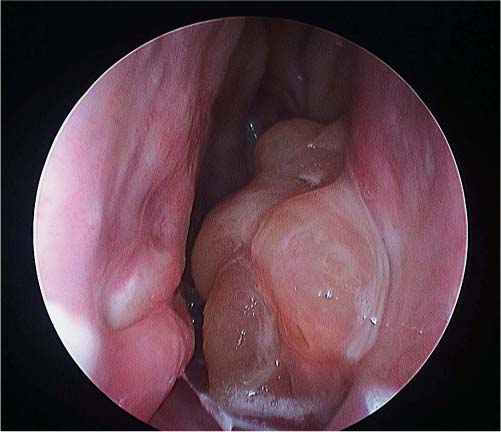
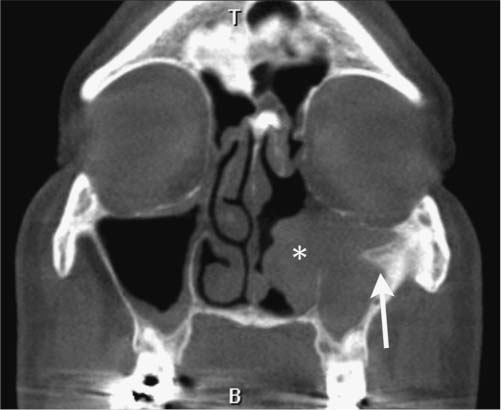
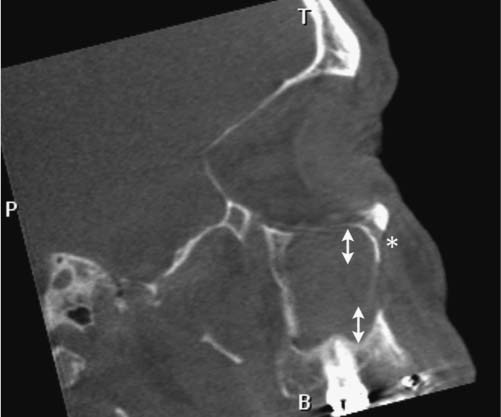
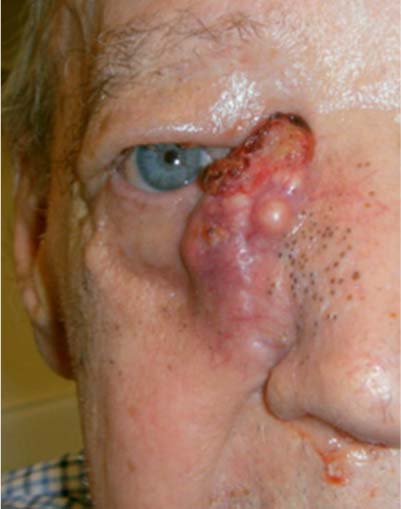
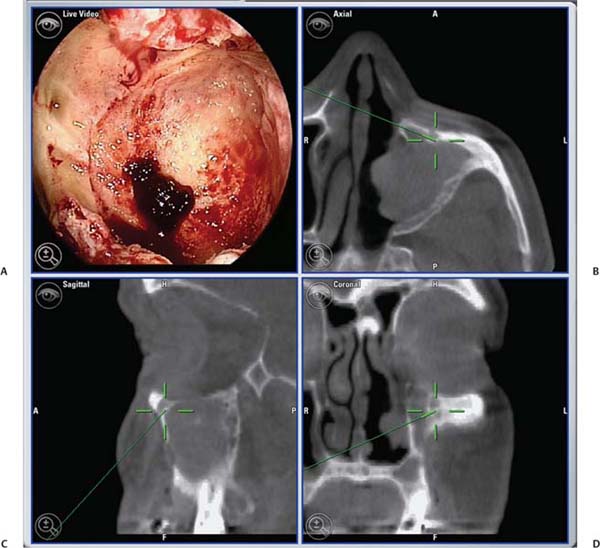
25 Learning from a Difficult Case: Accessing the Anterolateral Maxillary Sinus Aggressive exposure is not necessary for inflammatory disease. There are 90- and 120-degree microdebriders (Medtronic, Jacksonville, FL) to access the maxillary sinus via a wide antrostomy. The maxillary trephine technique can provide access without these angled instruments1 and a modified medial maxillectomy (MMM) can be performed for those with chronically dysfunctional sinus mucosa not being effectively treated via antrostomy alone.2,3 For tumor resection, exposure needs to be greater. Traditional surgical approaches include midface degloving and transfacial options. Transfacial approaches are hindered by the subsequent scar, alar retraction, and potential functional deformity to eye closure. Without significant bone removal, open access suffers from an increasingly narrow surgical corridor as the dissection proceeds posteriorly. Endoscopic approaches have the advantage of a wide field, near-vision dissection, while maintaining a narrow surgical approach. Visualization can be superior with an endoscopic approach compared with the microsurgical access.4 The removal of juvenile nasopharyngeal angiofibroma (JNA) and inverted papilloma (IP) are the case examples for successful endoscopic tumor removal. The literature is now filled with reports of low recurrence rates for these lesions when managed with an entirely endoscopic approach, even for extensive lesions.5–8 For lateral pathology, however, the use of angled endoscopes and instruments becomes necessary. This is especially true for more anterior and laterally based lesions (Fig. 25.1). The use of angled scopes greatly enhances the visualization, but angled instruments do not always afford the equivalent dissection technique compared with zero-degree surgery. As endoscopic techniques begin to encompass more extensive benign disease and even malignancy, more accurate dissection is required to improve our removal of pathology, control bleeding, and minimize morbidity.9 Working with curved instruments, angled endoscopes, and a single nostril often leaves the surgeon with curette only and “scraping” techniques for tumor removal. For the removal of large JNA, a posterior septectomy is often performed to improve access and the working field. Similarly, a superior septal window is used for extended surgery of the frontal sinus and recess. The subsequent functional impairment of either a posterior or superior septal window is minimal for most patients. Crusting, bleeding, support, and airflow changes do not appear to be a common cause of morbidity. An anterior septectomy is not feasible as the anterior septum plays an important role in the support of the external nose, forms part of the the nasal valve, and will often cause airflow changes with resultant crusting, whistling, and bleeding.10 The use of large pedicled septal flaps from skull base reconstruction9,11 has changed our approach for more anterior and lateral pathology. We use an anterior transseptal approach, created by the elevation of large septal flaps, a cartilaginous septal window, and reconstruction at the end of the procedure. The concept of a transseptal approach is not entirely novel. Wormald et al have discussed the concept of nonop-posing septal incisions to provide additional retraction and access to JNA.8 However, it has been our experience with large pedicled septal flaps9,12,13 in skull base surgery that has allowed an evolution in the way we manage extensive anterolateral sinonasal lesions. Additional anatomic studies have shown that even a large MMM or total medial maxillectomy (TMM) does not provide adequate access for many patients.14 Some pathologies have significant anterior attachments; for others, a transseptal approach is made in a stepwise fashion after defining the tumor origin. We utilize the following algorithm when accessing more anterolaterally based disease: transnasal direct, middle meatal antrostomy (MMA), modified medial maxillectomy (MMM),2 total medial maxillectomy15 (TMM; with removal of the lower lacrimal apparatus), and a combined transseptal MMM/TMM. Fig. 25.1 Endoscopic view of recurrent left inverted papilloma protruding into the left nasal cavity. A very active 72-year-old man with a Krouse stage 3 inverted papilloma of the left maxillary sinus presents with progressive nasal obstruction and postnasal mucoid discharge. He has had two previous endoscopic procedures then a further open lateral rhinotomy to remove the disease. It has now been 10 years since his first surgery, 2 years since his last procedure, and 18 months since last review. He has had a previous septoplasty, left middle turbinectomy, wide antrostomy, subtotal resection of his inferior turbinate, and a partial ethmoidectomy. Endoscopic examination easily identifies the tumor (Fig. 25.1) and the orbital floor and medial wall are not involved in the disease. Surgery was indicated for this patient because he had progressive symptoms, was otherwise fit with no comorbidities, and there was the potential for malignant change. Atypia, dysplasia, carcinoma-in-situ, and squamous cell carcinoma can all occur in papilloma. Malignant change within the inverted type of papilloma has been well documented.16 Krouse originally described a 9.1% rate of squamous cell carcinoma in his cohort.17 This has been supported by a recent meta-analysis of a published case series, which reported a synchronous carcinoma rate of 7.1% and a metachronous rate of 11%.18 It was widely speculated that atypical and dysplastic change was precancerous,17 but this is not supported by the literature. Degree of atypia, mitotic index, and recurrence appear to have little correlation with carcinoma transformation.19,20 Carcinoma-in-situ should be closely followed as these cases may represent precancerous change or undetected carcinoma from sampling errors.18 Fig. 25.2 Computed tomography evaluation: The recurrent left inverted papilloma protruding into the left nasal cavity can be seen (*). An area of osteitis and neo-osteogenesis is a landmark for the origin of the tumor (arrow). Computed tomography (CT) forms the basis for the assessment. The site of the tumor can be seen by a corresponding area of osteitis21,22 in the zygomatic recess of the left maxilla (Fig. 25.2). The previous anterior maxillary wall defect is also present (Fig. 25.3). The potential for tumor to have migrated in the premaxillary soft tissue should be recognized here. Similar seeding into extrasinus tissue, including the face, can occur through a lateral rhinotomy approach (Fig. 25.4). Considering the multiple previous procedures and open surgery, magnetic resonance imaging (MRI) was performed that confirmed tumor was confined to the maxillary sinus. Endoscopic transseptal surgery is performed under general controlled hypotensive anesthesia. Total intravenous anesthesia is associated with better mucosal hemostasis.23,24 Cotton pledgets containing adrenaline 1:1000 are placed in the nasal cavity over the areas of surgical access for 10 minutes before the surgical procedure. The septum is infiltrated with 1% lidocaine with adrenaline 1:100.000. Fig. 25.3 Computed tomography evaluation: The exit of the infraorbital nerve can be seen(*). The previous anterior open approach has fallen well short of exposing the area to be addressed(double-ended arrows). Fig. 25.4 Violation of natural barriers to spread: skin, dural, and peri-orbital can lead to tumor seeding and difficult to manage disease. Although squamous cell carcinoma conversion was suspected, this papilloma was benign on multiple biopsies. For some patients it is possible to predict the insertion site by a corresponding area of osteitis.21,22 Early planning in the need for a transseptal approach makes surgery more straightforward. But for many, the exact attachment of their sinonasal tumor is not evident until the time of surgery. The resection often follows a stepwise MMA, MMM, TMM, and then transseptal access, as the origin is elucidated. The decision to go to a transseptal approach should come early in the operation. Make this judgment on preoperative imaging and operative location of tumor attachment. Dissection will proceed very quickly via a transseptal approach compared with working with 70-degree scopes and angled instruments. The contralateral septal mucosa is generally chosen to raise the main mucoperiosteal/perichondrial flap. This ensures that it can be conveniently tucked away between the contralateral middle turbinate and posterior septum to avoid accidental injury during instrumentation on the operative side. The nasal floor incision begins far lateral in the inferior meatus. The superior incision is made as high as possible near the nasal dorsum. Both are brought forward to join a hemitransfixion incision. This mucoperichondrial/periosteal flap is pedicled posteriorly on the septal branch of the sphenopalatine artery. There is some foreshortening of the flap after elevation and the additional width is important for adequate reconstruction. A window of septal cartilage is removed beginning at the head of the inferior turbinate. The area usually comprises a 1.5 × 2 cm area that then allows both endoscope and instrument to work comfortably through the septum. The ipsilateral mucosa over this area is raised as an inverted “U” flap with a random blood supply based inferiorly from the nasal floor (see Video 25.1, which demonstrates the development and closure of a septal window). After MMM, 2-mm Kerrison rongeurs are used via the transseptal route to remove the remaining medial wall down to the nasal floor. Attention is then turned to removing the medial wall anteriorly. It is possible to resect the wall underneath the Hasner valve for access. But experience has found that this leads to inadequate exposure of the anterior wall. The lower lacrimal apparatus is resected and reconstructed at the end of the case. We drill away the bone at the attachment of most benign tumors as tumor pseudopods are often extended into the nearby bone.25 There is usually little active bleeding at the completion of tumor removal. The clean mucosal edges and rough diamond drilled bone offer good hemostasis (Fig. 25.5). A dissolvable hemostatic dressing (Surgiflo; Johnson & Johnson Medical, North Yorkshire, UK) is used to fill the cavity. Prior to closure, the lacrimal duct is exposed for a length of 6 to 8 mm. This membranous duct is then marsupialized. A formal dacryocystorhinostomy can be performed, but we have found this unnecessary in the majority of cases. Dissolvable sutures are used to reconstitute the septum. We do not replace the septal cartilage. It is usually removed piecemeal and is far less than that often removed during septoplasty. Thin Silastic (Dow Corning, Midland, MI/Barry, UK) sheets are placed on either side. They should be sized to include coverage of the nasal floor. One or two 3/0 Prolene (Ethicon, Somerville, NJ) transseptal sutures are used to secure the silastic sheets (see Video 25.2, which demonstrates the approach and resection for this patient). Patients are discharged on the same day or morning after depending on analgesic control. Oral antibiotics are used for 2 weeks and usually consist of amoxicillin and clavulanic acid (GlaxoSmithKline, Mississauga, Ontario, Canada) and clavulanic acid combination twice a day. Nasal irrigation begins at 24 to 36 hours postsurgery. This is performed with a high volume squeeze bottle (NeilMed Sinus Rinse; NailMed Pharmaceuticals, Inc., Santa Rosa, CA) twice a day only. Silastic sheets are removed 2 to 3 weeks postoperatively (see Video 