CHAPTER 88 Malignant Neoplasms of the Salivary Glands
Malignant neoplasms of the major and minor salivary glands are rare, comprising approximately 3% of all head and neck malignancies.1 The estimated incidence is only 0.9 per 100,000 in the United States, but the rate increases with age, peaking at ages 65 to 74 years.2 Less than 5% of all salivary gland tumors occur in the pediatric age group; however, salivary gland tumors in children are much more likely to be malignant than those of adults.
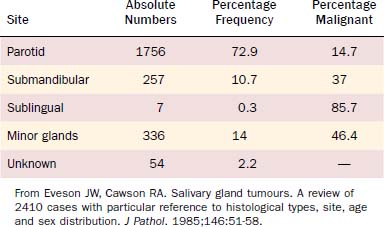
Of all salivary neoplasms (benign and malignant), the vast majority occurs in the parotid gland and the fewest in the sublingual gland. There is an interesting inverse relationship between the overall incidence of neoplasms by site and the percentage that are malignant (Table 88-1). In a review of 2410 cases of salivary gland tumors,3 73% occurred in the parotid, and of those, only 15% were malignant. On the other hand, minor salivary gland tumors constituted only 14% of the total number of cases but 46% were malignant. Likewise, submandibular gland neoplasms constituted 11% of the cases with 37% being malignant; sublingual gland neoplasms constituted only 0.3% with 86% being malignant.
The frequency of the different histologic types of salivary gland malignancy also varies depending on the gland and site. A number of studies have found the most common primary malignancy presenting in the salivary glands to be mucoepidermoid carcinoma.1,2,4 In one of the largest reviews of salivary gland neoplasms (2807 total), Spiro examined 1278 cases of malignant salivary gland tumors1 and reported that 34% were mucoepidermoid carcinoma (Table 88-2). The next most common type was adenoid cystic carcinoma (22%), followed by adenocarcinoma (a mixture of tumors which has more recently been subdivided, as described below) (18%), malignant mixed tumor (13%), acinic cell carcinoma (7%), and squamous cell (epidermoid) carcinoma (4%). When considering the types by anatomic site, mucoepidermoid carcinoma was the most frequent malignancy of the parotid, but adenoid cystic carcinoma was the most frequent of the submandibular and minor salivary glands. One exception to the latter was malignant minor salivary gland tumors arising in the nasal cavity and paranasal sinuses, in which case adenocarcinoma was the most common type as opposed to adenoid cystic carcinoma.
Table 88-2 Relative Incidence of Malignant Salivary Gland Neoplasms
| Histologic Type | Number | Percent |
|---|---|---|
| Mucoepidermoid | 439 | 34 |
| Adenoid cystic carcinoma | 281 | 22 |
| Adenocarcinoma NOS | 225 | 18 |
| Malignant mixed tumors | 161 | 13 |
| Acinic cell carcinoma | 84 | 7 |
| Squamous cell carcinoma | 53 | 4 |
| Other (anaplastic etc.) | 35 | 3 |
| TOTAL | 1278 |
From Spiro RH. Salivary neoplasms: overview of a 35-year experience with 2,807 patients. Head Neck Surg. 1986;8:177-184.
Evaluation of the Patient
Physical Examination
The clinical presentation of malignant salivary gland neoplasms can range from indolent asymptomatic masses to rapidly growing painful masses with progressive facial nerve paralysis. In Spiro’s review of 2807 salivary gland tumors, pain was a symptom in only 10% of malignant cases1 but was more frequently seen with malignant neoplasms than with benign ones. In general, episodic swelling and pain most often indicates salivary gland obstruction and inflammation, whereas constant pain is more worrisome for malignancy. However, sialadenitis can be secondary to obstruction by a salivary gland neoplasm; thus the latter must be considered in the evaluation of any salivary gland swelling with pain. Approximately 10% of parotid gland malignancies present with associated facial paralysis, and this portends a poor prognosis.5,6
Submandibular Gland
Bimanual (intraoral and external) palpation of any submandibular gland tumor should be performed to assess the extent of the tumor and to determine if there is fixation to adjacent structures such as the mandible or skin. A careful neurologic examination should also be performed to assess nerve involvement. In particular, worrisome signs of malignancy include numbness of the tongue (suggesting lingual nerve involvement), weakness of the tongue (suggesting hypoglossal nerve involvement), or weakness of the lower lip (suggesting facial nerve involvement). Careful examination of the neck is also important because 25% to 28% of submandibular malignancies will have metastases to regional lymph nodes.1,7
Sublingual Gland
The pair of sublingual glands is located in the floor of the mouth, one on each side of the frenulum, lateral to Wharton’s duct in the submucosal compartment. The drainage of these glands is via Bartholin’s ducts, which empty into Wharton’s ducts. As with submandibular gland tumors, bimanual palpation of the floor of the mouth is important to assess the extent and possible fixation of sublingual gland tumors to the mandible. Because the sublingual gland is intimately associated with the lingual and hypoglossal nerves, a careful neurologic examination is important as previously stated. Although tumors in this area are usually painless, the vast majority (86%) of sublingual gland tumors are malignant.1
Minor Salivary Glands
Minor salivary gland tissue is plentiful with estimates between 500 and 1000 glands along the upper aerodigestive tract. Although they are located in the submucosa throughout the oral cavity, oropharynx, nasal cavities, paranasal sinuses, pharynx, and larynx, the majority of them are located in the oral cavity, with the highest concentration being in the submucosa of the hard palate. As such, the site most frequently involved with minor salivary gland malignancies is the hard palate.8 Compared with the major salivary glands, minor salivary glands have minimal capsular tissue, making local invasion of tumors into surrounding tissue common. Patients with malignancies of the minor salivary glands most often present with a painless submucosal swelling, but there is frequently fixation of the overlying mucosa and ulceration. Approximately one quarter of patients will complain of local pain,9 and pain or paresthesia/anesthesia is concerning for nerve invasion. Because minor salivary glands are distributed throughout the mucosalized surfaces of the head and neck, malignancies of these glands may present in diverse ways depending on the location. Symptoms may include nasal airway obstruction, sinusitis, eustachian tube dysfunction, or hoarseness. A thorough head and neck evaluation including fiberoptic examination and cross-sectional imaging should be performed in all cases.
Radiologic Studies
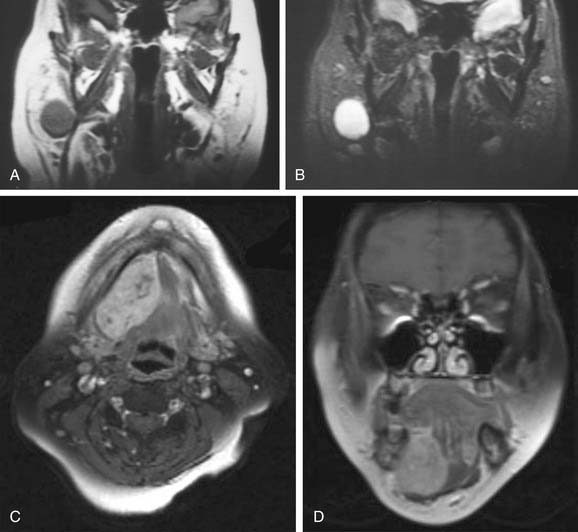
Magnetic resonance imaging (MRI) is the radiologic modality most often recommended to assess salivary gland neoplasms if there are no contraindications. Benign and malignant neoplasms of the parotid gland are well visualized on T1-weighted images because they are easily distinguished from the fatty parenchyma of the gland, which appears hyperintense. In general, benign epithelial neoplasms (such as pleomorphic adenomas) and low-grade malignancies have low T1-weighted and high T2-weighted signal intensities (Fig. 88-1A and B). High-grade carcinomas tend to have low-to-intermediate signal intensities on both T1-weighted and T2-weighted imaging (discussed later). The use of contrast material such as gadolinium and fat saturation of T1-weighted imaging provides additional information regarding the extent of a salivary gland malignancy (Fig. 88-1C and D). This is particularly useful in terms of assessing bone involvement and perineural spread. Bone marrow and cortex appear hypointense on fat-saturated images, and infiltrating tumor tissue appears hyperintense when enhanced with gadolinium. Enlarged foramina at the skull base and the presence of hyperintense enhanced tumor tissue are suggestive of perineural spread.
Some have proposed that T2-weighted imaging is helpful in distinguishing benign from malignant salivary gland neoplasms. Som and Biller10 examined 35 parotid tumors by MRI and made the following observations. Benign tumors and low-grade malignancies had low T1-weighted and high T2-weighted signal intensities, and they had clearly defined margins. High-grade malignancies, on the other hand, had low T1- and T2-weighted signal intensities and poorly defined margins. Freling and colleagues,204 however, did not make the same observations. They examined 116 patients with parotid masses, 30 of which were malignant tumors, and found no correlation between malignancy and signal intensity, heterogeneity, or radiographic margins on MRI. Malignant lesions could be discriminated from benign ones only when there was infiltration into adjacent structures. Among the malignant lesions, there was no correlation between tumor grade and MRI features. Thus MRI provides useful information about the extent of the disease, but histopathologic diagnosis is still required to distinguish benign from malignant processes.
Biopsy
Pretreatment diagnosis has been made possible by the use of fine-needle aspiration (FNA) biopsies. This diagnostic tool was established in the mid-to-late 1960s11–16 and has become a mainstay in the workup of salivary gland neoplasms. It is an extremely safe procedure that is well tolerated by patients and is considered to have no significant risk of tumor seeding to surrounding tissue.17
Although FNA biopsies of salivary neoplasms have proven to be extremely useful for preoperative planning, the interpretation of these biopsies can be difficult, resulting in diagnostic ambiguity and inaccuracy. A few entities have classic, pathognomonic features on needle aspiration. However, the variety of different tumor types and their widely overlapping histology usually necessitates the acquisition of biopsy or resection tissue for definitive diagnosis. Also, and most importantly, many malignant salivary gland tumors can only be diagnosable as such when the growth pattern (e.g., infiltration beyond the capsule into soft tissue, perineural invasion) is taken into consideration. In these cases, tumors can only be definitively diagnosed as malignant on surgical specimens, which adequately demonstrate the tumor architecture and its relationship to peripheral tissue. This is particularly true for low-grade malignant neoplasms such as polymorphous low-grade adenocarcinoma,18 myoepithelial carcinoma,19,20 and basal cell adenocarcinoma,21 which usually have bland-looking tumor cells and lack necrosis or abundant mitoses. Among tumors of the head and neck, FNA biopsies of major salivary gland tumors are considered to have the highest rate of error.22 By most reports, the sensitivity of FNA biopsies to diagnose a malignant neoplasm is much lower than the specificity.22–26 In other words, it is more common to misdiagnose a malignant tumor as benign than the reverse. A 5-year review of data from 6249 participant responses from the College of American Pathologists Interlaboratory Comparison Program in nongynecologic cytology revealed that FNA biopsies had a 68% sensitivity for diagnosing a salivary gland neoplasm as malignant, with an overall false-negative rate of 32%.27 The greatest number of false-negative diagnoses occurred in cases of lymphoma (false-negative rate = 57%), followed by acinic cell carcinoma (false-negative rate = 49%), low-grade mucoepidermoid carcinoma (false-negative rate = 43%), and adenoid cystic carcinoma (false-negative rate = 33%). In cases of benign neoplasms from the same study, the specificity of FNA biopsies was 91%, with an overall false-positive rate of 8%. The greatest false-positive rate was in cases of monomorphic adenoma (53%), which were most often diagnosed as adenoid cystic carcinoma. Pleomorphic adenomas, when diagnosed as malignant, were most often misinterpreted as adenoid cystic carcinoma, and Warthin’s tumors as lymphoma.
Although the utility and ease of FNA biopsies have resulted in a decline in the use of frozen section analysis intraoperatively, there are still several indications for frozen section studies, as reviewed by Seethala and colleagues28 and Westra.22 These include assessing the extent of tumor spread to local/regional tissues such as the nerves and lymph nodes, assessing surgical margins, and confirming/establishing the diagnosis in cases where the preoperative FNA biopsy was not diagnostic or was equivocal.
Staging
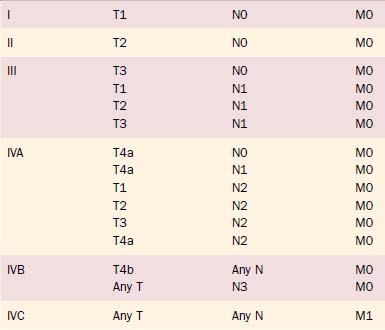
Clinical staging of salivary gland cancers is important for prognosis and treatment decisions. The 2003 TNM staging classification for major salivary gland cancers established by the American Joint Committee on Cancer (AJCC) is the classification most commonly used in the United States (Tables 88-3A and B). Minor salivary gland carcinomas are staged according to the anatomic site of origin (e.g., oral cavity, sinus, larynx). The staging guidelines are applicable to all forms of carcinoma; any nonepithelial tumor type is excluded.
Table 88-3A 2003 AJCC TNM Staging for Major Salivary Gland Cancer
| Primary Tumor (T) | |
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| T1 | Tumor is ≤2 cm in greatest dimension without extraparenchymal extension* |
| T2 | Tumor is >2 cm but not >4 cm in greatest dimension without extraparenchymal extension* |
| T3 | Tumor is >4 cm in greatest dimension and/or has extraparenchymal extension* |
| T4a | Tumor invades skin, mandible, ear canal, and/or facial nerve |
| T4b | Tumor invades skull base and/or pterygoid plates and/or encases carotid artery |
| Regional Lymph Nodes (N) | |
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Metastasis in a single ipsilateral lymph node, ≤3 cm in greatest dimension |
| N2a | Metastasis in a single ipsilateral lymph node, >3 cm but not >6 cm in greatest dimension |
| N2b | Metastases in multiple ipsilateral lymph nodes, none >6 cm in greatest dimension |
| N2c | Metastases in bilateral or contralateral lymph nodes, none >6 cm in greatest dimension |
| N3 | Metastasis in a lymph node, >6 cm in greatest dimension |
| Distant Metastasis (M) | |
| Mx | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
AJCC, American Joint Committee on Cancer.
* Extraparenchymal extension is clinical or macroscopic evidence of invasion of soft tissues. Microscopic evidence alone does not constitute extraparenchymal extension for classification purposes.
From Greene FL. AJCC Cancer Staging Manual. 6th ed. New York: Springer-Verlag, 2002.
Histopathology
Because salivary gland malignancies are remarkably diverse and heterogeneous, their behavior and resulting clinical management are highly dependent on their histologic type and, quite often, their grade. Thus knowledge of the types of tumors and their pathologic classification is critical for the clinician in order to provide proper treatment. Table 88-4 lists most of the critical pathology-related issues that the clinician needs to consider. The following section is intended to provide a succinct, but sufficient, discussion of the pathology of these tumors.
Table 88-4 Critical Pathologic Features for Assessing Malignant Salivary Gland Tumors
| Tumor Type | Unique Pathologic Issue(s) |
|---|---|
| Mucoepidermoid carcinoma | Grade |
| Adenoid cystic carcinoma | |
| Carcinoma ex pleomorphic adenoma | |
| Myoepithelial carcinoma | Grade |
| Basal cell adenocarcinoma | Grade |
| Adenocarcinoma NOS | Grade |
| Acinic cell carcinoma | |
| Small cell carcinoma | Primary vs. metastasis |
| Squamous cell carcinoma | Primary vs. metastasis |
| Lymphoma |
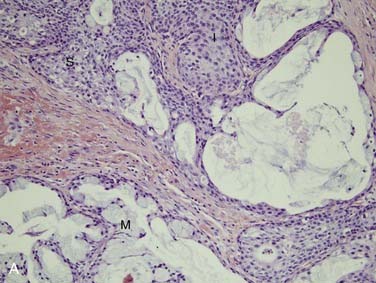
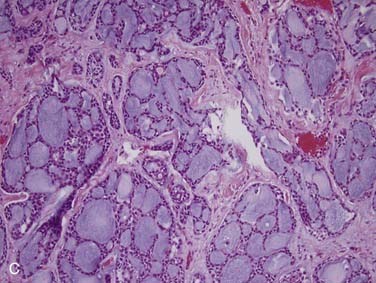
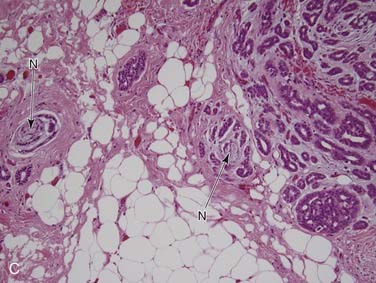
First, it is important to consider the normal histology of the glands because most tumors differentiate into the same cell types that are present in the normal gland. Salivary glands contain acini composed of either serous or mucous cells or a mixture of both. Serous cells are rounded or polygonal in shape and characteristically have abundant blue cytoplasmic granules, which are periodic acid–Schiff (PAS) positive. Mucous cells consist almost entirely of lightly basophilic intracytoplasmic mucus. The fluid secreted by the parotid gland is almost exclusively serous, while that from the sublingual gland is almost exclusively mucous and that from the submandibular gland is a mixture of serous and mucous. The ducts have cuboidal to columnar lining cells with abundant eosinophilic cytoplasm. They form tubular structures within the glands (Fig. 88-2). Both the acini and ducts have supportive cells called myoepithelial cells along their periphery. The parotid glands normally contain, on average, 10 to 20 intraglandular lymph nodes, a feature of great importance because many parotid masses represent metastases to these lymph nodes from primary skin or other cancers of the head and neck. These nodes have an otherwise typical appearance to any other nodal tissue in the body. The submandibular (also referred to as submaxillary) and sublingual glands have no intraglandular lymph nodes.
Neoplasms of the salivary gland can be roughly classified on the basis of the type of normal salivary gland cell toward which they differentiate. Neoplasms can differentiate toward the acinar, ductal, or myoepithelial cells. Practically speaking, though, most of them have dual differentiation—specifically, most salivary gland neoplasms have some myoepithelial differentiation.29 Also, most of the benign neoplasms have a malignant counterpart (e.g., pleomorphic adenoma and carcinoma ex pleomorphic adenoma, basal cell adenoma and basal cell adenocarcinoma, myoepithelioma and myoepithelial carcinoma). The number of different malignant epithelial tumors in the World Health Organization (WHO) classification has increased greatly over the past 50 years and now includes 24 entities.21 Between 21% and 46%3 of salivary gland tumors are malignant, including 15% to 32% of parotid, 41% to 45% of submandibular, 70% to 90% of sublingual, and 50% of minor salivary gland tumors.21 It is important to note that the incidence of particular histologic types is dependent on the site. For example, polymorphous low-grade adenocarcinoma virtually never occurs in the major glands.18,30
Mucoepidermoid Carcinoma
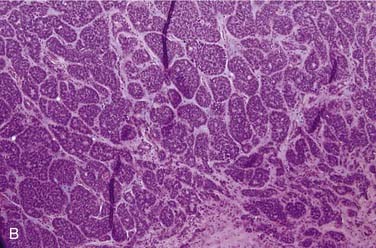
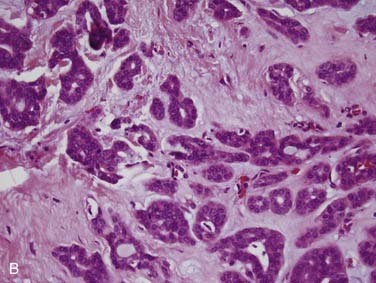
Mucoepidermoid carcinoma is the most common salivary gland malignancy.3,31–33 The majority of cases occur in the major salivary glands,34 but mucoepidermoid carcinoma can also arise from minor salivary glands in the oral cavity, particularly in the hard palate, buccal mucosa, lip, and retromolar trigone.35 Rarely, they can also arise intraosseously in the mandible and maxilla, but mucoepidermoid carcinomas of this location are considered odontogenic in origin and have a less aggressive clinical behavior.36,37 Clinically, mucoepidermoid carcinomas are slightly more common in women and have a mean age of approximately 45 years, but they can also occur in children.31 In fact, they are the most common pediatric salivary gland carcinoma.38 Patients usually present with a painless, slow-growing mass. Intraoral tumors may mimic a mucocele or vascular lesion clinically by presenting as a blue-red superficial nodule.3
Grossly, these tumors are not distinct. They usually have both solid and cystic components, often with mucinous material within the cysts.39 This is what sometimes imparts a bluish color to them, which can mimic the appearance of a mucocele in the oral cavity. Microscopically, their hallmark is the presence of three cell types: mucous, squamoid (or epidermoid), and intermediate (Fig. 88-3A). The architecture is usually a mixture of cystic (see Fig. 88-3B) and solid elements, the latter with sheets (see Fig. 88-3C), nests, or ductlike structures. The mucous cells have abundant, light blue mucin in their cytoplasm and nuclei displaced to the periphery. The mucin is usually obvious, but, in cases where it is scant, special stains such as PAS, mucicarmine, or Alcian blue can be used to highlight it. The squamoid cells are large with abundant pink cytoplasm, and, although they look somewhat squamous in appearance, true keratinization in mucoepidermoid carcinoma is rare.40 Intermediate cells typically have more modest amounts of pink or clear cytoplasm. The proportion of cell types varies quite a bit among tumors. Intermediate cells usually predominate and mucous cells usually line cystic spaces. Cytologic atypia varies from minimal to quite prominent.41–43 Immunohistochemistry is of limited to no utility in the diagnosis.39
The differential diagnosis, particularly in tumors arising along mucosal sites, includes necrotizing sialometaplasia, an uncommon non-neoplastic lesion of the hard palate with reactive changes in minor salivary glands44 and, more importantly, adenosquamous carcinoma, which is an aggressive variant of squamous cell carcinoma. Adenosquamous carcinomas are high grade, have definitive squamous differentiation usually with keratinization (unlike mucoepidermoid carcinoma), and often have surface mucosal squamous dysplasia, a feature that mucoepidermoid carcinoma does not have.39
Grading of mucoepidermoid carcinoma is important and correlates strongly with clinical behavior, although reproducibility and consistency are major issues, and no particular system has been universally accepted.41–4345 Clinical staging is as important as histologic grade, so the two should be considered in tandem. In general, low-grade lesions have a prominent cystic component and abundant well-differentiated mucous cells with little cytologic atypia and low mitotic activity. High-grade lesions are more solid with squamoid and intermediate cells predominating. They also have cytologic atypia, mitotic activity, necrosis, and infiltrative growth. A number of different grading schemes (well reviewed by Luna39) have been reported over the years. The most used grading system, originally designed by Auclair and colleagues,41,43 uses a three-tiered score based on a number of histologic features (Table 88-5). The original grading system was initially criticized for a tendency to “under grade” the tumors when others demonstrated that a significant number of low-grade mucoepidermoid carcinomas developed progressive disease. The later modification by Brandwein and colleagues42 (see Table 88-5) refined the grading system such that none of the tumors classified as low grade in their study went on to have progressive disease. Low-grade mucoepidermoid carcinoma, as strictly defined by their criteria, rarely ever metastasizes or results in death of the patient.41–43
Table 88-5 Grading Systems for Mucoepidermoid Carcinoma
| Parameter (Auclair et al, 1992) | Point Value |
|---|---|
| Cystic component <20% | +2 |
| Neural invasion | +2 |
| 4+ mitoses/10 HPF | +3 |
| Necrosis | +3 |
| Anaplasia | +4 |
| Grade | Point Score |
|---|---|
| Low (I) | 0-4 |
| Intermediate (II) | 5-6 |
| High (III) | 7 or more |
| Parameter (Brandwein et al, 2001) | Point Value |
|---|---|
| Cystic component <25% | +2 |
| Tumor front invades in small nests and islands | +2 |
| Pronounced nuclear atypia | +2 |
| Lymphatic and/or vascular invasion | +3 |
| Neural invasion | +3 |
| Necrosis | +3 |
| 4+ mitoses/10 HPF | +3 |
| Bony invasion | +3 |
| Grade | Point Score |
|---|---|
| Low (I) | 0 |
| Intermediate (II) | 2-3 |
| High (III) | 4 or more |
From Auclair PL, Goode RK, Ellis GL. Mucoepidermoid of intraoral salivary glands. Cancer. 1992;69:2021-2030; Brandwein MS, Ivanov K, Wallace DI, et al. Mucoepidermoid carcinoma. A clinicopathologic study of 80 cases with special reference to histological grading. Am J Surg Pathol. 2001;25(7):835-845.
In addition to grade, location of the primary tumor is potentially important in predicting clinical behavior. Several studies have shown that low-grade mucoepidermoid carcinomas of the submandibular gland recur and metastasize much more frequently than those of the parotid or minor salivary glands.40,43 Whether or not this represents truly different biology, it merits the aggressive and thorough resection of any submandibular gland primary malignancy, especially for known mucoepidermoid carcinoma.42
Adenoid Cystic Carcinoma
Adenoid cystic carcinoma (ACC) is one of the more common,3,32,33 and certainly more recognizable salivary gland tumors, being notorious for its infiltrative growth and slowly progressive behavior with recurrences and spread over a protracted course of many years.46 These tumors essentially occur with an even distribution among all salivary gland sites,32 although the total number of minor salivary gland cases outnumber those of major salivary glands when all sites are considered together.47–50 These malignancies occur with an equal incidence in men and women over a wide age range with the peak incidence between 50 and 60 years of age.47–4951
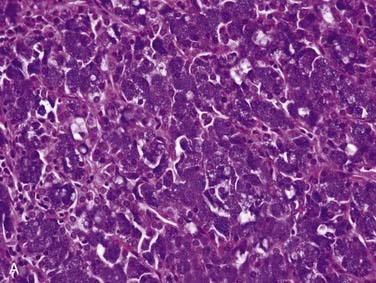
Grossly, ACCs are solid, light tan, firm and well-circumscribed, but unencapsulated, tumors. Microscopically, there are three growth patterns: tubular, cribriform, and solid. The tubular form consists of small tubules sitting in a pink, hyalinized, and hypocellular stroma (Fig. 88-4A). The solid variant has only rounded lobules of tumor cells with no, or almost no, glandlike structures and without a defined architecture (see Fig. 88-4B). The classic and most easily recognized pattern, though, is the cribriform (“Swiss cheese”-appearing) variant. Nests of cells are arranged around glandlike spaces that consist of PAS-positive blue or pink material (see Fig. 88-4C). The central spaces look like glandular lumina but are actually extracellular cavities containing reduplicated basement membrane (or ground substance) material52,53 and myxoid material that is produced by the tumor cells, rather than true epithelial mucin. The cells in ACC are markedly basaloid in appearance, with little cytoplasm and round-to-oval nuclei that are dark and hyperchromatic without nucleoli (see Fig. 88-4A).48 They are quite uniform in size and show little mitotic activity, except for the solid type, where mitotic activity can range from barely to extremely prominent. A rim of inconspicuous myoepithelial cells with clear cytoplasm is also usually present.29,54
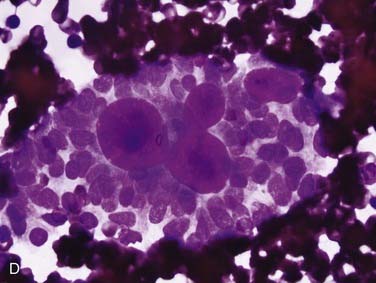
FNA frequently yields characteristic results in ACC. Aspirates have tumor cells with scant cytoplasm and round, regular nuclei in sheets and clusters. These features by themselves are not characteristic and actually are so bland that they might suggest a benign lesion. However, the finding of well-defined, round “cylinders” and/or spheres of acellular stroma accompanying these cells is typical, but not 100% specific, for ACC. The cells often “cling” to these rounded structures (see Fig. 88-4D).
Histologic grading has yielded conflicting results in the literature for predicting prognosis. Clinical stage, on the other hand, holds a great deal of prognostic information and, as such, should be considered as much or more than grade in clinical management.51 Also, as with mucoepidermoid carcinoma, ACC of the submandibular gland has a much more aggressive clinical course than ACC of other sites.49,51 The most accurate prognostic information has been garnered when tumors are not only graded by the predominant pattern (tubular, cribriform, or solid) but are more specifically graded as follows: tubular with or without some cribriform areas (grade I), cribriform with no tubular areas and with less than 30% solid areas (grade II), and cribriform with greater than 30% solid areas (grade III).49,55 A frequent histologic feature of ACC is perineural invasion, which is identified in approximately 70% to 75% of cases.49,56 Although somewhat inconsistent in the literature, many have correlated this finding with a worse prognosis, particularly when involving a major nerve trunk.46,57–59
The diagnosis of ACC is generally straightforward from routine hematoxylin and eosin examination. However, it can be difficult on small biopsies because one may not be able to appreciate the architecture and infiltrative growth. The differential diagnosis includes other salivary gland neoplasms (such as polymorphous low-grade adenocarcinoma or pleomorphic adenoma), basaloid squamous cell carcinoma, or high-grade neuroendocrine carcinoma.54 Although beyond the scope of this text, immunohistochemistry is not usually necessary but can occasionally be helpful in differentiating among these tumor types.60,61
Polymorphous Low-Grade Adenocarcinoma
Polymorphous low-grade adenocarcinoma (PLGA) is a unique, low-grade neoplasm first recognized as a distinct entity in the mid 1980s.62 PLGAs arise almost exclusively from the minor salivary glands and, in most series, are the second most common minor salivary gland carcinoma.35,46 The most common site for these neoplasms is the palate,63 particularly at the junction of the hard and soft palates. Other sites include the upper lip, buccal mucosa, and posterior third of the tongue.18 There are only rare reports of PLGA arising in major salivary glands.30 PLGA is twice as common in women and tends to present in the fourth to sixth decades as a slow-growing mass that may have been present for years.18,63 They are often asymptomatic.18 On gross examination, they are circumscribed, unencapsulated, pale yellow or tan masses that range from 1 to 3 cm. Microscopically, the architectural features of this tumor are quite variable, as the name would suggest. They are well circumscribed but unencapsulated and may appear as solid nests in lobules, cribriform glandlike structures, or ductlike arrangements. A common appearance within the tumor is concentric whorling of the nests around each other in a single-file arrangement. This has been termed the “eye of the storm” pattern (Fig. 88-5A). Stromal hyalinization with a slate-gray coloration is also characteristic.18 Tumor cells are quite regular with moderate eosinophilic cytoplasm and characteristic round-to-oval, extremely regular nuclei with open chromatin (see Fig. 88-5B). There is little mitotic activity and no necrosis. The periphery of the tumor shows markedly infiltrative growth and most cases have perineural invasion (see Fig. 88-5C).18,63,64
The differential diagnosis includes pleomorphic adenoma and ACC. The myxochondroid areas of pleomorphic adenoma are not seen in PLGA, and, although minor salivary gland pleomorphic adenomas are unencapsulated, they will not show the infiltrative growth pattern of PLGA and will not have perineural invasion. One particular immunostain that may be useful in difficult cases is worth mentioning here. Curiously, it has been recognized that almost all pleomorphic adenomas will be positive for glial fibrillary acidic protein (GFAP), whereas almost all PLGA are negative.65–68 ACC is important to differentiate from PLGA. This is done primarily by cytologic features because ACCs have basaloid cells with dark chromatin and little cytoplasm, whereas PLGA cells have moderate eosinophilic cytoplasm and nuclei with open chromatin.
PLGA is a low-grade malignancy, and histologic grading is not applicable. Conservative resection is the treatment of choice. Local recurrence occurs in 10% to 15% of patients.18,63,69 Because lymph node metastases are distinctly uncommon (literature rates of around 10% or less), neck dissection is only recommended with significant clinical adenopathy or needle-proven metastasis. This likely overestimates the occurrence, however. With strict histologic criteria for PLGA, nodal metastases are extremely rare.18 Distant metastases are equally as uncommon,70 and patients have an excellent long-term prognosis.18,63,69 In fact, few patients have been documented to die from this tumor and only after prolonged periods.18,63
Acinic Cell Carcinoma
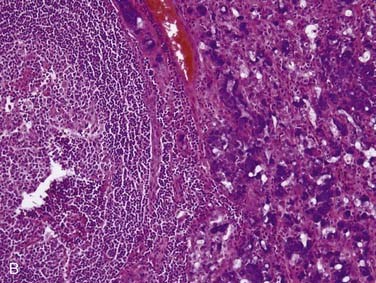
Acinic cell carcinoma, as the name implies, is a tumor with cells showing differentiation toward cells of the normal salivary gland acini (see Fig. 88-2). However, these tumors also show evidence of ductal and myoepithelial differentiation, an important feature to keep in mind. They are uncommon, comprising 1% to 3% of all tumors and approximately 10% of all malignant ones.3,32,33,71 More than 90% occur in the parotid,72,73 with the rest scattered among minor salivary gland sites and rarely the submandibular gland.71,74 Occurring over a wide age range from children to the elderly, they are relatively evenly distributed in the second through seventh decades75 with a peak in the third decade.73 Of note, these tumors are the second most common childhood salivary gland malignancy. They present as a slowly growing mass, which is only occasionally painful and rarely associated with a facial palsy.71,73
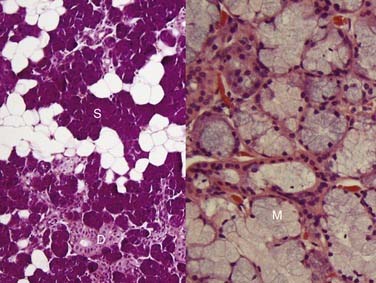
Grossly, acinic cell carcinoma presents as a single, usually circumscribed, rubbery, solid mass with up to one third showing cystic degeneration.75 Microscopically, they are highly variable, a feature of these tumors that has always led to angst in the diagnosing pathologist. The four principal histologic patterns are solid/lobular, microcystic, papillary-cystic, and follicular. Small tumors can be easily missed because the acinar cells are so well differentiated that they blend into the surrounding normal gland. Two features are classic, however. The first is the characteristic acinic cell, which has blue cytoplasm with abundant serous-type granules and a small, round, eccentrically placed nucleus (Fig. 88-6A). PAS stains will be strongly positive in these cells. A number of other cell types are seen including pink, clear, and vacuolated cells, such that most tumors are a mixture of different cell types (see Fig. 88-6A). The second classic feature is a dense lymphoid infiltrate with germinal centers (see Fig. 88-6B). The periphery of the tumors may or may not be infiltrative but are often “pushing” in nature (see Fig. 88-6C). Although one would assume that lack of an infiltrative border might signal a benign or low-grade lesion, there is no benign equivalent for this lesion (i.e., there is no known benign acinic cell neoplasm). “Dedifferentiation” has been described in a small subset of acinic cell carcinomas. This consists of tumors with areas of classic, “conventional” acinic cell carcinoma and an intermixed undifferentiated carcinoma in sheets with large, pleomorphic cells, brisk mitotic activity, and often necrosis.76–78
FNA biopsy of acinic cell carcinoma is fraught with difficulty because of the resemblance to normal tissue.27 The diagnosis rests on finding a cellular specimen with sheets and clusters of large acinar cells with abundant granular cytoplasm and central round, regular nuclei. One should not see normal ductal epithelial cells or significant amounts of adipose tissue (both of these latter findings are from normal salivary gland tissue), but there is often a prominent background component of lymphocytes.
Surgical resection with negative margins is the most important therapy. These tumors will recur in approximately one third of cases.71,73 Although classically regarded as low-grade malignancies, 10% to 15% of these tumors will metastasize locally to regional lymph nodes or distantly to the lung and bones. Acinic cell carcinomas are also notorious for recurrence and spread years beyond the primary presentation and for a protracted clinical course such that survival curves do not flatten out until after a decade.79 Survival is approximately 80% at 5 years and 70% at 10 years.71,75,79 Grading of these tumors has not correlated well with behavior, and there is not good agreement on histologic features within the proposed grading systems. Very bland, low-grade-appearing tumors can metastasize almost as frequently as tumors with more aggressive features. However, “dedifferentiation” (where there is a mixture of typical acinic cell carcinoma with a much higher-grade carcinoma) has consistently been associated with a poor outcome.76–78
Malignant Mixed Tumors
“Malignant mixed tumor” is the broad term that is used to encompass three different salivary gland malignancies: true malignant mixed tumor (or carcinosarcoma), carcinoma ex pleomorphic adenoma, and metastasizing mixed tumor. As a group, these account for approximately 3% to 5% of all salivary gland malignancies,3,32 and carcinoma ex pleomorphic adenoma is by far the most common of these.
True Salivary Gland Malignant Mixed Tumor (or Carcinosarcoma)
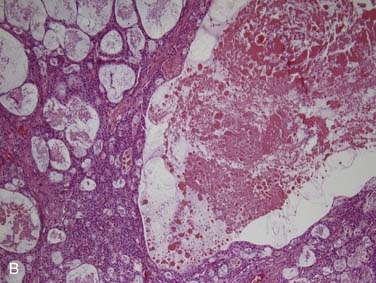
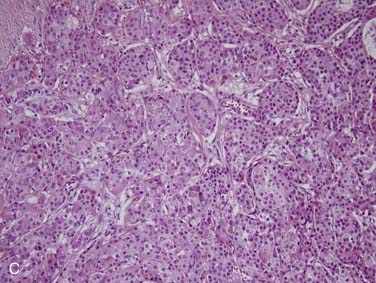
True salivary gland malignant mixed tumor (or carcinosarcoma) is a malignant neoplasm that consists of distinct carcinomatous and sarcomatous components. These tumors are rare,46 comprising approximately 1% of all salivary gland malignancies.32,35 The mean age of presentation is 58 years. Two thirds of cases arise in the parotid gland, approximately 15% in the submandibular gland, and 15% in the palate.80–84 Grossly, the findings are not distinct. The mass may be circumscribed or ill defined. It is commonly firm and tan-white with hemorrhage and necrosis and, on occasion, grittiness or calcification. Microscopically, there is an intimate admixture of the two components (carcinoma and sarcoma), but the amounts of each can vary widely. The carcinoma component can have any type of differentiation but typically takes the form of high-grade salivary duct carcinoma or undifferentiated carcinoma (Fig. 88-7A).80 The sarcomatous component is usually a chondrosarcoma (see Fig. 88-7B) or osteosarcoma,80 but fibrosarcoma, leiomyosarcoma,83 or even liposarcoma85 is possible. Treatment consists of wide local excision combined with radiotherapy. The tumors are aggressive with up to two thirds of patients dying of either local recurrence or, commonly, distant metastasis to the lungs or bone, usually within a period of 30 months.80
Carcinoma ex Pleomorphic Adenoma
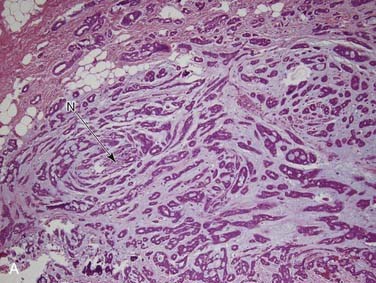
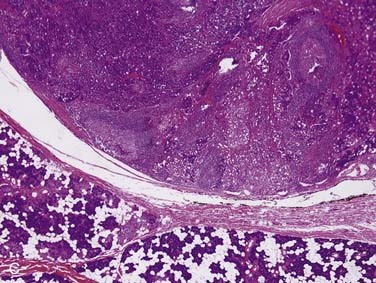
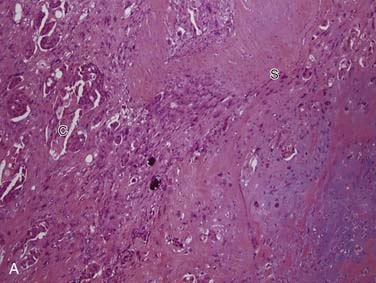
Carcinoma ex pleomorphic adenoma is defined as a pleomorphic adenoma (or mixed tumor) in which, or with which, a carcinoma is present. It accounts for more than 95% of malignant mixed tumors but is still relatively uncommon. The critical thing to remember is that this term encompasses a heterogeneous group of tumors because the carcinoma component can be of any form, from very low grade to the highest grade types.86 These tumors are most common in the parotid gland, followed by the submandibular gland, the minor salivary glands, and the sublingual gland.87 Although there is a wide age range, most patients are in their sixth and seventh decades, approximately 1 decade later than the peak age for pleomorphic adenomas. The classic history is a patient with a longstanding mass that suddenly undergoes rapid growth over a period of several months.87,88
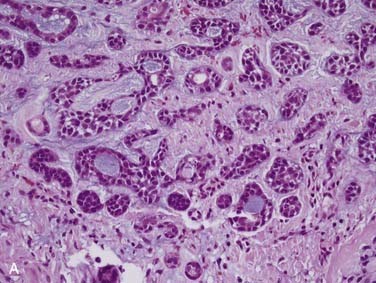
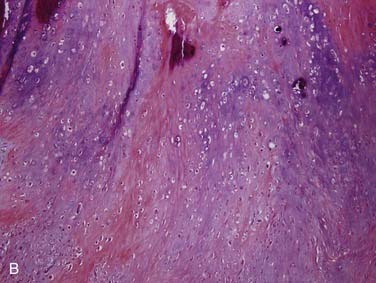
Grossly, these tumors can reach up to 25 cm and the average is more than twice that for pleomorphic adenomas.86 They usually consist of a tan-yellow, firm mass with ill-defined, infiltrative borders, and there may be a component of nodular, translucent blue or gray tumor representing the preexisting pleomorphic adenoma.88 Microscopically, the proportions of the two components vary quite a bit from tumor to tumor. The pleomorphic adenoma component has typical features with myxoid, blue stromal areas, a variable proliferation of epithelial elements, including ductlike structures and sheets of myoepithelial cells, and chondroid areas reminiscent of cartilage (Fig. 88-8A
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree