CHAPTER 102 Diagnostic Imaging of the Pharynx and Esophagus
Techniques
Conventional Radiography
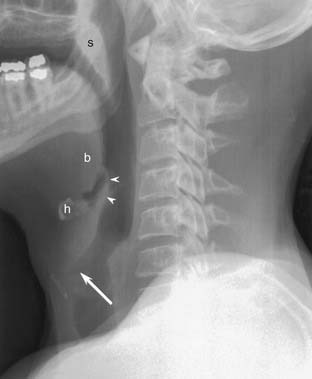
Conventional radiographs (plain films) of the neck are economical and readily available. They are particularly useful in pediatric patients with airway distress. The lateral projection provides the most information, and it will often be obtained without the frontal projection (Fig. 102-1). If the examination is performed to evaluate a radiopaque foreign body, the frontal projection should be included. Patients are instructed to say the letter “e” during exposure to bring the tongue forward and better demonstrate the oropharynx.1 If a hypopharyngeal lesion is being evaluated, the patient should blow through compressed lips to distend the hypopharynx. In children, the lateral radiograph should be obtained during peak inspiration to prevent redundancy of the prevertebral soft tissues that may simulate pathology.
Computed radiography and digital radiography are the modern counterparts to screen-film techniques. Instead of exposing a piece of film, the technologist captures an image on a reusable array of digital elements and reads the image directly into a computer. One advantage of these techniques is the ability to emphasize subtle soft tissue differences, even in suboptimally exposed images.2
Xeroradiography is a historical technique that clearly delineated the soft tissues of the neck.3 However, xeroradiography had a narrow range of applications, and it is no longer available at most institutions.
Pharyngoesophagram
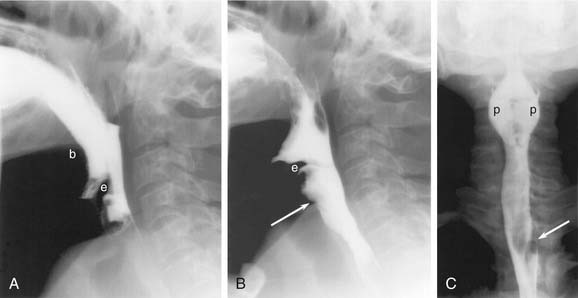
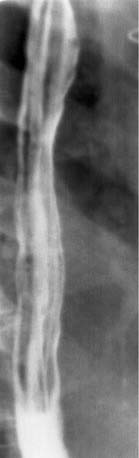
A complete esophagram has three phases: full-column (single contrast), air-contrast (double contrast), and mucosal relief.4 To obtain full-column images, the patient is given a thin suspension of barium by mouth. The pharynx is best imaged in the standing position, with rapid cineradiography (four to six images per second) in several projections (Fig. 102-2). In the frontal projection, the patient’s neck is extended to prevent the jaw from obscuring the pharynx. When digitally displayed, cine images reveal much of the swallowing dynamics. Video capture is still useful, however, to evaluate equivocal filling defects.
Full-column images of the esophagus (Fig. 102-3) are obtained in a prone oblique position, with the patient drinking barium suspension from a straw. There are two objectives to this portion of the examination: evaluate esophageal peristalsis and maximally dilate the esophagus to identify contour abnormalities. Peristalsis is evaluated with videotaped single swallows. Maximum dilatation is achieved with rapid swallows followed by a Valsalva maneuver. The prone position eliminates gravity as a factor in peristalsis.
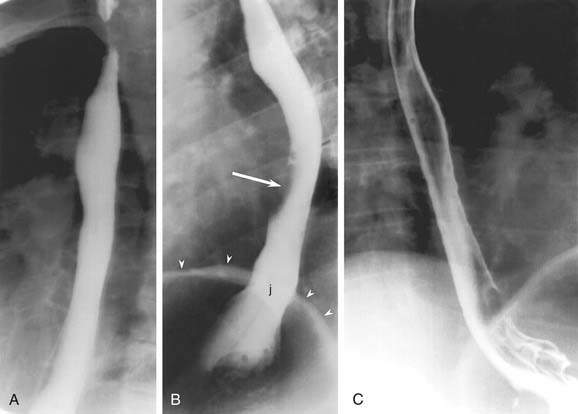
Air-contrast images of the esophagus (see Fig. 102-3) are obtained with the patient upright and in slightly left anterior obliquity. An effervescent agent is first administered, followed by a thick barium suspension. The barium coats the mucosal surface, whereas the gas from the effervescent agent distends the lumen. This provides exquisite mucosal detail and is most useful for the evaluation of small plaquelike mucosal tumors and the mucosal irregularities of esophagitis.5 If the patient is unable to undergo the air-contrast portion of the thoracic esophagram, prone full-column imaging should be obtained in two orthogonal planes as an alternative.
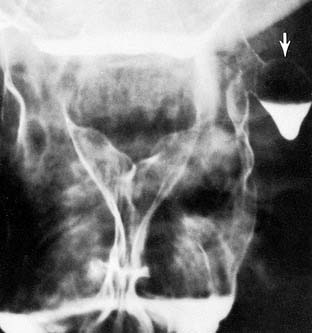
Air-contrast images of the pharynx are not always necessary because this region is amenable to direct inspection. However, in some cases, such as tumors arising in the hypopharynx, air-contrast images are of use. After the administration of thick barium suspension, phonation and a modified Valsalva maneuver are used to distend the pharynx (Fig. 102-4).
Mucosal-relief images of the esophagus (Fig. 102-5) are obtained after the administration of thick barium suspension, but without air distention. Esophageal varices and some mucosal lesions are best seen with mucosal relief. Only the distal esophagus and gastroesophageal junction need to be imaged in this phase of the examination.
Modified Barium Swallow
A modified barium swallow evaluates the coordination of the swallow reflex. It is most often used to determine a cause for, and evaluate the severity of, tracheal aspiration. The speech pathologist, using appropriate radiation safety precautions, administers barium suspensions of varying thickness while the radiologist observes fluoroscopically in the lateral projection. The entire examination is videotaped.6,7
Oral Contrast Agents
Barium suspension is the best-known fluoroscopic contrast agent, but some patients are not appropriate candidates for oral barium administration. Patients who may have a perforated pharynx or esophagus are at risk for barium extravasation into the soft tissues of the neck or chest. Extravasated barium may incite an inflammatory reaction or may become inspissated and fail to resorb.8 Water-soluble contrast agents (such as those used for intravenous CT contrast) may be used as an alternative. Unfortunately, water-soluble agents are not as dense as the barium suspension, so they are less sensitive to small leaks. If no leak is detected after the administration of a water-soluble agent, the examination should be repeated with barium.9
Ionic contrast agents have another disadvantage: if they are aspirated into the lungs, they may cause a chemical pneumonitis and pulmonary edema.10 Nonionic water-soluble agents are presumed to be safer and thus should be used if there is a risk of aspiration or tracheoesophageal fistula.
Computed Tomography
At most institutions, CT is the modality of choice for evaluating masses in the neck and chest. Two recent advances have dramatically changed the quality of CT imaging: multidetector scanners and helical acquisition. Multidetector scanners have several rows of photoreceptors, allowing the simultaneous acquisition of several slices. Helical techniques allow patients to move continuously through the scanner instead of stopping for each slice. These advances have dramatically decreased scan times and radiation doses while improving spatial resolution.11 This improved resolution allows multiplanar and three-dimensional reconstructions that rival magnetic resonance (MR) for clarity and diagnostic accuracy. The rapid scan times permit dynamic CT imaging, which can assess vascularity and other physiologic properties.12
CT is readily available and provides critical information about the extent and character of mass lesions. In known tumors, CT is used to determine the degree of invasion into surrounding deep tissues, the relationship of the neoplasm to critical structures such as the vocal cords and arteries, and the involvement of regional lymph nodes.13 CT is comparable to magnetic resonance imaging (MRI) for the evaluation of bone invasion,14 but MRI is preferred for the evaluation of soft tissue extent, particularly at the skull base.15 Disadvantages of CT include artifacts from dental amalgam and from patient body habitus, particularly at the shoulders.
Helical neck CT should be performed with a maximum slice thickness of 3 mm. Thinner slices may be required to delineate the extent of pathology, particularly in tumors near the larynx. A contrast bolus of 75 to 125 mL is used, with a delay of 30 to 60 seconds.16 Faster scanners require longer contrast delays, so revised protocols are required when equipment is upgraded. CT of the chest is performed with a thickness of 5 to 7 mm; additional intravenous contrast is usually not required. CT of the neck and chest should not be performed as a continuous acquisition because the arms are positioned differently for the two scans.
Magnetic Resonance Imaging
Unlike CT, which relies on ionizing radiation to create images, MRI uses a strong magnetic field and radiofrequency pulses to interrogate the patient’s tissues. It is particularly useful for evaluating tumors of the pharynx and larynx, but it is less useful in the thorax, where motion artifact and field distortions prevent a thorough evaluation of the mediastinum.17
Advantages of MRI include reduced artifacts from dental amalgam and body habitus, as well as the ability to directly image in any plane including oblique planes. The gadolinium-based intravenous contrast agents used in MRI have a substantially lower risk of allergic reaction than CT agents do.18 Disadvantages of MRI include the long duration of MRI studies, which sometimes results in motion artifact, especially in debilitated patients or patients with respiratory compromise. The small bore of the MRI magnet often induces claustrophobia. Many patients cannot go near the MRI magnet because of metallic or electronic implants that might be displaced or triggered by the rapid changes in the magnetic field.19 In particular, cardiac pacemakers/defibrillators, cochlear implants, and ferromagnetic aneurysm clips are relative contraindications to MRI.20 Metallic foreign bodies may also be of concern, depending on their location.21
Ultrasound
Transcutaneous sonographic evaluation of the neck22 has undergone a resurgence in popularity in patients with thyroid cancer and for guidance in interventional procedures. Although ultrasound can be used to evaluate superficial lymph nodes in the neck, it may overlook deep nodes and cannot be relied on for a complete evaluation of cervical tissues. Transesophageal echosonography provides information that is complimentary to CT and invaluable for the evaluation of the extent of esophageal lesions across tissue planes.23 All sonographic techniques are highly operator dependent, so experienced technologists and appropriately trained physicians are necessary to produce diagnostic-quality examinations.
Positron Emission Tomography-Computed Tomography
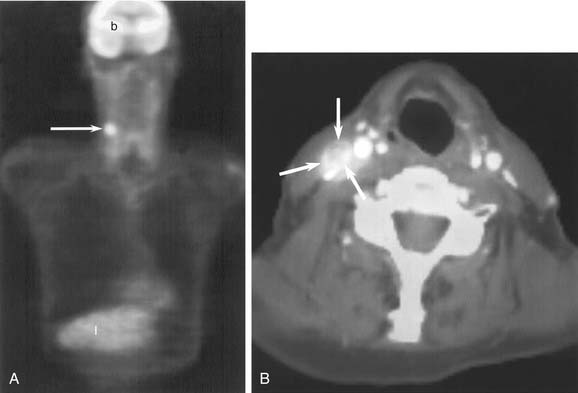
Combined 18F-fluorodeoxyglucose positron emission tomography and CT scanning (PET/CT) has revolutionized the care of head and neck cancer patients.24 PET is a functional imaging technique that relies on the increased metabolic uptake of glucose in tumors to identify unknown primary tumors, stage malignancies, search for metastatic disease, and evaluate recurrences.25,26 The major limitation of PET imaging is its poor spatial resolution, which is particularly significant in the head and neck. Combined PET/CT scanners use the high spatial resolution of CT with the functional information of PET to produce fused images that overcome this difficulty (Fig. 102-6). For the evaluation of head and neck cancers, PET/CT is superior to either PET or CT alone.27
PET/CT is used in the staging of cancer, the monitoring of treatment response, and the surveillance of treated patients.28 Optimal monitoring and surveillance schemata have not yet been established, but it is known that PET/CT should not be performed until at least 8 weeks after the conclusion of therapy to avoid both false-positive and false-negative results.29 Surveillance with PET/CT is particularly useful because of the low rate of false-negative studies.
The analysis of PET/CT scans is complicated and requires a radiologist with experience in both PET/CT and head and neck imaging. Physiologic uptake of FDG is prevalent in the head and neck, especially during contraction of muscles (e.g., neck and thyroarytenoid muscles), and may therefore be confused with tumor uptake.30 Inflammation, either postoperative or from infectious sources such as the teeth, palatine, or lingual tonsils, may also obscure the interpretation, resulting in false-positive reads and unnecessary clinic evaluations and biopsies.
Other Nuclear Medicine
Although PET/CT is the most commonly used nuclear medicine examination in the head and neck, other nuclear medicine tests are sometimes of use. Esophageal transit, gastroesophageal reflux, and gastric emptying can be studied with conventional nuclear medicine techniques.31,32 These studies are more sensitive than fluoroscopy for the presence of reflux, but the quantitative degree of reflux does not correlate well with symptoms. Radionuclide swallowing studies are used predominantly in the pediatric population.
Radiographic Anatomy
Pharynx
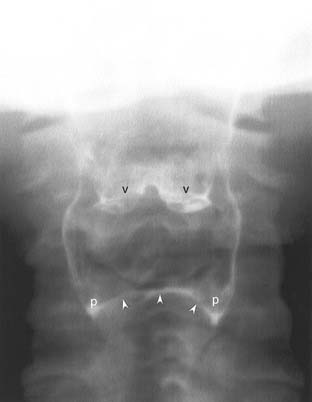
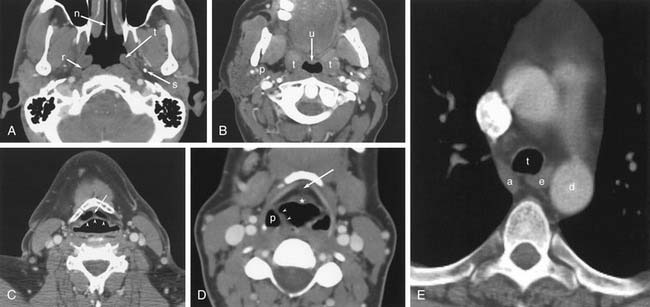
The radiographic anatomy of the pharyngeal lumen is best visualized with air-contrast fluoroscopic images (see Fig. 102-4). The mucosal surfaces of the epiglottis, vallecula, and pyriform sinuses are easily identified. Normal asymmetry of the lingual tonsils should not be confused with a vallecular mass. The epiglottis is best evaluated in the lateral projection, where its thickness can be directly assessed. The pyriform sinuses may be asymmetric, but a complete lack of filling is suspicious for tumor. Barium pooled between the posterior margin of the larynx and the posterior wall of the hypopharynx forms the postcricoid line (see Fig. 102-4). Disruption or irregularity of this line is a sign of tumor invasion. The cricopharyngeus muscle lies anterior to the sixth cervical vertebra. It may be seen as a slight indentation on the posterior wall of the hypopharynx, but it is often not visualized in normal individuals. The normal cross-sectional anatomy of the upper aerodigestive tract is demonstrated in Figures 102-7 and 102-8.33
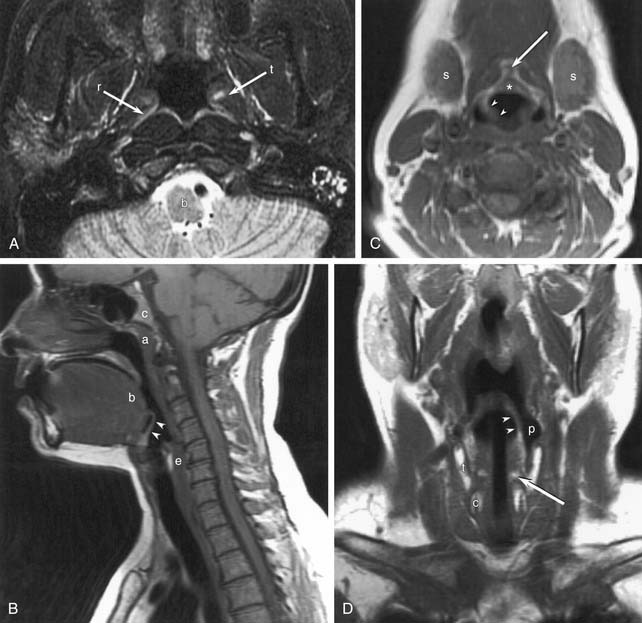
Figure 102-8. Normal magnetic resonance anatomy. A, T2-weighted axial image through the nasopharynx. Brainstem (b), torus tubarius (t), and fossa of Rosenmüller (r) are marked. B, Sagittal T1-weighted image through the neck and face. Adenoids (a), tongue base (b), clivus (c), epiglottis (arrowheads), and cervical esophagus (e) are marked. C, Axial T1-weighted image through the hypopharynx. The petiole of the epiglottis (*), the pre-epiglottic fat (arrow), the aryepiglottic fold (arrowheads), and the submandibular glands (s) are marked. Compare with Figure 102-7, D. D, T1-weighted coronal image of the neck. The aryepiglottic fold (arrowheads) forms the medial border of the pyriform sinus (p). Invasion of the thyroid (t) and cricoid (c) cartilages from hypopharyngeal tumors can be assessed in this plane. The arytenoid cartilages (arrow) are also seen.
Esophagus
The esophagus begins at the level of the sixth cervical vertebra. The cervical esophagus lies posterior to, and slightly to the left of, the trachea (see Fig. 102-7). Surrounding structures that may affect the cervical esophagus include the trachea, cervical spine, thyroid and parathyroid gland, and cervical lymph nodes. On CT, the anteroposterior diameter of the collapsed cervical esophagus should not exceed 16 mm, and its lateral dimension should not exceed 24 mm, except at the esophageal verge, where it may be more prominent.34 On full-column lateral projections, there is a normal mucosal irregularity of the anterior esophagus just below the cricoid cartilage (see Fig. 102-2). This is caused by lax mucosal folds overlying the ventral submucosal venous plexus, and it should not be mistaken for tumor invasion or a web.35 Unlike webs and tumors, the venous plexus changes shape during swallowing.
The thoracic esophagus lies anterior to the spine, anteromedial to the descending aorta (see Fig. 102-7). The phrenic ampulla, which is a normal widening of the esophageal lumen, is seen just above the gastroesophageal junction (Fig. 102-9). There are three normal indentations on the anterolateral esophagus: aortic arch, left mainstem bronchus, and left atrium (see Fig. 102-3). Other nearby structures include the descending aorta, aortic arch and great vessels, carina, mediastinal lymph nodes, and spine.
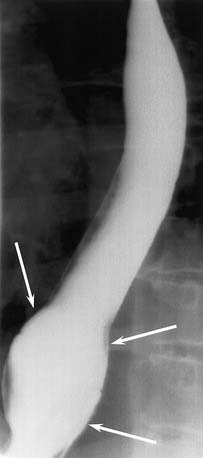
Figure 102-9. The phrenic ampulla is a normal widening of the distal esophagus just above the diaphragm.
Motility Disorders
Pharynx
The modified barium swallow is the most appropriate radiologic test to evaluate swallowing dysfunction.7 Although an esophagram provides some information about deglutition, the modified swallow uses barium of several different consistencies, which provides a more detailed evaluation. Functional endoscopic evaluation of swallowing, with or without sensory testing, has been proposed as an alternative to the modified swallow.36 However, the modified swallow provides a more physiologic environment because there is no endoscope to interfere with motility. During a modified swallow, patients can use protective maneuvers such as chin tuck and forced cough, which are not available during endoscopy. Furthermore, the modified swallow evaluates the upper phases of swallowing in more detail.37 Endoscopy and modified swallow are considered complementary examinations at most institutions.
The modified barium swallow can evaluate all phases of the swallow reflex.1,6,38 The tongue forms the oral bolus and then transports it from the oral cavity to the oropharynx. The soft palate elevates and approximates the posterior pharynx to prevent velopharyngeal reflux. The entire larynx elevates, followed by a peristaltic wave through the pharynx. The epiglottis inverts to deflect the bolus into the pyriform sinuses and protect the laryngeal vestibule. At the bottom of the hypopharynx, the cricopharyngeus muscle relaxes to permit the passage of the food bolus.
Abnormal Deglutition
Unilateral pharyngeal palsy causes an asymmetry in the pyriform sinuses because the contrast is thrown to the palsied side by the functioning pharyngeal constrictors.39 This asymmetry should not be misinterpreted as a filling defect from carcinoma in the unaffected side. A careful dynamic examination of several swallows may be necessary to prevent this diagnostic error.
Cricopharyngeal Dysfunction
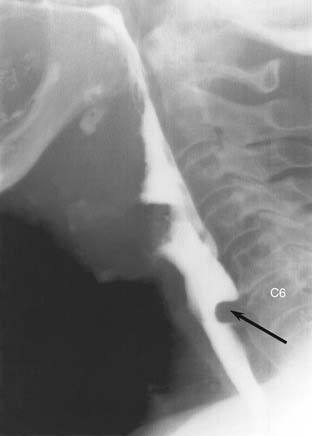
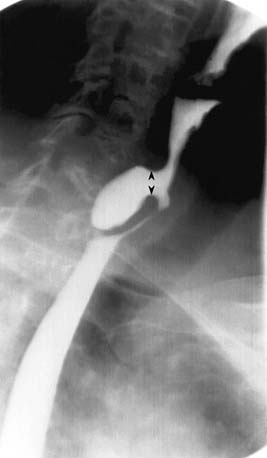
Unlike the other muscles of pharyngeal constriction, the cricopharyngeus remains contracted between swallows, acting as an upper esophageal sphincter. It normally relaxes during deglutition to allow the passage of the food bolus. When the muscle fails to completely relax (cricopharyngeal achalasia), there is a smooth posterior impression on the hypopharynx at the level of the C6 vertebra (Fig. 102-10). Unlike most retropharyngeal masses, the cricopharyngeus does not exceed 1 cm in vertical dimension. The most frequent cause of cricopharyngeal achalasia is cerebrovascular disease. Other causes include pseudobulbar palsy, nasopharyngeal carcinoma, poliomyelitis, thyroid myopathy, cervical vagotomy, polymyositis, dermatomyositis, oculopharyngeal syndrome, amyotrophic lateral sclerosis, and hiatus hernia, but many cases are idiopathic. Cricopharyngeal achalasia has been implicated in the development of Zenker’s diverticula.40
Incompetence of the upper esophageal sphincter is called cricopharyngeal chalasia. It manifests radiographically as a lack of cricopharyngeal impression between swallows. Cricopharyngeal chalasia is specific for myotonic dystrophy, although most patients with myotonic dystrophy have cricopharyngeal achalasia instead.41
Esophagus
Manometry is considered the reference standard for evaluation of esophageal dysmotility. The relative sensitivity of fluoroscopy, radionuclide scans, and manometry is controversial.31,32,42,43 However, radiographic techniques are less invasive and elicit less patient discomfort. Fluoroscopy has the additional advantage of identifying structural abnormalities.
Diminished Peristalsis
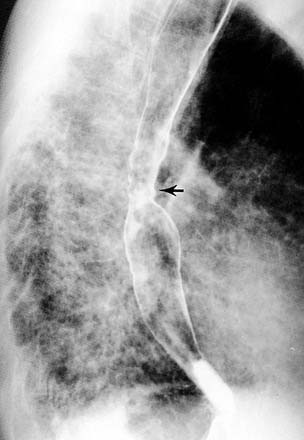
Achalasia is a neuromuscular disorder caused by degeneration of Auerbach’s plexus.44 Peristalsis fails, but the LES remains tight, so the esophagus progressively dilates. In severe cases a dilated esophagus with retained food can be appreciated on a chest film. On fluoroscopy, the distal esophagus has a conical (beaked) shape (Fig. 102-11). In early stages of the disease, the esophagus may be only minimally dilated, with dominant tertiary contractions. This is called vigorous achalasia, and it may mimic esophageal spasm (Fig. 102-12).45 Patients with achalasia are at increased risk of esophageal carcinoma, and radiographic screening may be indicated.46,47
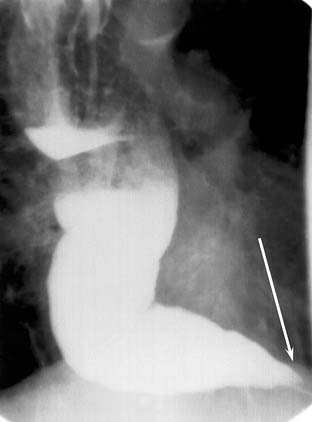
Figure 102-11. Achalasia. Oblique image of the lower mediastinum reveals a patulous esophagus with beaking of the gastroesophageal junction (arrow).
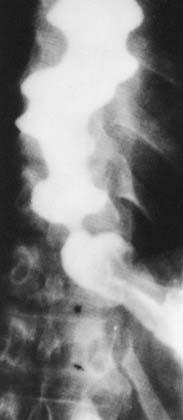
Figure 102-12. Vigorous achalasia. Barium esophagram shows dilation of the esophagus with multiple nonperistaltic contractions.
Impairment of the LES is a constant feature of achalasia, but it is not pathognomonic. Patients with diffuse esophageal spasm, presbyesophagus, or connective tissue diseases may also have impaired relaxation of the LES. Carcinoma of the distal esophagus or gastric cardia can mimic achalasia.48 Chagas’ disease, in which the parasite Trypanosoma cruzi affects the ganglion cells of the esophagus, may appear identical to achalasia on an esophagram.49 Other mimics of achalasia include central and peripheral neuropathies such as stroke, diabetes mellitus, and amyloidosis, and strictures from reflux esophagitis.40
Presbyesophagus is a failure of peristalsis associated with aging. It is frequently observed in older patients with dysphagia, but the relationships among reflux esophagitis, presbyesophagus, and dysphagia remain unclear.50 Presbyesophagus manifests as failure of the primary peristaltic wave with intermittent tertiary contractions.
Many other diseases can cause diminished peristalsis including severe esophagitis, diabetes, alcoholism, hyperthyroidism, anticholinergic medications, and surgical vagotomy.40
Connective Tissue Diseases
Scleroderma is the prototype for a connective tissue disease with esophageal manifestations. Peristalsis is absent in the distal two thirds of the esophagus (the portion with smooth muscle rather than striated muscle). This usually results in mild dilation of the distal esophagus. In contrast to achalasia, the LES becomes incompetent, allowing reflux (Fig. 102-13). Scleroderma has been associated with hiatus hernia, Candida esophagitis,51 aspiration pneumonia, esophageal adenocarcinoma,52 and reflux esophagitis with Barrett’s esophagus.53
Systemic lupus erythematosus, rheumatoid arthritis, and polymyositis have radiographic manifestations similar to scleroderma. Dermatomyositis, in contrast, predominantly affects the striated muscle of the pharynx and upper esophagus.40 Fluoroscopy and manometry are more useful than endoscopy in the evaluation of connective tissue disorders.54
Esophageal Spasm
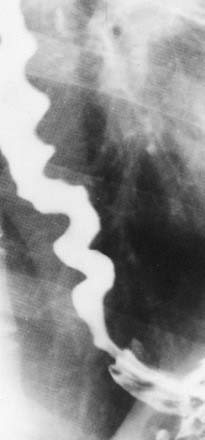
Patients with diffuse esophageal spasm have normal peristalsis in the proximal third of the esophagus, but contractions in the distal two thirds are uncoordinated and nonperistaltic. Possible radiographic manifestations include the corkscrew esophagus (a spiral column of contrast, Fig. 102-14), the rosary or shish-kebob (pouches separated by tight contractions), and diffuse narrowing. The diagnosis of diffuse esophageal spasm is made only when these radiographic findings are accompanied by substernal chest pain.
Nutcracker esophagus, also known as symptomatic esophageal spasm or hypertensive peristaltic esophagus, is characterized by high-contractility peristaltic waves causing chest pain. The esophagram is usually normal; this diagnosis is made with manometry.55,56
Diverticula
Zenker’s Diverticula
Zenker’s diverticulum is the most prevalent of the pharyngeal diverticula. It is a true diverticulum of the posterior pharyngeal wall, arising through Killian’s dehiscence just above the cricopharyngeus muscle. Patients generally present with halitosis and food regurgitation. Zenker’s diverticulum is best imaged fluoroscopically in the lateral projection (Fig. 102-15). Contrast is seen filling the diverticulum during swallowing, with variable retention between swallows. Large diverticula may compress the hypopharynx. Fluoroscopy is useful to delineate the size of the pouch and the diameter of its neck.
Pharyngeal Pouches
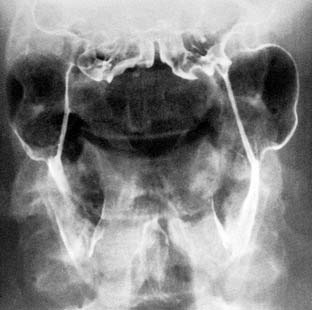
The pharyngeal wall may protrude out of its normal contour at the levels of the tonsillar pillar or thyrohyoid membrane. This “pharyngeal pouch” is usually asymptomatic, but patients may complain of dysphagia or regurgitation of food in severe cases. Pharyngeal pouches are best viewed in frontal projection, where they fill transiently (Fig. 102-16). Pouches should be distinguished from true pharyngeal diverticula, which present as persistent outpouchings of pharyngeal mucosa (Fig. 102-17).57 Pharyngeal diverticula are seen with chronically increased pharyngeal pressure, as in trumpet players and glass blowers.
Laryngoceles
A laryngocele is a diverticulum of the saccule of the laryngeal ventricle. Laryngoceles are classified as internal, external, or mixed, depending on whether they traverse the thyrohyoid membrane. Esophagram is usually normal, but the aryepiglottic folds may be displaced by the underlying mass. Laryngocele is best evaluated with CT or MR, where it appears as a well-defined, air-filled or fluid-filled mass in or around the larynx (Fig. 102-18).58 The density or signal of the mass varies with the degree of proteinaceous debris.