FIGURE 20.1 Coronal MRI of a 16-year-old boy with an extensive juvenile nasopharyngeal angiofibroma (JNA).
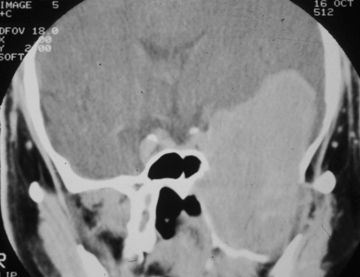
FIGURE 20.2 Coronal CT of a 58-year-old man with a giant schwannoma of V3.
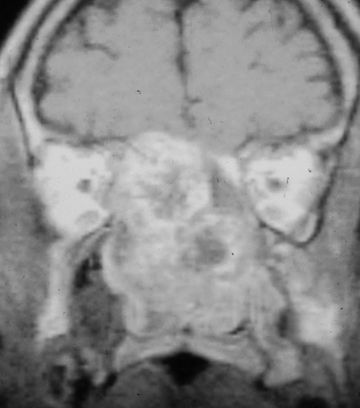
FIGURE 20.3 Coronal MRI of a 36-year-old man with a chondrosarcoma.
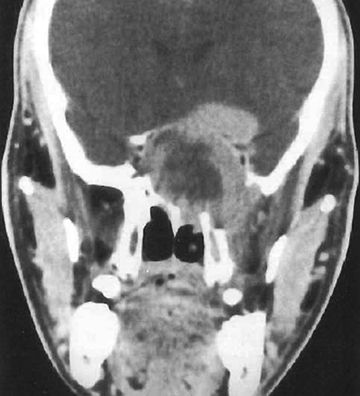
FIGURE 20.4 Coronal CT of a 26-year-old man with a clival chordoma, showing intra- and extracranial extent.
1.Extensive juvenile nasopharyngeal angiofibromas extending through the skull base to the middle cranial fossa and cavernous sinus
2.Carcinomas of the nasopharynx originating primarily on the lateral wall of the nasopharynx
3.Chordomas of the clivus and the surrounding structures, especially those with extension across the midline and to the craniovertebral junction
4.Sarcomas of the sphenoid rostrum
5.Transcranial lesions such as meningiomas, nerve sheath tumors, and congenital tumors extending to the infratemporal fossa and nasopharynx
6.Extensive neoplasms of the parotid with intracranial, infratemporal fossa as well as lateral pharyngeal extent
As more experience was gained, the FTA was used to address other neoplasms, and also certain nonneoplastic disorders including complex encephaloceles, CSF leaks, and advanced infectious processes of the central skull base that were refractory to other treatments.
CONTRAINDICATIONS
There are no absolute contraindications for the FTA, but this approach has been replaced by other less invasive approaches for smaller tumors. Alternative approaches include transnasal, transmaxillary, and infratemporal skull base approaches. In particular, endoscopic endonasal approaches provide good visualization and access to the central skull base for clival lesions. Relative contraindications include a desire to avoid facial scars or temporary paralysis of the temporal branches of the facial nerve.
PREOPERATIVE PLANNING
Radiologic imaging usually includes both CT and MRI due to the complementary information they provide. They can help differentiate between tumor invasion of a sinus and obstructed secretions. MRI can provide evidence of periorbital or dural invasion. Nowadays, radiologic imaging is performed according to protocol in anticipation of intraoperative image-guided navigation. (It is important to note that the FTA was introduced before image guidance was commercially available; at that time the wide exposure was needed in order to safely visualize and protect the vital neurovascular structures. Currently, image-guided navigation, along with other improvements in surgical technologies, sometimes makes it possible to use less invasive approaches. Refer to additional discussion below in Results.)
Orbital symptoms may prompt a complete ophthalmologic evaluation with assessment of visual fields. Electrical testing of motor cranial nerves is not necessary but may be useful in confirming subclinical deficits.
A short differential diagnosis can usually be established with radiologic imaging. Preoperative biopsy of accessible lesions is advised if not a vascular tumor. Deep-seated lesions can be biopsied using CT-guided fine needle aspiration.
Preoperative planning includes informed consent with a complete discussion of alternative treatments, risks of the FTA, and the relative merits and disadvantages of alternative surgical approaches.
SURGICAL TECHNIQUE
Preparation
The patient is positioned supine on the operating table, with the endotracheal tube secured using either a dental wire to the maxillary teeth or a circummandibular wire, on the side contralateral to the surgery. This is important because it provides airway security while allowing the head to be turned and repositioned as needed for best exposure during the procedure. The patient’s head rests on either a flat bed or a Mayfield horseshoe but usually not in fixation pins unless extensive intracranial microdissection is anticipated.
The hair will have been washed with antibacterial soap before arriving in the operating room, and the skin of the face and scalp are now prepped in sterile fashion. The nose and mouth are irrigated with antibiotic solution, usually clindamycin. Intravenous antibiotics are administered prophylactically. Antithromboembolism stockings are applied to the legs, and the body is appropriately padded to prevent pressure-related ischemia if the operation is expected to be long.
Electromyographic (EMG) monitoring of the frontalis branches of the ipsilateral facial nerve is routinely performed, as this facilitates rapid identification and tagging of these nerves (to be described in detail below). Other electrophysiologic monitoring (e.g., somatosensory evoked potential, electroencephalography) is also initiated at this time, if needed.
Incisions and Soft Tissue Flaps
Incisions for FTA involve a combination of a lateral rhinotomy incision, a hemicoronal incision with preauricular extension, and an inferior fornix incision of the eyelid. All these incisions are contiguous, joined by a horizontal incision across the zygomatic region (Fig. 20.5). This horizontal incision is the key to combining the facial access with the temporal access, giving the essential three-dimensional craniofacial and orbital exposure that is so crucial to maximizing the deeper planes of access (Fig. 20.6). Depending on the specific pathology and the extent of the tumor, an upper neck incision may also be useful for proximal control of the carotid artery or to manage concomitant neck disease.
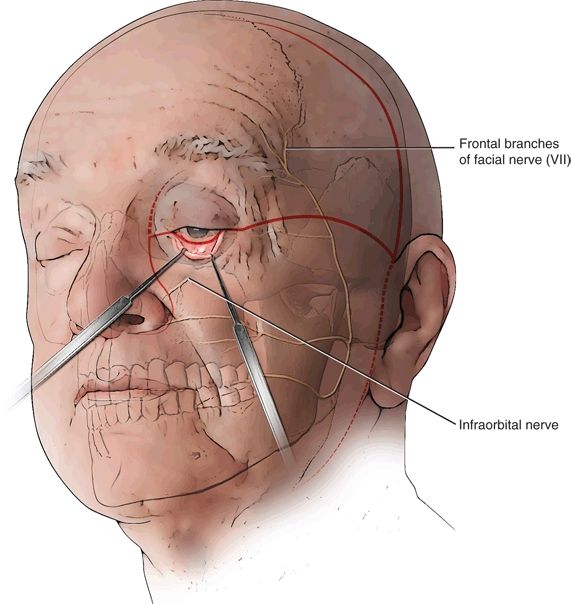
FIGURE 20.5 Incision design for facial translocation approach.
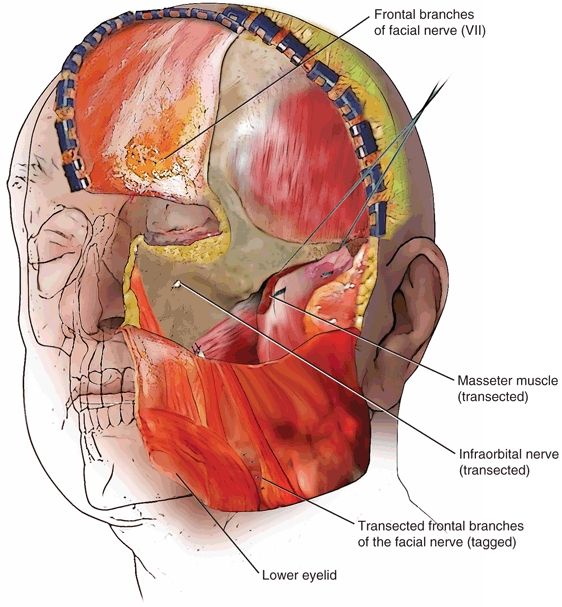
FIGURE 20.6 Exposure of the craniofacial skeleton after reflection of soft tissue envelope.
The incisions are marked, and injected with Xylocaine and epinephrine (1% and 1:100,000) to aid in hemostasis. The first incision is the horizontal incision across the temple, at the level of the zygoma; it begins just behind the lateral canthus and extends to the preauricular crease. This is carried through the skin and superficial subcutaneous tissues, which are gently retracted, and then using loupe magnification and EMG nerve stimulation, the frontalis branches of the facial nerve (“VIIf”) are identified and gently exposed for a short distance of 5 to 10 mm (Fig. 20.7). Typically, there are between three and six separate branches that cross over the zygoma at this level to innervate the frontalis muscle. Note that the innervation to the circular orbicularis oculi muscle enters below this line, and orbicularis function is therefore preserved.
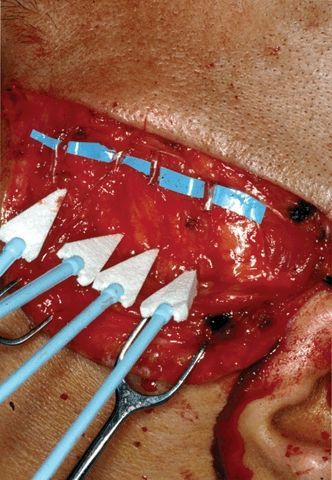
FIGURE 20.7 Intraoperative photo of horizontal temporal incision (left side of the patient), showing four separate frontalis branches of the facial nerve.
This horizontal incision was a source of controversy when the FTA was first introduced. (Other aspects of the operation that were initially criticized included concern over facial scars and the elective transection of the infraorbital nerve—to be addressed below.) It required a willingness to deliberately transect the upper branches of the facial nerve at the point where they cross the zygoma. However, as subsequent reports showed, the temporary forehead paralysis always recovers, provided the surgeon is meticulous in identifying and tagging these branches and in reattaching them at the end of the operation.
There are multiple ways to ensure that the forehead will become reanimated, but the essence is to be able to accurately find and recognize as many VII branches as are present and to then put them all back together at the end. An intimate knowledge of the layered anatomy of the temporal region is essential to this end.
I identify each VII nerve branch (again, using loupes and EMG), and transect it, placing a 7-0 nylon suture on each of the transected ends so that they can be easily found again at the end of the operation. Once all the branches have been found, tagged, and transected, the incision is then carried through the temporal pad of adipose tissue and down to the zygomatic arch itself.
The resulting lower facial soft tissue flap (“cheek and eyelid flap”) is then released in the plane of the parotid–masseteric fascia and reflected down to the level of the palate. This parotid–masseteric plane is deep to the parotid gland and the lower facial nerve branches, which are then out of harm’s way. This provides direct exposure of the masseter muscle and simple access to the condyle and coronoid processes, as well as the ramus, of the mandible.
In certain cases, the pathology may make it necessary to dissect and expose the proximal facial nerve, such as in deep-lobe parotid tumors, minor salivary gland tumors, and any tumors that extend into the lateral infratemporal fossa near the main trunk of VII. When this is necessary, the preauricular portion of the incision can simply be extended downward into a modified Blair incision, making this simple to accomplish. EMG monitoring of the lower branches of the facial nerve is of course still possible.
The next step is to perform the lateral rhinotomy, up to the level of the medial canthus (Fig. 20.6). The depth of incision extends to the periosteum of the nasal and maxillary bones, and the lower nose is mobilized off the pyriform aperture to expose the inferior aspect of the nasal cavity.
The fornix of the lower eyelid is then visualized, and the palpebral conjunctiva is incised. This incision is then extended medially, dividing the medial canthus, where it connects with the rhinotomy incision. It then extends laterally through the lateral canthus, and deeply down to the orbital rim and floor, until the eyelid is completely freed from the surrounding orbital skeleton in a subperiosteal plane. The nasolacrimal duct is divided as distally as possible at the medial orbital floor, and at this point, the entire lower eyelid is free to be retracted inferiorly (Fig. 20.8). In releasing the medial canthal ligament, it is helpful to use a marker or drill bit to mark the exact location of the canthal attachment to the nasolacrimal plate so that it can be accurately replaced at the end of the case.
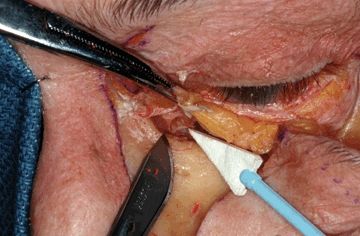
FIGURE 20.8 Cadaveric demonstration in which the lower eyelid has been released, and the rhinotomy incision has been completed, revealing the nasolacrimal duct just prior to its transection.
Next, the premaxillary soft tissue is elevated in the subperiosteal plane, and the infraorbital nerve is exposed. As with the frontalis branches of VII, the infraorbital nerve is tagged with fine sutures, is transected, and will be reattached at the completion of the operation, if it is not involved by tumor. To protect this nerve, one simply uses a fine elevator to enlarge the infraorbital foramen slightly, and the transected proximal end of the nerve is pushed through the foramen and up into the orbit, where it remains until the reconstruction.
Of note, the infraorbital artery, which travels with the nerve, may be quite significant here, and sometimes it has to be controlled with bipolar cautery, taking care not to injure the nerve. Having divided the infraorbital nerve, the premaxillary soft tissue is now readily mobilized down to the Le Fort I line, just above the maxillary teeth.
At this point, the skeletal features of the lower orbit, nose, maxilla, and zygoma are now fully exposed (see Fig. 20.6).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


