Herpes viruses infecting the retina manifest themselves differently depending upon the interaction between the virus and the host immune system. Most of them are known as necrotizing retinopathies, but nonnecrotizing forms should also be considered in atypical cases of chronic posterior uveitis.
 Necrotizing retinopathies are by far the most frequent clinical picture, with a spectrum of severity ranging from the acute retinal necrosis syndrome (almost always seen in healthy individuals) to the progressive outer retinal necrosis syndrome, (PORN), and cytomegalovirus (CMV) retinitis, both of which mainly affect severely immunocompromised patients.
Necrotizing retinopathies are by far the most frequent clinical picture, with a spectrum of severity ranging from the acute retinal necrosis syndrome (almost always seen in healthy individuals) to the progressive outer retinal necrosis syndrome, (PORN), and cytomegalovirus (CMV) retinitis, both of which mainly affect severely immunocompromised patients.
 Nonnecrotizing herpetic retinopathies are less frequent and have a number of different manifestations, including vitritis, occlusive vasculitis, papillitis, or macular edema.
Nonnecrotizing herpetic retinopathies are less frequent and have a number of different manifestations, including vitritis, occlusive vasculitis, papillitis, or macular edema.
ACUTE RETINAL NECROSIS SYNDROME
Acute retinal necrosis (ARN) syndrome is characterized by peripheral necrotizing retinitis, retinal vasculitis, a prominent inflammatory reaction in the vitreous, and a granulomatous anterior uveitis.
Epidemiology and Etiology
 ARN is a rare disease, occurring in 1 per 1.6 to 2.0 million people per year in Western countries.
ARN is a rare disease, occurring in 1 per 1.6 to 2.0 million people per year in Western countries.
 In most cases, ARN is caused by varicella zoster virus (VZV); however, herpes simplex viruses 1 (HSV-1) and 2 (HSV-2) also cause ARN.
In most cases, ARN is caused by varicella zoster virus (VZV); however, herpes simplex viruses 1 (HSV-1) and 2 (HSV-2) also cause ARN.
 HSV-2 is more prevalent in children and adolescents, while HSV-1 mainly occurs in young adults. VZV is usually found in the elderly.
HSV-2 is more prevalent in children and adolescents, while HSV-1 mainly occurs in young adults. VZV is usually found in the elderly.
Symptoms
 Acute, painless visual loss with floaters
Acute, painless visual loss with floaters
 65% of cases are unilateral at presentation; 33% of cases are bilateral (BARN, bilateral acute retinal necrosis)
65% of cases are unilateral at presentation; 33% of cases are bilateral (BARN, bilateral acute retinal necrosis)
 Conjunctival and ciliary injection of varying intensity
Conjunctival and ciliary injection of varying intensity
Signs (Figs. 9-1 to 9-6)
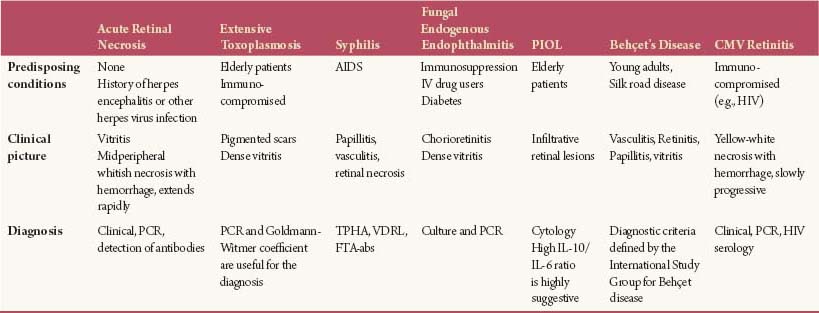
 Anterior granulomatous uveitis, which rarely can be associated with hypopyon or hyphema
Anterior granulomatous uveitis, which rarely can be associated with hypopyon or hyphema
 Intense vitritis
Intense vitritis
 Patchy or confluent areas of white or cream-colored retinal necrosis initially affecting the peripheral retina and then extending centripetally
Patchy or confluent areas of white or cream-colored retinal necrosis initially affecting the peripheral retina and then extending centripetally
 Occlusive vasculopathy
Occlusive vasculopathy
 Papillitis of varied intensity
Papillitis of varied intensity
 Immunocompetent, otherwise healthy young or middle-aged patients, with or without a remote history of herpes virus infection or herpetic encephalitis (an established risk factor for ARN)
Immunocompetent, otherwise healthy young or middle-aged patients, with or without a remote history of herpes virus infection or herpetic encephalitis (an established risk factor for ARN)
 ARN is a medical emergency. It is rapidly progressive and second eye involvement will naturally occur in almost 70% of patients in the absence of treatment. More than 50% of patients develop a rhegmatogenous retinal detachment (RRD) due to retinal atrophy secondary to necrosis and vitreous contraction.
ARN is a medical emergency. It is rapidly progressive and second eye involvement will naturally occur in almost 70% of patients in the absence of treatment. More than 50% of patients develop a rhegmatogenous retinal detachment (RRD) due to retinal atrophy secondary to necrosis and vitreous contraction.
Differential Diagnosis
 ARN needs to be differentiated from other nonviral necrotizing retinopathies, infectious or not (Table 9-1):
ARN needs to be differentiated from other nonviral necrotizing retinopathies, infectious or not (Table 9-1):
 Extensive Toxoplasma retino- choroidopathy
Extensive Toxoplasma retino- choroidopathy
 Syphilitic retinitis
Syphilitic retinitis
 Fungal endogenous endophthalmitis
Fungal endogenous endophthalmitis
 Primary intraocular lymphoma (PIOL)
Primary intraocular lymphoma (PIOL)
 Retinitis associated with Behçet’s disease
Retinitis associated with Behçet’s disease
 CMV retinitis (occurs only in severely immunocompromised patients)
CMV retinitis (occurs only in severely immunocompromised patients)
Diagnostic Evaluation
 ARN is primarily a clinical diagnosis.
ARN is primarily a clinical diagnosis.
 Because the natural disease course is usually devastating, ancillary tests confirm the etiology and/or eliminate nonviral causes, but they should never cause a delay in empirical treatment.
Because the natural disease course is usually devastating, ancillary tests confirm the etiology and/or eliminate nonviral causes, but they should never cause a delay in empirical treatment.
 Two different assays are performed in ocular samples (aqueous humor or vitreous):
Two different assays are performed in ocular samples (aqueous humor or vitreous):
 Polymerase chain reaction (PCR) for direct detection of viral DNA is highly sensitive with 80% to 96% positivity in both immunocompetent and immunocompromised hosts.
Polymerase chain reaction (PCR) for direct detection of viral DNA is highly sensitive with 80% to 96% positivity in both immunocompetent and immunocompromised hosts.
 Indirect detection of antibodies directed against virus proteins has 50% to 70% positivity in immunocompetent patients.
Indirect detection of antibodies directed against virus proteins has 50% to 70% positivity in immunocompetent patients.
 In cases of suspected meningitis, a brain MRI and a lumbar puncture should be performed without delay as simultaneous herpetic meningitis or encephalitis is possible.
In cases of suspected meningitis, a brain MRI and a lumbar puncture should be performed without delay as simultaneous herpetic meningitis or encephalitis is possible.
Treatment
 The primary treatment is medical. Traditionally, this has been administered in an inpatient setting during the acute phase; however, outpatient management has been reported recently but remains somewhat controversial.
The primary treatment is medical. Traditionally, this has been administered in an inpatient setting during the acute phase; however, outpatient management has been reported recently but remains somewhat controversial.
 The primary treatment is antiviral therapy, which is used to control viral replication and to reduce the risk of bilateralization. Intravitreal injections can be used in cases that are imminently threatening the macula or optic nerve.
The primary treatment is antiviral therapy, which is used to control viral replication and to reduce the risk of bilateralization. Intravitreal injections can be used in cases that are imminently threatening the macula or optic nerve.
 Anti-inflammatory therapy is used to minimize the deleterious effects associated with inflammation. Viral replication must be controlled with antiviral medications before administration of systemic corticosteroids.
Anti-inflammatory therapy is used to minimize the deleterious effects associated with inflammation. Viral replication must be controlled with antiviral medications before administration of systemic corticosteroids.
 Antithrombotic therapy should be considered in order to reduce vascular complications.
Antithrombotic therapy should be considered in order to reduce vascular complications.
Table 9-1. Necrotizing Retinopathies
 Laser retinopexy may reduce the occurrence of a RRD, but its efficacy remains controversial.
Laser retinopexy may reduce the occurrence of a RRD, but its efficacy remains controversial.
 Patients with vitreous hemorrhage secondary to retinal neovascularization or for RRD should have pars plana vitrectomy, with or without silicone oil tamponade. Early vitrectomy with intravitreal injection of antiviral medications and laser retinopexy can be performed to prevent RRD, although no improvement in final visual acuity has been reported.
Patients with vitreous hemorrhage secondary to retinal neovascularization or for RRD should have pars plana vitrectomy, with or without silicone oil tamponade. Early vitrectomy with intravitreal injection of antiviral medications and laser retinopexy can be performed to prevent RRD, although no improvement in final visual acuity has been reported.
 Treatment medications are outlined in Table 9-2.
Treatment medications are outlined in Table 9-2.
Prognosis
 Despite early recognition and institution of prompt therapy, the final visual outcome is generally poor.
Despite early recognition and institution of prompt therapy, the final visual outcome is generally poor.
 RRD and ischemic optic neuropathy (with subsequent optic nerve atrophy) preclude good final visual acuity.
RRD and ischemic optic neuropathy (with subsequent optic nerve atrophy) preclude good final visual acuity.
REFERENCES
Aizman A, Johnson MW, Elner SG. Treatment of acute retinal necrosis syndrome with oral antiviral medications. Ophthalmology. 2007;114:307–312.
Balansard B, Bodaghi B, Cassoux N, et al. Necrotizing retinopathies simulating acute retinal necrosis syndrome. Br J Ophthalmol. 2005;89(1):96–101.
Ganatra JB, Chandler D, Santos C, et al. Viral causes of the acute retinal necrosis syndrome. Am J Ophthalmol. 2000;129(2):166–172.
Holland, GN. Standard diagnostic criteria for the acute retinal necrosis syndrome. Executive Committee of the American Uveitis Society. Am J Ophthalmol. 1994;117(5):663–667.
Tibbetts MD, Shah CP, Young LH, et al. Treatment of acute retinal necrosis. Ophthalmology. 2010;117(4):818–824.
Wong R, Pavesio CE, Laidlaw DA, et al. Acute retinal necrosis: the effects of intravitreal foscarnet and virus type on outcome. Ophthalmology. 2010;117(3):556–560.
Table 9-2. Medical Treatment of ARN
| Active Drug | Dosages | |
| Antivirals | Acyclovir | 10 mg/kg/8 h (1500 mg/m2) IV or |
| 800 mg PO 5 times/day | ||
| Foscarnet | 90 mg/kg/12 h IV or/and intravitreal injections | |
| Ganciclovir | 5 mg/kg/12 h IV or/and intravitreal injections | |
| Valganciclovir | 900 mg/day b.i.d. | |
| Anti-inflammatory drugs | Methylprednisolone | 500 mg daily IV for 3 days |
| Prednisone | 0.5–1 mg/kg/day with a gradual taper | |
| Antithrombotic drugs | Standard heparin | |
| Aspirin (acetylsalicylic acid) | 100 mg/day |
Figure 9-1. Granulomatous anterior uveitis in a patient with ARN syndrome.
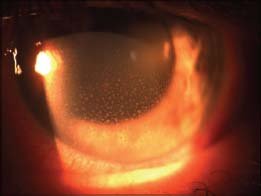
Figure 9-2. There are multiple white to yellow-white foci of peripheral retinal necrosis in a case of ARN syndrome. These occur in a circumferential fashion.
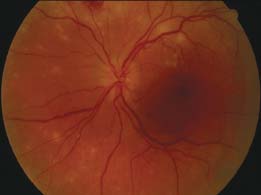
Figure 9-3. This 48-year-old patient presented with sudden onset of blurred vision and floaters. A. There are numerous focal white areas of retinitis and vasculitis in the periphery along with optic disk edema. B. Three days after the institution of therapy, the areas of retinitis appear slightly larger and better defined. Some scattered peripheral retinal hemorrhages are visible. C. In the late frames of the fluorescein angiogram, there is staining of the optic disk. D. The larger, superior patch of retinitis appears hypofluorescent centrally with a ring of late staining. (Courtesy of Paul Baker, MD.)
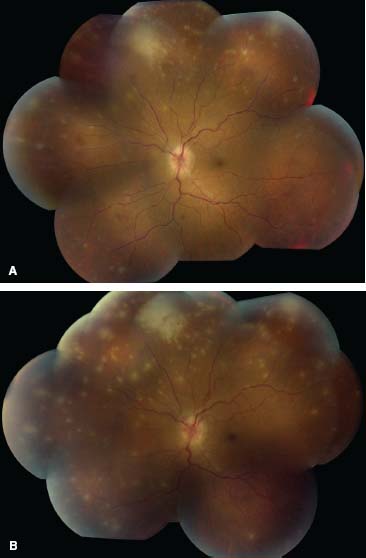
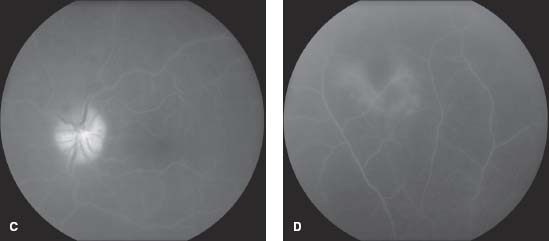
Figure 9-4. This person had extensive toxoplasma retinochoroiditis masquerading as ARN.
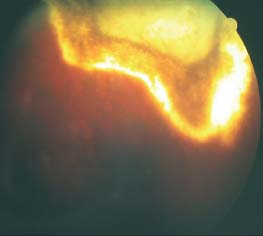
Figure 9-5. Pseudo-ARN syndrome with dense vitritis, retinal necrosis, and hemorrhages in a case of primary intraocular lymphoma.
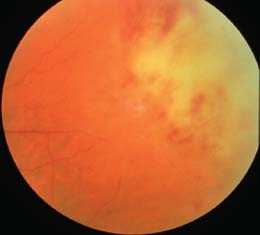
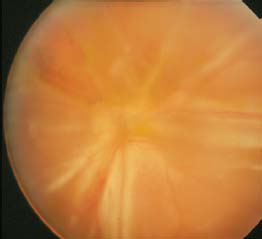
Figure 9-6. This patient with extensive ARN developed a retinal detachment.
PROGRESSIVE OUTER RETINAL NECROSIS
Progressive outer retinal necrosis (PORN) is the major form of necrotizing herpetic retinitis affecting immunocompromised patients. Early diagnosis and aggressive antiviral therapy is mandatory, but the visual prognosis generally remains poor.
Epidemiology and Etiology
 PORN is the most severe clinical form of herpetic retinitis. Although it appears to affect the outer retina, all retinal layers can become involved.
PORN is the most severe clinical form of herpetic retinitis. Although it appears to affect the outer retina, all retinal layers can become involved.
 VZV is the most common agent identified in patients with PORN, but a few cases of HSV-1–associated PORN have been described.
VZV is the most common agent identified in patients with PORN, but a few cases of HSV-1–associated PORN have been described.
 Patients are usually deeply immunocompromised. Most of them suffer from AIDS; however, it has also been described in patients who have had bone marrow transplantation and also those treated with high-dose systemic corticosteroids.
Patients are usually deeply immunocompromised. Most of them suffer from AIDS; however, it has also been described in patients who have had bone marrow transplantation and also those treated with high-dose systemic corticosteroids.
 PORN represents an ophthalmologic and medical emergency, as it usually is bilateral and progresses rapidly, and CNS involvement is possible. PORN usually has a poor visual outcome.
PORN represents an ophthalmologic and medical emergency, as it usually is bilateral and progresses rapidly, and CNS involvement is possible. PORN usually has a poor visual outcome.
Symptoms
 Painless decrease of visual acuity
Painless decrease of visual acuity
 Abrupt loss of central vision in those cases with initial macular involvement
Abrupt loss of central vision in those cases with initial macular involvement
 Constriction of visual fields
Constriction of visual fields
Signs (Figs. 9-7 to 9-9)
 Multifocal, poorly demarcated, deep retinal opacities of various sizes, without granular borders, scattered throughout the posterior pole and the midperipheral retina. The retinitis spreads outwardly and peripherally, becoming confluent within a few days.
Multifocal, poorly demarcated, deep retinal opacities of various sizes, without granular borders, scattered throughout the posterior pole and the midperipheral retina. The retinitis spreads outwardly and peripherally, becoming confluent within a few days.
 Retinal vasculitis and optic neuritis occur in less than 20% of cases.
Retinal vasculitis and optic neuritis occur in less than 20% of cases.
 The aqueous humor and the vitreous have minimal to no inflammation.
The aqueous humor and the vitreous have minimal to no inflammation.
 Extremely rapid progression to total retinal necrosis over the course of a few days.
Extremely rapid progression to total retinal necrosis over the course of a few days.
 The disease is bilateral at presentation in 70% of cases, and 80% will become bilateral within the first month.
The disease is bilateral at presentation in 70% of cases, and 80% will become bilateral within the first month.
 There is a history of recent or ongoing VZV infection in 75% of cases.
There is a history of recent or ongoing VZV infection in 75% of cases.
Differential Diagnosis
 CMV retinitis remains the most common opportunistic infection in AIDS, but the clinical picture and course is very different and it usually does not represent a diagnostic dilemma.
CMV retinitis remains the most common opportunistic infection in AIDS, but the clinical picture and course is very different and it usually does not represent a diagnostic dilemma.
 ARN syndrome is also a diagnosis to exclude. ARN occurs in otherwise healthy patients, affects the retinal vasculature, and is associated with vitritis (Table 9-3).
ARN syndrome is also a diagnosis to exclude. ARN occurs in otherwise healthy patients, affects the retinal vasculature, and is associated with vitritis (Table 9-3).
 Toxoplasma retinochoroiditis when affecting the elderly or immunosuppressed hosts may also produce a similar clinical picture.
Toxoplasma retinochoroiditis when affecting the elderly or immunosuppressed hosts may also produce a similar clinical picture.
Diagnostic Evaluation
 The classic clinical picture should suggest the diagnosis and prompt empirical treatment should be administered before ancillary tests are performed.
The classic clinical picture should suggest the diagnosis and prompt empirical treatment should be administered before ancillary tests are performed.
 Aqueous humor PCR should be performed for viral DNA identification and antibody production in order to confirm the pathogenic agent and eliminate other nonviral etiologies.
Aqueous humor PCR should be performed for viral DNA identification and antibody production in order to confirm the pathogenic agent and eliminate other nonviral etiologies.
 Brain MRI and lumbar puncture should be performed in every case of PORN, because extremely immunosuppressed patients have an increased risk of encephalitis.
Brain MRI and lumbar puncture should be performed in every case of PORN, because extremely immunosuppressed patients have an increased risk of encephalitis.
Treatment (Table 9-4)
 Patients with PORN must be treated in an inpatient setting.
Patients with PORN must be treated in an inpatient setting.
Table 9-3. Clinical Characteristics of ARN and PORN Syndromes
| ARN | PORN | |
| Host immune status | Immunocompetent or immunocompromised | Immunocompromised, mainly AIDS |
| Etiologic agent | VZV, HSV-1, HSV-2 | Mainly VZV |
| Retinal vasculitis | Present | Rare |
| Intraocular inflammation (KPs, AC cells, vitritis) | Present and very important | Rare |
| Bilaterality | 25% at presentation | 70% at presentation |
| Progression | Centripetally from peripheral foci of retinal necrosis to posterior pole | Centrifugally with rapid confluence |
| Prognosis | Poor | Extremely poor |
 Combined IV and intravitreal treatment with ganciclovir and foscarnet is the strategy of choice, as IV acyclovir does not treat PORN effectively.
Combined IV and intravitreal treatment with ganciclovir and foscarnet is the strategy of choice, as IV acyclovir does not treat PORN effectively.
 IV corticosteroids must be avoided in nearly all cases.
IV corticosteroids must be avoided in nearly all cases.
 Acetylsalicylic acid (100 mg per day) can be used to decrease platelet hyperaggregation that occurs in herpetic retinitis.
Acetylsalicylic acid (100 mg per day) can be used to decrease platelet hyperaggregation that occurs in herpetic retinitis.
 Prophylactic argon laser retinopexy delimitating necrotic retinal areas can be applied in an effort to reduce the high risk of RRD, although studies are inconclusive about its advantages.
Prophylactic argon laser retinopexy delimitating necrotic retinal areas can be applied in an effort to reduce the high risk of RRD, although studies are inconclusive about its advantages.
 Once RRD has occurred, vitrectomy with silicone oil tamponade is the management of choice. Some authors advocate performing early vitrectomy with intravitreal acyclovir lavage and laser demarcation to prevent RRD, although this has not led to an improvement in final visual acuity.
Once RRD has occurred, vitrectomy with silicone oil tamponade is the management of choice. Some authors advocate performing early vitrectomy with intravitreal acyclovir lavage and laser demarcation to prevent RRD, although this has not led to an improvement in final visual acuity.
 Patients should be tested for comorbid conditions, such as AIDS. If HIV is found, highly active antiretroviral therapy (HAART) should be promptly instituted.
Patients should be tested for comorbid conditions, such as AIDS. If HIV is found, highly active antiretroviral therapy (HAART) should be promptly instituted.
Prognosis
 Despite aggressive treatment, the prognosis is extremely poor. The final visual outcome is no light perception in almost 60% of eyes, mainly related to total retinal necrosis and RRD.
Despite aggressive treatment, the prognosis is extremely poor. The final visual outcome is no light perception in almost 60% of eyes, mainly related to total retinal necrosis and RRD.
Table 9-4. Medical Treatment of PORN
| Active Drug | Dose and Route of Administration | Toxicity |
| Ganciclovir | 5 mg/kg IV b.i.d. for 3 weeks, then 5 mg/kg daily IVT injections: 1–2 mg/0.05 mL | Blood cells and liver |
| Foscarnet | 180 mg/kg/day IV divided into 2–3 infusions for 3 weeks, then 90–120 mg/kg/day IVT injections: 1.2–2.4 mg/0.05 mL | Renal failure |
| Valgancyclovir | 900 mg/day PO, after IV induction phase, as a maintenance regimen | Blood cells and liver |
| Valacyclovir | 1 g PO t.i.d. as maintenance treatment | Renal failure |
 Ischemic optic neuropathy with further optic atrophy also precludes good visual restoration.
Ischemic optic neuropathy with further optic atrophy also precludes good visual restoration.
REFERENCES
Benz MS, Glaser JS, Davis JL. Progressive outer retinal necrosis in immunocompetent patients treated initially for optic neuropathy with systemic corticosteroids. Am J Ophthalmol. 2003;135(4):551–553.
Chau Tran TH, et al. Successful treatment with combination of systemic antiviral drugs and intravitreal ganciclovir injections in the management of severe necrotizing herpetic retinitis. Ocul Immunol Inflamm. 2003;11(2):141–144.
Engstrom RE, Jr., et al. The progressive outer retinal necrosis syndrome. A variant of necrotizing herpetic retinopathy in patients with AIDS. Ophthalmology. 1994;101(9):1488–1502.
Forster DJ, et al. Rapidly progressive outer retinal necrosis in the acquired immunodeficiency syndrome. Am J Ophthalmol. 1990;110(4):341–348.
Moorthy RS, et al. Management of varicella zoster virus retinitis in AIDS. Br J Ophthalmol. 1997;81(3):189–194.
Figure 9-7. VZV-associated PORN with retinal necrosis and retinal hemorrhages in an AIDS patient.
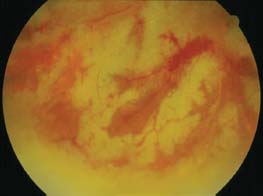
Figure 9-8. Multifocal areas of outer retinal necrosis in a patient with PORN.
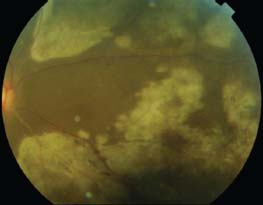
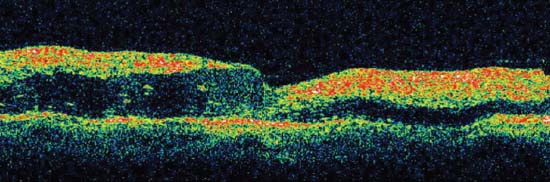
Figure 9-9. The OCT shows full thickness retinal involvement, most prominent in the outer retina. (Courtesy of Sunir J. Garg, MD, and Heather Shelsta, MD.)
CONGENITAL RUBELLA SYNDROME
P. Vijayalakshmi
Rubella is a mild systemic viral illness transmitted by respiratory droplets, and has an incubation period of 2 weeks. When a pregnant woman gets infected for the first time, the virus is transmitted transplacentally and it can lead to miscarriage, stillbirth, or to an infant with multiple systemic abnormalities. The range of congenital defects differs according to the gestational age of the child and the earlier in gestation that the infection occurs, the worse the damage; infection during the first 12 weeks usually results in cardiac and ocular involvement, while infection during the 12th to 28th weeks results in deafness and pulmonary artery stenosis. The consequences of rubella infection in utero are collectively termed congenital rubella syndrome (CRS).
Etiology and Epidemiology
 Rubella is a single-stranded RNA virus and humans are the only host. The World Health Organization estimates that more than 100,000 children are born with CRS each year worldwide, most of them in developing countries.
Rubella is a single-stranded RNA virus and humans are the only host. The World Health Organization estimates that more than 100,000 children are born with CRS each year worldwide, most of them in developing countries.
 Vaccination tremendously reduces the occurrence of CRS.
Vaccination tremendously reduces the occurrence of CRS.
Symptoms
 The symptoms depend on the organ systems involved and on the severity of damage. Parents often note leukocoria.
The symptoms depend on the organ systems involved and on the severity of damage. Parents often note leukocoria.
Signs (Figs. 9-10 to 9-16)
 Cardiac
Cardiac
 Patent ductus arteriosus
Patent ductus arteriosus
 Pulmonary artery stenosis
Pulmonary artery stenosis
 Atrial and ventricular septal defects
Atrial and ventricular septal defects
 Deafness
Deafness
 The most common finding is progressive sensorineural deafness, which occurs in 44% of cases.
The most common finding is progressive sensorineural deafness, which occurs in 44% of cases.
 Vestibular function is rarely impaired.
Vestibular function is rarely impaired.
 Brain damage
Brain damage
 Moderate to severe mental retardation
Moderate to severe mental retardation
 Spastic diplegia
Spastic diplegia
 Microcephaly
Microcephaly
 Schizophrenia-like clinical picture
Schizophrenia-like clinical picture
 Intrauterine growth retardation
Intrauterine growth retardation
 Failure to thrive
Failure to thrive
 Hepatosplenomegaly
Hepatosplenomegaly
 Insulin-dependent diabetes mellitus
Insulin-dependent diabetes mellitus
 Meningoencephalitis
Meningoencephalitis
 Ocular signs
Ocular signs
 Cataract (usually bilateral, occasionally unilateral) occurs in the majority of patients.
Cataract (usually bilateral, occasionally unilateral) occurs in the majority of patients.
 Salt and pepper retinopathy: occurs in approximately 20% of patients. The appearance ranges from a fine stippling of the retinal pigment epithelium (RPE) to dark, patchy areas, and are most prominent in the posterior pole.
Salt and pepper retinopathy: occurs in approximately 20% of patients. The appearance ranges from a fine stippling of the retinal pigment epithelium (RPE) to dark, patchy areas, and are most prominent in the posterior pole.
 Congenital glaucoma (in 10% of cases)
Congenital glaucoma (in 10% of cases)
 Microphthalmos (occurs in 10% of cases)
Microphthalmos (occurs in 10% of cases)
 Corneal edema in the absence of a raised IOP
Corneal edema in the absence of a raised IOP
 Nystagmus
Nystagmus
 Strabismus
Strabismus
 Optic atrophy
Optic atrophy
 Dacryostenosis
Dacryostenosis
Differential Diagnosis
 Other TORCH infections
Other TORCH infections
 Toxoplasma gondii
Toxoplasma gondii
 Others (syphilis, HIV, West Nile virus, varicella zoster, Epstein-Barr virus)
Others (syphilis, HIV, West Nile virus, varicella zoster, Epstein-Barr virus)
 Rubella
Rubella
 Cytomegalovirus
Cytomegalovirus
 Herpes simplex virus
Herpes simplex virus
Diagnostic Evaluation
 The serum of an infant is tested for rubella-specific IgM. Older infants may need additional investigations such as IgG avidity test, RT-PCR for demonstration of virus in lens matter and other body fluids such as serum, throat secretions, and urine.
The serum of an infant is tested for rubella-specific IgM. Older infants may need additional investigations such as IgG avidity test, RT-PCR for demonstration of virus in lens matter and other body fluids such as serum, throat secretions, and urine.
Treatment
 The treatment is directed to the affected organs and tissues. If a cataract is present, lens aspiration is performed in conjunction with primary posterior capsulorrhexis and anterior vitrectomy. Intraocular lenses (IOLs) are usually avoided in young children. A severe inflammatory response can follow cataract surgery and requires intensive therapy with topical steroids with or without systemic steroids. Glaucoma is initially managed medically followed by glaucoma surgery if needed.
The treatment is directed to the affected organs and tissues. If a cataract is present, lens aspiration is performed in conjunction with primary posterior capsulorrhexis and anterior vitrectomy. Intraocular lenses (IOLs) are usually avoided in young children. A severe inflammatory response can follow cataract surgery and requires intensive therapy with topical steroids with or without systemic steroids. Glaucoma is initially managed medically followed by glaucoma surgery if needed.
Prognosis
 Poor when multiple systems are involved, and these children need a multidisciplinary team of a pediatrician, neurologist, ophthalmologist, otolaryngologist, and rehabilitation personnel.
Poor when multiple systems are involved, and these children need a multidisciplinary team of a pediatrician, neurologist, ophthalmologist, otolaryngologist, and rehabilitation personnel.
REFERENCES
Vijayalakshmi P, Rajasundari TA, Prasad NM, et al. Prevalence of eye signs in congenital rubella syndrome in South India: a role for population screening. Br J Ophthalmol. 2007;91(11):1467–1470.
Vijayalakshmi P, Srivastava KK, Poornima B, et al. Visual outcome of cataract surgery in children with congenital rubella syndrome. J AAPOS. 2003;7(2):91–95.
Vijaylakshmi P, Muthukkaruppan VR, Rajasundari A, et al. Evaluation of a commercial rubella IgM assay for use on oral fluid samples for diagnosis and surveillance of congenital rubella syndrome and postnatal rubella. J Clin Virol. 2006;37(4):265–268.
Figure 9-10. This child has congenital rubella syndrome. Although full term, the baby is small for his gestational age. There is leukocoria due to a cataract in the left eye.
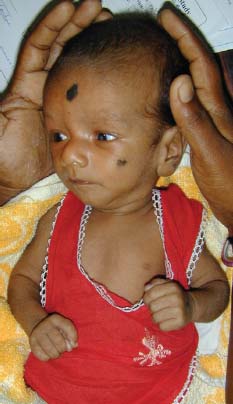
Figure 9-11. A. This child had bilateral cornea clouding and is small for gestational age. B. Another child with cloudy corneas.
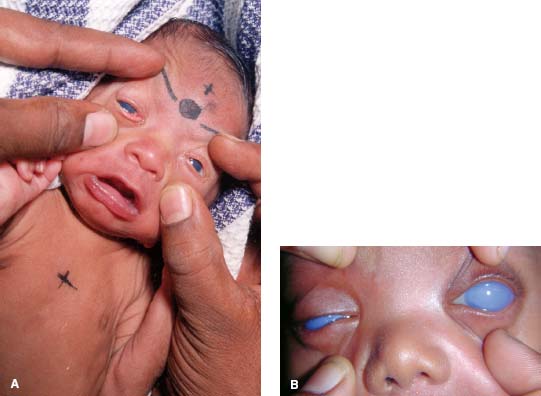
Figure 9-12. This child has bilateral microphthalmos.
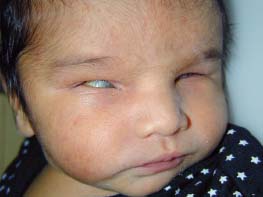
Figure 9-13. This child has corneal scarring and buphthalmos due to congenital glaucoma.
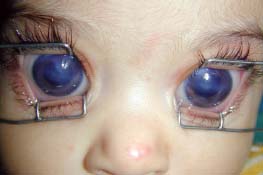
Figure 9-14. There is a mature cataract in the right eye and an early cataract in the left eye.
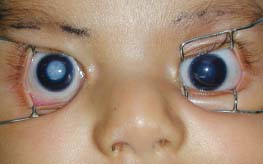
Figure 9-15. This child has a bilateral posterior capsular cataract.
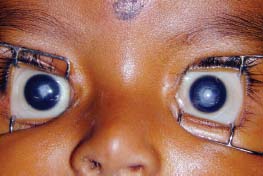
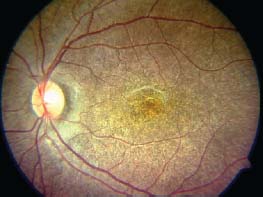
Figure 9-16. There is diffuse mottling of the RPE, giving a salt and pepper fundus in rubella retinopathy.
WEST NILE VIRUS
Sunir J. Garg and Moncef Khairallah
West Nile virus is a single-stranded RNA virus transmitted by mosquitoes. It causes a bilateral uveitis, with multifocal chorioretinal lesions that have a target-shaped and/or linear appearance.
Etiology and Epidemiology
 West Nile virus was first identified in Uganda, and is endemic to many parts of the world.
West Nile virus was first identified in Uganda, and is endemic to many parts of the world.
 Although birds are the natural host of the virus, the virus is transmitted by mosquitoes. West Nile virus is in the same virus family as yellow fever, dengue fever, and Japanese encephalitis.
Although birds are the natural host of the virus, the virus is transmitted by mosquitoes. West Nile virus is in the same virus family as yellow fever, dengue fever, and Japanese encephalitis.
 Patients older than 50 years and those with diabetes may be more susceptible to ocular manifestations.
Patients older than 50 years and those with diabetes may be more susceptible to ocular manifestations.
Symptoms
 Only 20% of infected people have systemic symptoms such as fever, headache, nausea and vomiting, malaise, myalgias, joint pain, vertigo, confusion, aphasia and ataxia, lymphadenopathy, and skin rashes.
Only 20% of infected people have systemic symptoms such as fever, headache, nausea and vomiting, malaise, myalgias, joint pain, vertigo, confusion, aphasia and ataxia, lymphadenopathy, and skin rashes.
 Less than 1% of infected people develop severe CNS symptoms, including mental status changes, sensory and motor neuropathies, and encephalitis.
Less than 1% of infected people develop severe CNS symptoms, including mental status changes, sensory and motor neuropathies, and encephalitis.
 Patients with ocular involvement get blurry vision, floaters, photophobia, and peripheral field loss.
Patients with ocular involvement get blurry vision, floaters, photophobia, and peripheral field loss.
Signs (Figs. 9-17 to 9-19)
 Patients have bilateral ocular involvement.
Patients have bilateral ocular involvement.
 The most characteristic finding is multiple, discrete, circular cream-colored lesions scattered throughout the mid-periphery, periphery, and posterior pole that become pigmented and “punched out” over time.
The most characteristic finding is multiple, discrete, circular cream-colored lesions scattered throughout the mid-periphery, periphery, and posterior pole that become pigmented and “punched out” over time.
 In diabetic patients, these chorioretinal lesions are more prevalent in the posterior pole, and tend to be larger and more numerous.
In diabetic patients, these chorioretinal lesions are more prevalent in the posterior pole, and tend to be larger and more numerous.
 Linear clustering of chorioretinal lesions, following the course of the retinal nerve fiber layer, is a common finding.
Linear clustering of chorioretinal lesions, following the course of the retinal nerve fiber layer, is a common finding.
 Retinal hemorrhages
Retinal hemorrhages
 Bilateral vitritis
Bilateral vitritis
 Transient mild anterior chamber cells
Transient mild anterior chamber cells
 Retinal arteriolar narrowing and/or occlusion, vascular sheathing
Retinal arteriolar narrowing and/or occlusion, vascular sheathing
 Optic disk edema and optic atrophy
Optic disk edema and optic atrophy
 Patients may develop choroidal neovascular membranes late in the course of the disease
Patients may develop choroidal neovascular membranes late in the course of the disease
Differential Diagnosis
 Tuberculosis (TB)
Tuberculosis (TB)
 Syphilis
Syphilis
 Sarcoidosis
Sarcoidosis
 Systemic lupus erythematosus
Systemic lupus erythematosus
 Herpes virus
Herpes virus
 Lyme disease
Lyme disease
 Epstein-Barr virus
Epstein-Barr virus
 Ocular histoplasmosis
Ocular histoplasmosis
 Multifocal choroiditis
Multifocal choroiditis
 Vogt-Koyanagi-Harada disease
Vogt-Koyanagi-Harada disease
 Rift Valley fever
Rift Valley fever
 Rubella
Rubella
Diagnostic Evaluation
 Clinical exam is the most helpful.
Clinical exam is the most helpful.
 Fluorescein angiography (FA) can demonstrate the chorioretinal lesions. In early stages of infection, the angiogram can shows early blockage with late staining. Chronic lesions will show “target-shaped” focal areas of hypofluorescence surrounded by hyperfluorescence.
Fluorescein angiography (FA) can demonstrate the chorioretinal lesions. In early stages of infection, the angiogram can shows early blockage with late staining. Chronic lesions will show “target-shaped” focal areas of hypofluorescence surrounded by hyperfluorescence.
 Indocyanine green angiography (ICGA) shows more lesions in the form of hypofluorescent spots that those appreciated clinically or by fluorescein angiography.
Indocyanine green angiography (ICGA) shows more lesions in the form of hypofluorescent spots that those appreciated clinically or by fluorescein angiography.
 Visual field testing can show nonspecific field defects.
Visual field testing can show nonspecific field defects.
 MRI can demonstrate myelitis (a nonspecific sign).
MRI can demonstrate myelitis (a nonspecific sign).
 IgM and IgG can be elevated, both in the serum and in cerebrospinal fluid.
IgM and IgG can be elevated, both in the serum and in cerebrospinal fluid.
Treatment
 Supportive, as this is a self-limited disease
Supportive, as this is a self-limited disease
 Topical steroids can be used to treat anterior segment inflammation.
Topical steroids can be used to treat anterior segment inflammation.
Prognosis
 Generally good, with most patients retaining good central acuity.
Generally good, with most patients retaining good central acuity.
 Patients with more significant vascular occlusion can experience significant vision loss to the 20/400 range.
Patients with more significant vascular occlusion can experience significant vision loss to the 20/400 range.
REFERENCES
Chan CK, Limstrom SA, Tarasewicz DG, et al. Ocular features of West Nile virus infection in North America: a study of 14 eyes. Ophthalmology. 2006;113:1539–1546.
Khairallah M, Ben Yahia S, Attia S, et al. Linear pattern of West Nile virus-associated chorioretinitis is related to retinal nerve fibres organization. Eye (Lond). 2007; 21(7):952–955.
Khairallah M, Ben Yahia S, Ladjimi A, et al. Chorioretinal involvement in patients in patients with West Nile virus infections. Ophthalmology. 2004;111(11):2065–2070.
Khairallah M, Yahia SB, Letaief M, et al. A prospective evaluation of factors associated with chorioretinitis in patients with West Nile virus infection. Ocul Immunol Inflamm. 2007;15(6):435–439.
Figure 9-17. Fundus photograph (A) and fluorescein angiogram (B) of the left eye of a 64-year-old diabetic woman with serologically proven West Nile virus infection show numerous small and large atrophic chorioretinal lesions in the posterior pole and mid-periphery. There is also nonproliferative diabetic retinopathy.
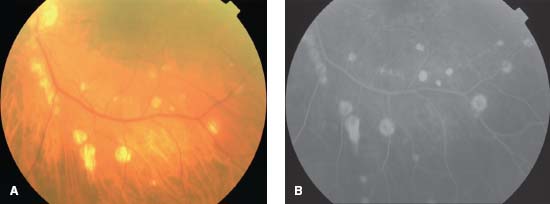
Figure 9-18. A. Red-free fundus photograph of the left eye of a diabetic man with serologically proven West Nile virus infection shows chorioretinal lesions extending superotemporally in a linear pattern from the optic disk. Note the presence of multifocal retinal arterial sheathing. There is also nonproliferative diabetic retinopathy. B. Fluorescein angiogram of the same eye shows central hypofluorescence and peripheral hyperfluorescence of the chorioretinal lesions. C. Late-phase ICGA of the same eye shows well-delineated hypocyanescent choroidal lesions, which are more numerous than those appreciated clinically or by FA.
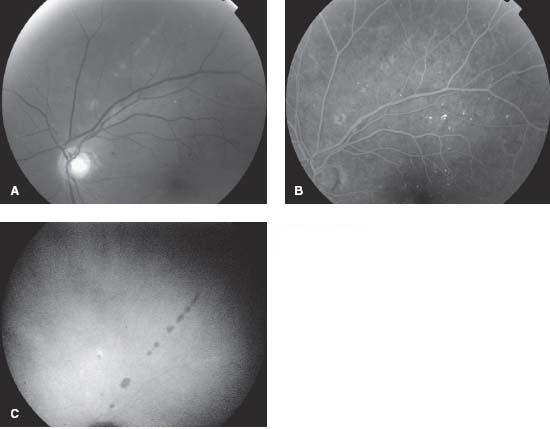
Figure 9-19. A. Fundus photograph of the left eye of a 58-year-old man with serologically proven West Nile virus infection shows multiple, inactive chorioretinal lesions of various sizes. Note the linear clustering of several chorioretinal lesions (curvilinear pattern in the vicinity of inferior major temporal vessels, and a radial pattern superiorly). There also are features of nonproliferate diabetic retinopathy. B. Fluorescein angiogram of the same eye shows more lesions than that observed clinically. Several lesions show the typical “target-like appearance” with central hypofluorescence and peripheral hyperfluorescence.
CHIKUNGUNYA
S. Lalitha Prajna and S. R. Rathinam
Chikungunya is a self-limited viral illness characterized by fever, fatigue, rash, arthralgias, and myalgias. However, more recent outbreaks have been associated with sight, as well as life-threatening complications.
Etiology and Epidemiology
 It is an arthropod-borne, single-stranded RNA alphavirus, belonging to the family Togaviridae and it is transmitted by the bite of the mosquito Aedes aegyptei.
It is an arthropod-borne, single-stranded RNA alphavirus, belonging to the family Togaviridae and it is transmitted by the bite of the mosquito Aedes aegyptei.
 It is endemic in parts of Africa and Asia. Major epidemic outbreaks occurred in 2005.
It is endemic in parts of Africa and Asia. Major epidemic outbreaks occurred in 2005.
Symptoms
 Systemic
Systemic
 Acute fever, chills, headache, fatigue, nausea, vomiting, myalgias, and a diffuse maculopapular rash.
Acute fever, chills, headache, fatigue, nausea, vomiting, myalgias, and a diffuse maculopapular rash.
 Polyarticular and migratory joint pains
Polyarticular and migratory joint pains
 Ocular
Ocular
 Photophobia, red eye, blurred vision, floaters, and retro-orbital pain
Photophobia, red eye, blurred vision, floaters, and retro-orbital pain
 Loss of vision, color vision deficits, central or centrocecal scotoma, and peripheral field defects.
Loss of vision, color vision deficits, central or centrocecal scotoma, and peripheral field defects.
Signs (Figs. 9-20 to 9-22)
 Nodular episcleritis
Nodular episcleritis
 Mild granulomatous or nongranulomatous anterior uveitis
Mild granulomatous or nongranulomatous anterior uveitis
 Pigmented, diffuse keratic precipitates either over the central or the entire corneal endothelium, and stromal edema.
Pigmented, diffuse keratic precipitates either over the central or the entire corneal endothelium, and stromal edema.
 Chikungunya retinitis. This can be differentiated from herpetic retinitis as Chikungunya retinitis has markedly less vitreous reaction and typically has posterior pole involvement, whereas herpetic retinitis in immunocompetent individuals has significant vitritis and multiple, focal lesions of retinitis in the retinal periphery.
Chikungunya retinitis. This can be differentiated from herpetic retinitis as Chikungunya retinitis has markedly less vitreous reaction and typically has posterior pole involvement, whereas herpetic retinitis in immunocompetent individuals has significant vitritis and multiple, focal lesions of retinitis in the retinal periphery.
 Optic neuritis, neuroretinitis, and retrobulbar neuritis.
Optic neuritis, neuroretinitis, and retrobulbar neuritis.
Differential Diagnosis
 Toxoplasmosis
Toxoplasmosis
 Herpetic viral retinitis
Herpetic viral retinitis
 Syphilis
Syphilis
 Cat-scratch disease
Cat-scratch disease
 Dengue fever
Dengue fever
 CMV retinitis
CMV retinitis
Diagnostic Evaluation
 Virus isolation and RT-PCR are useful during the initial viremic phase, whereas serologies (IgM antibody) are useful after 10 days of infection.
Virus isolation and RT-PCR are useful during the initial viremic phase, whereas serologies (IgM antibody) are useful after 10 days of infection.
Treatment
 Systemic
Systemic
 Treatment is mainly supportive and includes rest, hydration, nonsteroidal anti-inflammatory agents, acetaminophen (paracetamol), and corticosteroids for refractory arthritis. Chloroquine has also been used to treat the joint pain.
Treatment is mainly supportive and includes rest, hydration, nonsteroidal anti-inflammatory agents, acetaminophen (paracetamol), and corticosteroids for refractory arthritis. Chloroquine has also been used to treat the joint pain.
 Ocular
Ocular
 Topical steroid eye drops for anterior uveitis.
Topical steroid eye drops for anterior uveitis.
 IV/oral acyclovir and oral prednisone may be used to treat confluent retinitis. However, the efficacy of acyclovir against this virus has not yet been established.
IV/oral acyclovir and oral prednisone may be used to treat confluent retinitis. However, the efficacy of acyclovir against this virus has not yet been established.
Prognosis
 The visual prognosis is good in cases of anterior uveitis, but may be poor in cases of posterior segment involvement.
The visual prognosis is good in cases of anterior uveitis, but may be poor in cases of posterior segment involvement.
REFERENCES
Lalitha P, Rathinam S, Banushree K, et al. Ocular involvement associated with an epidemic outbreak of chikungunya virus infection. Am J Ophthalmol. 2007;144(4):552–556.
Mahendradas P, Ranganna SK, Shetty R, et al. Ocular manifestations associated with chikungunya. Ophthalmology. 2008;115(2):287–291.
Figure 9-20. There are multiple, diffuse, pigmented keratic precipitates in chikungunya anterior uveitis.
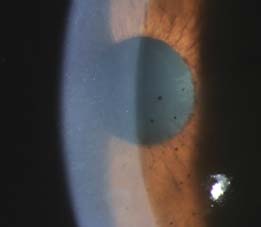
Figure 9-21. This patient has macular retinitis with areas of outer retinal whitening and hard exudates in the outer plexiform layer.
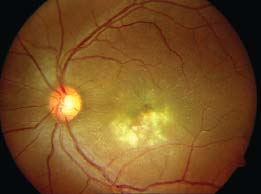
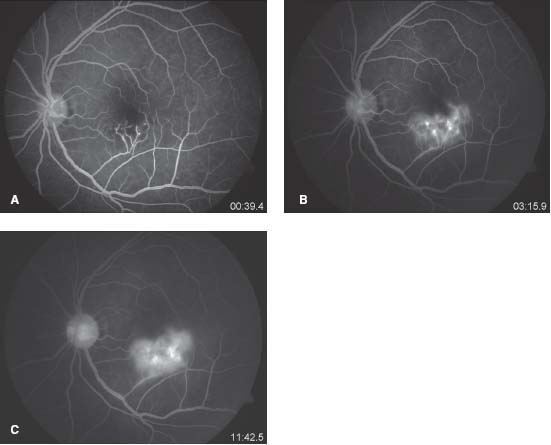
Figure 9-22. A. This frame of the late venous phase of the fluorescein angiogram shows irregular hypofluorescence in the inferior macula, with enlargement of foveal avascular zone. The hypofluorescence is due to reduced perfusion as a result of capillary closure and also due to blocked fluorescence secondary to the retinitis. B. Later frame of the angiogram demonstrates leakage from the vasculitis secondary to retinitis. C. Late phase shows continued leakage from an altered inner blood retinal barrier secondary to vasculitis and retinitis.
SPIROCHETES
SYPHILIS
Julie Gueudry, Bahram Bodaghi, and Phuc LeHoang
Syphilis is a sexually transmitted disease. It has been called “the great masquerader” due to its ability, particularly in the tertiary stage, to mimic numerous types of uveitis. Ocular syphilis is an uncommon but diagnostically important manifestation of the disease and it can result in major complications without appropriate treatment.
Etiology and Epidemiology
 Syphilis is caused by the spirochete Treponema pallidum.
Syphilis is caused by the spirochete Treponema pallidum.
 It is transmitted almost exclusively by sexual contact.
It is transmitted almost exclusively by sexual contact.
 There have been recent syphilis outbreaks, especially in patients with AIDS. Patients with HIV are more likely to contract syphilis, and may have more rapid disease progression.
There have been recent syphilis outbreaks, especially in patients with AIDS. Patients with HIV are more likely to contract syphilis, and may have more rapid disease progression.
 Approximately 84% of cases occur in males.
Approximately 84% of cases occur in males.
 Ocular findings can occur in primary, secondary, latent, and tertiary syphilis.
Ocular findings can occur in primary, secondary, latent, and tertiary syphilis.
Symptoms
 Pain, redness, photophobia
Pain, redness, photophobia
 Decreased vision, floaters
Decreased vision, floaters
 Skin changes and mucous membrane lesions
Skin changes and mucous membrane lesions
 Fever, headache
Fever, headache
Signs (Figs. 9-23 to 9-29)
 Ocular
Ocular
 Primary syphilis
Primary syphilis
 Manifestations are limited to chancre of the eyelid and the conjunctiva.
Manifestations are limited to chancre of the eyelid and the conjunctiva.
 Secondary syphilis (unilateral or bilateral)
Secondary syphilis (unilateral or bilateral)
 Cutaneous rash of the eyelids and blepharitis are common, as is anterior uveitis.
Cutaneous rash of the eyelids and blepharitis are common, as is anterior uveitis.
 Conjunctivitis mimicking trachoma has been reported.
Conjunctivitis mimicking trachoma has been reported.
 Dacryocystitis and dacryoadenitis are less common.
Dacryocystitis and dacryoadenitis are less common.
 Keratitis, episcleritis, and scleritis have been described.
Keratitis, episcleritis, and scleritis have been described.
 Chorioretinitis, neuroretinitis, papillitis, exudative retinal detachment, vasculitis, and acute posterior placoid chorioretinopathy occur later during the secondary stage.
Chorioretinitis, neuroretinitis, papillitis, exudative retinal detachment, vasculitis, and acute posterior placoid chorioretinopathy occur later during the secondary stage.
 Tertiary syphilis (unilateral or bilateral)
Tertiary syphilis (unilateral or bilateral)
 In addition to the other signs seen in secondary disease, gummas of the eyelid, interstitial keratitis, pseudo-retinitis pigmentosa, optic atrophy, and Argyll Robertson pupil can be seen.
In addition to the other signs seen in secondary disease, gummas of the eyelid, interstitial keratitis, pseudo-retinitis pigmentosa, optic atrophy, and Argyll Robertson pupil can be seen.
 Argyll Robertson pupils are bilateral small pupils that do not constrict when exposed to light, but do constrict when the eye accommodates.
Argyll Robertson pupils are bilateral small pupils that do not constrict when exposed to light, but do constrict when the eye accommodates.
 If the patient has HIV, the syphilis manifests itself differently. They can get yellow, placoid, subretinal lesions (acute syphilitic posterior placoid chorioretinopathy). They can also just have vitritis.
If the patient has HIV, the syphilis manifests itself differently. They can get yellow, placoid, subretinal lesions (acute syphilitic posterior placoid chorioretinopathy). They can also just have vitritis.
 Systemic
Systemic
 Primary syphilis occurs 2 weeks to 2 months after inoculation.
Primary syphilis occurs 2 weeks to 2 months after inoculation.
 The chancre (a painless ulcer) begins at the inoculation site, and may have associated regional lymphadenopathy.
The chancre (a painless ulcer) begins at the inoculation site, and may have associated regional lymphadenopathy.
 Secondary syphilis occurs 1 to 3 months after untreated primary syphilis.
Secondary syphilis occurs 1 to 3 months after untreated primary syphilis.
 A maculopapular or pustular cutaneous rash, often on the trunk, palms, and soles, commonly occurs.
A maculopapular or pustular cutaneous rash, often on the trunk, palms, and soles, commonly occurs.
 Mucous membrane lesions and generalized lymphadenopathy are also fairly common.
Mucous membrane lesions and generalized lymphadenopathy are also fairly common.
 Acute syphilitic meningitis is a less common finding.
Acute syphilitic meningitis is a less common finding.
 Latent syphilis
Latent syphilis
 No clinical manifestations
No clinical manifestations
 Tertiary syphilis occurs months to years after untreated secondary syphilis.
Tertiary syphilis occurs months to years after untreated secondary syphilis.
 Benign tertiary syphilis with gummas of the skin and bone can occur.
Benign tertiary syphilis with gummas of the skin and bone can occur.
 Cardiovascular involvement (aortitis, aortic valve insufficiency, and cardiovascular aneurysms) cause significant morbidity and mortality.
Cardiovascular involvement (aortitis, aortic valve insufficiency, and cardiovascular aneurysms) cause significant morbidity and mortality.
 Neurosyphilis occurs in 5% to 10% of patients, and includes meningitis, headaches, sensorimotor loss, cranial nerve palsies, and seizures.
Neurosyphilis occurs in 5% to 10% of patients, and includes meningitis, headaches, sensorimotor loss, cranial nerve palsies, and seizures.
 Congenital syphilis is rare and results from transplacental transmission. Only 60% of infected fetuses become newborns.
Congenital syphilis is rare and results from transplacental transmission. Only 60% of infected fetuses become newborns.
 Systemic manifestations include rhinitis within the first few months of birth, and a maculopapular rash. Late findings include notched incisors (Hutchinson’s teeth), saddle-nose deformity, and sensorineural hearing loss.
Systemic manifestations include rhinitis within the first few months of birth, and a maculopapular rash. Late findings include notched incisors (Hutchinson’s teeth), saddle-nose deformity, and sensorineural hearing loss.
 It can present with various ocular findings, including:
It can present with various ocular findings, including:
 Bilateral interstitial keratitis
Bilateral interstitial keratitis
 Acute or chronic uveitis
Acute or chronic uveitis
 Secondary cataract
Secondary cataract
 Salt and pepper chorioretinitis
Salt and pepper chorioretinitis
Differential Diagnosis
 As syphilis can look like all forms of ocular inflammation, it should be considered in all kinds of uveitis.
As syphilis can look like all forms of ocular inflammation, it should be considered in all kinds of uveitis.
Diagnostic Evaluation
 Laboratory tests should include both nontreponemal and treponemal tests.
Laboratory tests should include both nontreponemal and treponemal tests.
 The nontreponemal tests, Venereal Disease Research Laboratory (VDRL) and rapid plasmin reagin (RPR) are nonspecific for Treponema pallidum, so false-positive are possible. These tests are also used to assess treatment response.
The nontreponemal tests, Venereal Disease Research Laboratory (VDRL) and rapid plasmin reagin (RPR) are nonspecific for Treponema pallidum, so false-positive are possible. These tests are also used to assess treatment response.
 The main treponemal test is fluorescent Treponema antibody–absorption (FTA-Abs). It is specific for Treponema pallidum, so false-positive results are less common, but may still occur during pregnancy and in patients with autoimmune disorders. Once infected, patients will be positive for life.
The main treponemal test is fluorescent Treponema antibody–absorption (FTA-Abs). It is specific for Treponema pallidum, so false-positive results are less common, but may still occur during pregnancy and in patients with autoimmune disorders. Once infected, patients will be positive for life.
 HIV serology should be obtained as coinfection is possible.
HIV serology should be obtained as coinfection is possible.
 Dark field microscopy, electron microscopy, and immunofluorescence can also be used.
Dark field microscopy, electron microscopy, and immunofluorescence can also be used.
 Lumbar puncture should be considered to exclude asymptomatic neurosyphilis.
Lumbar puncture should be considered to exclude asymptomatic neurosyphilis.
 Patients with ocular syphilis should have CSF evaluation for VDRL titers, total protein, and cell counts.
Patients with ocular syphilis should have CSF evaluation for VDRL titers, total protein, and cell counts.
Treatment
 Uveitis should be considered to be a manifestation of neurosyphilis and treated accordingly, even though this point remains controversial.
Uveitis should be considered to be a manifestation of neurosyphilis and treated accordingly, even though this point remains controversial.
 The recommended treatment for neurosyphilis is penicillin G sodium (18 to 24 million units IV daily) or penicillin G procaine (2.4 million units IM daily) plus probenecid PO for 10 to 14 days.
The recommended treatment for neurosyphilis is penicillin G sodium (18 to 24 million units IV daily) or penicillin G procaine (2.4 million units IM daily) plus probenecid PO for 10 to 14 days.
 Ceftriaxone (2 g/day IV or IM for 10 to 14 days) may be considered in penicillin-allergic patients. However, cross-sensitivity exists and this agent has not been tested for the management of neurosyphilis. Some authors recommend penicillin desensitization for these patients prior to starting the cephalosporin.
Ceftriaxone (2 g/day IV or IM for 10 to 14 days) may be considered in penicillin-allergic patients. However, cross-sensitivity exists and this agent has not been tested for the management of neurosyphilis. Some authors recommend penicillin desensitization for these patients prior to starting the cephalosporin.
 Cycloplegics and topical corticosteroids may be useful if anterior segment inflammation is present, but they should be considered solely an adjunct to antibiotic therapy.
Cycloplegics and topical corticosteroids may be useful if anterior segment inflammation is present, but they should be considered solely an adjunct to antibiotic therapy.
 Patients should be monitored for a Jarisch-Herxheimer reaction, which occurs most often in secondary syphilis treated with penicillin (prophylaxis with corticosteroids may help in some cases).
Patients should be monitored for a Jarisch-Herxheimer reaction, which occurs most often in secondary syphilis treated with penicillin (prophylaxis with corticosteroids may help in some cases).
 Patients should also be evaluated and treated for other sexually transmitted diseases, including HIV, gonorrhea, and chlamydia. In the United States, patients with sexually transmitted diseases should be reported to the local Board of Health.
Patients should also be evaluated and treated for other sexually transmitted diseases, including HIV, gonorrhea, and chlamydia. In the United States, patients with sexually transmitted diseases should be reported to the local Board of Health.
Prognosis
 If patients receive prompt therapy, the prognosis is usually good with full visual recovery.
If patients receive prompt therapy, the prognosis is usually good with full visual recovery.
 Retinitis may resolve without scarring within a few days after initiation of specific antibiotics.
Retinitis may resolve without scarring within a few days after initiation of specific antibiotics.
 If untreated, chronic progressive intraocular inflammation and complications may occur.
If untreated, chronic progressive intraocular inflammation and complications may occur.
REFERENCES
Aldave AJ, King JA, Cunningham ET Jr. Ocular syphilis. Curr Opin Ophthalmol. 2001;12(6):433–441.
Chao JR, Khurana RN, Fawzi AA, et al. Syphilis: reemergence of an old adversary. Ophthalmology. 2006;113(11):2074–2079.
Tran TH, Cassoux N, Bodaghi B, et al. Syphilitic uveitis in patients infected with human immunodeficiency virus. Graefes Arch Clin Exp Ophthalmol. 2005;243(9):863–869.
Figure 9-23. Slit-lamp photograph showing interstitial keratitis associated with congenital syphilis.
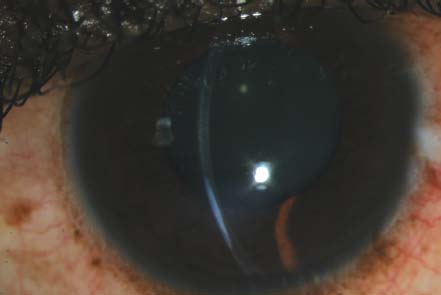
Figure 9-24. Fundus photographs of a patient with uveitis associated with syphilis. A. There is vitritis that is obscuring the posterior pole. B. A peripheral retinal infiltrate is present nasally.
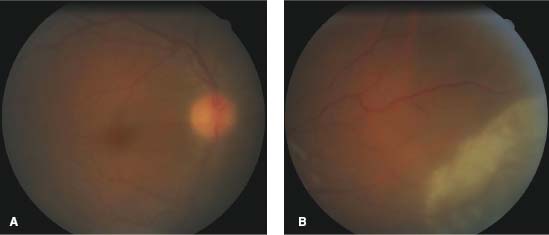
Figure 9-25. Fundus photograph of a patient with resolved uveitis due to syphilis reveals salt and pepper chorioretinitis.
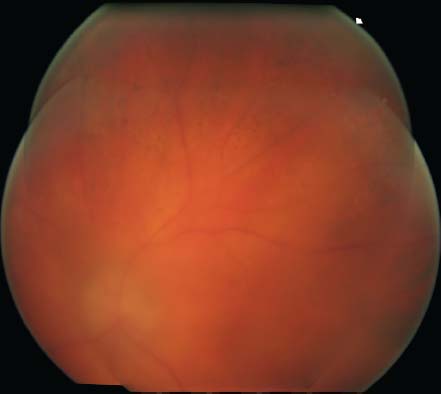
Figure 9-26. Color fundus photograph (A), red-free photograph (B), FA (C, D), and ICG angiography (E, F) of an acute syphilitic posterior placoid chorioretinitis, often seen in patients with HIV.
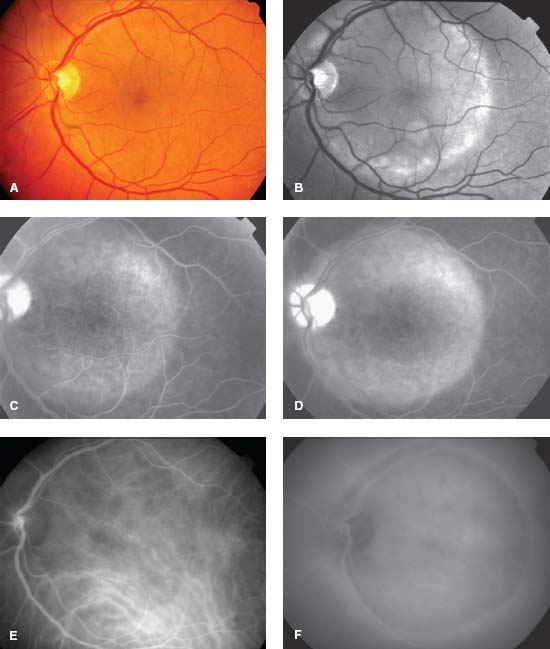
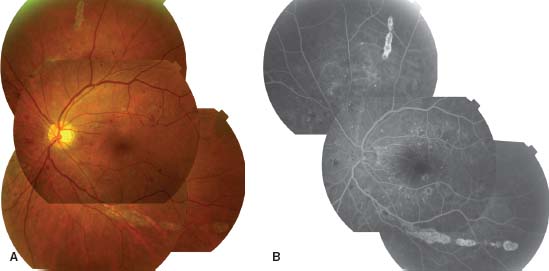
Figure 9-27. Perlèche (angular stomatitis) in a patient with syphilitic uveitis.
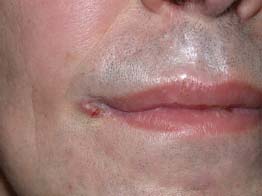
Figure 9-28. This 44-year-old man presented with bilateral decreased vision. He had a recent skin rash that was diagnosed as mononucleosis. His vision was 20/80 OD and 20/800 OS. A. Color fundus photo of the right eye shows large, patchy, multifocal yellow areas in the outer retina/choroid. B. The left eye has a similar, placoid lesion involving the macula. The fluorescein angiogram shows early mottled hyperfluorescence (C) with late staining of the optic disk, outer retina, and choriod, as well as leakage into the subretinal space (D, E). Diagnostic workup found him to be positive for HIV as well as for syphilis. (Courtesy of Paul Baker, MD.)
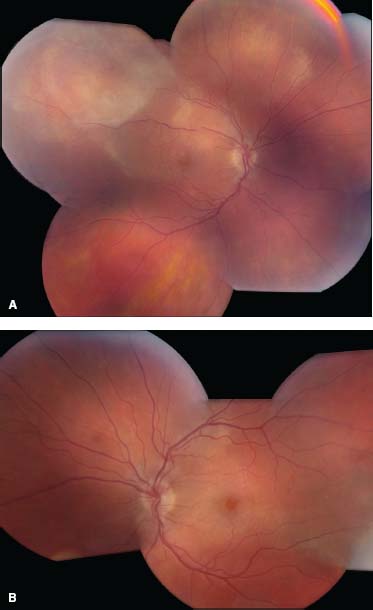
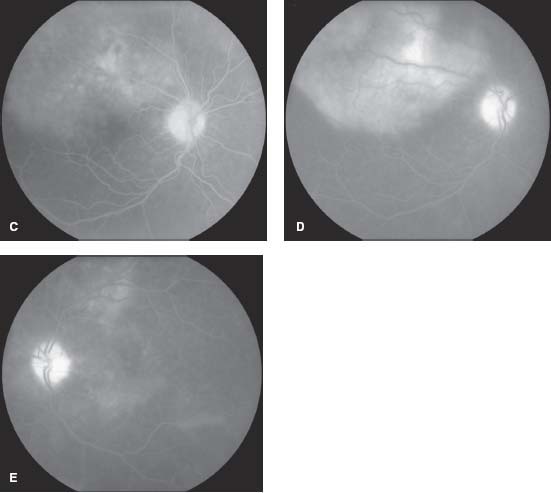
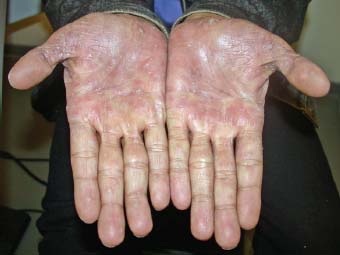
Figure 9-29. This patient with secondary syphilis has multiple, raised, erythematous lesions on the palms of the hands.
LYME DISEASE
Sunir J. Garg
Lyme disease is a multisystem disease that can cause eye, skin, joint, heart, and neurologic problems.
Etiology and Epidemiology
 It is caused by a spirochete, Borrelia burgdorferi, which is transmitted by the Ixodes deer tick.
It is caused by a spirochete, Borrelia burgdorferi, which is transmitted by the Ixodes deer tick.
 In the United States, tick-endemic areas are along the Northeast Coast, the upper Midwest, and in northern California and Oregon.
In the United States, tick-endemic areas are along the Northeast Coast, the upper Midwest, and in northern California and Oregon.
 Lyme disease has also been reported in Europe, Australia, China, and Japan.
Lyme disease has also been reported in Europe, Australia, China, and Japan.
 Infected people will often have a history of camping or hiking.
Infected people will often have a history of camping or hiking.
Symptoms
 These patients can develop essentially all types of ocular inflammation, ranging from a mild conjunctivitis in the early stages, to retinal vasculitis and optic neuropathy. They can also develop cranial neuropathies.
These patients can develop essentially all types of ocular inflammation, ranging from a mild conjunctivitis in the early stages, to retinal vasculitis and optic neuropathy. They can also develop cranial neuropathies.
 Early in the disease course, patients get flu-like symptoms, but they can develop significant arthritis, cardiac problems, headaches, and changes in mental status.
Early in the disease course, patients get flu-like symptoms, but they can develop significant arthritis, cardiac problems, headaches, and changes in mental status.
 Approximately 50% of patients will recall being bitten by a tick.
Approximately 50% of patients will recall being bitten by a tick.
Signs (Figs. 9-30 to 9-33)
 Lyme disease is divided into three stages:
Lyme disease is divided into three stages:
 Stage 1: Patients have flu-like symptoms, including fevers, myalgias, and arthralgias, often with regional lymphadenopathy. Three-fourths of patients will have erythema chronicum migrans. It starts as a red papule that enlarges to form a ring or target-shaped rash. If untreated, these lesions often grow to several centimeters to nearly a meter in size, and then fade over the course of 3 to 4 weeks. Although it occurs in up to two-thirds of patients, many people may not recall having this sign. Early eye findings include conjunctivitis and episcleritis. In Europe, patients are infected with Borrelia afzelii, which can cause a lymphocytoma (of the earlobe or areola) and acrodermatitis chronica atrophicans (a violaceous area of atrophy followed by fibrosis on the dorsal aspect of the hands and feet).
Stage 1: Patients have flu-like symptoms, including fevers, myalgias, and arthralgias, often with regional lymphadenopathy. Three-fourths of patients will have erythema chronicum migrans. It starts as a red papule that enlarges to form a ring or target-shaped rash. If untreated, these lesions often grow to several centimeters to nearly a meter in size, and then fade over the course of 3 to 4 weeks. Although it occurs in up to two-thirds of patients, many people may not recall having this sign. Early eye findings include conjunctivitis and episcleritis. In Europe, patients are infected with Borrelia afzelii, which can cause a lymphocytoma (of the earlobe or areola) and acrodermatitis chronica atrophicans (a violaceous area of atrophy followed by fibrosis on the dorsal aspect of the hands and feet).
 Stage 2: This stage occurs 2 to 3 weeks to several years after infection. Patients may develop cardiac conduction defects (heart block), followed later by severe arthritis. Neurologically, they can get severe headaches, meningitis, and peripheral and cranial neuropathies, including a seventh nerve palsy. The ocular signs include conjunctivitis, keratitis, iridocyclitis, intermediate uveitis, vitritis, choroiditis, vasculitis, serous retinal detachments, and optic neuritis. They may also have diplopia from a third, fourth, fifth, or sixth nerve palsy, and may develop orbital myositis.
Stage 2: This stage occurs 2 to 3 weeks to several years after infection. Patients may develop cardiac conduction defects (heart block), followed later by severe arthritis. Neurologically, they can get severe headaches, meningitis, and peripheral and cranial neuropathies, including a seventh nerve palsy. The ocular signs include conjunctivitis, keratitis, iridocyclitis, intermediate uveitis, vitritis, choroiditis, vasculitis, serous retinal detachments, and optic neuritis. They may also have diplopia from a third, fourth, fifth, or sixth nerve palsy, and may develop orbital myositis.
 Stage 3: These patients have prolonged arthritis and encephalitis. As a result, they can have chronic fatigue and can develop ataxia and dementia. Although they can develop any of the eye findings listed above, bilateral keratitis is the most common manifestation.
Stage 3: These patients have prolonged arthritis and encephalitis. As a result, they can have chronic fatigue and can develop ataxia and dementia. Although they can develop any of the eye findings listed above, bilateral keratitis is the most common manifestation.
Differential Diagnosis
 Syphilis: If one cannot remember what Lyme does, it is a spirochete like syphilis, and has similar manifestations.
Syphilis: If one cannot remember what Lyme does, it is a spirochete like syphilis, and has similar manifestations.
 Sarcoidosis
Sarcoidosis
 TB
TB
 Intermediate uveitis
Intermediate uveitis
 Vogt-Koyanagi-Harada syndrome
Vogt-Koyanagi-Harada syndrome
Diagnostic Evaluation
 Lab tests for Lyme disease are good, but they are most helpful when combined with a high clinical suspicion.
Lab tests for Lyme disease are good, but they are most helpful when combined with a high clinical suspicion.
 Antibodies to Lyme disease are present for several weeks after infection. ELISA testing (followed by a Western blot) is useful. False negatives are common, both in the early stage of the disease and after treatment with antibiotics. False positives also occur, especially if the patient has had syphilis. (Because Lyme disease and syphilis are both spirochetes, there is some cross-reactivity with the tests, so one should order both syphilis and Lyme titers).
Antibodies to Lyme disease are present for several weeks after infection. ELISA testing (followed by a Western blot) is useful. False negatives are common, both in the early stage of the disease and after treatment with antibiotics. False positives also occur, especially if the patient has had syphilis. (Because Lyme disease and syphilis are both spirochetes, there is some cross-reactivity with the tests, so one should order both syphilis and Lyme titers).
 In Lyme endemic areas, 10% to 15% of patients may be antibody positive. If so, positive serology along with erythema chronicum migrans or one of the classic manifestations of Lyme disease makes the diagnosis. If not an endemic area, the patient should have two or more of the classic symptoms before the diagnosis is made.
In Lyme endemic areas, 10% to 15% of patients may be antibody positive. If so, positive serology along with erythema chronicum migrans or one of the classic manifestations of Lyme disease makes the diagnosis. If not an endemic area, the patient should have two or more of the classic symptoms before the diagnosis is made.
 Cerebrospinal fluid may show pleocytosis, but it is frequently unremarkable.
Cerebrospinal fluid may show pleocytosis, but it is frequently unremarkable.
Treatment
 Antibiotics: For very early stage disease, treatment with oral amoxicillin, doxycycline, or erythromycin may be considered. For later stages, consider IV penicillin or ceftriaxone, and patients may need long-term treatment. If the patient has a penicillin allergy, use doxycycline.
Antibiotics: For very early stage disease, treatment with oral amoxicillin, doxycycline, or erythromycin may be considered. For later stages, consider IV penicillin or ceftriaxone, and patients may need long-term treatment. If the patient has a penicillin allergy, use doxycycline.
Prognosis
 Approximately one-fourth to one-third of patients with untreated erythema chronicum migrans will develop stage 2 disease. Less than 10% of these patients will develop chronic arthritis or neurologic disease. Once severe arthritis or neurologic symptoms occur, the symptoms persist despite antibiotic therapy.
Approximately one-fourth to one-third of patients with untreated erythema chronicum migrans will develop stage 2 disease. Less than 10% of these patients will develop chronic arthritis or neurologic disease. Once severe arthritis or neurologic symptoms occur, the symptoms persist despite antibiotic therapy.
REFERENCES
Mikkilä HO, Seppälä IJ, Viljanen MK, et al. The expanding clinical spectrum of ocular lyme borreliosis. Ophthalmology. 2000;107(3):581–587.
Stanek G, Strle F. Lyme borreliosis. Lancet. 2003;362:1639–1647.
Stanek G, Strle F. Lyme borreliosis: a European perspective on diagnosis and clinical management. Curr Opin Infect Dis. 2009;22(5):450–454.
Figure 9-30. This patient had intermediate uveitis due to Lyme disease. There are snowballs present inferiorly.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


