Ocular Angiography in Uveitis
Maurice F. Rabb
Wico W. Lai
Tom Jennings
Fluorescein angiography was initially developed to study retinal vascular flow patterns but now also serves as an important way to diagnose ocular fundus disease. Although often used today in ophthalmology, this technique is not particularly useful in defining the choroidal circulation.1 The rapid leakage of fluorescein from the choriocapillaris and the scattering of fluorescence by pigmented layers of the fundus have limited its ability to study the choroid in both the normal and pathologic states. Indocyanine green (ICG), a water-soluble tricarboncyanine dye, possesses unique optical and biophysical properties, including its near-infrared absorption peak and its high plasma protein-binding capacity. Coupled with the development of high-resolution imaging systems, ICG angiography has become a useful adjunct or alternative to fluorescein angiography in imaging the choroidal circulation.
In this chapter, we describe the characteristic angiographic findings in many common uveitic syndromes using abnormal hypofluorescence or hyperfluorescence patterns with both fluorescein (Table 1)2,3 and ICG (Fig. 1).4 Many of these clinical entities and their treatments are described in more detail in other chapters in these volumes.
INTERPRETATION OF HYPOFLUORESCENCE AND HYPERFLUORESCENCE
HYPOFLUORESCENCE
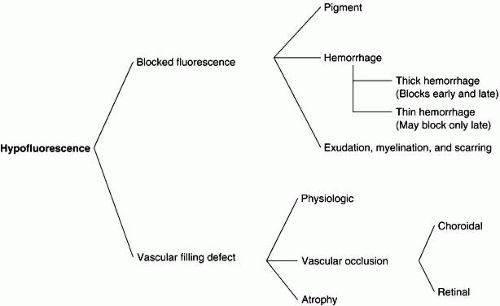
Hypofluorescence may be caused by blocked fluorescence, which is synonymous with transmission decrease, or by a vascular filling defect. Blocked fluorescein corresponds to the size, shape, and location of the blocking material seen ophthalmoscopically. Any hypofluorescence that does not correspond to the ophthalmoscopic picture must then be considered a vascular filling defect.
With ICG angiography, hypofluorescence needs to be analyzed by taking into consideration its evolution during the different phases of the angiogram. It is important to distinguish between hypofluorescence observed in both the early and late phases and when it is observed only in the later phases. The blockage depends on the nature of the blocking material and on the background choroidal fluorescence.
Blocked Fluorescence
Retinal. Any opacification in front of the retinal vessels that reduces media clarity causes blocked retinal vascular hypofluorescence on fluorescein angiogram (FA). Many inflammatory conditions produce hazy media, causing poor visualization of fundus detail. Fibrotic or gliotic membranes and inflammatory material on the retina or involving its inner layers obstruct the view of the retinal vasculature and cause blocked fluorescence.
CHOROIDAL.
Hypofluorescence can result from blocked choroidal vasculature due to fluid, exudate, hemorrhage, pigment, or other material accumulating in front of the choroidal vasculature deep to the retinal vasculature. Any opaque substance located beneath the retina but in front of the choroid blocks fluorescence of the choroidal vasculature; however, it does not block retinal vascular fluorescence. Fluorescence can be blocked by substances such as fluid in retinal pigment epithelial detachments and inflammatory material in acute multifocal posterior placoid pigment epitheliopathy (AMPPPE).
In general, the blockage to ICG is less than that to fluorescein. For example, although blood at any level can lead to blockage throughout the FA, blockage may occur only in the later phases in ICG angiography. However, inflammatory diseases that primarily affect the choroid, such as birdshot chorioretinopathy and multifocal choroiditis, may result in profound and more dramatic blockage than that observed with FA.
Vascular Filling Defect
RETINAL.
A vascular filling defect is the other cause of hypofluorescence in the retina if blocked fluorescence is not present. For a retinal vascular filling defect to be diagnosed, the location of the defect must be determined. It may arise from inflammatory obstruction of the artery, vein, or capillary or from a combination of these. Eales’ disease, sarcoidosis, and other inflammatory diseases also cause peripheral capillary nonperfusion.
CHOROIDAL.
In ICG angiography, vascular filling defects have been subdivided into those that are physiologic, those resulting from a vascular occlusion, and those that are caused by tissue atrophy (see Fig. 1).4 Physiologic filling defects may be the result of choroidal watershed zones (early defects) and choroidal vascular silhouette against the background choroidal fluorescence (late defect). In contrast to FA, ICG studies are better at delineating choroidal vascular occlusion than are retinal vascular defects. Patients with AMPPPE may show persistent vascular filling defects after resolution of the condition. Choroidal vascular atrophy may develop in patients with a condition such as age-related macular degeneration, which results in vascular bed closure from scar formation.
HYPERFLUORESCENCE
Hyperfluorescence is any abnormally light area on the positive print of an angiogram. There are three types of hyperfluorescence in uveitis: transmitted fluorescence, abnormal vessels, and leakage.
Transmitted Fluorescence
Increased visibility of fluorescence from the choroidal vasculature caused by the absence of pigment in the pigment epithelium is called transmitted fluorescence (i.e., a pigment epithelial window defect) on FA. Various types of inflammation at the level of the pigment epithelium, choriocapillaris, or choroid produce pigment epithelial window defects such as AMPPPE, serpiginous choroiditis, and histoplasmosis. Each of these inflammatory conditions adversely affects the pigment epithelium, resulting in depigmentation and atrophy. Window defect is a very frequent finding in a fundus that has undergone choroidal inflammation.
In ICG angiography, transmitted fluorescence may present as one of two forms depending on the integrity of the choriocapillaris. When the choriocapillaris is intact, transmission hyperfluorescence of the larger choroidal vessels is noted in the early phases of the angiogram. In the late phases of the study, the fluorescence pattern appears normal. In conditions associated with atrophy of the choriocapillaris, hyperfluorescence of the larger choroidal vessels is noted in the early phase of the angiogram. However, this area may become relatively hypofluorescent in the later phases because of decreased or absent perfusion of the choriocapillaris.
Abnormal Vessels
RETINAL AND DISC.
There are many diseases that produce neovascularization. Retinal ischemia is theorized to be the primary pathogenic stimulus. Most of the disease processes resulting in neovascularization show some degree of ischemia by fluorescein angiography, with capillary nonperfusion an almost constant finding. The diseases that are known to result in neovascularization are phlebitis (Eales’ disease), severe uveitis, pars planitis, sarcoidosis, and Behçet’s disease.
CHOROIDAL.
Fluorescein angiography has proved to be of great value as an aid in the recognition and demonstration of normal and abnormal vascular patterns in the subretinal and choroidal tissues. The demonstration of subretinal neovascularization (SRNV) by fluorescein angiography (early lacyhyperfluorescence) represents one of the most significant advances in ophthalmic knowledge.
Although ICG angiography can identify abnormal vessels in the retina, it is more helpful in imaging changes in the choroidal circulation and is most useful in situations in which occult or poorly defined SRNV is present. Often the neovascular complex is obscured by blood or fluid and is poorly visualized on FA. ICG angiography has been shown to be efficacious in identifying occult SRNV and has increased the likelihood of identifying potentially treatable lesions in patients who are otherwise considered ineligible for laser treatment under conventional guidelines.5,6
Leakage
VITREOUS.
In uveitis, there are two major causes of vitreous leakage seen on fluorescein angiography: (1) neovascularization and fibrosis and (2) intraocular inflammation. Disc and retinal neovascularization may be associated with uveal inflammation. Inflammation of the retina or the disc, such as that seen in toxoplasmic chorioretinitis, is an example of intraocular inflammation leading to vitreous leakage.
DISC.
Inflammations of the optic nerve head, such as papillitis, vasculitis, and papillophlebitis, show a fuzzy hyperfluorescence of the disc in the later phases of the FA because of the staining of the edematous inflamed tissue.
RETINAL.
In the late stages of the normal angiogram, the retinal vessels contain a minimal concentration of fluorescein and the retina is normally dark. Retinal hyperfluorescence in the late stages indicates extravasation of fluorescein into the retinal extravascular space. The leakage occurs in a cystoid or noncystoid pattern and arises from leakage of retinal or choroidal vessels. Similar findings may be observed in ICG angiograms.
In the macula, cystoid retinal edema assumes a stellate or flower-petal appearance because of the obliquity of the outer plexiform layer. Outside the macula, there is a honeycomb appearance. Causes of cystoid edema rising from retinal vessels include inflammatory conditions, such as uveitis, chorioretinitis, and aphakia (Irvine-Gass syndrome). Cystoid retinal edema may be caused by leakage from choroidal vessels, such as with SRNV. Inflammation, such as toxoplasmic retinitis, is one of the most common causes of noncystoid retinal edema. On the FA, the lesion leaks fluorescein profusely into the surrounding retina and overlying vitreous, resulting in a fuzzy cotton-ball fluorescence. Marked staining of the retina (noncystoid edema) is then apparent.
When a large retinal vessel is inflamed, as in phlebitis or vasculitis, the endothelium of the involved large retinal vessel leaks fluorescein, resulting in hyperfluorescence. On the angiogram, this appears as a line of hyperfluorescence along the wall of the vessel. Retinal vessels traversing an area of chorioretinitis show reactive perivascular staining.
CHOROIDAL.
Choroidal inflammatory diseases, as in focal or massive choroiditis (Harada’s syndrome), can change or damage the overlying pigment epithelium sufficiently to produce a sensory retinal detachment. In some cases, marked leakage occurs, which results in heavy pooling (defined as leakage of fluorescein dye) under the sensory retinal detachment.
UVEITIS OF UNKNOWN ETIOLOGY
VOGT-KOYANAGI-HARADA SYNDROME
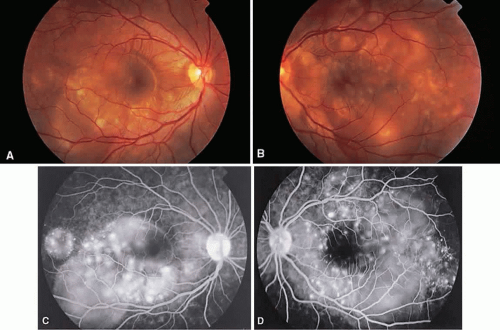
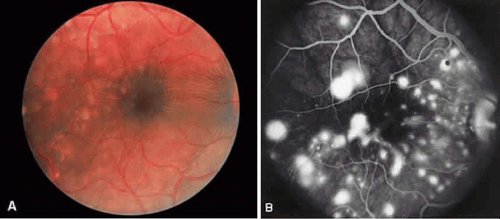
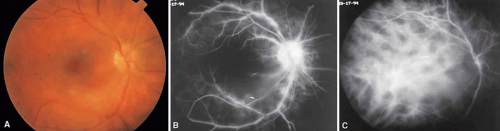
Vogt-Koyanagi-Harada syndrome, also known as uveomeningoencephalitis syndrome, is a systemic disease often found in young, darkly pigmented whites, blacks, native Indians, Hispanics, and Asians. It presents as an anterior and posterior uveitis with disc hyperemia, disc edema, and exudative retinal detachment (Figs. 2A and B and 3A). Systemic signs and symptoms include headache, hearing loss, poliosis, vitiligo, nuchal rigidity, seizures, and even coma. SRNV and disciform scars are late complications. The differential diagnosis should include AMPPPE and sympathetic ophthalmia.
There is a characteristic fluorescein angiographic appearance in the early phase showing multiple discrete hyperfluorescent dots at the retinal pigment epithelial level that enlarge over time (see Fig. 2C and D). In the late phase, if there is a serous detachment, the fluorescein pools beneath the subretinal space (see Fig. 3B). The edematous disc hyperfluoresces in the late phase. Generally, the retinal vessels are unaffected.7,8 Retinochoroidal anastomoses also have been documented by fluorescein angiography.9
ICG angiography shows multiple hypofluorescent spots in the posterior pole (see Fig. 3C). These spots may coalesce and obscure the filling of large choroidal vessels. When serous retinal detachment develops, the marked hyperfluorescence observed on FA is not as prominently noted in ICG angiography, presumably because of the highly protein-bound nature of ICG. In fact, diffuse late hypofluorescence may be observed in patients with serous retinal detachment.10 Ill-defined areas of hyperfluorescence corresponding to diffuse choroidal staining may be seen in some cases in the late phases of the angiogram. The optic disc may stain with ICG in the acute phase but usually is minimal compared to that observed on FA.
SYMPATHETIC OPHTHALMIA
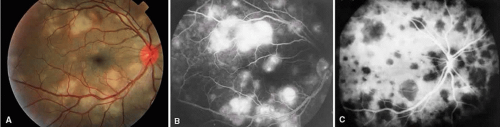
Sympathetic ophthalmia is a rare, bilateral granulomatous, paninflammatory disease that can occur several weeks to years after an ocular injury. Sympathetic ophthalmia is hypothesized to be an autoimmune reaction against a normally sequestered antigen unmasked by an injury. Retinal findings in sympathetic ophthalmia are perivasculitis, papillitis, and yellowish subretinal nodules known as Dalen-Fuchs nodules. Dalen-Fuchs nodules are believed to be transformed retinal pigment epithelium cells or T lymphocytes or both. Areas of choroiditis or granuloma simulating AMPPPE may be seen. Pigmentary disturbance may be noted in the chronic phase of the disease (Fig. 4A and B). Choroidal neovascularization also may develop (see Fig. 4C).
One of two possible fluorescein angiographic findings may be evident. The more common one is identical to Vogt-Koyanagi-Harada syndrome, with early multiple pinpoint retinal pigment epithelial leaks that enlarge with time.11 The second, less common, manifestation is early choroidal hypofluorescence of the Dalen-Fuchs nodules followed by hyperfluorescence in the later phase.12 When exudative retinal detachment occurs, there are confluent areas of subretinal fluorescein pooling.
Two patterns have been identified on ICG angiography.13 The first pattern, observed in a patient with chronic disease, showed hypofluorescent spots in both the intermediate and late phases of the angiogram. The second pattern, reported in a patient with recent onset of the disease, showed hypofluorescent spots in the intermediate phase but faded in the late phase of the angiogram.
POSTERIOR SCLERITIS
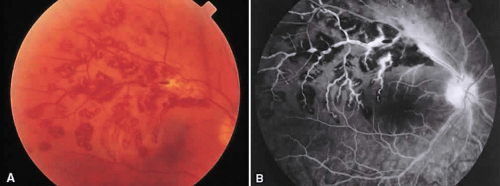
Posterior scleritis is a generally unilateral condition that usually is seen in patients 40 to 60 years old. Patients present with pain, decreased vision, and decreased visual fields. It generally is associated with anterior scleritis. Posterior scleritis may cause exudative retinal detachment resembling Harada’s disease (Fig. 5A), cystoid macular edema, retinal pigment epithelial detachments, choroidal folds, and subretinal masses. Thickening of the choroid and posterior sclera can be seen with ultrasound, magnetic resonance imaging, and computed tomography.
Fluorescein angiography can show several manifestations. Frequently, the angiographic picture of posterior scleritis is identical to that of Vogt-Koyanagi-Harada syndrome, except that posterior scleritis usually is unilateral. Multiple early pinpoint retinal pigment epithelial leaks with late staining of the subretinal fluid can be seen (see Fig. 5B).14 Another pattern is early multifocal choriocapillaris filling defects followed by overlying retinal pigment epithelial staining, late staining of the subretinal fluid, and disc staining.15 Cystoid macular edema16 and multiple discrete retinal pigment epithelial detachments can be seen.
ICG angiography shows diffuse zonal hyperfluorescence in the intermediate and late phases. Pinpoint areas of hyperfluorescence also may be seen in the areas of zonal hyperfluorescence in the later phases of the angiogram. Delayed choroidal perfusion and hypofluorescent spots present up to the intermediate phase and faded in the late phase of the angiogram may be visualized. These spots are noted to be smaller and more irregular in size than those observed in Vogt-Koyanagi-Harada syndrome.17
RETICULUM CELL SARCOMA
Reticulum cell sarcoma, a non-Hodgkin’s lym-phoma, is a systemic disease that can affect many organs, including the eyes and central nervous system. Seventy-five percent of the patients with ocular and central nervous system involvement have no other manifestation. Patients with ocular reticulum cell sarcoma tend to be older and have painless loss of vision. Findings include vitreous cells that are not responsive to corticosteroids, multiple subretinal pigment epithelial detachments with yellow infiltrates, exudative retinal detachment, and retinal vascular sheathing. On fluorescein angiography, patients with reticulum cell sarcoma with multiple retinal pigment epithelial detachments have an irregular, incomplete staining of these detachments.18
BEHÇET’S DISEASE
Behçet’s disease is a systemic occlusive vasculitis that presents predominately in young middle Eastern and Japanese men. The classic features include acute hypopyon, iritis, aphthous stomatitis, and genital ulceration. Skin lesions and strokes also occur frequently. There often is an acute recurrent bilateral panuveitis. Ocular findings include retinal vasculitis (Fig. 6A) with an occlusive arteritis, vitritis, macular edema, ischemic retinitis, ischemic optic neuropathy, peripheral neovascularization, and occasionally SRNV.
On fluorescein angiography (see Fig. 6B), during the active phases of the disease, capillary dropout and dilated retinal capillaries are seen. Dilated retinal capillaries (particularly peripapillary capillaries) leak dye and cause retinal and disc staining.19 Cystoid macular edema,19 SRNV, and disciform scars often are seen.20 Leakage of peripheral capillaries can be seen in patients with normal-appearing fundi.21
SARCOIDOSIS
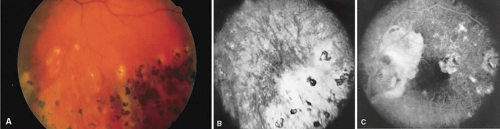
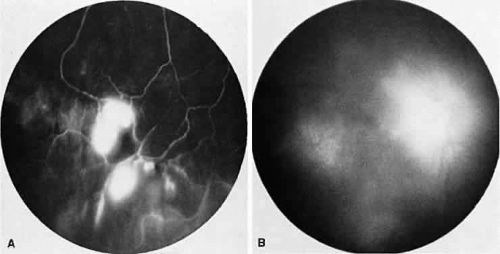
Sarcoidosis is a systemic, idiopathic, noncaseating, granulomatous disease that affects various organs, including the eye, brain, lung, and skin. Ocular findings may include conjunctival nodules; anterior iridocyclitis; vitreous cells; retinal, optic nerve, and choroidal granulomas (Fig. 7A); retinal vasculitis (venules are preferentially affected); cystoid macular edema; retinal vessel occlusion; and disc and retinal neovascularization.
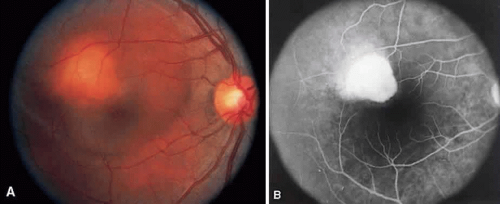 Fig. 7. Sarcoidosis. A. Choroidal granuloma. B. Fluorescein angiogram shows staining of the choroidal granuloma. |
The FA reflects the various clinical entities. Retinal venular walls stain, particularly where there are perivenular exudates.25,26,27 More extensive venous involvement can produce a picture of dilated veins and perivenous leakage.27 Peripheral neovascularization occurs near areas of retinal capillary nonperfusion.25,28 Optic disc granulomas and optic disc neovascularization both leak extensively (Fig. 8A); sarcoid retinal lesions also stain (see Fig. 7B).27 In disc edema, the disc is hyperfluorescent and leaks fluorescein (see Fig. 8B).25
Four main patterns can be identified with ICG angiography. The first and most common pattern is hypofluorescent dark spots in the early and intermediate phases of the angiogram. These spots either become isofluorescent or remain hypofluorescent in the late phases. The second pattern is focal hyperfluorescent spots seen in the intermediate and late phases. The third pattern is fuzzy choroidal vessels due to perivascular choroidal leakage in the intermediate phase. Finally, the fourth pattern is characterized by diffuse zonal hyperfluorescence representing choroidal staining in the late phase of the angiogram. The latter two patterns resolved after systemic corticosteroid treatment.29
INTERMEDIATE UVEITIS
Intermediate uveitis is also known as pars planitis, peripheral uveitis, and chronic cyclitis. Intermediate uveitis is an inflammatory disease that affects young adults, causing symptoms of photophobia, floaters, and blurry vision. Clinically, mild anterior chamber inflammation, vitreous cells, vitreous snowballs, inflammatory membranes on the pars plana, phlebitis, cystoid macular edema, and, rarely, choroidal and retinal neovascularization are seen.
On fluorescein angiography, there is venular wall staining (Fig. 9),30 hyperfluorescence, and leakage of the peripheral inflammatory membranes.31 Cystoid macular edema often is evident.30 Optic disc, peripheral retinal, and subretinal32,33,34 neovascularization are rare.
BIRDSHOT RETINOCHOROIDOPATHY
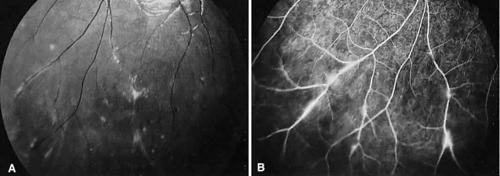
Birdshot retinochoroidopathy (also known as vitiliginous chorioretinitis) presents bilaterally, generally in middle-aged women, causing floaters and decreased vision, night blindness, and color blindness. Clinically, there are patches of postequatorial choroidal and retinal pigment epithelial depigmentation (Fig. 10A), vitreous cells, macular and disc edema, and venous sheathing. SRNV frequently is a late sequela. Often central vision may be preserved in at least one eye.
On fluorescein angiography, retinal vessel staining, disc leakage, and cystoid macular edema are found (see Fig. 10B). There often is generalized hypofluorescence of the retinal vessels and increased circulation time.35,36 Surprisingly, the patches of depigmentation may appear normal on angiography, although there can be mild late hyperfluorescence.35,36 Posterior pole choroidal hyperfluorescent lesions that correspond to the areas of depigmentation and SRNV also can be seen.37
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree