Immunology of Ocular Infection
Daniel Sand
Laura Tonjes Muller
Stefan D. Trocmé
The discipline of immunology boomed in 1880, when Louis Pasteur showed the ability to attenuate pathogens and use them for vaccination.1 However, the concept of immunology dates back to ancient China, where children inhaled powders made from crusts of skin lesions of patients recovering from smallpox.2 In Pasteur’s time, the study of immunology focused on the prevention, diagnosis, and cure of infectious diseases. As the science progressed, it was realized that the infecting organism may induce the immune system to cause disease through autoimmunity, anaphylaxis, immune complex deposition, or delayed type hypersensitivity.
As more knowledge was gained regarding immunology, the way the immune system interacted with the eye was found to be different from systemic manifestations of immunology, and it was determined that the eye is an immunologically privileged organ. This privilege is thought to have evolved as a protective mechanism to preserve the integrity of the visual axis, to maintain clarity, and to prevent blindness.3 The presence of an intraocular immunosuppressive microenvironment is most apparent in the anterior chamber and has been termed anterior chamber-associated immune deviation (ACAID).4 Included in this deviation is a blunted cytotoxic T-lymphocyte response leading to a several-day delay in delayed type hypersensitivity (DTH) as compared with a skin infection. This response requires the spleen to be intact and functional.5 In addition, enucleation abolishes ACAID.6 CD4+ T cells are needed and play a crucial role in suppression of DTH, and it is thought that this is via a Th2 immune response.7 The Th1 immune response is suppressed by the Th2 immune response. Interestingly a CD8+ T Reg population also suppresses the delayed type hypersensitivity response via integrin alpha-E (CD 103).8
The environment in the eye is also ideal for a different immune response to antigens including the immunosuppressive nature of aqueous humor. Aqueous humor contains several immunosuppressive factors including somatostatin9 and TGF-beta 210 among others. Fas ligand is a powerful way by which apoptosis is induced in activated immune cells to protect the eye and visual axis. Fas ligand is expressed on the corneal epithelium and endothelium as well as the iris, ciliary body, and retina.11
Other features unique to the eye are the lack of major histocompatability class II molecules on corneal cells and the lack of blood and lymph vessels in the cornea.12 Because of these and other factors, the eye is a unique organ and has distinct responses to infection. In this chapter, the eye’s defense system, the organism’s offense system, and the abnormal and normal responses of the immune system to infection are discussed.
THE HOST’S DEFENSES
There are three main lines of host defense against microorganisms. The first includes the barrier effect of skin, tears, and conjunctiva.13 The second is the blood-borne nonspecific (natural) defense that is present in a host before it is exposed to infectious microbes or other foreign macromolecules, including complement, phagocytes, and their derivatives (interferons alpha and beta and tumor necrosis factor). The third is specific (acquired) immune defenses, including lymphocytes and their products (including antibodies and lymphocyte-derived cytokines, such as interferon gamma), that are able to adapt their response to the attack.
PHYSICAL BARRIERS
The physical barriers include the skin, tear contents, and conjunctiva. The initial interaction a potentially infectious organism may have is with the eyelashes. The eyelashes, or cilia, are sensitive to contact with external objects that initiate a blink reflex to mechanically protect the eye.14 Blinking also causes quicker tear drainage via the lacrimal pump mechanism15, which in turn washes away any potential infectious organism. If the bacterium reaches the skin, it does not survive long because of the direct inhibitory effect of the lactic acid and the fatty acids in sweat and sebaceous secretions and the low pH that these substances generate. Desquamation of the skin aids by shedding the adherent microorganisms.
If an organism is able to bypass the lids and lashes and has successfully reached the ocular surface, it interacts with various proteins, including lysozyme, lactoferrin, and lipocalin, which are present in the tear film and aid with nonimmunologic host defense. Lysozyme, which accounts for 40% of tear proteins, is a bacteriolytic enzyme that disrupts the peptidoglycan cell wall of susceptible organisms through a mechanism similar to that of penicillin.16 Although most Gram-positive bacteria are affected by lysozyme, Staphylococcus aureus is an exception, because the lysozyme-susceptible site in the cell wall is blocked structurally from lysozyme attack. Propionibacterium is another exception. There is evidence that lysozyme levels decrease with age and with aqueous tear deficiency. This may contribute to the higher rates of outer eye infection in these groups.16 Beta-lysin is another bacteriocidal protein found in tears, and it has been shown to act synergistically with lysozyme.17 Lactoferrin makes up 25% of the proteins in tears, and in normal human tears lactoferrin concentration is 2.2 mg/mL.18 It enhances the function of natural killer cells, deprives bacteria such as staphylococci of iron, and may even have a direct effect on certain strains of bacteria.19 It also makes biofilms more susceptible to the bacteriolytic lysozyme.20. A biofilm is an exopolysaccharide glycocalyx polymer that allows for protection from humoral and cellular immunity. It can also allow for protection from surfactants and antibiotics through dilution of the antibiotic and binding of antibiotics by exopolymers.21 Lipocalin was recognized as playing a role in the nonimmunologic defense against microorganisms and viruses.22 Its function in the tear film is thought to be related to cysteine proteinase inhibition, which regulates protein metabolism and protects ocular tissue from proteolytic attack from bacteria and viruses.23 Lipocalin’s antimicrobial activity further appears to work via interfering with microbial siderophores because of its high affinity for them.24
If the organism is able to make it through the lashes, past the skin and tear film, it reaches yet another barrier, the conjunctiva. Mucin secreted from goblet cells in the conjunctiva can inhibit the penetration of cells by viruses through competition with cell surface receptors for the viral neuraminidase.25 The conjunctiva is also able to increase blood flow in the presence of irritants that may represent potentially infectious agents. The blood contains products that participate in natural and humoral immunity.
BLOOD-BORNE NONSPECIFIC (NATURAL) IMMUNITY
Nonspecific immunity includes defense mechanisms present before a person is exposed to infectious microbes or other foreign macromolecules; it has no specificity and no memory.26 Included are phagocytic cells, eosinophils, natural killer cells, and various blood-borne molecules. Innate immunity is the phylogenetically oldest defense against microbes and gives them the opportunity to evolve strategies to resist innate immunity.27
For the factors of innate immunity to be available at the site of potential infection, the factors must be brought to the area of interest through the bloodstream. The acute inflammatory response to bacteria or tissue injury is characterized by capillary dilation and increased capillary permeability. This transfers to the conjunctival epithelium the neutrophils, which play a crucial role in the defense against pyogenic bacteria, such as pneumococci and streptococci. Neutrophils work via two different mechanisms by which they kill pathogens: the oxygen-dependent and the oxygen-independent mechanism. The oxygen-dependent mechanism works via production of reactive oxygen species secondary to activation of NADPH oxidase as well as production of hypochlorite (HOCl) by myeloperoxidase. The oxygen-independent mechanism uses defensins, lysozyme, and major basic protein among others.
In addition to furthering the transfer of leukocytes, the increased capillary permeability brings about a massive transudation of bactericidal factors contained in the serum: C-reactive protein, defensins, and the complement system (Fig. 28-1). These factors aid in the adherence of bacteria to the polymorphonuclear leukocyte and ultimately in phagocytosis.2 Defensins, for example, are considered to be one of the earliest peptide effectors of innate immunity.28 They are released by neutrophils and are present in tears and in the ocular mucosa.29 Defensins have antimicrobial activity against Gram-positive and Gram-negative bacteria, fungi, and viruses and accelerate wound healing by their mitogenic effect on epithelial cells and fibroblasts. There are three different pathways for the complement system: the classical, lectin, and alternative pathways. Ultimately a cascade of a series of complement components is activated to form the membrane attack complex, which causes cell lysis and destruction of the pathogen.
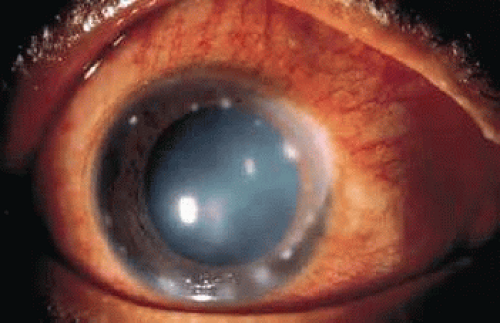
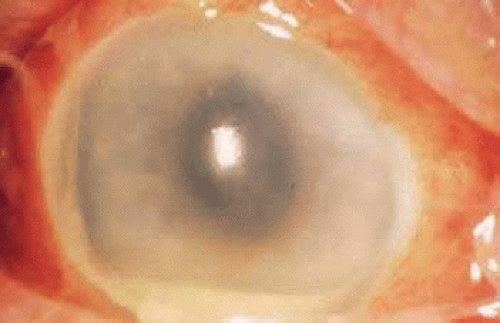 Figure 28-1. Acute inflammation with capillary dilation and leukocytes in the anterior chamber (hypopyon). |
Many types of innate immunity occur in the eye. One example is the immune response to lipopolysaccharide or teichoic acids, found on microbe cell walls but not on mammalian cells. Recognition of lipopolysaccharide by CD4-positive lymphocytes leads to the release of tumor necrosis factor (TNF) and IL-12, activating neutrophils and natural killer cells leading to macrophage activation and inflammation.
SPECIFIC IMMUNE DEFENSES
Specific immune responses are classified into two types: humoral immunity, which is mediated by antibodies produced by B lymphocytes; and cell-mediated immunity (CMI), which is mediated by T lymphocytes.2 In humoral immunity, immunocompetent cells recognize an antigen and then must go to a central processing site, the lymphoid tissue, to produce antibodies. There are interrelated central processing sites throughout the body called mucous membrane–associated lymphoid tissues (e.g., in the lungs, gastrointestinal tract, and lacrimal gland). They lead to antibody distribution in all sites despite antigenic insult at only one site. Knop and Knop30 described conjunctiva-associated lymphoid tissue that contained all components, including follicular spots, to be necessary for a complete immune response. Before the demonstration of conjunctiva-associated lymphoid tissue, the predominant teaching was that in human eyes, after antigen sensitization in the mucosa-associated lymphoid tissue (Peyer’s patches of the intestine), immunoglobulin A (IgA) committed precursor B cells and sensitized T cells to reach the general circulation and eventually reside in the lacrimal gland.31 Beneath the surface epithelium of the lacrimal gland, the B cells undergo further antigen stimulation and become plasma cells that secrete IgA and IgG.
Immunoglobulins are present at a higher concentration in tears than in serum.14 However, the concentration of IgA in tears is directly proportional to the tear flow rate. A diminished tear flow rate leads to increased IgA concentration while a person sleeps. This higher concentration of IgA can protect the ocular surface from residual organisms by neutralizing toxins and viruses and inhibiting the adherence of bacteria to mucosal surfaces, thus limiting an organism’s ability to colonize the eye.32 Whereas IgG can actually activate the classical complement pathway, IgA has been shown to activate complement by the alternative pathway when coated onto a plastic surface. However, alternative complement activation cannot be achieved when IgA is bound to antigen.33
Cell-mediated immunity also plays an important role locally and systemically in the eye’s defense against microorganisms. When a T lymphocyte becomes sensitized to a bacterial antigen, it releases a soluble factor (lymphokine) that can invest the macrophage with the power to destroy ingested organisms. The sensitized T lymphocyte also releases factors that can aggregate macrophages at the site of insult and hinder their departure from the site. Chemotactic factors for neutrophils, basophils, and eosinophils are also released.2 The leukocyte infiltrate may confine the pathogen and prevent its entrance into the interior of the eye, but may also contribute to the necrotizing inflammation of the corneal stroma seen in Gram-negative bacteria and herpes simplex virus (HSV).34
In CMI, several cytokines and interleukins play a role. IL-2 and its receptor have been found to be important mediators of the ocular inflammatory response, and a study by Nussenblatt et al35 used IL-2 humanized anti-Tac antibody (Zenapax*RM [daclizumab], Hoffman La Roche, Nutley, NJ) in patients with chronic noninfectious uveitis. The mechanism of action of cyclosporin is also related to the inhibition of the production and release of IL-2. IL-2 is necessary for the induction of cytotoxic T lymphocytes in response to alloantigenic challenge and plays a major role in cellular and humoral immune response. Chemokines are produced by nearly all human cells. IL-8 has been shown to selectively induce neutrophil and eosinophil chemotaxis and degranulation in response to HSV and adenovirus.36
OFFENSIVE STRATEGIES OF THE ORGANISM
For ocular infection to occur, microorganisms must penetrate the host’s barriers and be able to multiply without overly harming the host from whom it must derive shelter and nutrients. Exploitation of a host’s intrinsic regulatory functions is an obvious way for microbes to accomplish this goal. Among an organism’s offensive strategy are adherence, extracellular products to aid with invasion, production of toxins, and prevention of its own destruction by becoming unrecognizable or by blocking phagocytosis.
The first offensive strategy of bacteria is adherence to the host cell. Pili are fibrous organelles on the surface of Gram-negative bacteria that aid in the adherence of the bacteria to the cells.37 They are required for colonization and are critical in the first steps in the development of disease.38 Pili are produced by plasmids (R factors), which can also contribute directly to virulence by converting the organism to phagocytosis-resistant forms. R factors are transferable between organisms and lead to one microorganism carrying combinations of different copies of R factors and contribute to antibiotic resistance.39 Different types of pili have also been shown to exist on Gram-positive bacteria.40 Staphylococcus aureus uses a different technique to adhere to the corneal surface. Certain strains of S. aureus produce proteins called adhesins, which have specific collagen binding sites. One study showed that adhesins aided in the pathogenesis of keratitis, especially with the use of contact lenses.41 Many bacteria contain extracellular products that help the organism to invade tissue. Gram-positive organisms invade tissue with the help of hyaluronidase, which depolymerizes hyaluronic acid.42 Phospholipases are another mechanism by which the organism can destroy cellular membranes.
Group A streptococci produce fibrinolysins and streptokinase to digest fibrinogen and lyse fibrin clots. S. aureus produces alpha-toxin, lipase, protein A, exotoxins (extracellular bacterial toxins), leukocidins (bacterial toxins that destroy polymorphonuclear leukocytes), and coagulases (enzymes that accelerate the formation of blood clots), all of which help to invade the tissue. Other microbes produce exotoxins that are toxic to all cells of the reticuloendothelial system and help in tissue breakdown and invasion.43
Another technique that microbes use is to hide from the immune system and antibiotics. When antibiotics are present, some microorganisms, such as S. aureus, can also undergo transition to L forms, which lack the cell wall susceptible to antibiotics. Once the antibiotic has been withdrawn, the organism can re-emerge as fully virulent.44 L-phase organisms bear strong resemblance to mycoplasma in their resistance to penicillin, lack of cell wall, and colony morphology.45 Structural lipids prevent destruction of certain organisms, including mycobacteria, Listeria species, monocytogenes, and Francisella tularensis, from phagocytic digestion even once they are inside the phagolysosomes. The lipopolysaccharide endotoxins of Gram-negative bacteria are a major constituent of their cell walls. Although innate immunity responds to lipopolysaccharide, alterations in the structure of lipopolysaccharide impair the ability of the host to respond to certain Gram-negative bacteria.
Another method of preventing destruction is by blocking phagocytosis. S. aureus often produces fibrin-containing abscesses that act as an osmotic barrier to prevent the entrance of immunoglobulins into the focus of infection. Much of the surface of each S. aureus organism is covered with a unique substance called protein A, which helps to prevent phagocytosis of the bacterium.46 The antibody-binding fragment (Fab), which is normally the combining end of the antibody molecule, cannot combine with protein A; only the opposite end, the complement-binding fragment (Fc), combines with it. This reversed attachment blocks phagocytosis by macrophages and polymorphonuclear leukocytes. However, it also activates macrophages to release IL-1, increase phagocytosis, and increase TNF-alpha.46 Other microbes have other types of antiphagocytic factors as integral parts of their structure, for example, pneumococcal capsules and the M proteins of streptococcal cell walls that repel the host phagocytes by negative chemotaxis.47 The M proteins also bind complement regulatory and inhibitory factors leading to suppression of the complement pathway.48
NORMAL OCULAR FLORA
Under normal conditions, eyes interact with many microorganisms. The eyelid skin is colonized with nonpathogenic organisms similar to those in the upper respiratory tract, on the skin, and elsewhere. Under normal conditions, there is no growth of microorganisms from conjunctival cultures in 70% of people.49 The 30% of people who do have growth from conjunctival cultures generally are colonized with the same organisms that grow on the eyelids and eyelashes. These include Gram-positive organisms, such as Staphylococcus species, Streptococcus species, Propionibacterium acnes, and Corynebacterium species; and Gram-negative organisms, such as Haemophilus influenzae, Moraxella species, and Neisseria meningitides.14 Occasionally, Enterobacteriaceae species are found in elderly persons.50 The nonpathogenic organisms prevent the reintroduction of the pathogenic organisms.
GRAM-POSITIVE BACTERIA
Ocular bacterial infections can be caused by various organisms. Gram-positive cocci, including staphylococci and streptococci, account for most ocular infections.51 Gram-positive bacilli, including Bacillus cereus, Corynebacterium species, Listeria species, Clostridium species, and P. acnes, can also cause ocular infection. Because the cocci account for a much higher proportion of infections, they are discussed in greater detail.
Staphylococcus aureus is a Gram-positive coccus that causes most cases of bacterial conjunctivitis and many of the culture-positive corneal ulcers.26 The traditional findings in S. aureus keratitis are a well-defined abscess with stromal infiltrates from the leukocytes attracted to fight infection. Clinical findings from staphylococcal blepharitis include poliosis, madarosis, and trichiasis, which reflects direct damage from the organisms to the glands of Zeis or the meibomian glands and the body’s response (inflammatory and immunologic) to the microorganisms and their products (Fig. 28-2). In addition, ocular manifestations of S. aureus can be the result of types III and IV hypersensitivity reactions. The various presentations with this organism show the complex interaction of the immune system with different factors of the infecting organism.
Staphylococcus aureus produces lipase to invade tissue. Decreasing the amount of lipase production with the use of tetracycline can aid in the treatment of blepharitis even at doses that do not decrease the growth of the bacteria.52
Staphylococci can cause immune-related disease with type III hypersensitivity reactions. Type III hypersensitivity is caused by an abnormal amount of antibody-antigen immune complexes deposited in the involved tissue. When antigens such as ribitol teichoic acid53 from the staphylococci bind to antibodies, neutrophils are attracted and can damage the tissues in the vicinity.
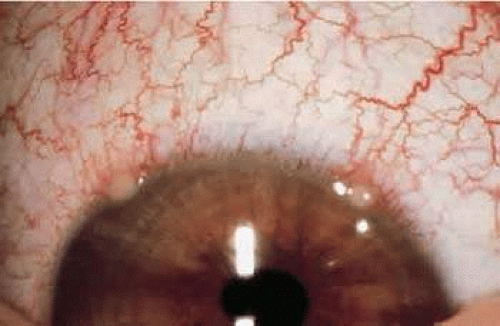
This destruction manifests as white infiltrates at the limbus with excavation from damage to the cornea from the leukocyte products. The limbal distribution of these infiltrates results from the positioning of the lid margin and the presence of the appropriate amount of antigen and antibody in this area, caused by the lack of blood flow in the center of the cornea (Fig. 28-3). The infiltrates may regress without scarring if no necrosis has occurred, or it may progress to a breakdown of the overlying epithelium and finally to a peripheral ulcer. The lesions were once known as catarrhal ulcers, which is a misnomer because the destroyed cornea does not produce mucus, but the denuded surface may have increased adherence to mucus produced by goblet cells in the conjunctiva.
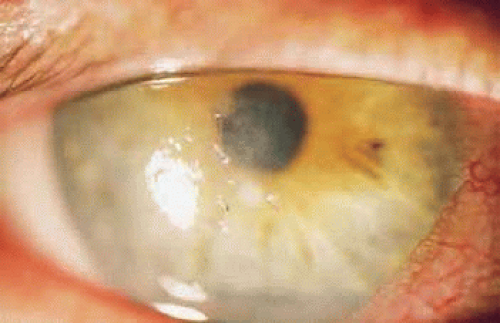
Type IV hypersensitivity may also be seen with S. aureus in the form of phlyctenules. They represent CD4-positive delayed type hypersensitivity to a microbial antigen. Worldwide, most phlyctenules are caused by hypersensitivity to the tubercle bacillus,54 but in the United States, the lesion is more often associated with staphylococcal blepharoconjunctivitis. Other reported causes are Candida albicans, Coccidioides immitis, chlamydia, and nematodes. Phlyctenules are seen clinically as elevated limbal lesions that migrate toward the central cornea with a leash of vessels (Fig. 28-4). Late in the 10- to 14-day course, the nodular lesion ulcerates. Scarring is limited to the cornea and leaves a pathognomonic limbus-based triangular scar with the apex pointing toward the center of the cornea. Phlyctenules are more common in the first two decades of life.54 This can be explained by the high degree of cellular immunity in the early years of life.55 Another characteristic feature of severe cases is a wedge-shaped or fascicular pannus, found more often inferiorly than superiorly.
Streptococci are responsible for a significant percentage of ocular infections. Streptococcus pneumoniae can cause corneal ulceration through cytolysin, a protein that activates and releases degradative enzymes.56
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree