CHAPTER 198 Differential Diagnosis of Neck Masses
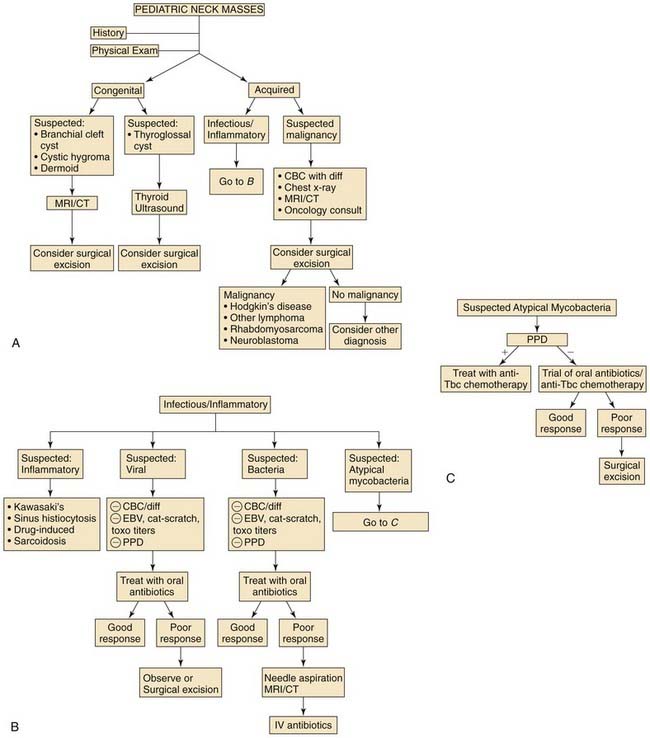
The presentation of a neck mass in a pediatric patient has many diagnostic possibilities not normally encountered in an adult (Fig. 198-1). Neoplastic disease, especially malignant, is the primary consideration in the evaluation of a neck mass in an adult. In contrast, diagnosis of a pediatric neck mass can be categorized, depending on the location, growth factors, and the child’s age. The possibility of malignancy should always be considered in the differential diagnosis in pediatric patients; however, it is not as overriding a concern as in adults.
The two major categories of pediatric neck masses include congenital and acquired lesions. Congenital masses have a high incidence (>50% in some series),1 which initially differentiates them from acquired masses. The primary type of acquired masses is inflammatory (acute and chronic), which further narrows the differential diagnosis. Benign and malignant lesions are rare in the pediatric patient.
Physical Examination
In examination of abnormal lymph nodes or a neck mass, the location is important for determining the site of the precipitating infection or the primary source of a malignant neoplasm. For example, infection in the nasopharynx tends to drain into the posterior cervical region, whereas tonsillitis may cause enlargement in the anterior cervical region. Moussatos2 and Putney3 noted an increased incidence of malignancy in the posterior cervical triangle; in contrast, Torsiglieri and colleagues1 reported an increased incidence in the supraclavicular region.
Congenital Masses
Branchial Cleft Cysts
Although the definitive mechanism for the development of branchial cleft cysts is unclear, it is suspected that these congenital masses result from the cervical sinus of His becoming entrapped without an external or internal opening. An epithelium-lined cyst results. Others suggest that these cysts develop from epithelial rests of tissue from Waldeyer’s ring.4 Branchial cleft cysts are relatively common; in one series, they comprise one third of congenital neck masses.1
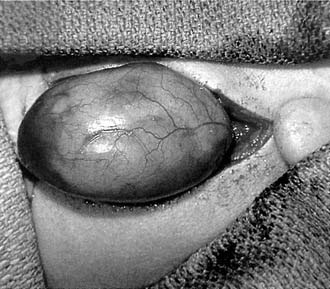
Branchial cleft cysts typically are seen as nontender, fluctuant masses that may become inflamed and abscess during an upper respiratory infection (Fig. 198-2). First branchial cleft cysts, although rare, typically present near the angle of the mandible (Fig. 198-3). Second branchial cysts are found high in the neck and deep to the anterior border of the sternocleidomastoid muscle. Third branchial cleft cysts, also rare, are seen near the upper pole of the thyroid gland. Other symptoms, depending on the size of the cyst, include dysphagia, dyspnea, and stridor.
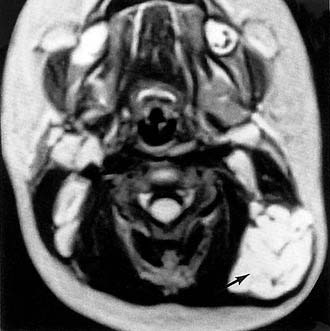
Radiologic evaluation of a branchial cleft cyst may include ultrasound, CT, and MRI. Ultrasound shows a fluid-filled cyst and can differentiate cystic lesions from solid masses. CT and MRI also confirm the cystic characteristics of the mass and, more importantly, delineate the relationship of the cyst to surrounding structures (Fig. 198-4).
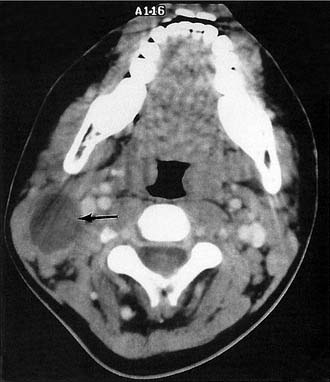
Figure 198-4. Axial computed tomography of the neck shows a branchial cleft cyst in the hypolucent area (arrow).
Thyroglossal Duct Cysts
Thyroglossal duct cysts form in a persistent thyroid descent tract that begins as an elongation of the thyroid diverticulum. Beginning at the foramen caecum of the tongue, this tract may extend to the thyroid gland itself, where some tissue may persist in the region of a pyramidal lobe. These remnants are cysts and are not associated with a cutaneous sinus or fistula tract unless surgically drained and inadequately excised. They comprise approximately one third of congenital neck masses in children.1
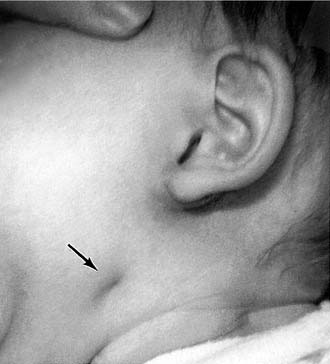
Most thyroglossal duct cysts are seen in the midline near the level of the hyoid bone because the tract passes just anterior to the hyoid bone (Fig. 198-5). Some cysts may present laterally and are not infrequently found superior to the hyoid or as low as the level of the thyroid gland. Thyroglossal duct cysts that occur off the midline may be difficult to differentiate from branchial cleft cysts, an important factor in their surgical excision. Other unusual presentations of thyroglossal duct cysts include formation on either side of the hyoid bone as dumbbell-shaped lesions and, rarely, as cystic lesions in the larynx. Thyroid tissue is found in surgical specimens in up to 45% of cases.5
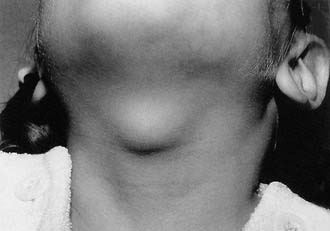
Figure 198-5. Thyroglossal duct cysts are often found in the midline of the neck at or near the hyoid bone.
In cases in which a thyroglossal duct cyst is suspected, it is important to differentiate ectopic thyroid from a cyst. Although only 10% of ectopic thyroid is found in the neck, it may represent the only thyroid tissue in 75% of patients.6 Children with ectopic thyroid may be mildly hypothyroid; however, excision of this tissue necessitates hormone replacement for the remainder of the patient’s life. Thyroid ultrasound and radionucleotide scanning can differentiate ectopic thyroid from a thyroglossal duct cyst; ultrasound is easier to perform, less expensive, and does not use radioisotopes.
The Sistrunk operation is the standard method of thyroglossal duct cyst excision. The cyst is excised along with a cuff of tissue including the center portion of the hyoid bone. Incision and drainage of an infected cyst should be avoided because violation of the cyst capsule may invite recurrence. Recurrence rates of nearly 10% have been reported in children who have undergone uncomplicated Sistrunk operations.1
Lymphatic Malformations (Lymphangiomas)
A lymphatic malformation presents as a soft, smooth, nontender mass that is compressible and can be transilluminated (Fig. 198-6). Typically, lymphatic malformations fluctuate in size as a result of infection or hemorrhage. They mostly impact the cosmetic appearance of the child. Depending on the size and location of the mass, there may be respiratory compromise and difficulty in feeding.
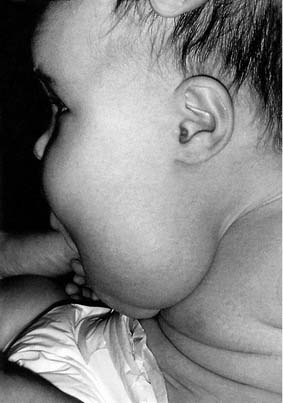
Figure 198-6. Lymphangiomas of the neck may cause significant cosmetic deformity and may impair respiration or alimentation.
Some experts recommend staging of surgical excision in extensive cases.7 Management with radiotherapy has not been effective.8 Macrocystic lesions can be treated with sclerotherapy using alcohol. An experimental lyophilized streptococcal compound, OK-432, has been used successfully to sclerose macrocystic lesions.9
Hemangiomas
Hemangiomas are proliferative endothelial lesions rather than true neoplasms. Hemangiomas present at birth, grow rapidly during the first year of life, and begin to slowly involute at 18 to 24 months of age. Hemangiomas occur in up to 10% of children with a female-to-male ratio of 2 : 1.10
Hemangiomas are seen as red or bluish soft masses that frequently have a skin component. Typically, these masses are compressible and increase with straining or crying. With large hemangiomas, a bruit may be heard over the lesion. CT or MRI with contrast often confirms the diagnosis of a vascular lesion (Fig. 198-7).
Because hemangiomas typically involute after several years, the usual management is observation, unless there is functional impairment, bleeding, skin necrosis, or a coagulopathy caused by thrombocytopenia. Use of systemic corticosteroids may be helpful in the management of complications such as skin ulceration, dysphagia, dyspnea, thrombocytopenia, or cardiac failure.11–14 Surgical excision or laser therapy may be helpful in cases of incomplete involution or when mild functional or cosmetic abnormalities remain. Management with radiotherapy may lead to malignant transformation, and the use of sclerosing agents and cryotherapy has produced poor results.8,15
Teratomas
Teratomas in the head and neck are rare, comprising only 3.5% of all teratomas in one series.16 Although there is a female-to-male preponderance of 6 : 1 in other body regions, the female-to-male ratio is equal in the head and neck.17 Teratomas arise from pluripotential cells and consist of tissues foreign to the site from which they arise. A history of maternal polyhydramnios has been noted in up to 18% of neonates who have a cervical teratoma (Fig. 198-8).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree