As of 2007, over one million people in the United States, and over 33 million people worldwide are infected with the HIV virus. Men make up 75% of affected patients, and men who have sex with men and racial minorities are disproportionally affected. The HIV virus itself can cause retinopathy. Many of the more serious ocular manifestations of AIDS are due to opportunistic infections.
HIV RETINOPATHY
H uman immunodeficiency virus (HIV) retinopathy is the most common ocular manifestation of HIV.
Epidemiology and Etiology
 It is uncommon if the CD4 is above 200, but it occurs in approximately 50% of patients with a CD4 less than 50.
It is uncommon if the CD4 is above 200, but it occurs in approximately 50% of patients with a CD4 less than 50.
 On histopathology, loss of pericytes, narrowing of the capillary lumen, and thickening of the basement membrane are the earliest manifestations of the disease.
On histopathology, loss of pericytes, narrowing of the capillary lumen, and thickening of the basement membrane are the earliest manifestations of the disease.
Symptoms
 Patients are typically asymptomatic, but they may describe alterations of color vision, visual field changes, and reduced contrast sensitivity.
Patients are typically asymptomatic, but they may describe alterations of color vision, visual field changes, and reduced contrast sensitivity.
Signs
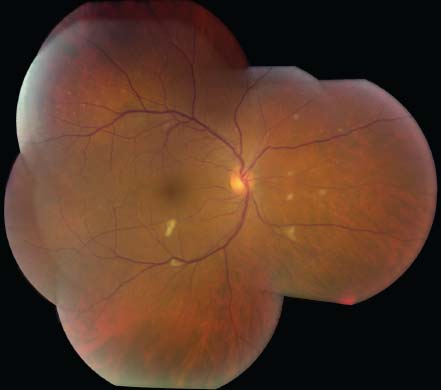
 Cotton wool spots (Fig. 11-1)
Cotton wool spots (Fig. 11-1)
 Intraretinal hemorrhages
Intraretinal hemorrhages
 Capillary microaneurysms, nonperfusion, and telangiectasis
Capillary microaneurysms, nonperfusion, and telangiectasis
Differential Diagnosis
 Diabetic retinopathy
Diabetic retinopathy
 Hypertensive retinopathy
Hypertensive retinopathy
 Radiation retinopathy
Radiation retinopathy
 Venous occlusive disease
Venous occlusive disease
 Collagen vascular disease (lupus)
Collagen vascular disease (lupus)
Prognosis
 The prognosis is almost uniformly good.
The prognosis is almost uniformly good.
Treatment
 No specific treatment is required, and it usually regresses with highly active antiretroviral therapy (HAART).
No specific treatment is required, and it usually regresses with highly active antiretroviral therapy (HAART).
REFERENCES
Holland GN. AIDS and ophthalmology: the first quarter century. Am J Ophthalmol. 2008;145(3):397–408.
Jabs DA. Ocular manifestations of HIV infection. Trans Am Ophthalmol Soc. 1995;93:623–683.
Figure 11-1. A few scattered cotton wool spots in a patient with HIV.
CYTOMEGALOVIRUS RETINITIS
Cytomegalovirus (CMV) retinitis is the most common ocular opportunistic infection in AIDS, and is characterized by yellow-white retinal necrosis and perivascular inflammation.
Epidemiology and Etiology
 The primary risk factor is the degree of immune compromise. Patients with a CD4 count below 50 cells/μL have more than a fourfold higher risk of developing CMV retinitis compared to patients with CD4 greater than 100 cells/μL.
The primary risk factor is the degree of immune compromise. Patients with a CD4 count below 50 cells/μL have more than a fourfold higher risk of developing CMV retinitis compared to patients with CD4 greater than 100 cells/μL.
 CMV retinitis is the most common ocular opportunistic infection in patients with AIDS, and is itself an AIDS-defining illness.
CMV retinitis is the most common ocular opportunistic infection in patients with AIDS, and is itself an AIDS-defining illness.
 In the pre-HAART era, it occurred in up to 40% of patients with AIDS and most patients died within 1 to 2 years after diagnosis. Fortunately, there has been a dramatic decrease in incidence in HAART era.
In the pre-HAART era, it occurred in up to 40% of patients with AIDS and most patients died within 1 to 2 years after diagnosis. Fortunately, there has been a dramatic decrease in incidence in HAART era.
 Rhegmatogenous retinal detachments (RRDs) are also an important cause of vision loss in patients with CMV retinitis, developing in approximately 20% of patients.
Rhegmatogenous retinal detachments (RRDs) are also an important cause of vision loss in patients with CMV retinitis, developing in approximately 20% of patients.
Symptoms
 Painless vision loss
Painless vision loss
 Floaters
Floaters
 Photopsias
Photopsias
Signs (Figs. 11-2 to 11-7)
 Perivascular inflammation with irregular patches of fluffy white retinitis and necrosis with associated scattered hemorrhages. It usually starts as a single lesion and then spreads outward as granular dots from that focus.
Perivascular inflammation with irregular patches of fluffy white retinitis and necrosis with associated scattered hemorrhages. It usually starts as a single lesion and then spreads outward as granular dots from that focus.
 Patients often have a granular lesion with multiple dots around the edge (representing advancing lesions) with central clearing that leads to a stippled retinal pigment epithelium.
Patients often have a granular lesion with multiple dots around the edge (representing advancing lesions) with central clearing that leads to a stippled retinal pigment epithelium.
 Additional features include:
Additional features include:
 Frosted branch angiitis
Frosted branch angiitis
 Chronic vitritis
Chronic vitritis
 Cystoid macular edema
Cystoid macular edema
 Progression typically occurs via expansion of previous retinal lesions, which can advance at a rate of up to 250 microns per week.
Progression typically occurs via expansion of previous retinal lesions, which can advance at a rate of up to 250 microns per week.
 It typically advances faster anteriorly than posteriorly.
It typically advances faster anteriorly than posteriorly.
 Vision loss occurs via macular or optic nerve involvement from primary retinitis or secondarily with macular edema associated with paramacular involvement or due to retinal detachment.
Vision loss occurs via macular or optic nerve involvement from primary retinitis or secondarily with macular edema associated with paramacular involvement or due to retinal detachment.
Differential Diagnosis
 Acute retinal necrosis (ARN) starts as multiple lesions in the mid-peripheral that rapidly progress and is associated with substantial vitritis.
Acute retinal necrosis (ARN) starts as multiple lesions in the mid-peripheral that rapidly progress and is associated with substantial vitritis.
 Progressive outer retinal necrosis (PORN)
Progressive outer retinal necrosis (PORN)
 Syphilis
Syphilis
 Toxoplasmosis
Toxoplasmosis
Diagnostic Evaluation
 Fundus photos can be used to follow disease progression.
Fundus photos can be used to follow disease progression.
 Fluorescein angiography (FA) demonstrates mottled hyperfluorescence of the expanding borders. There can be late staining of the retinal vessels near the area of infection.
Fluorescein angiography (FA) demonstrates mottled hyperfluorescence of the expanding borders. There can be late staining of the retinal vessels near the area of infection.
 Autofluorescence: Advancing border often displays hyperautofluorescence.
Autofluorescence: Advancing border often displays hyperautofluorescence.
 AC tap or vitreous tap with polymerase chain reaction (PCR) may be considered.
AC tap or vitreous tap with polymerase chain reaction (PCR) may be considered.
Treatment
 HAART with immune recovery arrests CMV retinitis progression.
HAART with immune recovery arrests CMV retinitis progression.
 Without HAART, 50% of patients experience CMV reactivation at maintenance dose of anti-CMV therapy.
Without HAART, 50% of patients experience CMV reactivation at maintenance dose of anti-CMV therapy.
 The clinician must consider concurrent CMV disease in other organ systems.
The clinician must consider concurrent CMV disease in other organ systems.
 Systemic therapy
Systemic therapy
 Gancyclovir (5 mg/kg IV b.i.d.) for an induction period of 2 weeks followed by long-term maintenance (5 mg/kg IV q.d.)
Gancyclovir (5 mg/kg IV b.i.d.) for an induction period of 2 weeks followed by long-term maintenance (5 mg/kg IV q.d.)
 Foscarnet (90 mg/kg IV b.i.d. or 60 mg/kg IV t.i.d.) for an induction period of 2 weeks followed by maintenance (90 mg/kg q.d.)
Foscarnet (90 mg/kg IV b.i.d. or 60 mg/kg IV t.i.d.) for an induction period of 2 weeks followed by maintenance (90 mg/kg q.d.)
 Cidofovir (5 mg/kg every week) for an induction period of 2 weeks followed by long-term maintenance (3 to 5 mg/kg IV q2wk)
Cidofovir (5 mg/kg every week) for an induction period of 2 weeks followed by long-term maintenance (3 to 5 mg/kg IV q2wk)
 Valgancyclovir (900 mg PO b.i.d.) is an orally administered prodrug of gancyclovir, which has an induction period of 2 weeks followed by maintenance (900 mg PO q.d.)
Valgancyclovir (900 mg PO b.i.d.) is an orally administered prodrug of gancyclovir, which has an induction period of 2 weeks followed by maintenance (900 mg PO q.d.)
 Intravitreal therapy can be useful as systemic therapy can cause bone marrow suppression (gancyclovir and valgancyclovir) and renal toxicity (foscarnet and cidofovir). However, intravitreal injections need to be performed frequently. If a patient needs long-term therapy and systemic treatment cannot be continued, a gancyclovir implant (see below) should be considered.
Intravitreal therapy can be useful as systemic therapy can cause bone marrow suppression (gancyclovir and valgancyclovir) and renal toxicity (foscarnet and cidofovir). However, intravitreal injections need to be performed frequently. If a patient needs long-term therapy and systemic treatment cannot be continued, a gancyclovir implant (see below) should be considered.
 Gancyclovir (2 mg/0.05 mL)
Gancyclovir (2 mg/0.05 mL)
 Foscarnet (1.2 mg/0.05 mL)
Foscarnet (1.2 mg/0.05 mL)
 Fomivirsen (330 g intravitreal every week); withdrawn from the market
Fomivirsen (330 g intravitreal every week); withdrawn from the market
 Combination
Combination
 Surgical therapy
Surgical therapy
 Gancyclovir implant (Vitrasert, Bausch and Lomb) is surgically placed in the pars plana and delivers gancyclovir for 6 to 8 months at + 4 concentration of IV injection.
Gancyclovir implant (Vitrasert, Bausch and Lomb) is surgically placed in the pars plana and delivers gancyclovir for 6 to 8 months at + 4 concentration of IV injection.
 May be a good option in patients with resistance and unilateral disease.
May be a good option in patients with resistance and unilateral disease.
 Retinal detachments in these cases are often complex with multiple retinal holes and often require vitrectomy with silicone oil tamponade.
Retinal detachments in these cases are often complex with multiple retinal holes and often require vitrectomy with silicone oil tamponade.
 Resistance
Resistance
 Phenotypic and genotypic resistance can occur to gancyclovir, foscarnet, and cidofovir.
Phenotypic and genotypic resistance can occur to gancyclovir, foscarnet, and cidofovir.
 Gancyclovir resistance often secondary to mutations in the UL97 gene.
Gancyclovir resistance often secondary to mutations in the UL97 gene.
 Rate of resistance has declined in HAART era (from 28% in pre-HAART to approximately 9% in HAART era).
Rate of resistance has declined in HAART era (from 28% in pre-HAART to approximately 9% in HAART era).
 Reports exist of gancyclovir-resistant CMV in serum with susceptible CMV in aqueous and vitreous samples.
Reports exist of gancyclovir-resistant CMV in serum with susceptible CMV in aqueous and vitreous samples.
Prognosis
 Dependent on extent of retina involved, but has dramatically improved in HAART era.
Dependent on extent of retina involved, but has dramatically improved in HAART era.
REFERENCES
Jabs DA, van Natta ML, Thorne JE, et al. Course of cytomegalovirus retinitis in the era of highly active antiretroviral therapy: Second eye involvement and retinal detachment. Ophthalmology. 2004;111:2232–2239.
Musch DC, Martin DF, Gordon JF, et al. Treatment of cytomegalovirus retinitis with a sustained-release ganciclovir implant. The Ganciclovir Implant Study Group. N Engl J Med. 1997;337(2):83–90.
Studies of Ocular Complications of AIDS Research Group, in collaboration with the AIDS Clinical Trials Group. Mortality in patients with the acquired immunodeficiency syndrome treated with either foscarnet or ganciclovir for cytomegalovirus retinitis. N Engl J Med. 1992;326(4):213–220.
Figure 11-2. Active CMV retinitis in a patient who underwent bone marrow transplantation.
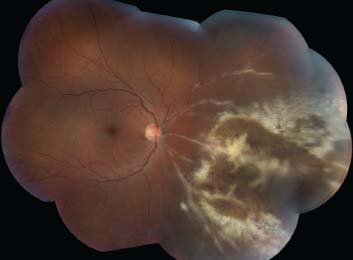
Figure 11-3. CMV retinitis in this case is limited to perimacular area with necrotizing retinitis along the inferior arcade, intraretinal hemorrhage, and granularity surrounding fovea.
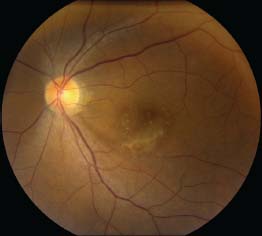
Figure 11-4. This patient with bilateral CMV retinitis confirmed with PCR has extensive white areas of retinitis in the mid-periphery with scattered hemorrhages in both eyes.
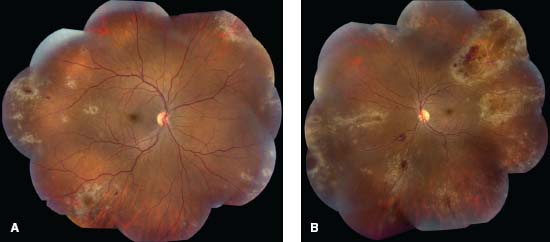
Figure 11-5. Note the area of CMV retinitis superior to fovea (upper right) and vascular staining and leakage on FA (upper left). Fundus autofluorescence shows hyperautofluorescence in the corresponding area (lower left). Lower right frame shows resolution of retinitis following ganciclovir implant.
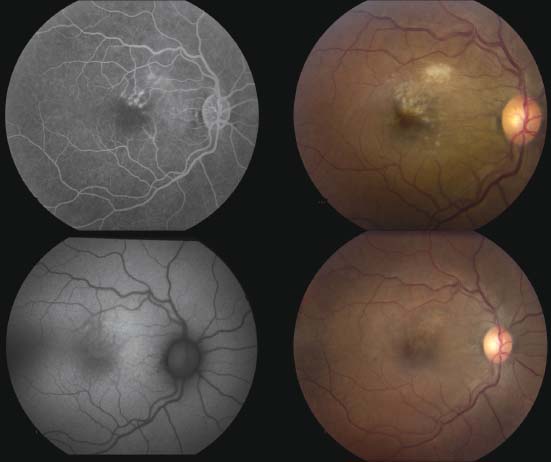
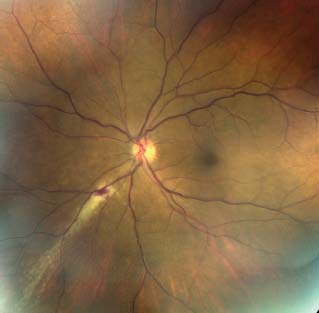
Figure 11-6. CMV retinitis with “brush-fire” appearance along the inferonasal quadrant in a patient with HIV.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


