Fig. 12.1
Super-selective catheterization of the ophthalmic artery (Reproduced with permission from Jabbour et al. [33])
12.4 Current Results
Abramson’s initial report was that of a phase I/II trial of SSIOAC with melphalan [9]. Ten patients with advanced retinoblastoma (Reese-Ellsworth Group V) who met criteria for enucleation were enrolled in an effort to attain globe salvage. Nine of the ten patients had successful OA cannulation. Clinical response to chemotherapy was seen in all cases (Fig. 12.2), with favorable effects on vitreous seeding noted. The number of treatment sessions varied between 2 and 6 to the same eye. An additional chemotherapeutic agent, carboplatin, was used in combination with the melphalan in selective cases. A total of 27 ophthalmic artery cannulations were performed. At the end of the study, 2 of 9 eyes had been enucleated for suspected tumor recurrence. Median follow-up time was 7.5 months. No adverse anesthesia or vascular complications from the catheterization process were reported.
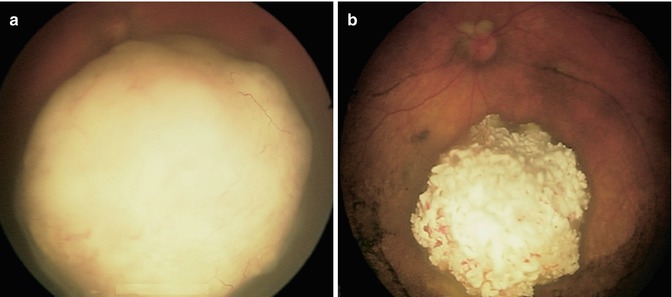

Fig. 12.2
Before (a) and 3 weeks after (b) a single dose of melphalan (3 mg) delivered via SSIOAC (Reproduced with permission from Abramson et al. [9])
In a subsequent publication from the group, Gobin et al. published the largest series of patients treated with SSIOAC to date, performing a total of 289 catheterizations in 95 eyes – 83 of which were RE Group V (Fig. 12.3) [10]. Only 41 % of these eyes had not received prior therapy. All patients were alive at the end of the study, with two developing systemic metastases. They found ocular event-free survival at 2 years to be 70.0 % (95 % confidence interval (CI), 57.9–82.2 %) in all eyes, 81.7 % (95 % CI, 66.8–96.6) in eyes receiving IAC as primary therapy, and 58.4 % (95 % CI, 39.5–77.2 %) in eyes that had been previously treated with intravenous chemotherapy and/or external beam radiotherapy. In all, 77 % of eyes required additional treatment during or after IAC, with 20 % ultimately having to be enucleated secondary to tumor growth or insufficient tumor regression. Median follow-up time was 13 months.
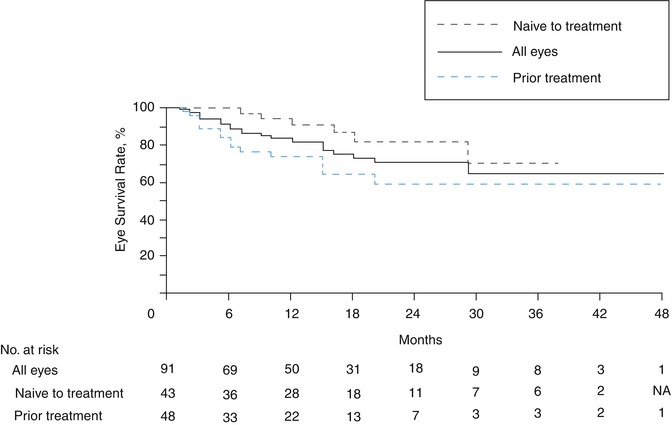

Fig. 12.3
Kaplan-Meier curve of eye survival until enucleation or external beam radiation therapy in eyes treated with SSIOAC (Reproduced with permission from Gobin et al. [10])
The same group has also reported SSIOAC with three-drug therapy (simultaneous carboplatin, topotecan, and melphalan) in eyes that had tumors refractory to treatment with systemic chemotherapy, SSIOAC with 1 or 2 agents, or external beam radiation [11]. The eye salvage rate was 88 % (23 of 26 eyes) at a mean follow-up of 14 months. Eleven of 26 eyes (35 %) developed disease recurrence and were treated with enucleation (n = 3), or focal therapy (n = 8) including plaque brachytherapy (n = 3).
Shields et al. published a series of 38 catheterizations on an initial cohort of 17 retinoblastoma patients – 13 of which had SSIAOC as their primary treatment [12]. A 5 mg dose of melphalan was used. Ocular salvage was achieved in 8 of the 12 eyes successfully treated with primary SSIOAC. Failures occurred in four of the six Group E eyes (International Classification). Additional therapeutic measures were needed to control disease in some eyes.
Muen et al. reported outcomes of 15 eyes in 14 patients previously treated with systemic chemotherapy or local therapy with refractory or recurrent retinoblastoma [13]. After two or three intra-arterial treatments with melphalan, tumor control was achieved in 12 eyes. Venturi et al. treated 41 eyes in 38 patients with 140 SSIOAC procedures between 2008 and 2010 [14]. Two patients had failed catheterizations and two patients were lost to follow-up. Of the 37 remaining eyes, 8 were enucleated, 7 of which had received no prior treatment. In a subsequent study of 48 eyes in 43 patients, the authors successfully saved 65 % of the eyes; however, only 21 eyes (44 %) were treated with SSIOAC using melphalan alone [15]. Additional chemotherapies – local and systemic – focal therapies, and brachytherapy were needed. Success rates were higher among previously treated eyes as opposed to newly diagnosed eyes, 78 % versus 48 %.
Thampi and colleagues treated 20 eyes in 16 patients; a total of 40 procedures were performed, ranging from 1 to 5 per patient [16]. The dose of melphalan was adjusted based on the age of the patient. At median follow-up of 14.5 months, ranging 1–29 months, radiotherapy and enucleation had been avoided in 86 % (6/7) of Groups A-C eyes and 38 % (5/13) of Groups D and E eyes.
12.5 Visual Outcome
Visual outcomes in treated eyes are of great interest, although little data have been published to date. Brodie et al. have monitored electroretinograms (ERGs) during sequential cycles of SSIOAC as surrogate for visual outcomes in this predominantly preverbal population. Retinal function has been observed to improve initially, remain stable, and later decline [17]. Tsimpida and colleagues looked at 12 eyes with refractory retinoblastoma that had good visual potential based on healthy foveolas as noted by ophthalmoscopy and optical coherence tomography prior to SSIOAC [18]. Five out of 12 eyes (42 %) sustained a reduction in visual acuity, with 2 suffering severe vision loss. Reasons for vision loss included diffuse retinal detachment (n = 1), diffuse choroidal ischemia (n = 2), and sectoral choroidal ischemia involving the fovea (n = 2). Four of the five eyes sustaining vision loss, however, had been previously treated with either EBRT or plaque brachytherapy, suggesting that previous radiation may predispose SSIOAC patients to visual loss. However, two patients with macula-sparing choroidal ischemia had no prior radiation exposure, which supports prior evidence that SSIOAC can lead to vasculopathy. Posttreatment ERGs deteriorated in four of eight eyes. Additional prospective, long-term follow-up studies on survival, metastasis rates, ocular survival, visual outcomes, and ocular toxicities are still needed.
12.6 Confounding Factors
SSIOAC for the treatment of retinoblastoma has quickly disseminated worldwide. Though the outcomes (eye salvage rates) appear promising, the reported case series have many confounding factors that limit the generalizability of this technique. Many patients have been treated previously, concurrently, or subsequently with other therapies. Various doses and agents have been utilized, sometimes with dose titrated to effect. It is difficult to assess the overall efficacy of SSIOAC, and thus, the singular effect of SSIOAC remains unknown.
12.7 Complications
Reported complications following SSIOAC are limited to case reports, as this therapy has not yet been evaluated as part of a large prospective retinoblastoma clinical trial. As such, these reports have been sporadic, including sectoral choroidal occlusion, retinal arteriolar emboli, retinal detachment, vitreous hemorrhage, transient retinal ischemia, ophthalmic artery obstruction, and cataract formation. Periocular inflammation and edema, cranial nerve III palsies, strabismus, and eyelash loss are reported orbital and adnexal side effects [10, 13, 14, 19–22]. Additionally, Fallaha et al. documented retinal vascular precipitates during the administration of melphalan into the ophthalmic artery [23].
Histopathology studies of eyes with retinoblastoma treated with SSIOAC have documented findings that have been attributed to the toxicities of SSIAOC. Eagle and coworkers examined eight eyes enucleated after SSIOAC and observed ischemic atrophy involving the outer retina and choroid (n = 4) (Fig. 12.4), orbital vascular occlusion and sub-endothelial smooth muscle hyperplasia (n = 1), and thrombosed blood vessels involving the retrobulbar ciliary arteries (n = 5), scleral emissary canals (n = 1), small choroidal vessels (n = 1), and CRA (n = 1) [24]. Intravascular birefringent foreign material was noted in five eyes and classified as cellulose fibers (n = 3), synthetic fabric fibers (n = 1), or unknown composition (n = 2). Other histopathology studies have focused principally on residual viable tumor [25–27]. In these eyes that were enucleated for progression of disease, partial response to IAC was seen in most cases, although many also had areas of non-necrotic, viable tumor and vitreous seeding still present. None of these papers reported evidence of ocular toxicity to the surrounding tissues from IAC.
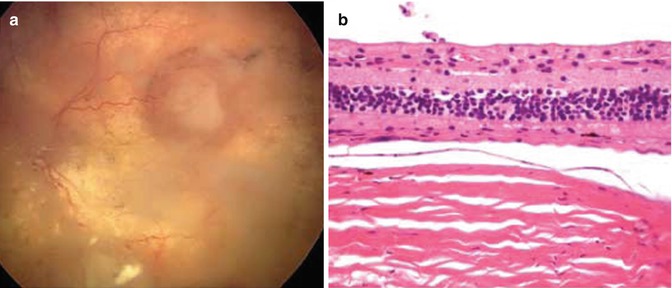

Fig. 12.4
Fundus photograph showing chorioretinal atrophy and foci of viable retinoblastoma tumor (a). Histopathology of the same patient showing severe choroidal atrophy and loss of outer nuclear layer, photoreceptors, and retinal pigment epithelium (b) (Reproduced with permission from Eagle et al. [24])
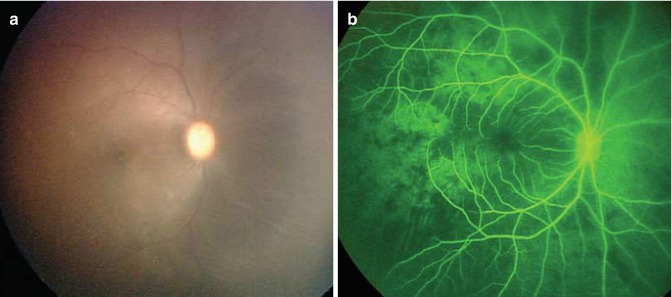
Melphalan, a potent alkylator, has known vascular toxicities [28]. In a 2011 editorial, Wilson et al. attributed the reported vascular complications of SSIOAC to concentration of melphalan being used [29]. A preclinical model was developed to study the vascular sequelae of SSIOAC [30]. Using techniques and protocols similar to those previously published, a cohort of six non-human primates were treated with SSIOAC, and real-time ophthalmoscopic findings were documented during each infusion. Visible pulsatile pallor of the optic nerve, choroidal blanching, and retinal arterial narrowing were observed (Fig. 12.5). Sectoral choroidal non-perfusion and diffuse capillary dropout were seen on fluorescein angiography immediately following chemotherapy infusion (Fig. 12.5). Orbital and ocular histopathology revealed drug-induced toxicity to retinal endothelial cells as well as technique-induced changes to the orbital vasculature, including intimal hyperplasia, fracturing of the internal elastic lamina, and arterial wall dissection involving the ophthalmic, and central retinal arteries [31].
 < div class='tao-gold-member'>
< div class='tao-gold-member'>





Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