25.3). Exposed cartilage and bone appears to heal extremely well under the Silastic and avoids crusting. They are thin and so allow patients to breathe and irrigate with minimal interference. For inverted papilloma, a collation of literature reports recurrence rates of 12.8% for endoscopic resection, 17.0% for lateral rhinotomy, and 34.2% for other limited resections.18 Selection bias may exist for some of the published reports, but the Krouse Staging System used to provide a comparison basis is well accepted in the literature (Table 25.1). Fig. 25.5 (A–D) Intraoperative images from image guidance system (IGS; not necessary for this type of surgery), but demonstrates well the area of resection and the angle of approach. Here the IGS probe is on the internal exit point of the infraorbital nerve. Other approaches exist to access the anterolateral maxillary sinus. Endoscopic maxillotomy has been described and can provide similar access.26 This procedure involves the removal of the medial buttress via osteotomies (see Video 25.4). The lacrimal apparatus is disrupted as with a TMM. The anterior superior alveolar nerve, the canine root, and potential loss of lateral support of the alar cartilage to the piriform aperture can all occur with this approach. Performed endoscopically, the resultant alar retraction and collapse is limited compared with similar lateral rhinotomy approaches, but can still occur. The maxillary trephine has been popularized by Wormald and can provide either additional instrument or endoscopic access through the anterior maxilla.26 This is an excellent adjunct to a lateral infratemporal fossa or lateral maxillary lesion. However, for those pathologies involving the anterior wall itself, the trephine does not improve surgical access and will come through tumor in its approach.
 Case Presentation
Case Presentation
History
Radiologic Evaluation
Operative Technique
Postoperative Care
 Other Approaches
Other Approaches
T1 | Tumor isolated to one area of the nasal cavity without extension to the paranasal sinuses |
T2 | Tumor involves medial wall of the maxillary sinus, ethmoid sinuses, and/or ostiomeatal complex. |
T3 | Tumor involves the superior, inferior, posterior, anterior, or lateral walls of the maxillary sinus; frontal sinus; or sphenoid sinus. |
T4 | Tumor with extra-sinonasal extent or malignancy |
 Conclusion
Conclusion
Most maxillary sinus masses can be successfully managed via direct single or simple bilateral transnasal access. However, when pathology arises anterior and lateral to the inferior orbital nerve, the transseptal approach greatly enhances the endoscopic surgeon’s ability to completely remove disease. Direct, straight ahead, endoscopic dissection is possible for even these most anterolateral lesions. The morbidity from anterior septal reconstruction must be weighed against the need for access and complete tumor removal. The transseptal approach affords access to areas previously thought inaccessible with an endoscopic technique.
• Converting to a transseptal access early affords quicker subsequent dissection.
• Oversize the contralateral septal flap. Do not hesitate to start at the squamous-mucosal junction.
• Silastic splints are essential as they prevent fibrinous adhesions and protect the flaps.
• Secondary bone pain is common at5 to7 days after aggressive drilling in the maxillary sinus. Nonsteroidal antiinflammatory drugs(NSAIDs) are effective analgesia rather than opioids.
• Do not fear the lower lacrimal system. Remove the inferior nasolacrimal system to remove disease. Robust superior reconstruction is possible.
• The extent of exposure and its subsequent morbidity should be dictated by the disease.
• Simple nonopposing incisions seem appealing and maybe appropriate for a rectractor, but as a working channel it is awkward, does not easily accommodate both instruments, results in traumatized mucosa, and blood has a tendency to run down the endoscope.