CHAPTER 197 Infections of the Airway in Children
Croup (Laryngotracheitis)
The term croup is from the ancient Scottish language that refers to the harsh, barking cough that may be present in those with a number of different acquired or congenital disorders.1,2 Several terms have been used synonymously with croup, leading to confusion about the exact nature of the disease. These terms include croup, viral croup, laryngotracheitis, laryngotracheobronchitis, pseudomembranous croup, spasmodic croup, and postintubation croup. This infectious disease primarily involves the vocal folds or glottis and the subglottis. Because viruses predominantly cause this entity, the most descriptive and widely used terms are either viral croup or, more properly, viral laryngotracheitis. Viral laryngotracheitis, or croup, is the most common form of infectious upper airway tract obstruction in children. It is estimated that this condition accounts for 90% of infectious airway obstructions and that 3% to 5% of children have at least one episode of croup. This condition can be recurrent in approximately 5% of children.3 Most children with croup have mild-spectrum symptoms but some children (1.5% to 15%) with more severe spectrum croup require hospital admission for airway obstructive symptoms, and during hospitalization 1% to 5% may require intubation.4 Parainfluenza viruses 1 and 2 primarily are implicated in this disease (up to 75%), but influenza A and B, respiratory syncytial virus, measles, adenovirus, varicella, and herpes simplex virus type 1 all have been isolated.5 Measles involvement in those with croup historically leads to an aggressive form of the disease.6 More recently, influenza viruses and herpes simplex viruses have both been implicated in more severe or prolonged courses of croup.7,8
Viral croup transmission occurs by direct contact and exposure to nasopharyngeal secretions.9 The incubation period before onset of clinical symptoms is 2 to 6 days for parainfluenza virus type 1, and children may continue to shed the virus for up to 2 weeks.10 After acquisition, the viral infection involves the nasopharynx, producing nasal symptoms. Local spread to the larynx and trachea ensues. The mucosa of the vocal folds and subglottis become erythematous and swollen. Lymphocytes, histiocytes, and polymorphonuclear leukocytes infiltrate the lamina propria, submucosa, and adventitia. In addition, generalized mucosal edema is present. The duration of illness usually is from 3 to 5 days. The crucial factor in those with viral croup is the amount of swelling in the subglottic area. The subglottis is the narrowest portion of the upper respiratory tract and, because of the complete cartilaginous cricoid ring, any edema that occurs in the subglottis does so at the expense of the airway lumen. Because stridor does not occur until there is already significant airway obstruction and because any decrease in cross-sectional area is proportional to the square of the radius of the airway lumen, any further decrease in airway size caused by mucous plugging or crusting may lead to rapid and complete airway obstruction.
Clinical Features
Viral croup may present with only mild symptoms of airway obstruction, but it also can be severe with life-threatening airway obstruction. Many different schemes have been developed to assess the spectrum and severity of croup in an effort to assist in determining how to best manage the different presentations of croup.1,5,10 Regardless of the scheme used, it is important to consider the following parameters when evaluating children with croup or airway obstruction in general: (1) stridor—inspiratory versus expiratory; (2) respiratory rate; (3) chest retractions; (4) air entry into the chest by auscultation (decreased with severe obstruction); (5) anxiety or restlessness; (6) color or cyanosis; (7) oxygen desaturation by pulse oximetry; and (8) level of consciousness.
Diagnosis
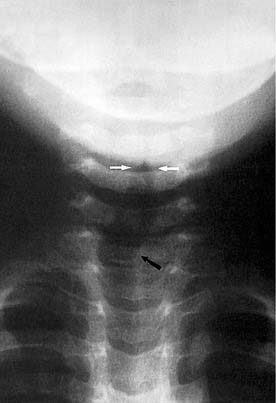
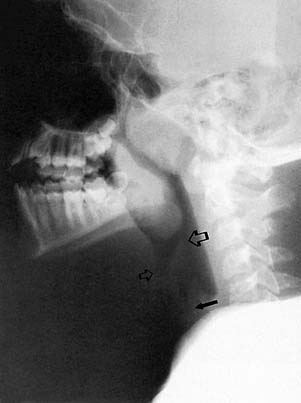
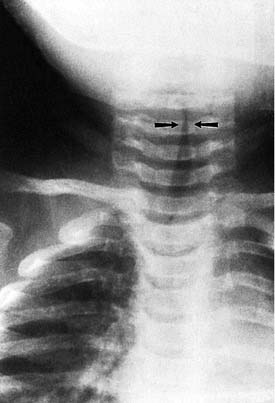
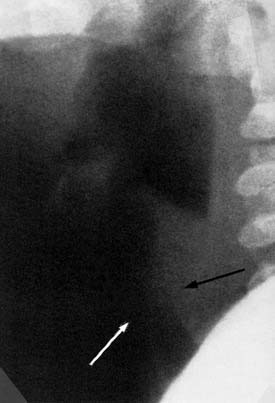
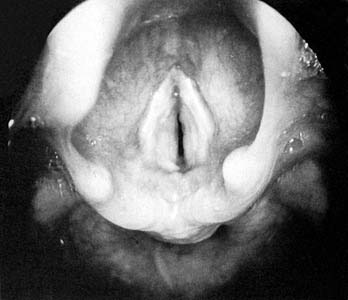
The diagnosis of croup is highly suggested by history and physical examination alone. Detailed questioning about possible foreign body ingestion should be made, along with any previous intubation history. Radiographic imaging of the airway traditionally has been helpful in evaluating those with airway disorders (Figs. 197-1 and 197-2). The diagnosis can be confirmed radiographically by anteroposterior (AP) and lateral (high kilovoltage) films of the upper airway. The frontal AP view classically shows a “steeple sign” in the subglottic area, whereas the lateral view is characterized by haziness of the same area (Figs. 197-3 and 197-4). Flexible fiberoptic laryngoscopy occasionally may be helpful in establishing the correct diagnosis of viral croup. This procedure should be performed with utmost care if there are moderate-to-severe airway obstructive symptoms. In patients with severe airway obstruction, the diagnosis may be confirmed by direct laryngoscopy at the time of intubation (Figs. 197-5 and 197-6).
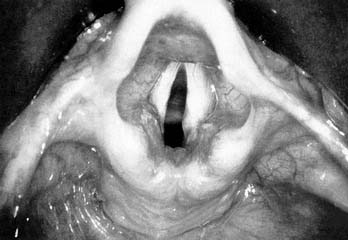
Figure 197-5. Normal child larynx with crisp vocal folds and widely patent subglottis visualized by a rigid rod telescope.
Differential Diagnosis
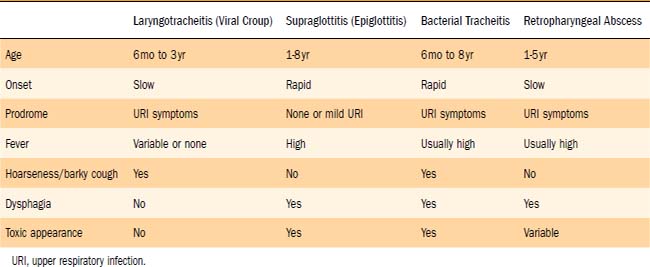
The most important differential diagnosis to consider in children with suspected croup is supraglottitis (Table 197-1). Thereafter, the possible presence of any airway foreign body, particularly in the larynx or subglottis, must be considered. Subglottic hemangiomas may have similar symptoms in infants younger than 6 months of age. Recurrent episodes of a crouplike illness may occur not only in those with congenital subglottic narrowing, but also in children with acquired subglottic stenosis. Therefore a history of intubation may be important. Viral croup in the presence of any preexisting laryngeal or subglottic narrowing may lead to early or recurrent stridor and symptoms of croup. Atypical or recurrent symptoms of croup always demand close attention for other etiologies of airway compromise in addition to viral croup. Gastroesophageal reflux also may be a contributing cause of recurrent croup.11 When viral croup is not responding to appropriate therapy and when children exhibit an increasingly febrile or toxic appearance, bacterial superinfection in the subglottis or trachea should be suspected. This may occur in 10% to 15% of patients hospitalized with more severe croup. Bacterial tracheitis actually may originate as a complication of viral croup.12
Spasmodic Croup
Spasmodic croup is a poorly understood entity that is manifested by the sudden onset of stridor, usually at night, in a healthy child between 1 and 3 years of age with no concomitant upper respiratory tract infection. A barking cough and dyspnea accompany the stridor. These episodes may be recurrent in nature and usually resolve spontaneously or with cool humid air within several hours. The vocal folds may be involved with spasm, and mild, pale transient vocal fold and subglottic edema may be present. Some of the same viruses that cause viral croup may be involved with causing spasmodic croup. An increasing body of evidence suggests an association between spasmodic croup and recurrent croup with nonspecific bronchial hyperactivity, asthma, and allergic disease.13–16 Spasmodic croup and viral croup also may well represent two ends of a broad spectrum in the clinical presentation of a single disease.17
Management
Management strategies for viral croup have included glucocorticoids, racemic epinephrine, heliox, and mist. Although viral croup is the most common form of airway obstruction in young children, many controversies remain and there is no consensus regarding optimal treatment. Recently, croup clinical pathways have been devised to promote consistent, safe, and effective management that might result in reduced hospital admissions and earlier hospital discharge.18
Overwhelming evidence is present to support the use of glucocorticoids in both outpatient and inpatient settings in the management of moderate to severe decrease in croup.19–22 Segal and others23 have documented an 86% decrease in hospital admissions for treatment of croup when steroids were used. The exact mechanism of action of glucocorticoids in croup is unknown, but they probably decrease endothelial permeability, thus leading to decreased mucosal edema. This effect commonly takes at least 3 hours. In mild to moderate cases, oral prednisolone 2 mg/kg/day in two divided doses may be used in the outpatient setting or home for approximately 3 days. This regimen is particularly useful in milder, recurrent cases of croup, with parents keeping prednisolone at home to use when symptoms occur.
In the emergency center, dexamethasone has been extensively used in doses from 0.15 mg/kg to 0.60 mg/kg, with maximum doses of 1 mg/kg for moderate to severe croup.22,24 Oral administration is as effective or better than intramuscular injection or nebulized therapy.15,22 Nebulized budesonide has also been compared with dexamethasone but has had no clear clinical advantage.24 If the symptoms of croup are severe enough to warrant the use of racemic epinephrine, then glucocorticoids are helpful in preventing any associated mucosal rebound edema 2 to 3 hours later. Glucocorticoids have had a significant impact in the outpatient setting but have also greatly decreased the severity of symptoms and the duration of hospitalization.22,25,26 At this point it appears that an initial dexamethasone dose of 0.60 mg/kg orally is preferred because of its low dose and ease of administration.27 Further doses of prednisolone (2 mg/kg/day) or dexamethasone (1 mg/kg/day) in divided doses may be used to sustain the glucocorticoid effect in either inpatients or outpatients. Intravenous administration is preferable in serious inpatient disease. Prolonged use of glucocorticoids should be monitored carefully, particularly when antibiotics may be used, to prevent candida overgrowth in the subglottis or trachea.28 In the absence of clinical evidence of an emerging bacterial process, there is no support for the use of antibiotics in the management of viral croup.
Racemic epinephrine in an equal mixture of the d- and l-isomers of epinephrine has been used in the United States and Canada since 197129 but has become available and accepted in other countries as well. It has been shown that l-epinephrine is at least as effective as racemic epinephrine, is readily available worldwide, has no increased side effects, and is less expensive than d-epinephrine.30 The α-adrenergic effects of epinephrine cause mucosal vasoconstriction, leading to decreased edema in the subglottic region. Using 0.5 mL of a 2.25% solution of racemic epinephrine diluted in 3 mL of saline delivered by a nebulizer results in rapid clinical improvement of moderate to severe respiratory distress within 10 to 30 minutes.17,31 Alternatively, 5 mL of a 1 : 1000 solution of l-epinephrine produces equivalent results. This effect wanes and disappears by 2 hours after administration, with rebound mucosal edema in some patients. Because of this temporary effect and possible rebound, children should be observed closely after administering racemic epinephrine for 2 to 3 hours. Recent reports document the safe use of racemic epinephrine in an outpatient setting. These children were observed for at least 2 to 3 hours, were significantly improved, could easily return to the hospital if needed, and were closely followed as outpatients.32–35 Racemic epinephrine has become widely used in outpatient settings for moderate to severe croup within similar observation criteria described in the studies. In an intensive care setting it can be used every 30 minutes, but it is usually spaced every 3 to 4 hours in a regular hospital unit. Glucocorticoid administration is important in any setting to prevent or diminish the mucosal rebound potential of racemic epinephrine. Racemic epinephrine should be used with great caution in patients with tachycardia or cardiac anomalies such as tetralogy of Fallot or idiopathic subaortic stenosis.
Heliox, a helium-oxygen mixture, may be helpful in severe croup. It decreases the work of breathing by promoting laminar flow through the partially obstructed airway. One study has shown similar improvement in croup scores with heliox administration when compared with patients given epinephrine.36 Heliox may be used as an adjunct to all other medical therapy for croup.
Humidified air or mist has historically been used to treat croup before other medical therapy was available. Humidifiers or steam showers at home and mist tents or blow-by mist in hospital settings presumably moisten secretions, make for easier expectoration, and prevent drying or crusting that may further compromise a narrow airway. There may also be soothing of the involved mucosa, making children more comfortable and less likely to increase submucosal inflammation by vigorous coughing. However, only several studies have investigated the effectiveness of moist air in the management of croup and have demonstrated no benefit over control patients.22,26,37,38 Furthermore, the issue of warm versus cool mist has not been settled. Despite the lack of objective benefit, strong anecdotal experience may support the continued use of mist, particularly in severe hospitalized cases of viral croup or in low-humidity environments. Mist tents should be avoided or used with caution because they may provoke more anxiety in children and therefore blow-by mist may be a better alternative. Well-designed studies are necessary to define the role of mist in the management of viral croup.
Supraglottitis (Epiglottitis)
Since Michel’s original description of “angina epiglottidea anterior” in 1878,39 acute epiglottitis, or supraglottitis as it is more correctly called, has become recognized as a rapidly progressive, life-threatening airway emergency. Supraglottitis predominantly affected children in what may be termed the prevaccine era. A precipitous decline in supraglottitis in children has occurred since the introduction of the Haemophilus influenzae type b (HIB) vaccine.40–42 Before widespread use of the HIB vaccination, the incidence of HIB among children ranged from 6 per 100,000 in Canada43 to 34 per 100,000 in Switzerland.44 In 1988 conjugated HIB vaccine was introduced with recommendations for universal use in children by 2 years of age in 1990 and immunized levels of at least 90% by 1995. Invasive HIB disease, primarily supraglottitis and meningitis, has declined nationally in the United States by 95%.45 Immunization works by producing antibodies to the polysaccharide capsule of HIB. Supraglottitis may infrequently occur even when immunization has occurred because of failure of antibody production.45 Acute supraglottitis remains a rare infection. Its importance lies in its high mortality if the diagnosis is not made promptly.
Supraglottitis is a cellulitis of all the supraglottic structures (not just the epiglottis) that may completely obstruct the airway. As the supraglottic edema increases, the epiglottis curls posteriorly and inferiorly as a result of diffuse inflammatory infiltration of the lingual surface of the epiglottis (Fig. 197-7
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree