this condition. The introduction of macrolides as agents for controlling CS in the 1990s dramatically improved outcome. Sinusitis, however, characterized by recalcitrant inflammation of the eosinophil-dominant mucosa became recognized,3 and this type of inflammation showed poor results even with macrolide treatment. The main locus in the early stage of this type of sinusitis was in the ethmoid cells, although pansinus involvement occurred later. This type of sinusitis was highly associated with bronchial asthma and could be controlled only by steroids. The number of such cases is still increasing and ESS is used in its treatment. The use of macrolides for treatment of these patients could reduce the number of patients undergoing ESS. Yet there are still patients who will require the surgery, especially eosinophil-dominant infiltration cases. Concomitant treatment of CS with preand postoperative macrolide treatment and the addition of steroids treatment is now the choice of treatment for this condition. The process of CS seen in Japanese is presented in Fig. 23.1. Fig. 23.1 The history of sinusitis treatment in Japan. (1) Before 1970, sinusitis developed mainly because of an anatomic abnormality in lateral nasal wall. Inflammation was located in the maxillary sinus. Neutrophil-dominant infiltration is present at the sinus mucosa. This type of sinusitis is a good indication for macrolide treatment and endoscopic sinus surgery. (2) Sinusitis accompanied by allergic rhinitis increased after 1970. Insufficient ventilation and drainage due to mucosal swelling in the nose seems to be the cause. This type is often accompanied by severe cases of allergic rhinitis and is observed frequently in pediatric cases. Concomitant antiallergic drug usage is effective. (3) Sinusitis caused by recalcitrant inflammation of the eosinophil-dominant mucosa was recognized in the 1990s. The main locus is in the ethmoid cells, followed by pansinus involvement; eosinophil infiltration is dominant. This type of sinusitis is highly associated with bronchial asthma. Poor results are obtained with macrolide therapy. Early in the 17th century, the Japanese feudal government broke off all relations with foreign countries, prohibited foreign travel, and entered an era of isolation. Japan’s modernization began in 1867 following the abandonment of the isolation policy. Until then, Japanese patients had been treated with traditional Chinese herbal medicine. For Japan, modernization meant Westernization. In the late 19th century, Japanese doctors, such as Okonogi, Kanasugi, and Kako who had studied otolaryngology in Germany, introduced Western techniques for the treatment of sinusitis. Dr. Okada was the first man to study at overseas universities at the government’s expense. After coming back from 4 years’ study in Berlin, Munich, and Vienna, he became the first professor of the Department of Otolaryngology, University of Tokyo Hospital in 1898. Dr. Watsuji, the first professor of otolaryngology at the University of Kyoto Hospital, also studied in Berlin. He developed a sinus operation, which is similar to Denker’s approach except that the inferior turbinate is left intact. He developed his approach in 1905—the same year as Denker. Thus, in Japan this approach has been named the Watsuji–Denker method. Professor Kubo studied abroad at Killian’s clinic in Freiburg before he became a professor at Fukuoka University Hospital. He was the first surgeon to report cases of postoperative maxillary cyst in the world.4 In the early 20th century, the prevalence and severity of sinusitis in Japan was high. Because of the lack of effective drugs, most patients underwent surgery for the treatment of sinusitis. The Caldwell–Luc method was used for maxillary sinusitis and Killian’s method for frontal sinusitis. However, once the importance of ethmoid cells in controlling CS was established, surgeons soon introduced the ethmoidectomy. Dr. K. Takahashi was the first surgeon to perform an endonasal ethmoidectomy in Japan (1918). Kuwana (1918) and Hirogami (1926) developed an ethmoidectomy through the maxillary sinus via the Caldwell–Luc approach. Dr. Kurosu developed the pansinectomy in 1929, which gave access to the ethmoid, frontal, and sphenoid sinuses through the maxillary sinus, nostril, and facial skin. Professor Nishihata at Keio University Hospital developed a combined sinectomy via the Caldwell–Luc approach and the endonasal approach in 1931. The latter method had been performed widely in Japan until the introduction of ESS in the 1980s. Professor R. Takahashi of Jikei University Hospital had performed an endonasal sinus operation in 19505 and tried to popularize the procedure. This method performed without the aid of a scope of any kind, required keen eyes and great surgical skill. This limited its use as a general procedure. The first report of ESS was done by Dr. Kaneko, one of Professor Takahashi’s pupils, in 1980.6 Since then, Professors Ohnishi and Moriyama of the Department of Otolaryngology at Jikei University Hospital have contributed to the teaching of ESS in Japan. As the cause of CS had not yet been established, effective conservative or medical treatment was not available until the introduction of macrolides. Antibiotics, antiinflammatory enzymes, and antihistamines had been used with limited effect. In the 1960s, the prevalence of sinusitis in children was as high as 30 to 40%. After 1980, it was reduced to 3 to 4%. In addition, the severity of sinusitis was high until 1980. Moderate and severe cases amounted to 40% in those days; it was not until 1995 that the rate was reduced.7 Sinobronchial syndrome (SBS) is defined as the coexisting pathologies of CS and nonspecific chronic lower airway inflammation, which includes chronic bronchitis, bronchiectasis, and diffuse panbronchiolitis (DPB). Allergic inflammation, such as bronchial asthma and allergic rhinitis, are not included in this category. Parenchymal lesions, such as pneumonia, are also not included. Typical SBS cases show pansinus lesions in diagnostic imaging and are classified as resistant-to-treatment. The history of SBS dates back to the end of the 19th century.8 However, it has been within the past 30 years that the cause of this syndrome has been found to be an abnormality of the systemic airway defense mechanism. The incidence of family occurrence is extremely high; it amounts to two-thirds in SBS patients.9 B54 in the B locus of human leukocyte antigen (HLA) is a characteristic antigen in mongoloids and is highly recognized in SBS patients. The incidence of B54 in Japanese control patients is ≥10%; in DPB patients it is 50 to 70%.9 In patients with bronchiectasis or chronic bronchitis, the prevalence is also high, although less than that of DPB. So, a portion of SBS patients may be regarded as having a genetic disease. DPB is sometimes seen in Caucasoids, but is common in Mongoloids. Nevertheless, B54 is not found in DPB patients in Korea, but HLA A11 is highly identified.10 The disease susceptibility gene may exist between the two HLA loci on chromosome 6. This genetic theory explains how both nasal and bronchial symptoms occur simultaneously. Yet the bronchiolar region is regarded as “silent zone” in view of symptomatology. Therefore, the difference of reactivity and standby capacity between the upper and lower airway tissues may be attributed to a time lag in manifestation. DPB was first reported in Japan in 1969. Cases were reported in the English literature in 1983.11 DPB is a diffuse chronic inflammation of the respiratory bronchiole and their peribronchiolar regions. It is considered a small airway disease because it usually involves terminal bronchiole and more proximal airways. DPB is accompanied by CS without exception. Before the introduction of macrolide therapy, DPB had the following characteristics: 1. Prognosis of the patient was poor; the 5-year survival rate was as low as 63% and the death rate in each year was 10%.12 2. The results of a Caldwell–Luc operation for the sinus lesion were not good compared with other types of CS.13 3. Complete cure after sinus surgery was rare. 4. The lower airway symptoms did not necessarily improve, even in the patients whose sinus symptoms improved. The introduction of macrolide therapy along with ESS made SBS a curable condition. Japan was the first country to begin macrolide therapy. Macrolide therapy was started as the result of an accidental discovery by Dr. Kudoh. He had treated a 52-year-old male DPB patient with steroids and various antibiotics for 5 years without any effect and the patient discontinued consultation. Two years later in 1980 the patient came to see him with minimal symptoms and his PaO2 had improved from 55 to 85 torr. Dr. Kudoh was surprised at the patient’s improved condition and learned that he had consulted Dr. Miyazawa in Matsumoto City (200 km northwest to Tokyo) for 2 years. His prescribed drugs were almost the same as those Dr. Kudoh had prescribed in his earlier treatment, except for the addition of 600 mg a day of erythromycin (EM). Normally we would think that such a low dose would not work. But Dr. Kudoh could not help but believe what he saw and accepted the validity of this regimen. To confirm this treatment, the first open trial of low-dose, long-term EM therapy was started immediately. After 6 months to 3 years of treatment with 600 mg of EM, symptoms and clinical parameters were markedly improved in 18 patients. FEV 1 and PaO2 were significantly increased on average from 1.61 to 2.17 L (p < 0.01) and from 65.2 to 75.1 mm Hg (p < 0.01), respectively. Small nodular shadows on chest x-ray (CXR) films disappeared in more than 60% of the cases.14 A subsequent national-level controlled study with placebo was performed in 1990 with significantly better results.15 In the 1970s before EM treatment, the overall 5-year survival rate was 63% as stated earlier. Between 1980 and 1984, fluoroquinolones were started for treating Pseudomonas aeruginosa, but the survival rate was still limited to 72%. After 1985, when EM therapy was introduced, the 5-year survival rate was 91%.12 As a result, EM treatment became a standard therapy for chronic lower airway inflammatory disease including DPB. As DPB always accompanies CS, EM therapy for DPB also then affects sinus pathology. Nasal symptoms get better during EM therapy. The first EM trial for CS was done in 1990 among patients with CS-associated DPB. EM showed remarkable effectiveness.16 The next trial was performed by Kikuchi and others17 in 26 adult patients with CS regardless of the presence of lower airway inflammatory diseases in 1991. They were treated with 400 to 600 mg of EM per day for an average of 7.9 months. Rhinorrhea was reduced in 60%, postnasal drip in 50%, nasal obstruction in 60%, hyposmia in 11.8%, and a sense of dullness in the head was absent in all the study’s patients. Rhinoscopy showed reduced mucosal swelling in 10.5%, lower volume of rhinorrhea in 80%, better quality of rhinorrhea in 70%, and reduced postnasal drip in 85.7%. Clinical efficacy was unrelated with sensitivity of EM for bacteria identified. No significant side effects were noted in any of the patients during this therapy; however, this was not due to EM’s antibacterial activity. The same authors then proceeded to study 130 CS patients, including 21 pediatric patients.18 EM at 400 to 600 mg per day for adults and at 200 to 300 mg per day for children was administered orally for an average of 5.4 months and at least 3 months. Subjective symptoms—excluding dysosmia—and objective findings—excluding mucosal swelling—were improved after treatment. The results of this EM study were as follows: 1. Adequate clinical effect was obtained with half or less of ordinary dose. 2. The clinical course is slowly, but surely improved with time and reliable effect was obtained at 2 to 3 months. 3. This treatment is more effective in cases of hypersecretion-type CS. 4. The clinical effect in children was somewhat less than that found in adults. (Successive studies have yielded better outcomes in pediatric cases.)19 5. Clinical efficacy was unrelated with the presence of lower airway diseases or history of sinus surgery. A subsequent trial was done with new 14-membered macrolides. Clarithromycin (CAM) and roxithromycin (RXM) also showed effectiveness for CS. The first article describing the use of macrolides in patients with CS appeared in the English literature in 1996. Hashiba and Baba20 treated 45 adult CS patients with CAM, 400 mg daily for 8 to 12 weeks. Twenty (44%) of these patients had undergone previous sinus surgery. Improvements in symptoms and rhinoscopic findings were directly related to the duration of macrolide therapy. The investigators noted improvement rates of 5%, 48%, 63%, and 71%, respectively, after 2, 4, 8, and 12 weeks of therapy. After 12 weeks of therapy, 64% of patients had reduced viscosity of nasal secretions, 56% had reduced quantity of nasal secretions, 62% had decreased postnasal drip, and 51% had reduced nasal obstruction. Clinical benefit in patients with CS also was observed following long-term administration of roxithromycin, 150 mg daily.21 Roxithromycin was tried on polyps associated with chronic rhinosinusitis.22 Forty adult patients were enrolled and were grouped according to whether they received azelastine or not. Duration of medication was at least 2 months. More than half the cases experienced shrinkage of nasal polyps in various extents in both groups. The rate of polyp disappearance was 16% in each group. A number of studies have been done to ascertain macrolides mechanism of action, outside their antibacterial effect. As DPB is a typical neutrophil-derived inflammatory condition, some novel actions relating neutrophils were examined. Macrolides inhibit accumulation and activation of neutrophils, suppress expression of adhesion molecules, block production of proinflammatory cytokines, and hamper lymphocyte activation. It has recently been found that even sub-MIC of 14-membered ring macrolides exhibits inhibitory effects on biofilm formation and the expression of virulence factors such as pyocyanine, elastase, and proteases. Macrolides inhibit water secretion by blocking Cl–– channels via epithelial cells.23 They also inhibit mucin secretion.24 The novel actions of macrolides are summarized in Table 23.1. The pathologic mechanism of developing CS is complicated. Where the ostia of the sinuses presents in the middle meatus is called the ostiomeatal complex (OMC). If this part is blocked, ventilator and drainage impairment will occur and the gas composition and pH in the sinuses will change. As a result, bacterial proliferation occurs, and the mucosal lamina propria in the sinuses thickens. The blockade of the ostia is then reinforced, which creates a “vicious circle.” Any treatment of CS aims to break this cycle. Antral lavage can do this by clearing accumulated fluids in the sinuses, whereas antibiotics act specifically against bacterial proliferation. Topical management including a topical vasoconstrictor helps to open the ostium; whereas mucolytics improve mucociliary clearance. Macrolides (1) inhibit the accumulation of inflammatory cells, (2) block cytokine production, (3) decrease water and mucin secretion, (4) restore mucociliary clearance, and (5) hamper biofilm formation. There are many different treatment options for CS. Among medical treatments, macrolide treatment produces the most improvement with the exception of steroid treatment. With macrolide treatment, many patients can avoid surgery. Nevertheless, in mild or moderate cases of CS, a combination of drugs has proven to be the most effective treatment. The low-dose long-term administration of macrolide should be used for recalcitrant cases. For the treatment of CS, macro-lide therapy should be used for hypersecretion-type CS, a refractory case, and for certain postoperative conditions. In 1998, the following clinical guidelines for long-term macrolide therapy was announced by the Society for Novel Actions of Macrolides Study Group (Table 23.2).25 1. What macrolides should be used? The 14-membered macrolides (EM, CAM, RXM) 2. What are the appropriate doses to prescribe? Half of an ordinary dose is recommended. For adults, daily doses are EM 400 to 600 mg, CAM 200 mg, and RXM 150 mg. For children, daily doses are EM 8 to 12 mg/kg and CAM 4 to 8 mg/kg. 3. How long should they be used? For 3 months at first. At 3 months the efficacy of the drug should be evaluated. If ineffective, change to another treatment. It is important to shorten the duration of medication to avoid adverse effects and the development of resistant bacterial strains. Even if effective, medication should be stopped.
 History of Sinusitis Treatment in Japan
History of Sinusitis Treatment in Japan
 The Impact of Sinobronchial Syndrome
The Impact of Sinobronchial Syndrome
 Macrolide Treatment
Macrolide Treatment
1. On host |
Inhibit cytokine production |
Inhibit active oxygen generation |
Inhibit production of chemotactic factors, such as lipopolysaccharide (LPS) and tumor necrosis factor-alpha (TNF-α) from fibroblasts |
Suppress the activation of nuclear factor-kappa B and activator protein-1 |
Accelerate both differentiation and proliferation of monocytes-macrophage system cells |
Enhance intrinsic glucocorticoid production |
Inhibit chloride channel opening |
Suppress mucin production |
2. On bacteria |
Potentiate serum sensitivity |
Suppress expression of virulence factors (pyocyanine, elastase, protease) |
Restrain production of biofilm |
Inhibit quorum sensing system |
Fourteen-membered macrolides (EM, CAM, RXM) are recommended. |
Half of an ordinary dose is recommended. For adults, daily doses are EM 400–600 mg, CAM 200 mg, RXM 150 mg. For children, EM 8–12 mg/kg, CAM 4–8 mg/kg |
Macrolides are used for 3 months at first. At 3 months the efficacy of the drug should be evaluated. If failed, change to another treatment. Even if effective, medication should be stopped at 6 months to prevent adverse effects and the development of resistant bacterial strains. |
If symptoms recur, treatment should be restarted. Tachypraxis has not been recognized so far. |
In the following conditions, additional treatments may be required: the presence of type 1 allergy or bronchial asthma, obstruction at the OMC, coexistence of polyps, and acute exacerbation. Additional treatments include surgery and steroids. |
Abbreviations: EM, erythromycin; CAM, clarithromycin; RXM, roxithromycin; OMC, ostiomeatal complex.
4. If effective, when should the treatment be stopped?
Even if effective, stop at 6 months.
5. What should be done if the symptoms recur?
Macrolide treatment should be restarted. Tachypraxis has not been recognized so far.
6. Under what conditions may additional treatment be required?
The presence of type 1 allergy or bronchial asthma, obstruction at the OMC, coexistence of polyps, and acute exacerbation limit the efficacy of macrolides. In such conditions, additional appropriate treatment, such as surgery or the administration of steroids, may be required.
This guideline was also incorporated with no modification in 2006 in the Japan Rhinologic Society’s Practical Guidelines for Sinusitis.26
Indications in children are different from those in adults. In pediatric CS, signs and symptoms are unstable and bacterial infection is prominent, whereas the incidence of irreversible mucosal lesions including polyps is low. CS may spontaneously remit during adolescence. Thus, long-term treatment is not the sole option. Indications of pediatric CS are the presence of purulent or mucopurulent nasal discharge and if its duration is longer than one month. Evaluation should be done at 2 months and the term of treatment should be as brief as possible.27
Side effects reported were gastrointestinal upset and abnormality of liver function. These adverse effects will occur within 3 months at which point the medication should be stopped. The incidence of an adverse effect was reported as 7.2% with EM, 2.5% with CAM, and 1.9% with RXM.28 The cessation of treatment due to side effects was 2.8% with EM and 0% with CAM and RXM in Japan. Severe adverse effects have not been recognized so far. Hence, macrolide therapy is considered a safe treatment.
One possible problem posited in macrolide treatment is the increase in the prevalence of resistant strains of bacteria. Mean types of bacteria during macrolide treatment were investigated by Hashiba and others.29 Twenty-seven patients were included in the study. No numerical change was confirmed during and at the end of the treatment compared with baseline. Maeda et al presented 26 patients with chronic lower airway inflammation who had been administered macrolides for more than 10 years.30 Adverse effects were observed in only two cases (dysgeusia and raised liver enzyme level); they disappeared soon after cessation and never reappeared in the next administration. Long-term administration of macrolides had no effect on sensitivity of identified bacteria from the sputa to β-lactum antibiotics.
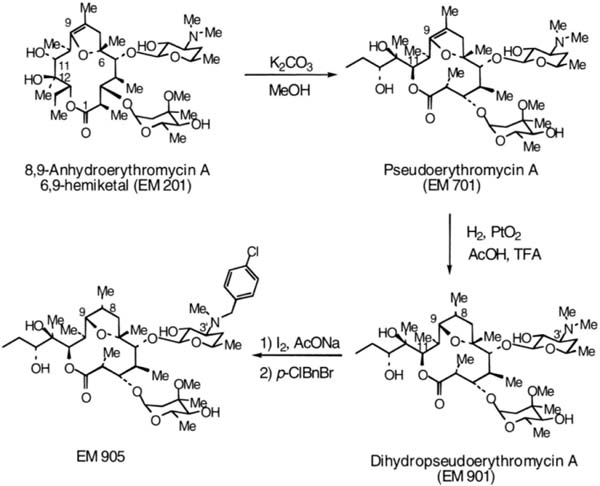
To date, there has been no direct evidence for increased antibiotic-resistant bacteria in the nasal and oral cavity. And to the best of my knowledge, there have been no reports of patients treated with long-term macrolides who experienced life-threatening infectious disease with macrolide-resistant bacteria. Nevertheless, new antiinflammatory macrolides without antimicrobial activity should continue to be developed. EM201 was developed as a metabolic product of EM; it has no antibiotic effect, but still has motilin-like action. From this product, EM 703 was created as a new macrolide derivative, which reinforced antiinflammatory activity but was weak in solubility and stability. Then the EM 900 series compounds (901, 905, 914, and 939) were developed (Fig. 23.2). They have no influence on CYP3A4. They are now being investigated in vitro; their induction potency of monocytes on macrophages has been shown to increase 30 times as much as that of EM.31 Further investigation of this series is required.
Fig. 23.2 Synthesis of the EM 900 series.
As mentioned here earlier, macrolide therapy has reduced the rate of ESS. In cases of extensive mucosal pathology and those of obstructed OMC that are usually resistant to macrolide therapy, removal of polyps is very useful. Macrolide therapy is continued in the postoperative course. Patients who are candidates for surgery are recommended to have macrolides prior to the surgical procedure. Although not published before, minimizing inflammation by macrolides decreased volume of bleeding during surgery. With the introduction of postoperative macrolide therapy, the symptomatic improvement rate after ESS has become 90% compared with 80% obtained before.32
 Eosinophilic Sinusitis
Eosinophilic Sinusitis
A histologic study by Iino et al indicated that macrolide treatment is effective in sinusitis with lymphocyte-dominant or neutrophil-infiltrating patterns of submucosal infiltrating cells, but not effective in sinusitis with an eosinophil-dominant pattern.33 Some cases recur even after the combination treatment of macrolide therapy and ESS. A retrospective analysis of postoperative cases revealed that the cases associated with bronchial asthma or with eosinophil-dominant infiltration in the sinus tissue had poor results.34 Haruna and Moriyama35 proposed the term, eosinophilic sinusitis to describe the infiltration of activated eosinophil in the sinuses. Diffuse eosinophil-dominant polyposis and eosinophil mucin rhinosinusitis (Ferguson36) are terms that describe similar conditions. Eosinophilic sinusitis includes sinusitis combined with nonatopic asthma, aspirin-induced asthma, Churg–Strauss syndrome, allergic fungal sinusitis, and eosinophil-infiltration sinusitis without lower airway lesions.
The clinical characteristics of eosinophilic sinusitis are as follows:
1. Age at onset is ~40.
2. This type of sinusitis is usually involved bilaterally and associated with multiple edematous polyps.
3. Stronger pathologic changes are present around the middle turbinate and less in the inferior turbinate.
4. The majority of patients complain of dysosmia.
5. Accumulation of thick mucus with eosinophil infiltration is observed.
6. This condition is remotely related to allergic rhinitis and shows varied quantities of immunoglobulin E (IgE).
7. Bronchial asthma and aspirin-induced asthma are accompanied by the condition.
8. Eosinophil count in blood and value of eosinophilic cation protein (ECP) in serum and nasal mucosa are increased.
9. The ethmoid sinuses are mainly involved in the early stages followed by pansinus involvement.
10. This disease is resistant to treatment, especially to surgical therapy.
11. The administration of systemic steroids is effective for this condition.
Ishitoya et al37 proposed the following diagnostic criteria for eosinophilic sinusitis. According to these criteria, eosinophilic sinusitis accounts for 15% of the CS that requires surgery.
1. Computed tomography (CT) findings: ethmoid/maxillary (E/M) score ratio*≧1 and score of ethmoid cells ≧3 *E/M score ratio = (CT score of anterior ethmoid cells + score of posterior ethmoid cells)/(score of maxillary sinus × 2) (after Lund–McKay score)
2. Eosinophil count in blood: ≧6% or 400/μL
3. Eosinophil count* in nasal polyps: ≧350 in the field of view at 400-fold magnification. *In a polyp specimen that has been stained with hematoxylin-eosin, five areas where eosinophils are found in a higher percentage are selected and the number of eosinophils is counted at 400-fold magnification and the mean of the value of the top three areas is obtained.
4. The presence of typical clinical findings: dysosmia, nasal obstruction, thick mucous discharge, etc.
5. All asthmatic patients have CS. About 10% of asthmatic patients have aspirin sensitivity. The condition is characterized by raised leukotrene (LT) s in the serum and urine.
 Maxillary Sinus Surgery and Postoperative Treatment
Maxillary Sinus Surgery and Postoperative Treatment
For maxillary sinus lesions, a wide opening of the natural ostium (NO) and middle meatus is essential for ensuring reliable working space during surgery. If there are polyps in the middle meatus, they should be removed first. The middle meatus is widened by resection of the uncinate process. For performing uncinectomy we Japanese use a Kurosu elevator (Fig. 23.3A) instead of the Freer elevator widely used in Western countries. For easier management of the NO we remove the ethmoidal bulla and open the other anterior ethmoidal cells. If there is no ethmoidal involvement, opening of the (third) basal lamella of the ethmoid is not necessary; however, we usually open it to secure the ventilation route of the maxillary sinus in the postoperative period. In this initial stage, the NO can be seen in over half the patients even with a straight, zero-degree endoscope. (The procedure used to identify and widen the NO is shown in Video 23.1).
To enlarge the NO, the knife specialized for the membranous portion of the medial wall of the maxillary sinus (Fig. 23.3B) is used followed by retrograde cutting forceps (Fig. 23.3C) for forward or vertical enlargement and straight cutting forceps (Fig. 23.3D) for backward widening. The position of the knife cut is on the membranous portion of the medial wall at a height between both ends of the middle turbinate. The window should have a diameter of not less than 1 cm. Care should be taken not to injure the nasolacrimal duct during a forward enlargement procedure. Removal of the mucosa in the sinus is unnecessary. Instead, the edematous swollen membrane is trimmed with a microdebrider, Nishihata’s greatly curved spoon forceps (Fig. 23.3E), or a malleable suction tube without exposing bony surface.
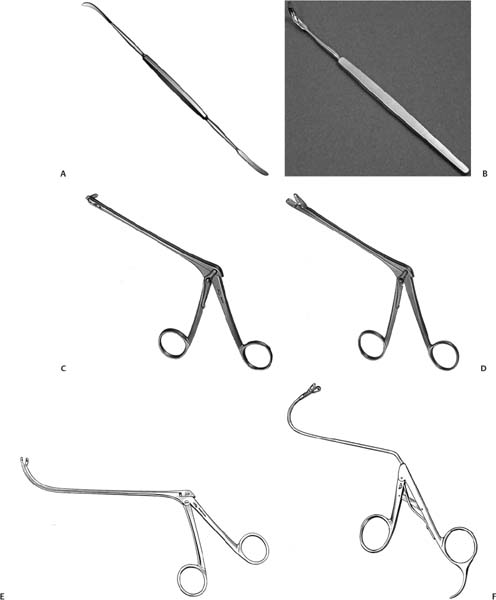
Fig. 23.3 (A–F) Instruments for surgery. (A) Kurosu elevator. (B) Membranous portion knife. (C) Ashikawa’s retrograde cutting forceps (opening to the right). (D) Ashikawa’s straight cutting forceps. (E) Nishihata’s greatly curved spoon forceps. (F) Kikawada’s spoon forceps (135 bent).
The anteroinferior portion of the sinus is the difficult part to handle because it is out of reach of instruments through the middle meatus. If pathology is present there, we have two options. One is to use Kikawada’s spoon forceps (135 degrees) (Fig. 23.3F), which can put the unreachable within reach and another is to make a hole at the lateral wall of the inferior meatus as anterior as possible. This hole is big enough to pass through suction tubes, slender curved forceps, or a microdebrider. We usually create a hole of 5-mm width. The trimming procedure is continued with forceps, suction tubes, or a microdebrider passed through this control hole. Illumination is provided by a 70-degree endoscope set in the middle meatus. If the alveolar recess of the sinus is well developed, the management of the anteroinferior part is still difficult even with the creation of an inferior meatal hole. In such cases, a small incision is made at the gingivobuccal sulcus and a small hole around 5-mm width is created at the anterior wall of the sinus with a chisel or drill. The trimming is performed through the hole in the same fashion as from the inferior meatal window. If there are tenacious fluid, fungus balls, caseous masses, or foreign bodies, irrigation with saline is effective.
The surgical procedure for eosinophilic sinusitis is almost the same as that for CS. Important reminders are (1) to suction the pooled thick fluid in the sinuses as completely as possible, (2) to avoid adhesion due to the formation of multiple wound surfaces at the olfactory cleft and superior meatus, and (3) to make a single sinonasal cavity by widening ostia of sinuses and removing ethmoid cell walls.
Postoperative treatment is very important. We usually ask patients to visit weekly for the first postoperative month, biweekly the next month, monthly for the next 4 months, and then once evert 3 months until one year of non-eosinophilic sinusitis. Because mild and moderate cases are controlled by macrolide therapy, the patient who undergoes surgery needs intense follow-up and requires treatment during the postoperative period. Mucociliary function is recovered by 3 months after surgery. At each visit, endoscopic observation of the nose and sinuses is mandatory. In the early postoperative stage, excessive fibrin masses and crusts are suctioned and removed gently. Bleeding should be avoided as much as possible so as not to prolong the healing process. Secretions collected in the sinus are flushed with saline. Small polyps, edematous mucosa, and granulations are managed by placing cottons soaked with adrenaline and steroids followed by aerosol therapy. We remove polyps at every visit after the postoperative 3 weeks. Topical steroids and oral low-dose macrolides are applied for 2 months postoperatively. Eosinophilic sinusitis often re-occurs even after the postoperative third month. For this condition, peroral administration of steroids for some days soon after the operation and regular postoperative therapy is applied along with administration of topical steroids and leukotriene receptor antagonists for as long as 3 to 6 months. As for aspirin-sensitive asthma (ASA), the addition of topical cromolyn helps to prevent recurrence after surgery. Oral steroids (prednisolone 20–30 mg/day) are administered for several days to offset the recurrence of polyps.
The postoperative improvement rate was 88% in patients with lymphocyte-dominant sinus mucosa; 69% in patients with eosinophil-dominant mucosa.34 If patients with histopathologically diagnosed eosinophilic sinusitis were divided into two groups according to whether they had bronchial asthma, the percentage of patients without nasal symptoms after surgery was 38% for the asthma group compared with 89% for the non-asthma group.38
 Postoperative Maxillary Cyst
Postoperative Maxillary Cyst
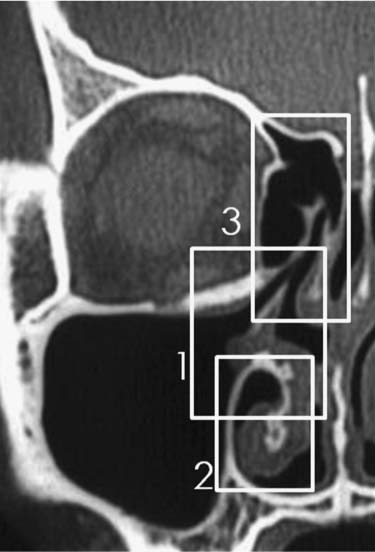
Maxillary sinus surgery via the Caldwell–Luc approach has a complication of postoperative maxillary cyst (POMC). The first report on POMC was presented by Professor Kubo as “Postoperative Wangenzyste” in 1927.3 Before the introduction of ESS, we were busy surgically removing POMCs. Because of the absence of prospective studies, the incidence of POMC is not known. From an imaging study of POMCs, latent asymptomatic cysts have been found in other area in 34% of POMC cases.39 Although there has been no study on whether these asymptomatic cysts present with symptoms later, Iinuma estimated this rate as 3.4%.40 In my experience, this figure seems reasonable and can be applied to the incidence of POMC. The most prevalent period of occurrence is 11 to 15 years postoperatively. A POMC is most often found at the anteromedial part of the maxillary sinus. A monocyst is present in more than 50% of cases, followed by double (30%) and triple (10%) cysts.41 There are several theories on POMC development, but two are considered to be reasonable proposals. The retention cyst theory posits that the remnant sinus mucosa is surrounded by granulation tissue at the postoperative period, and as a result, pathologic closed cavity develops. The invagination theory proposes that the upper and medial wall of the maxillary sinus invaginated to the center of the sinus and the traffic route to the middle meatus became stenotic and formed a closed cavity. Patients with a POMC complain of pressure symptoms according to the localization of the cyst, cheek swelling, cheek pain, nasal obstruction, toothache, gingival swelling, palatal swelling, epiphora, diplopia, and proptosis. There is a short acute exacerbation stage and a long intermittent stage. Bony erosion develops in the acute stage. Even in the intermittent stage, a bony destruction process will develop gradually due to prostaglandin E2 (PGE2) and collagenase in the accumulated fluid. When such pressure symptoms appear, the history of sinus surgery and the scar at the gingivobuccal sulcus indicate the diagnosis of POMC. CT and/or magnetic resonance imaging (MRI) are essential tools for determining the treatment modality. After acute exacerbation with antimicrobials, the endoscopic endonasal opening of the cyst is the current surgical choice. The exceptions are far laterally placed cysts or cysts with an extremely thick bony wall. In creating a large opening at the inferior meatus, identification of the nasolacrimal duct outlet is important to avoid injuring it. (In Video 23.2, the author shows how to identify the nasolacrimal duct outlet using dye infusion.)
The introduction of ESS and the resultant decrease in the number of Caldwell–Luc operations reduced the numbers of POMCs. This is a good trend for patients with CS. The chance, however, of surgery for POMC is still quite high. In the case of a dentigerous cyst, the cyst needs total removal because of high recurrence rate after simple cystostomy and the risk of a keratinizing primordial cyst. (The procedure of removing the cyst wall of a dentigerous cyst via the middle meatus and inferior meatus is shown in Video 23.3.)
 Conclusion
Conclusion
Until 20 years ago, the sinusitis refractory to treatment was sinobronchial syndrome. Introduction of macrolide therapy and ESS was able to control this condition. Recently, eosinophilic sinusitis has become a treatment challenge. Extensive and minute surgery along with elaborate postoperative treatment and the administration of steroids is the treatment protocol at present.
• CS can be divided into three types: (1) with lymphocytes or neutrophil dominant infiltration in the mucosa, (2) associated with allergic rhinitis, (3) associated with bronchial asthma or with eosinophil-dominant infiltration in the mucosa.
• Recommended treatment for type 1 is macrolide treatment; in unresponsive cases ESS is recommended. Recommended treatment for type 2 is macrolide treatment with antiallergic drugs.
• Recommended treatment for type 3 is ESS with macrolide treatment in the pre- and postoperative period along with a short course of systemic steroids in the postoperative period and at recurrence of polyps.
• Creating a large hole in the membranous portion of the medial wall of the maxillary sinus and ethmoidectomy is a key point in maxillary sinus surgery. Bony surface exposure should be avoided.
• Decreased numbers of postoperative maxillary cysts is due to the abandonment or restriction of the Caldwell–Luc operation.