Intraocular Masquerade Syndromes
Devron H. Char
When Duke-Elder’s System of Ophthalmology was published in 1966, the term ocular masquerade syndromes was not included.1 The term was first used in the ophthalmic literature in 1967 to describe conjunctival malignancies that simulate chronic conjunctivitis.2,3 Most ophthalmologists now use the term for any malignant process that simulates benign disease.
This chapter discusses only intraocular masquerade lesions. In previous editions of this chapter, I considered any noninflammatory condition that simulated uveitis to be a masquerade syndrome. In this edition, although I am using this broad definition, I have emphasized neoplastic-simulating lesions because more harm occurs when a malignant process is mistaken for a benign lesion.
Increased awareness that malignant intraocular processes can simulate benign lesions (most commonly in adults as a unilateral media opacity or in children younger than 2 years of age with intraocular inflammation), together with the development of better diagnostic techniques, has decreased the frequency of incorrect diagnoses. However, diagnostic problems still occur even with the use of ultrasonography, computed tomography (CT), optical coherence tomography (OCT), and magnetic resonance imaging (MRI). Occasionally, a correct diagnosis is not established until a cytologic, histopathologic, or newer, molecular-based laboratory examination is performed.
ADULT NEOPLASTIC PROCESSES
In adults, the three most common intraocular malignancies that can simulate intraocular inflammation are uveal melanoma, metastatic tumors, and intraocular lymphoma.4
Uveal Melanoma
Any elderly patient with a unilateral media opacity and inflammation has a uveal melanoma until proven otherwise with appropriate imaging studies and, when necessary, invasive diagnostic techniques.4 Although unapparent intraocular foreign bodies are a much more common cause of unilateral cataract, in as many as 10% of blind eyes submitted to eye pathology laboratories, an undiagnosed uveal melanoma is found. All adults with unexplained unilateral media opacity require a thorough evaluation, including a diagnostic ultrasound examination.5,6 We have managed many very large uveal melanomas that were discovered only after cataract extraction and were not detected at the time of ultrasonic biometry to measure axial length.
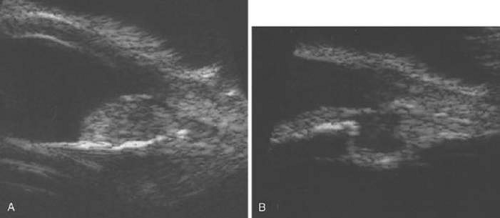
Necrotic melanomas account for approximately 5% of uveal melanomas; in most cases, there is intraocular inflammation accompanying the cataract.7,8,9 Figures 53-1 and 53-2 demonstrate a typical case. The patient had a long history of unilateral decreased vision. The eye had become painful 1 month before admission, and he was referred for evaluation of uveitis. Clinically, there was a dense, unilateral cataract with significant intraocular inflammation, which was manifested as a ciliary flush with 2+ cells and flare. Media opacity obscured all fundus detail. An immersion B-scan demonstrated a large intraocular tumor that was most consistent with a uveal melanoma. The eye was removed, and the diagnosis was confirmed histologically.10
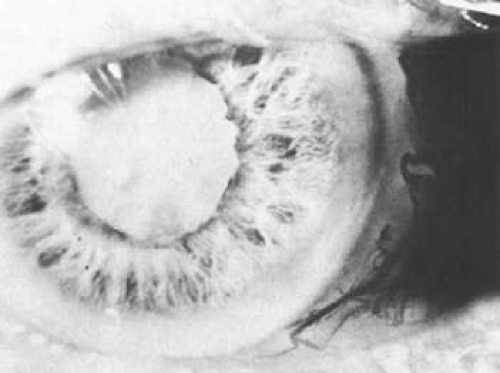 Figure 53-1. A unilateral media opacity in an adult, requiring that either a foreign body or an intraocular tumor be ruled out. The finding of a large episcleral (sentinel) vessel was consistent with either an intraocular tumor or an inflammation. An immersion B-scan helped determine the diagnosis (see Fig. 53-2). |
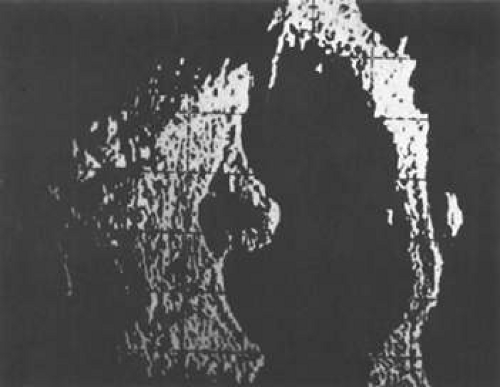 Figure 53-2. Immersion B-scan of the patient in Figure 53-1 demonstrates a uveal melanoma. |
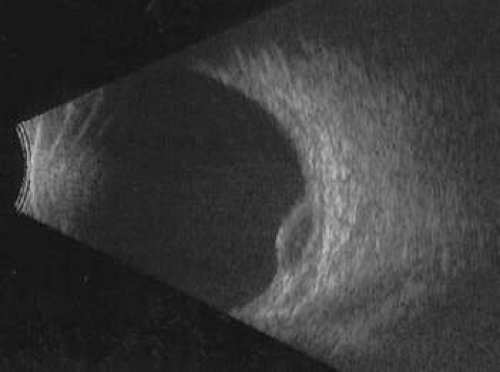
Unfortunately, imaging studies are not always diagnostic. The A- and B-scans shown in Figures 53-3 and 53-4 were from a patient with unilateral media opacity referred for evaluation and treatment of a presumed uveal melanoma. Occasionally, if the cataractous lens is either scanned tangentially or is luxated, an artifactual diagnosis of a melanoma is made. A repeat ultrasound at our institution was negative for tumor, and the referral ophthalmologist who performed a cataract extraction noted that no tumor was present and that the patient had an excellent visual outcome. In contrast, the patient shown in Figure 53-5 was referred with a presumed uveal melanoma with secondary inflammation and a history similar to that described for the patient shown in Figure 53-111 Ultrasound and CT examinations were not diagnostic. Because the eye was blind and painful, it was enucleated, and an extremely necrotic uveal melanoma was noted histologically. In some necrotic melanomas, the fundus cannot be visualized and the diagnosis cannot be established with imaging studies. Very rarely, a necrotic uveal melanoma can produce sufficient pigment dispersion into the vitreous to obscure the correct diagnosis.12
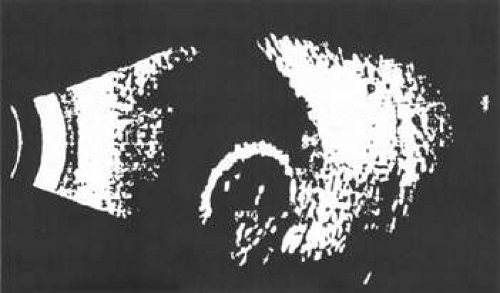 Figure 53-3. B-scan in a patient with dense cataract, showing a possible tumor. Tangential cuts through a cataractous lens can produce a melanoma-like pattern (pseudomelanoma). |
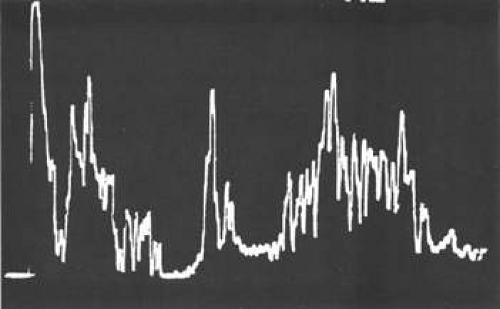 Figure 53-4. A-scan through the area shown in Figure 53-3 shows a pseudomelanoma pattern. What distinguished this from a melanoma was a positive-angle kappa (climbing posterior spike). This patient did not have an eye tumor and had 20/20 vision after cataract removal. |
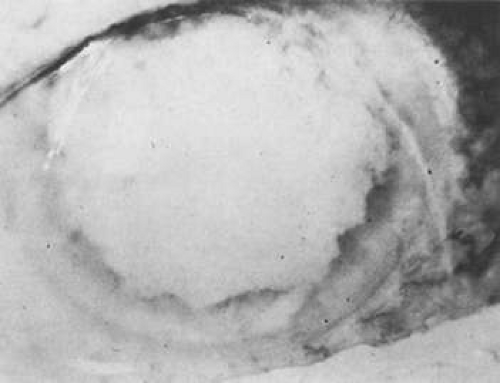 Figure 53-5. Clinical appearance of an eye with inflammation and pain. Ultrasound and CT were not diagnostic. Histologic examination revealed a necrotic melanoma. |
The role of MRI in the evaluation of intraocular masquerade syndromes is unclear. There was initial enthusiasm about the possibility of differentiating atypical uveal melanomas from hemorrhagic-simulating lesions based on the unique paramagnetic properties of melanin.13 In relation to vitreous, a uveal melanoma typically has a bright signal on a T1 image and a dark signal on a T2 image. In contrast, hemorrhagic processes tend to be either bright or dark on a T1 image and bright on a T2 image.14 Two problems with diagnostic adjuvant is that most of the difficult cases have a uveal melanoma with hemorrhage and that combination of findings often obviates the utility of MRI in this setting.
Unfortunately, there are several problems with MRI for uveal melanoma diagnosis. First, several investigators have shown that unless high field-strength (3 Tesla), thin-section scans with surface coils are used, the quality of data obtained is suboptimal.15 Second, even in a state-of-the-art center, we have had several cases in which we were unable to differentiate between a uveal melanoma and a simulating lesion on the basis of MRI data. We obtain MRI studies in older patients with opaque media, equivocal ultrasonographic data, and large masses. In some of these cases, the MRI is diagnostic of either a melanoma or an extramacular disciform (hemorrhagic) process. It is also a useful adjunctive study if localized extraocular extension is suspected. In equivocal cases, however, we proceed to using invasive diagnostic techniques, such as fine-needle aspiration biopsy (FNAB) (see Metastatic Tumors).
In addition to producing media opacity, inflammation, and cataract, uveal melanomas can also produce other signs that may lead to the incorrect diagnosis of uveitis. Anterior melanomas, especially those that involve the ciliary body, often produce dilated episcleral vessels (sentinel vessels) that occasionally have been misdiagnosed as scleritis (Fig. 53-6).
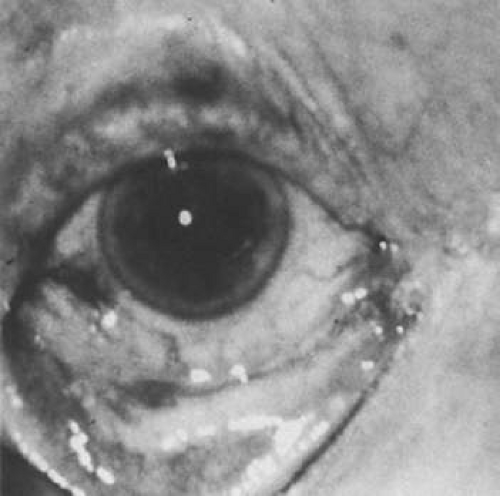 Figure 53-6. Sentinel vessels present over a ciliary body melanoma. (Reproduced with permission from Char DH: Clinical Ocular Oncology. 2nd ed. Philadelphia: Lippincott-Raven Publishers, 1996.) |
Iris melanomas can produce anterior chamber cell and flare, and if the tumor is located in the extreme iris periphery, it may be missed during a cursory examination. Iris melanomas, especially those in a circumferential or ring configuration, are often difficult to diagnose and may simulate either an anterior uveitis or glaucoma.16 Newer, higher-megahertz (high-frequency) ultrasound equipment is better for imaging anterior uveal tumors (Fig. 53-7). It is also possible to image iris tumors with OCT.17 The limitations with current generation OCT in this setting is its inability to image the ciliary body. One of the key issues in an iris melanoma is whether ciliary body involvement is present, and that is better delineated with high-frequency ultrasound.
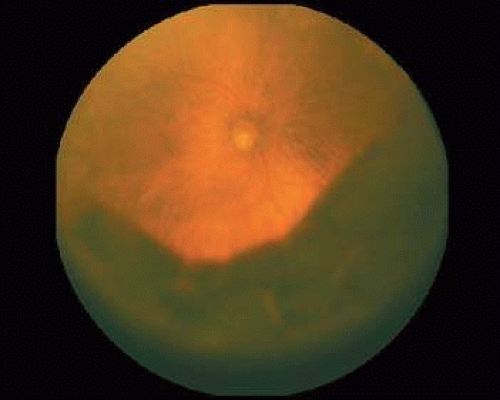
If direct ophthalmoscopy is used, a circumferential peripapillary melanoma can simulate optic neuritis (Fig. 53-8).
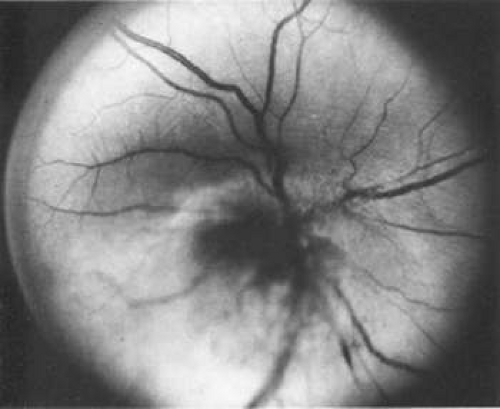 Figure 53-8. A peripapillary melanoma simulating an optic neuritis. (Reproduced with permission from Char DH: Clinical Ocular Oncology. 2nd ed. Philadelphia: Lippincott-Raven Publishers, 1996.) |
Pseudomelanoma
Melanomas can masquerade as uveitis, and some inflammations or hemorrhagic processes can simulate melanoma. These processes include posterior scleritis, focal choroidal hemorrhage or detachments, and focal scleral thinning. Figures 53-9 and 53-10 demonstrate focal scleral thinning in the area of the superior rectus insertion in a patient with rheumatoid scleritis that produced a pseudomelanoma. An apparent elevated pigmented mass was noted on upgaze, but no tumor was observed when the eye was in primary position. Posterior scleritis is often not painful, although some of these patients have deep, piercing pain and increased discomfort during eye movement. Ultrasound usually is diagnostic, demonstrating fluid inside of Tenon’s capsule (Fig. 53-11). A focal hemorrhagic choroidal detachment after cataract surgery can simulate a melanoma. These lesions usually have a bright area of hemorrhage at their base, whereas melanomas typically do not have associated hemorrhage until they break through Bruch’s membrane (most commonly, such tumors are more than 5 mm thick). The differential diagnosis of uveal melanoma is discussed elsewhere in these volumes.
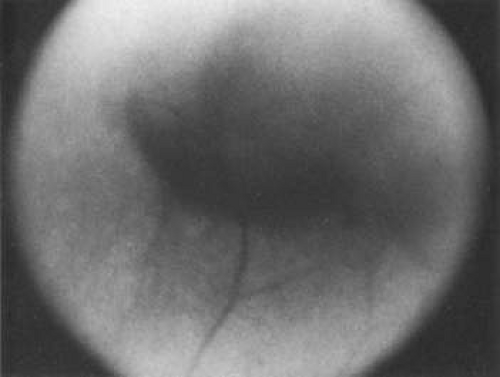 Figure 53-9. A patient with rheumatoid arthritis and weakened sclera with a pseudomelanoma on upgaze. |
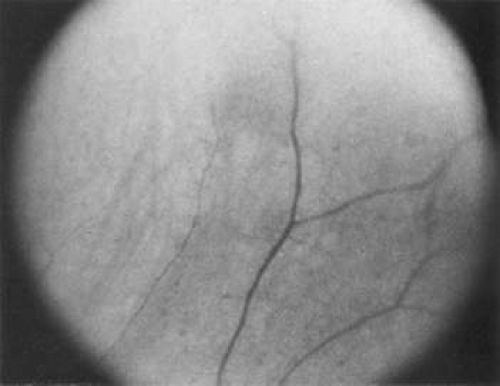 Figure 53-10. Lesion shown in Figure 53-8 disappeared when the patient’s eye was in the primary position. (Note vessel pattern in Figs. 53-9 and 53-10.) |
Other hemorrhagic processes can simulate choroidal melanomas. The patient in Figure 53-12 was referred with a large melanoma. He had been on too high a dose of Coumadin, and this was a diffuse choroidal hemorrhage (Fig. 53-12).
Less commonly trauma can simulate a melanoma, although there are reports of melanomas arising from a scar. The patient shown in Figures 53-13 and 53-14 had a prior history of minor trauma. As shown in the high-frequency ultrasound, this was traumatic uveal prolapsed (Fig. 53-14).
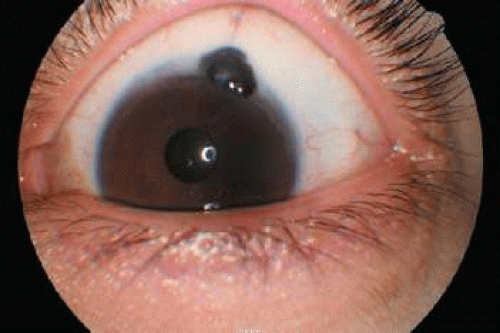 Figure 53-13. Patient referred with an anterior uveal melanoma with extraocular extension. This was iris prolapse. |
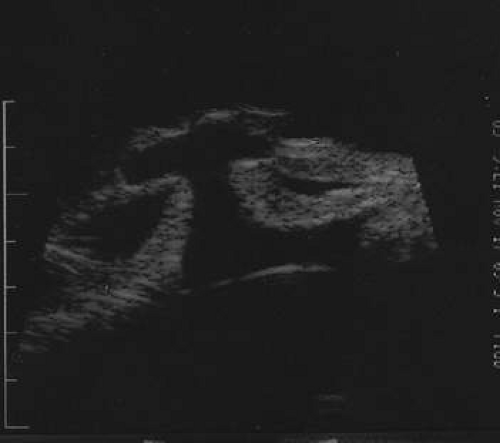 Figure 53-14. The high-frequency ultrasound of the case shown in Figure 53-13 demonstrating iris prolapse. |
Metastatic Tumors
Choroidal metastases can also produce signs that mimic uveitis. As many as 50% of patients with metastases to the eye may present to the ophthalmologist before the primary malignancy is discovered.18 Patients with renal and lung carcinomas have the highest frequency of presenting with eye symptoms as the initial complaint associated with the neoplasm; in contrast, more than 90% of breast carcinomas have a known primary at the time ocular metastases are noted. Usually, intraocular metastases are solid, posterior, amelanotic, choroidal tumors; however, in less than 10% of cases, they can simulate a benign intraocular inflammation. At least five patterns of inflammatory masquerade syndromes can occur with intraocular metastases.
Tumors may produce a pseudoscleritis.
Patients with metastatic carcinoma may present with a diffuse infiltration of the iris (Fig. 53-15).
In rare cases, an optic nerve metastasis can simulate either an optic neuritis or an acute anterior ischemic neuropathy (Fig. 53-16).
Isolated metastatic cells in the vitreous or anterior chamber may occur as the only manifestation of an ocular metastasis.19
Figure 53-17 shows a patient with bilateral pseudohypopyon as the first stigmata of widespread disease.
Most commonly, pseudohypopyons in adults develop in association with either a systemic lymphoma or leukemic relapse. Rarely, metastatic cutaneous melanoma can produce this pattern; however, usually these latter cells are pigmented (Fig. 53-18). Much more frequently, vitreous cells in patients with known metastases are benign, originating from either infectious or endogenous uveitis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree