CHAPTER 96 Malignant Neoplasms of the Oral Cavity
Etiology
Tobacco and alcohol consumption are considered the most common preventable risk factors associated with the development of oral cavity squamous cell carcinoma. In addition, this relationship is synergistic, with alcohol serving as a promoter for the carcinogenic effects of tobacco. When compared with nonsmokers, smoking confers a 1.9-fold risk to males and 3-fold risk to females for development of head and neck squamous cell carcinoma. The risk is directly proportional to the years spent smoking and the number of cigarettes smoked per day. Alcohol alone confers a 1.7-fold risk to males drinking 1 to 2 drinks per day compared with nondrinkers. This risk rises to more than 3-fold for heavy drinkers. Individuals who both smoke (two packs per day) and drink (four units of alcohol per day) are 35 times more likely to develop cancer when compared with controls.1 The reported incident rates of developing head and neck cancer based on smoking status are higher in males than females, respectively (nonsmokers 24.4 : 4.8, former smokers 36.9 : 17.2, and current smokers 147.3 : 75.7); however, hazard ratios suggest smoking leads to a higher proportion of females (12.96 for females, 5.45 for males) eventually developing head and neck cancer with the same risk exposure.2 Users of smokeless tobacco have a 4-fold increased risk of oral cavity carcinoma compared with nonusers.3
Tobacco is the leading preventable cause of death in the United States and is responsible for one out of every five deaths.4 As of 1999, approximately one quarter of U.S. adults used tobacco products (25.7% of males and 21.5% of females).5 The evidence supporting the benefit for head and neck cancer patients to cease smoking is compelling. In a study by Moore, 40% of patients who continued to smoke after definitive treatment for an oral cavity malignancy went on to recur or develop a second head and neck malignancy. For patients who stopped smoking after treatment, only 6% went on to develop a recurrence.6 Induction of specific p53 mutations within upper aerodigestive tract tumors has been noted in patients with a history of tobacco and alcohol use.7
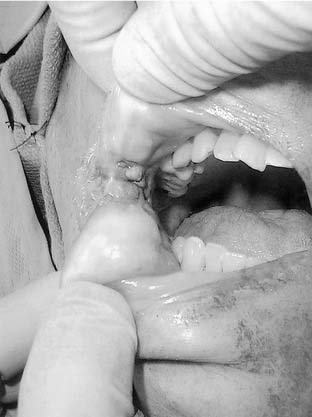
When smoking status in patients with head and neck squamous cell carcinoma is assessed, differences exist between the two populations (smoker vs. nonsmoker). Koch and colleagues noted that nonsmokers were represented by a disproportionate number of women and were more frequently at the extremes of age (younger than 30 or older than 85 years of age). Tumors from nonsmokers presented more frequently in the oral cavity, specifically within the oral tongue, buccal mucosa, and alveolar ridge. Smokers presented more frequently with tumors of the larynx, hypopharynx, and floor of the mouth (Fig. 96-1). Common genetic alterations such as loss of heterozygosity at 3p, 4q, and 11q13 and the overall number of chromosomal microsatellite (repeated base sequences) losses were significantly more likely in the tumors of smokers. In addition, the rate of p53 mutations was markedly increased in this group of patients.8 Smoking and habitual use of alcohol have also been associated with p15 gene methylation.9 Former smokers, defined as those individuals who had quit more than 10 years earlier, demonstrate a genetic profile more consistent with nonsmokers.8 Additionally, multiple mesenchyme-associated, stroma-specific, genetic alterations may have a significant role in the development and progression of smoking-related head and neck cancers.10
In India and Southeast Asia, the product of the Areca catechu tree, known as a betel nut, is chewed in a habitual manner and acts as a mild stimulant similar to that of coffee. The nut is chewed in combination with lime and cured tobacco as a mixture known as a quid. The long-term use of the betel nut quid can be destructive to oral mucosa and dentition and has been demonstrated to be highly carcinogenic.11 Another habit associated with oral malignancy is that of reverse smoking, where the lighted portion of the tobacco product is within the mouth during inhalation. The risk of hard palate carcinoma is 47 times greater in reverse smokers compared with nonsmokers.12
The human papillomavirus (HPV) is an epitheliotropic virus detected to varying degrees within samples of oral cavity squamous cell carcinoma. Infection alone is not considered sufficient for malignant conversion; however, results of multiple studies suggest a role of HPV in a subset of head and neck squamous cell carcinoma.13
Environmental ultraviolet light exposure has been associated with the development of lip cancer. The projection of the lower lip, as it relates to this solar exposure, has been implicated in the pathogenesis of squamous cell carcinomas arising on the vermilion border of the lower lip (Fig. 96-2).14 Pipe smoking has also been associated with the development of lip carcinoma. Mechanical irritation, thermal injury, and chemical exposure are suspected etiologic factors contributing to lower lip cancer development in pipe-smoking patients.15
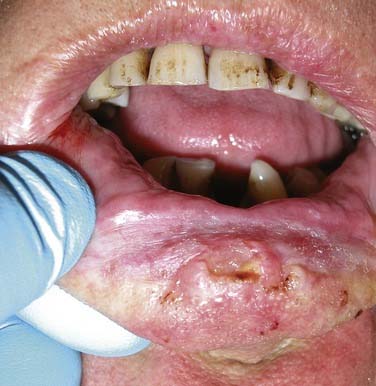
Figure 96-2. Lower lip ulcerative squamous cell carcinoma extending toward the gingivobuccal sulcus.
Other entities associated with oral malignancy include Plummer-Vinson syndrome (achlorhydria, iron deficiency anemia, and mucosal atrophy of the mouth, pharynx, and esophagus); chronic infection with syphilis; ill-fitting dentures; and long-term immunosuppression (30-fold increase with renal transplant). Although evidence linking HIV infection to squamous cell carcinoma of the head and neck is lacking, Kaposi’s sarcoma may arise in the oral cavity.16
Molecular Biology
H-ras, a member of the ras gene family, encodes a plasma protein (p21), which has a role in the transduction of mitogenic signals to the intracellular environment. A synergism between H-ras and HPV is believed to play a role in the induction of squamous cell carcinoma. In human studies, 22% of oral squamous cell carcinomas expressed H-ras, whereas 11% expressed H-ras and HPV. All specimens of oral verrucous carcinoma that expressed H-ras (25%) demonstrated evidence of HPV DNA.17
HPV is a mucosotropic virus that has been closely associated with cervical carcinoma in women, such that HPV 16 and 18 are considered carcinogenic. In oral cavity specimens, variable detection has been reported with rates ranging from 19% to 78%.18,19 Findings of HPV 16 within normal tissue at the margins of tumor specimens have suggested that infection is not a sole event preceding malignant transformation. Rather, it has been postulated that HPV may play a role in the early events of carcinogenesis. HPV oncoproteins E6 and E7 have the ability to bind and degrade tumor suppressor gene products of p53 and pRB, respectively. This binding can impair the capacity of the cell cycle to arrest for the repair of DNA damage and results in an accumulation of genetic changes assisting transformation.13
Smith and colleagues detected a significant increase in the presence of HPV DNA within oral cavity carcinoma samples (15%) compared with controls (5%). In addition, they noted that HPV (odds ratio [OR]-3.7) was a risk factor for carcinoma, independent of tobacco (OR-2.63) and alcohol use (OR-2.57).20 Maden and coworkers noted an increased risk for oral cavity carcinoma with the presence of HPV 6 and HPV 16. Similar to Smith and colleagues, they also demonstrated HPV infection to be a risk factor, independent of age and tobacco and alcohol use.21
Costa and colleagues examined resectable oral cavity carcinomas for biomarkers predictive of chemosensitivity before primary surgical treatment. They found that tumors negative for glutathione S-transferase (GST-π) were more responsive to cisplatin and 5-fluorouracil than tumors that were GST-π-positive. They also noted that lack of bcl-2 expression was consistent with an improved 3-year disease-free survival. No relationship between p53 activity and chemotherapeutic response was observed.22
Overexpression of mutant p53 has been associated with carcinogenesis at multiple sites. Point mutations in p53 have been reported in up to 45% of head and neck carcinomas. Brennan and colleagues have evaluated the concept of primary site margin assessment of specimens on the basis of p53 staining to guide the need for further treatment.23,24 Koch and others have noted that p53 mutation is a key event in the malignant transformation of greater than 50% of head and neck squamous cell carcinomas in smokers.8
Her2-Neu overexpression has been demonstrated in a subset of squamous cell carcinomas.25 Expression of Her2-Neu and heat shock protein (HSP)-27 have been associated with regional treatment recurrence in patients with oral cavity and oropharyngeal carcinomas treated with primary radiotherapy.26
Koch’s tumor progression model has demonstrated that 3p is a site of early loss of heterozygosity, 4q is a late event, and 11q13 is the site of the cyclin D1 gene. Reviews of the head and neck literature have suggested the role for p53, EGFR, TGF-α, and cyclin D1 in predicting clinical outcome for patients with squamous cell carcinoma.27
The Akt receptor has been correlated with the histologic progression of oral premalignant lesions to oral carcinoma. Massarelli and colleagues28 demonstrated that phosphylated-Akt, when highly expressed, is associated with recurrence and shorter disease-free survival independent of a patient’s overall staging.
Epidemiology
Oral cavity malignancies (excluding lip lesions) account for 14% of all head and neck cancers.29 In a review (1985-1996) of the National Cancer Data Base (NCDB), patients diagnosed with an oral cavity cancer had a mean age at presentation of 64 years with a male predominance (60%). Squamous cell carcinoma (SCC) represented the majority of lesions (86.3%), and adenocarcinoma (5.9%), verrucous carcinoma (2.0%), lymphoma (1.5%), and Kaposi’s sarcoma (1.5%) accounted for the remainder. At the time of diagnosis, 55% of patients had early-stage lesions (stage I-II). Patients 35 years old or younger were more likely to present with oral tongue SCC than older patients (76.1% vs. 33%) and less likely to present with a floor-of-mouth SCC (10.5% vs. 35.9%). Of note, the NCDB data did not reveal a poorer overall prognosis for those patients diagnosed at a younger age. In fact, younger patients demonstrated a better 5-year survival (63.7%) than older stage-matched patients (51% for 36 to 65 years of age and 47.6% for older than 65 years of age). Blacks and lower income patients more frequently presented with advanced-stage lesions. Five-year survival was found to be worse for males, blacks, and older patients (older than 65 years). Adenocarcinoma presented more commonly in women than men and was most frequently diagnosed on the hard palate.30
A study by Schantz and Yu31 noted that the incidence for oral tongue carcinoma in patients younger than 40 years of age increased 60% from 1973 to 1984 and has since stabilized. During this same timeframe the incidence of carcinoma at all other oral cavity subsites remained constant or decreased.32 The rise was related to patients born during the years 1938 to 1948. Despite this increase, the 5-year determinant survival of these patients was better than that for older individuals. Although the exact cause for the increase was considered unclear, etiologies of increased smokeless tobacco and marijuana use and human papillomavirus (HPV) infection were considered possible factors.19
Anatomy
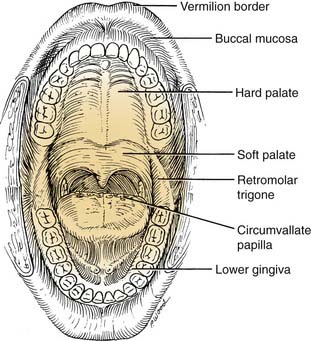
Because of the pivotal role the oral anatomy has on articulation and deglutition, treatment of an oral malignancy can have a significant impact on a patient’s quality of life. An understanding of the complex anatomic relationships among the salivary glands, maxilla, mandible, tongue function, and dentition are paramount to achieving satisfactory oncologic and functional results. The oral cavity extends from the vermilion border of the lip, posterosuperiorly to the hard palate–soft palate junction, inferiorly to circumvallate papillae (linea terminalis), and laterally to the anterior tonsillar pillars. It is divided into seven specific subsites. These subsites are the lips, dentoalveolar ridges, oral tongue, retromolar trigone, floor of mouth, buccal mucosa, and hard palate (Fig. 96-3).
The local, regional, and distant spread of oral cavity malignancies is dependent on the course of the neurovascular anatomy, lymphatic pathways, and the fascial planes of the head and neck. The latter serves as a barrier to the direct spread of tumor and can influence the pattern of local and regional lymphatic spread. In addition, perineural and angioinvasion can act as a conduit for the spread of head and neck malignancies. When present, these histologic findings can have a profound impact on the patient’s prognosis and long-term survival.33,34 Patients previously treated with either surgery or radiation for an oral malignancy may demonstrate atypical patterns of local invasion and regional spread of tumor.35 A brief discussion of the vascular and lymphatic anatomy pertinent to the oral cavity while reviewing the unique attributes of the oral cavity subsites is necessary before considering diagnostic and treatment options.
Arteriovenous Anatomy
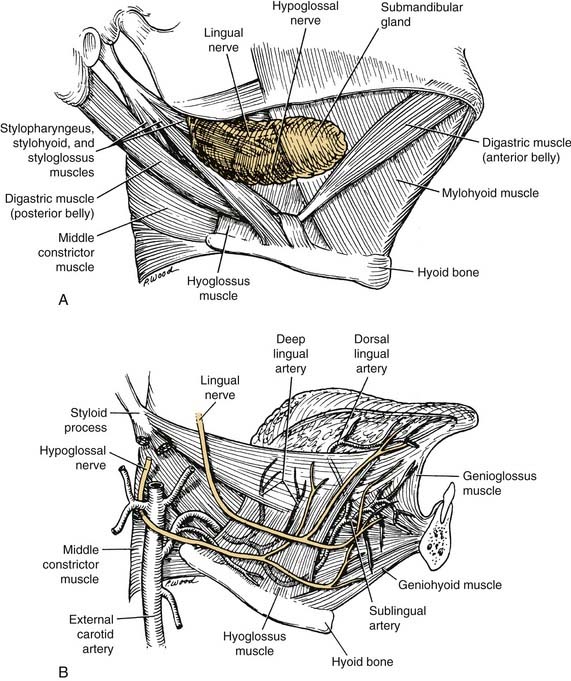
The arterial supply to the oral cavity includes multiple contributions from the external carotid artery. The lingual artery provides the majority of the vascular supply to the oral tongue and tongue base. Identification of the artery within the neck requires the exposure of the floor of the submandibular region. The artery is found deep to the hyoglossus muscle and requires the division of the muscle for maximal exposure. Superficial to the hyoglossus and deep to the mylohyoid muscle is the hypoglossal nerve and lingual veins (Fig. 96-4). An understanding of this relationship can aid in the localization of the artery when necessary for microvascular reconstruction or selective ligation. The vein, which closely approximates the nerve in this region, is prone to injury during dissection and necessitates care when attempting to obtain hemostasis if hypoglossal nerve injury is to be avoided.
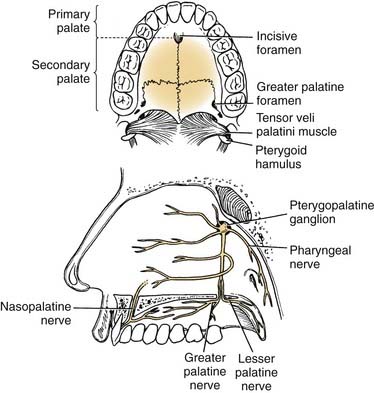
The hard palate blood supply is derived from the greater palatine and the superior alveolar arteries. After branching off the descending palatine artery at the greater palatine foramen, in the region medial to the second maxillary molar, the artery runs anteromedially within the soft tissue of the hard palate (Fig. 96-5). The venous drainage is to the pterygoid plexus and subsequently to the internal jugular venous system. The superior alveolar arteries (anterior, middle, posterior) arise as terminal branches after the transition of the internal maxillary artery to sphenopalatine artery at the pterygopalatine fossa. These arteries provide blood supply to the maxillary gingiva, alveolar ridge, and dentition.
Subsites of the Oral Cavity
Lips
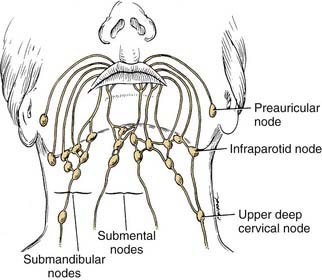
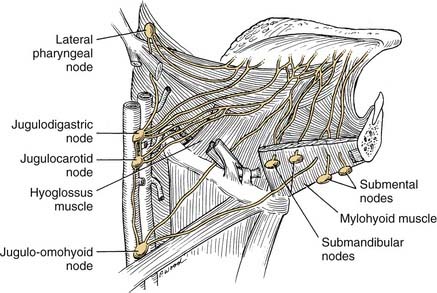
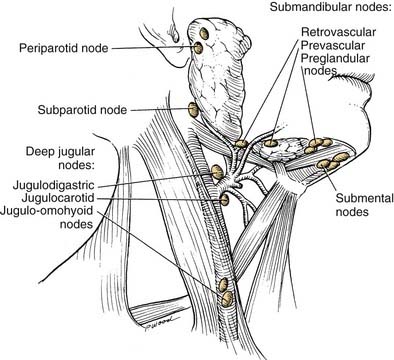
The lips represent a transition from external skin to internal mucous membrane that occurs at the vermilion border. The underlying musculature of the orbicularis oris, innervated by the facial nerve, creates a circumferential ring that allows the mouth to have a sphincter-like function. The sensation for the upper lip is supplied by the infraorbital nerve (CN V2), whereas the lower lip is provided by the mental nerve (CN V3). Lymphatics from both the upper and lower lips drain primarily to the submandibular lymph nodes, yet midline lower lip lesions may present with submental lymphatic spread. In addition, the upper lip may drain to preauricular, infraparotid, and perifacial lymph nodes (Fig. 96-6).
Alveolar Ridge
Oral Tongue
The lymphatic drainage of the oral tongue varies by the region within the tongue. The tip drains preferentially to submental nodes, whereas the lateral tongue drains primarily to the levels I and II (Fig. 96-7). However, it is important to note that a defined lymphatic pathway from the lateral tongue does exist and drains directly to the level III/IV nodal group. The base of the tongue drains to the upper cervical lymphatics. The lack of anastomoses between the anterior tongue lymphatics results in lateralized oral tongue lesions tending to drain ipsilaterally. This is not the case with base-of-tongue lesions where crossover and bilateral cervical lymphatic spread may readily occur.
Retromolar Trigone
This region is represented by the mucosa overlying the ascending ramus of the mandible toward the coronoid process. It is continuous with the buccal mucosa laterally and the anterior tonsillar pillar medially. The superior extent is the maxillary tuberosity and the anterior margin is the posterior aspect of the second mandibular molar. The same considerations that were true for alveolar lesions exist for this anatomic site, given the close approximation of mucosa to the underlying mandible. Lower lip paresthesia may be an indication of perineural invasion at the level of the mandibular foramen with these lesions. The sensation to this region is provided by the lesser palatine nerve and branches of the glossopharyngeal nerve. It is the presence of CN IX that causes patients with lesions within this area to present with referred otalgia. The primary lymphatic drainage for this region is to the upper cervical-jugulodigastric nodal group (Fig. 96-8).
Pathology
Certain histopathologic findings have significant implications on treatment. Tumor thickness, in particular with oral tongue carcinoma, has been the subject of many reports. Tumor thickness has been shown to have a direct relationship to the incidence of regional metastatic spread and survivorship.36 The degree of differentiation and the presence of vascular or perineural invasion have important prognostic implications and may warrant the use of postoperative radiation therapy.
Premalignant Lesions
Leukoplakia is white, mucosal-based keratotic plaque that cannot be wiped free from the underlying tissue. This is a clinical term without a definitive histologic definition. Leukoplakic lesions may demonstrate parakeratosis, hyperkeratosis, and acanthosis on histologic examination. Paradoxically, an increased risk of malignant transformation of leukoplakic lesions is seen more commonly in nonsmokers compared with smokers. If a leukoplakic lesion is associated with an area of dysplasia (in 1% to 3% of all lesions), the risk of progression to malignancy increases seven-fold. Banoczy followed 670 patients with leukoplakic lesions for 3 years and noted that 31% of lesions disappeared, 30% improved, 25% experienced no change, and 7.5% demonstrated local spread. Only 6% of lesions demonstrated eventual progression to squamous cell carcinoma.37 Syphilitic leukoplakias seen in tertiary syphilis have a higher reported rate of malignant transformation.38
Erythroplakia is a red mucosal plaque that does not arise from any obvious mechanical or inflammatory cause and persists after removal of possible etiologic factors. The associated risk for progression to carcinoma is significantly greater than that for leukoplakic lesions. Shafer and Waldron39 resected erythroplakic lesions in 58 patients and found that 91% had evidence of invasive, carcinoma in situ, or severe dysplasia within specimens.
Lichen planus, in particular the erosive subtype, has also been associated with the development of oral carcinoma. The etiology of lichen planus is unknown. T-cell lymphocyte infiltration is noted in this lesion, yet the development of lichen planus is not typically associated with immunologic disorders. The typical patient is a female in the fourth decade of life. Lesions demonstrate a lacy pattern of white striae. Atrophic lesions are red and smooth, whereas erosive lesions have depressed margins and are covered by a layer of fibrinous exudate. An estimated 1% risk of malignant change over 10 years has been reported.38
Squamous Cell Carcinoma and Variants
Several variants of squamous cell carcinoma exist and may be encountered in the oral cavity.
Sarcomatoid squamous cell carcinoma demonstrates a heterogeneous appearance with spindle-shaped cells interwoven with squamous cells. The cytologic behavior of this variant is considered aggressive with a reported metastatic rate of 37%.40 Basaloid carcinoma is also considered a high-grade variant with reported 64% incidence of regional metastases and 44% incidence of distant metastases, and it is associated with 38% mortality at 17 months.41 However, one report notes that when clinical T and N staging are matched, the 5-year disease-free survival associated with oral basaloid carcinoma is similar to that of poorly differentiated and moderately differentiated oral carcinomas.42
Other Pathologies
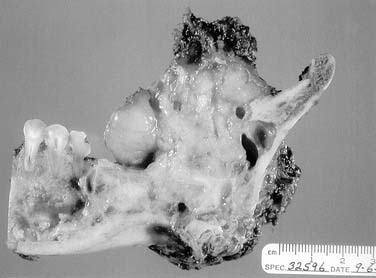
Sarcomas of the oral cavity usually arise in the mandible or hard palate and include osteosarcoma (Fig. 96-9), chondrosarcoma, malignant fibrous histiosarcoma, rhabdomyosarcoma, and liposarcoma. Patel and colleagues demonstrated a 5-year local control rate for head and neck osteosarcoma of 78% and overall survival of 70%. Patients presented with osteosarcoma within the maxilla and mandible with approximately equal frequency, 45% to 41%, respectively. Surgery was performed in all patients, and neoadjuvant chemotherapy was used in 68% of patients. Positive surgical margins were found to be the only significant negative predictor with respect to disease-specific survival.43
Kaposi’s sarcoma (KS) is the most common HIV-associated malignancy. The head and neck is the site for 63% of all presentations. KS may present as a mucosal or cutaneous lesion with a nodular or macular appearance and most commonly occurs on the hard palate. Despite being considered an incurable disorder, a variety of treatment options that may offer remission for limited disease are available. Local therapies include radiation, intralesional injection of chemotherapy, cryotherapy, alitretinoin gel, laser therapy, and surgical excision. Systemic chemotherapy is reserved for advanced disease.16
Desmoplastic neurotrophic melanoma is a nonpigmented lesion of the lower lip that presents as an ulceration and has a high incidence for perineural invasion. Mucosal melanoma typically presents as a pigmented lesion of the oral cavity that may be associated with preexisting melanosis. Pigmented lesions of the oral cavity should be considered for biopsy to exclude this diagnosis.44
Diagnostic Evaluation
History and Physical Examination
The head and neck physical examination should allow for accurate staging of the tumor, assess the patient’s functional capacity before treatment, and include a careful search for synchronous upper aerodigestive tract cancers. The physician should evaluate the dimensions of the index lesion and the potential anatomy involved by direct spread of tumor (Fig. 96-10). The lesion should be palpated to assess for fixation to the underlying periosteum suggesting potential mandibular or maxillary involvement. Determination of midline extension, regional lymphatic spread, and the need for reconstruction should be considered. The appropriateness of specific donor sites for free and/or pedicled flap reconstruction should also be assessed.
Future screening for oral cancer may be possible with outpatient biomarker laboratory assays. St. John and colleagues45 have described the use of an assay for salivary interleukin (IL)-8 and serum IL-6 with promising results in patients with oral cavity and oropharyngeal carcinoma.
Preoperative Assessment
Imaging to assess the primary extent of tumor and cervical lymph nodes includes either computed tomography (CT) or magnetic resonance imaging (MRI). CT is best for demonstrating cortical bone erosion and lymph node metastases. MRI is favored for demonstrating soft tissue invasion by tumor and extension into medullary bone. A Panorex may be obtained preoperatively in patients with suspected mandibular invasion and can assist dental evaluation. Dentascan imaging, using a dental CT-based software program, can be helpful in assessing the likelihood of mandibular invasion, with a reported sensitivity of 95%.46 Goerres and colleagues noted the assessment of cortical invasion of the mandible was best assessed by conventional contrast-enhanced CT (as well as the CT component of PET-CT) with a sensitivity of 100% and accuracy of 97%.47 The extent of imaging is frequently driven by physician preference and the complexity of the presenting lesion. Patients with T1 lesions, without clinical lymphadenopathy, may not require extensive imaging to aid operative decision making.
Staging
Staging for oral cavity malignancies is defined by the American Joint Committee on Cancer (AJCC)48 and follows TNM (primary Tumor, regional Nodal metastases, distant Metastasis) staging format (Table 96-1).
Prognosis
Patients with tumors that display “pushing” margins (when the peripheral invasive margin is assessed as it relates to the surrounding soft tissue) as opposed to “infiltrative” growth patterns have better outcomes.49 In addition, the depth of invasion of oral squamous cell carcinoma and its relationship to the risk of regional metastasis and 5-year survival has been examined. For lesions with a depth of invasion less than 2 mm, 13% of patients demonstrated regional metastases and a 95% 5-year survival was noted. For lesions with a 2- to 9-mm depth of invasion, 46% of patients had regional metastases and the 5-year survival rate decreased to 85%. When depth of invasion increased to greater than 9 mm, the risk of lymph node metastasis increased to 65% and the 5-year survival decreased to 65%.50
Loss of cadherin expression (E-cad and P-cad) as it relates to oral cavity squamous carcinoma correlates with the invasive potential of a cancer and locoregional disease recurrence. In particular, P-cad expression is considered an independent prognostic marker.51 Marcus and colleagues52 demonstrated that monocyte chemoattractant protein 1 was up-regulated in oral cavity carcinoma and the resultant increase in macrophage content was statistically associated with regional metastasis, extracapsular spread, and advanced-stage disease. Tissue inhibitor of metalloproteinase 1 and collagen type 11 alpha-1 have also been demonstrated to be increased in expression in oral cavity and oropharyngeal cancers that are metastatic.53
Laimer and colleagues54 noted that a high STAT1 activation (>35%) in oral cavity carcinoma specimens was associated with a negative lymph node status and a better prognosis in patients receiving adjuvant chemotherapy when compared with nonexpressing counterparts. Xie and colleagues55 noted that patients with higher numeric aberrations of chromosome X and 11 had shorter disease-specific survival. Podoplanin, a glycoprotein important in lymphangiogenesis, is expressed preferentially in oral tongue carcinoma. High levels of podoplanin have been shown to be predictive of lymph node metastasis and shortened disease-specific survival.56 Additionally, intratumoral lymphangiogenesis in patients with early-stage oral carcinoma has been correlated with locoregional recurrence.57
The 5-year distant metastatic rate in patients with oral carcinoma after treatment is approximately 10%. This rate increases from 6.6% in patients with locoregional control to 21.4% in patients who develop a locoregional recurrence. Additional risk factors noted at the time of initial treatment associated with the development of distant metastasis in the two groups were greater than or equal to five positive lymph nodes, extracapsular spread, poor differentiation, and advanced pathologic stage (III or IV).58
In a study examining whether oral tongue carcinoma was associated with a worse prognosis when compared with other oral cavity subsites, Bell and colleagues demonstrated no significant difference in disease-free survival between the groups. Risk factors that were found to statistically affect overall and disease-specific survival were tumor grade and overall staging.59
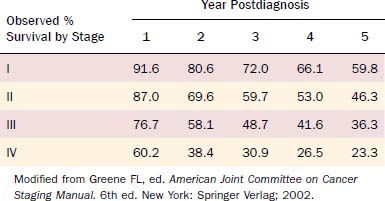
Table 96-2 demonstrates the 5-year observed survival rates for patients with oral cavity squamous cell carcinoma stratified by stage as reported by the AJCC for 1985 to 1991.
Second Primary Tumors
The overall incidence for synchronous and metachronous lesions for patients diagnosed with a primary head and neck malignancy is approximately 14%. Of these malignancies, 80% are metachronous cancers with 50% of cases presenting within the first 2 years of initial treatment of the primary tumor. For patients with oral cavity and oropharyngeal malignancies, the site of a second malignancy is most frequently in the cervical esophagus. New symptoms of dysphagia or odynophagia in this patient population should prompt a diagnostic evaluation with barium swallow or esophagoscopy.60 There appears to be a bimodal pattern for second primary tumors in the esophagus, with an early group developing tumors within 2 years of treatment of the index primary tumor and a later group that develops esophageal cancer greater than 5 years after primary treatment.61
Treatment Considerations
Perioperative antibiotics, in cases with aerodigestive tract exposure to the operative field, can decrease infectious complications associated with surgical intervention. Prophylactic antibiotics are most effective when administered immediately before surgery and for up to 24 hours postoperatively. Extended use of antibiotics in postsurgical patients has not been shown to decrease the risk of infectious complications or fistula formation. The inappropriate use of antibiotics increases the risk for pseudomembranous colitis and the emergence of resistant organisms. Selection of a prophylactic antibiotic for clean-contaminated head and neck surgery should provide adequate gram-positive in addition to anaerobic coverage.62
Multiple general statements can be made concerning the surgical management of oral cancer as a whole. A detailed discussion of the treatment of each oral cavity subsite follows within this chapter. For early-stage lesions of the oral cavity (T1/T2), transoral resection tends to be possible with complete tumor removal and adequate margin control. Primary closure, healing by secondary intention, or placement of a split-thickness skin graft may be used for reconstruction of small defects. Larger and more posteriorly located lesions may require a pull-through or mandibulotomy-based technique to aid exposure for resection and reconstruction. When the tumor closely approximates the mandible, marginal versus segmental mandibulectomy needs to be considered. Reconstructive options for advanced T-stage lesions include microvascular free tissue or pedicled flap reconstruction. Traditionally, T4b lesions have been considered inoperable. Liao and colleagues demonstrated equivalent results with T4a and selected T4b lesions (without carotid encasement and skull base extension) when aggressive surgical management of the primary site and regional lymphatics with postoperative radiation or chemoradiation therapy was performed. Free tissue reconstruction was necessary in 95% of patients. Local control, disease-free, and overall survival numbers were similar.63
Surgery—Treatment of the Primary Lesion
Lip
The majority of neoplastic lesions that affect this subsite present on the lower (88% to 95%) as opposed to the upper lip (2% to 7%) or the commissure (1%) (Fig. 96-11). For the lower lip, squamous cell carcinoma predominates, whereas basal cell carcinoma disproportionately arises on the upper lip. In addition to basal and squamous cell carcinoma, the differential diagnosis of a lip lesion includes keratoacanthoma, minor salivary gland tumors, malignant melanoma, and tumors of mesenchymal origin (malignant fibrous histiocytoma, leiomyosarcoma, fibrosarcoma, angiosarcoma, and rhabdomyosarcoma).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree