CHAPTER 106 Diagnostic Imaging of the Larynx
The radiologist plays an important role in the evaluation of upper airway disorders in children and adults by providing unique and useful diagnostic information directly affecting the treatment of the patient.1,2 In the past, evaluation of the airway included plain-film radiography, barium studies, and fluoroscopy. Now computed tomography (CT) and magnetic resonance imaging (MRI) have become the procedures of choice for defining mass lesions and traumatic abnormalities. These procedures can supplement the findings at laryngoscopy when additional diagnostic information is required to plan treatment.
Normal Airway
Technique
Plain films remain an effective and inexpensive screening examination for acute airway obstruction. Except in rare circumstances, plain-film examination of the airway should include anteroposterior and lateral radiography of the pharynx and laryngotracheal air column. In cooperative patients, these films should be exposed during inspiration with the patient upright because acute respiratory obstruction may be exacerbated in the recumbent position. Although conventional soft tissue techniques are often adequate, high kilovoltage magnification technique with selective filtration better visualizes the airway with improved air/soft tissue interfaces.3–5 Xeroradiography has been abandoned in clinical practice.6,7 When plain films are inconclusive, fluoroscopy and barium studies of the esophagus may be required for diagnosis. Abnormal vocal cord motion or swallowing mechanisms, vascular impressions, mass lesions, or nonopaque foreign bodies are best evaluated by these methods. Cross-sectional imaging may also be helpful in chronic airway disease.
Anatomy
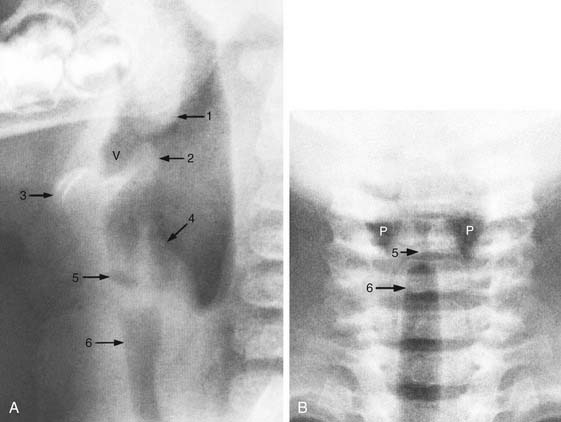
Lateral radiography is excellent for identifying the tongue, adenoids, tonsils, epiglottis, aryepiglottic folds, pyriform sinuses, laryngeal ventricle, and subglottic trachea (Fig. 106-1A). The anteroposterior airway is superb for examining the glottic and subglottic areas (see Fig. 106-1B). During quiet inspiration, the vocal cords are abducted and the width of the upper airway almost equals that of the trachea. During phonation of the vowel “e,” the vocal cords adduct, resulting in narrowing of the glottic area. However, narrowing of the subglottic area should be considered abnormal.
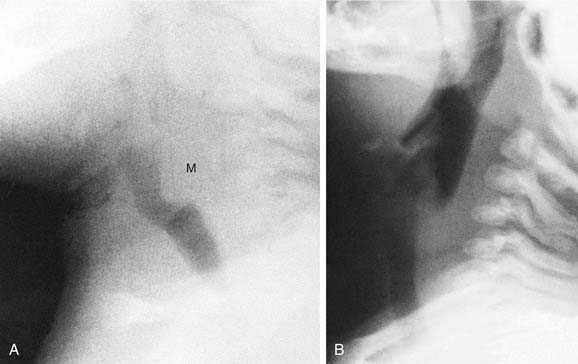
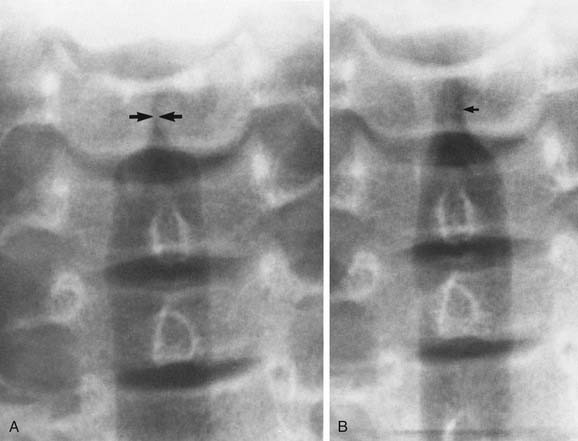
The airway may show considerable variability in children, unlike in adults. Therefore knowledge of the normal airway anatomy is essential before attempting to recognize pathology. This requires that the patient be properly positioned for the study. Lateral airway radiography should be obtained as much as possible in full inspiration with the neck extended. If the study is performed during expiration or with forward flexion of the neck, the retropharyngeal soft tissue in children bulges anteriorly and may simulate a retropharyngeal mass (Fig. 106-2).8
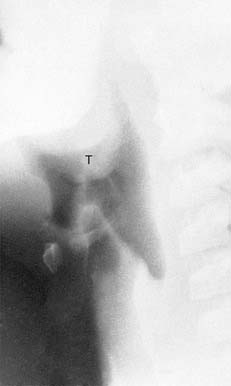
The size of the adenoids and tonsils in children and irregular cartilage ossification in adults can present significant problems in evaluation of the airway. In newborns and infants, the tonsils and adenoids are normally sparse; lymphoid tissue can be identified radiographically in all children at 6 months of age.9 After age 6 months, the tonsils and adenoids vary considerably in size and may encroach on the nasopharynx or oropharynx, suggesting a pathologic soft tissue mass. In most cases these structures, even when large, are normal and are frequently noted as incidental findings on plain-film radiography or cross-sectional imaging scout views or sagittal views. Occasionally, however, they can be associated with airway obstruction (Fig. 106-3).10,11 The adenoids are largest at 7 years to 10 years of age and then decline by the seventh decade.12 In most instances, adenoidal size can be evaluated subjectively, but objective methods of assessing adenoidal size using an adenoidal-to-nasopharyngeal ratio have been described.12,13 The final decision as to whether adenoid or tonsillar tissue is of symptomatic importance is clinical, not radiographic.
Cartilage of the neck may pose another problem because in certain adults, normal thyroid and cricoid calcifications can be irregular and incomplete, thereby simulating a foreign body or neoplastic destruction.14 These normal calcifications can be seen occasionally in older children, but as a rule the only cartilage calcified routinely in the younger child is the hyoid bone. Any other radiopaque structure is usually abnormal.
Cross-sectional imaging also adequately shows the airway. Configuration of the airway on axial or transverse images varies depending on the level of the image. At the level of the epiglottis and aryepiglottic folds, the airway is elliptic. Approaching the false cords, the airway narrows and assumes a tear-drop shape. The airway becomes elliptic at the true cords. The term rima glottidis refers to the airway at the level of the true vocal cords. The intermembranous portion of the rima glottidis (glottis vocalis) consists of the ventral 60% of the cords, and the dorsal intercartilaginous portion (glottis respiratoria) consists of the portion between the arytenoid cartilages.15 Below the cricoid cartilage, the airway appears circular. The posterior membrane of the trachea may posteriorly flatten, and the normal esophagus occasionally indents the airway silhouette.16 Nodular projections into the airway or asymmetric tracheal wall thickening should be viewed suspiciously as subglottic tumor involvement. Multirow detector CT imaging has recently allowed computer-generated, three-dimensional visualization of the airway lumen and contiguous structures by reconstructing two-dimensional data. Named “virtual laryngoscopy,” this technique offers noninvasive interactive evaluation of the tracheal lumen that correlates well with conventional rigid endoscopy (Fig. 106-4).17,18
Inflammatory Disease of the Larynx
Epiglottitis
Epiglottitis is an inflammation of the epiglottis caused by the bacteria Haemophilus influenzae. The entire supraglottic airway may be involved, but the epiglottis and aryepiglottic folds are most extensively involved. These normally well-defined thin structures become edematous, enlarged, and unsharp, resulting in a rounded, thumblike density in place of the epiglottis. The edema often encroaches on the vallecula and rarely may extend to the posterior pharyngeal wall. In addition, the hypopharynx and pyriform sinuses are usually mildly to moderately overdistended.19–22 The remainder of the airway is fairly normal, although at least 25% of children with epiglottitis have subglottic narrowing.23
The changes of epiglottitis are shown best on lateral radiography (Fig. 106-5). Extensive manipulation of the patient should be avoided because of the possibility of inducing glottic spasm. Cross-sectional imaging is unnecessary.
Enlargement of the epiglottis may result from various other disorders including irritation from a foreign body or burn, tumors such as epiglottic cysts or neoplasms (e.g., lymphoma), granulomatous disease (e.g., sarcoidosis, tuberculosis, Wegener’s granulomatosis), and angioneurotic edema.24,25 The radiographic findings should therefore be correlated with the patient’s clinical history (Fig. 106-6). On occasion, an omega epiglottis—a normal anatomic variant in children in which the epiglottis is floppy, vertically positioned, and resembles the capital Greek letter “omega”—may be misdiagnosed as epiglottitis.26 An important distinguishing feature is the absence of thickened aryepiglottic folds or other edematous changes.
Croup
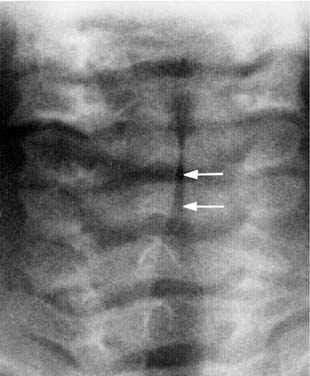
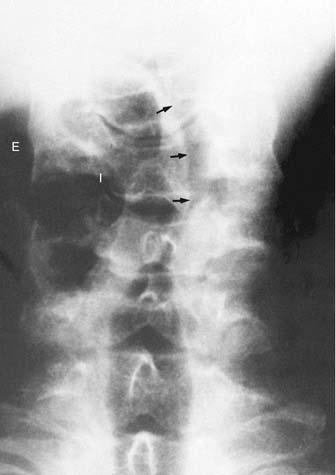
Croup is an inflammation of the subglottic larynx usually caused by parainfluenza virus type 1. It typically occurs in young children. Radiographic studies are not indicated or obtained routinely in patients with croup but are useful in confusing cases, primarily to exclude other causes of stridor. The radiographic changes are the result of inflammatory edema affecting the larynx and subglottic tissue. Radiographically, the frontal view is most helpful in the diagnosis (Fig. 106-7). Symmetric subglottic airway narrowing or “penciling” of the airway is the major radiographic finding.19–22 In contrast to congenital subglottic stenosis, narrowing of the subglottic portion of the trachea is not fixed and may improve on expiration.27 The lateral view of the neck appears less helpful, although the narrowing may be noted. This projection shows hypopharyngeal airway distention, but more importantly it establishes that the epiglottis and aryepiglottic folds are normal.
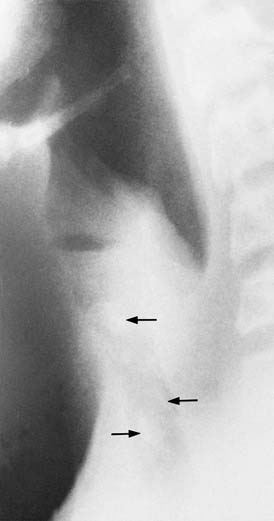
Membranous or bacterial croup and viral croup may present with similar symptoms; however, membranous croup is characterized by diffuse inflammation of the larynx, trachea, and bronchi with adherent exudate and mucus on the surface of the upper tracheal mucosa.28 Radiography shows subglottic narrowing and multiple tracheal soft tissue excrescences (Fig. 106-8). These intraluminal lesions can be mistaken for foreign bodies, so clinical correlation is required for diagnosis.
Retropharyngeal Abscess
The retropharyngeal space lies posterior to the larynx between the middle and deep layers of the deep cervical fascia. It extends from the base of the skull to the mediastinum and frequently serves as a conduit for spread of disease from the neck into the chest.29,30 Retropharyngeal abscess results from suppuration of retropharyngeal lymph nodes in patients with upper respiratory tract infection or from perforation of the pharynx or upper esophagus by a foreign body. If the abscess compresses the larynx and upper trachea, symptoms of upper airway obstruction develop.
Lateral soft tissue radiography of the neck will show fixed thickening of the retropharyngeal soft tissues, anterior displacement of the airway, reversal of normal cervical lordosis, and occasionally gas bubbles within the abscess (Fig. 106-9).19,21,27 The retropharyngeal space should not exceed 7 mm as measured from the most anterior aspect of C2 to the posterior pharyngeal wall. At C6, the thickness of the retropharyngeal tissues should not be greater than 14 mm in children and 22 mm in adults.31 Fluoroscopy or barium esophagography may be necessary to confirm that the radiographic changes did not occur with expiration or with the head in a flexed position.
CT and MRI superbly evaluate the retropharyngeal space. Suppurative adenopathy is usually limited to the suprahyoid region of this space and spares the midline. As infection becomes extranodal, the entire width of the retropharyngeal space frequently becomes thickened. Inflammation can then easily track into the infrahyoid region. The full extent of the process can be imaged by CT or MRI before therapy.29 Abscess will appear hypodense on CT, hypointense on T1-weighted, and hyperintense on T2-weighted magnetic resonance (MR) images, reflecting the presence of liquefaction (Fig. 106-10). Ring enhancement may be seen following administration of intravenous contrast. Differentiation between retropharyngeal abscess and adenitis is difficult on CT because both processes cause hypodense regions within the inflammatory mass. Some authors advocate the use of ultrasound in this evaluation.32 If air bubbles or a foreign body is present within the mass, the diagnosis of a retropharyngeal abscess is more likely.
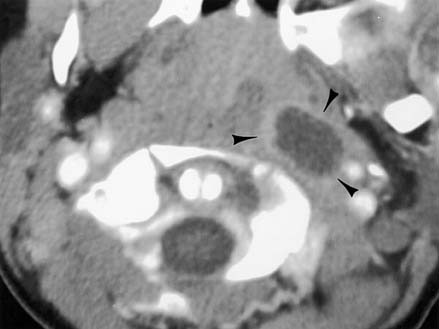
Figure 106-10. Retropharyngeal abscess. Enhanced CT image of the neck demonstrates a fluid collection in the left retropharyngeal space (arrowheads).
Thickening of the retropharyngeal soft tissues also can occur secondary to bleeding and edema from cervical spine trauma, lymphadenopathy (e.g., lymphoma, tuberculosis), or retropharyngeal tumors (e.g., cystic hygroma, neuroblastoma, hemangioma, retropharyngeal goiter, cancer).27,33–35 The imaging findings should therefore be correlated with the clinical history.
Laryngomalacia
Laryngomalacia or congenital flaccid larynx is one of the most common causes of inspiratory stridor in the neonate and infant.21 Laryngoscopy shows flaccidity of the epiglottis, aryepiglottic folds, or the entire larynx, which collapses during inspiration. The corresponding radiographic findings are best shown with fluoroscopy. Hypopharyngeal overdistention with associated collapse of the aryepiglottic folds and epiglottis is observed on inspiration. Paradoxic narrowing of the subglottic portion of the airway may also be seen as the patient inspires. The primary reasons for laryngoscopy and radiography are to exclude other causes of congenital stridor (e.g., cysts, webs, tumors, stenoses). In most children, the symptoms disappear by 1 year of age.
Vocal Cord Paralysis
Paralysis of the vocal cord may be caused by any process involving the vagus nerve or its recurrent laryngeal branch between the jugular foramen and its entrance into the larynx. Almost 75% of patients have unilateral paralysis.36 Almost 90% of paralyses are caused by lesions that compress the nerve along peripherally located segments, and only 10% originate in the central nervous system or before the nerve exits the jugular foramen; central paralyses are accompanied by other cranial neuropathies.37 The peripheral location of most lesions reflects the long and redundant routes of the recurrent laryngeal nerves. Specific causes include neoplasm (36%); postoperative complications, such as from parathyroid and thyroid surgery (25%); and inflammation (13%). Some cases are idiopathic.36 Congenital central nervous system anomalies are often associated with childhood vocal cord paralysis.38 Although laryngeal cancers can paralyze the cord by direct invasion of the thyroarytenoid muscle, infiltration of the recurrent or superior laryngeal nerves by local tumor is rare.39
Although plain-film radiography, fluoroscopy, and CT can evaluate vocal cord mobility, abnormal vocal cord motion is generally diagnosed by laryngoscopy (Fig. 106-11).40,41 However, in patients with vocal cord paralysis secondary to neoplasm, imaging helps show the extent of neoplasm and its relationship to the adjacent cartilaginous structures.42,43 Radiologic evaluation of the isolated paralyzed cord should entail imaging of the entire vagus nerve from the skull base to the pulmonary hila. CT is excellent for evaluating the neck and chest; MRI is superior for the skull base (Fig. 106-12).36,44,45, Imaging signs of cord paralysis include paramedian position of the cords, displaced arytenoid cartilage, ipsilateral dilation of the pyriform sinus, tilting of the thyroid cartilage, and prominent laryngeal ventricle.37 Dynamic MRI using an open-bore configuration, with the patient seated, has recently been explored as a technique for evaluating swallowing and other laryngeal and pharyngeal functions.46
Benign Laryngeal Masses
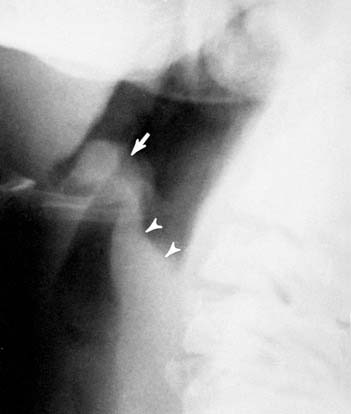
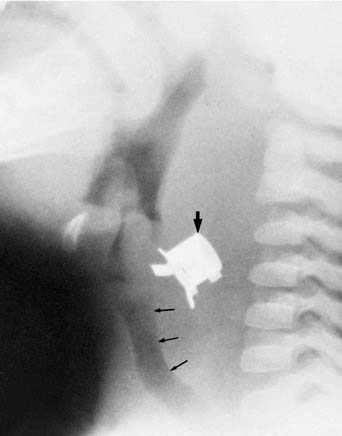
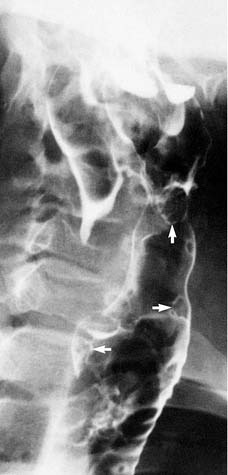
Although rare in adults, subglottic hemangioma is the most common laryngeal and upper tracheal neoplasm in the newborn and infant. The lesion typically appears as a well-defined mass in the posterior or lateral portion of the subglottic airway (Fig. 106-13).25,47 The subglottic narrowing is usually eccentric; however, circumferential narrowing suggestive of croup may also be seen. Additional hemangiomas may occur on the skin or elsewhere in the body.
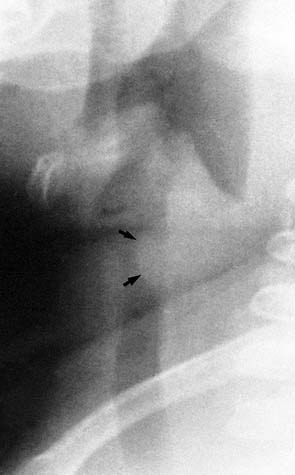
Figure 106-13. Subglottic hemangioma. Lateral view shows a soft tissue mass (arrows) in the posterior portion of the subglottic airway.
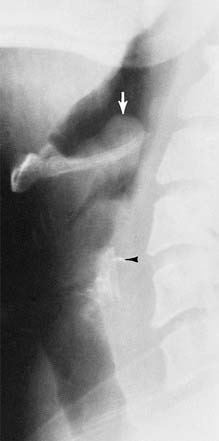
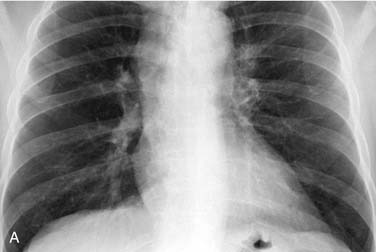
Squamous papillomas, the most frequent laryngeal tumors in children, have also been reported in adults.48,49 The imaging appearance of papillomatosis is contingent on the size and location of the lesions. Generally single or multiple intraluminal soft tissue nodules are seen in the glottis or in the tracheal air column (Fig. 106-14). Because papillomas may extend to the bronchial tree or pulmonary parenchyma, chest CT is extremely important in defining the extent of disease. Localized areas of atelectasis, air trapping, or pneumonia have been reported. Papillomas also may appear as well-defined discrete pulmonary nodules that eventually cavitate, forming multiple thin-walled cystic lesions.50,51
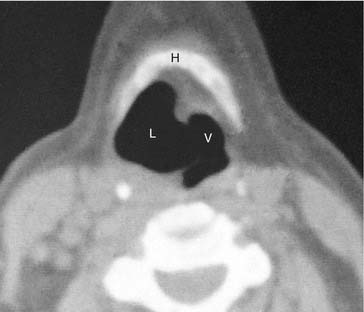
A laryngocele is an abnormal elongation and expansion of the saccule of the laryngeal ventricle and represents more than 20% of submucosal lesions of the larynx.52,53 Almost 25% are bilateral.54 Although laryngoceles are usually asymptomatic, large lesions may be associated with airway obstruction and vocal cord paralysis.55 Laryngoceles within the paraglottic space and confined by the thyroid lamina are termed internal. These lesions may present on laryngoscopy as soft, localized bulges of the aryepiglottic fold and may be mistaken clinically for submucosal neoplasms.56–58 Lesions that pierce the thyrohyoid membrane to present in the lateral neck are termed external. Most cases are a combination of external and internal lesions and are referred to as mixed. Regardless of position, laryngoceles are usually visualized as sharply defined air-containing structures on imaging (Fig. 106-15).59 A modified Valsalva’s maneuver occasionally improves visualization of these lesions. Laryngeal mucocele is a fluid-filled laryngocele caused by obstruction of the ostium of laryngeal saccule and may appear as a soft tissue mass. These lesions are overwhelmingly benign; however, up to 15% may be associated with a small ventricular cancer.57,60,61 Rarely, cystic lesions such as cystadenomas can mimic a laryngeal mucocele.62
CT can be helpful in showing fluid-containing and air-containing laryngoceles (Figs. 106-16 and 106-17).56 If the mass is of soft tissue density because it contains either mucoid or purulent material, the distinction from neoplasm may be more difficult. However, its location and smooth surface in conjunction with a healthy mucosa on laryngoscopy should suggest a laryngocele. CT may also help show the external component of a laryngocele that is not apparent on physical examination. An enhancing wall usually indicates infection. The endolaryngeal origin of this lesion differentiates it from a lateral thyroglossal duct cyst.57
Laryngeal Carcinoma
Nearly all mucosal tumors are diagnosed by direct inspection and biopsy; imaging provides crucial information about involvement of deep structures such as the paraglottic space, cartilage, and lymph nodes. CT is a proven imaging technique for evaluating patients with laryngeal carcinoma.63–67 In most patients, CT shows more extensive disease than is initially appreciated by laryngoscopy. The cross-sectional imaging provided by CT allows evaluation of the intrinsic and deep soft tissues of the larynx and the cartilaginous skeleton.
MRI provides anatomic information on the neck that compares favorably with CT.68–72 Advantages of MRI include its excellent soft tissue contrast, which is superior to CT. Multiplanar display enables coronal, transverse, and sagittal anatomic formatting, whereas CT is usually limited to the transverse plane. Newer multirow detector CT scanners allow for multiplanar reconstructions; however, these views are derived from data obtained in the transverse plane. MRI uses no ionizing radiation and is not plagued by artifacts caused by beam hardening, dental amalgam, or poor beam penetration of the shoulders. MRI does have limitations, however. It is slower than CT, and therefore motion artifacts from breathing, carotid artery pulsations, and swallowing may degrade images.73–75 This should be considered when choosing a modality in laryngeal cancer patients, who are often elderly and may have other medical debilities (e.g., chronic obstructive pulmonary disease).74 MRI does not image cortical bone or calcifications well. Furthermore, MRI is contraindicated in patients with cardiac pacemakers, metallic cochlear implants, and cerebral aneurysm clips.76–79
The decision for selecting radiotherapy, conservation surgery, or total laryngectomy depends on an accurate delineation of tumor extent.80,81 CT and MRI are excellent noninvasive methods capable of three-dimensional (3D) anatomic display of portions of the larynx that are not well examined by laryngoscopy. Submucosal extension of tumor and cartilaginous destruction that are not suspected clinically can be assessed. Because imaging modalities have different strengths and weaknesses, the choice of CT or MRI should be tailored to the patient. Furthermore, imaging is not a substitute for but is complementary to laryngoscopy. Although imaging may show advanced mucosal abnormalities, minor mucosal abnormalities and abnormalities of intrinsic motion are best studied by laryngoscopy.82
Neoplasm is identifiable on CT as an area of increased soft tissue density that alters the normal symmetric laryngeal anatomy. Similarly, the hallmark of an abnormal larynx on MRI is asymmetry. The superb soft tissue contrast provides additional information. On T1-weighted images, fat signal is hyperintense and differs significantly from the mucosa and muscles in appearance. Signals from lymph nodes and infiltrating tumor, which are less intense than fat, are well visualized on T1-weighted images. On T2-weighted images, muscle signal is less intense than mucosa, fat, and many tumors. Carcinomas within the mucosa and muscles can be well delineated.83 Neither CT nor MRI findings are histologically specific, and similar appearances can be produced with hemorrhage, edema, inflammation, or fibrosis. Therefore, examination should be correlated with the clinical history and should be performed before laryngeal biopsy or at least 48 hours after biopsy to avoid confusion with postbiopsy edema and hemorrhage.
Technique
Spiral (helical) CT using multidetector technology and volumetric data sets has largely replaced conventional dynamic CT (slice-by-slice acquisition) in most medical centers.69,84 Spiral CT permits rapid scanning of large volumes of tissues during quiet respiration.85 Spiral images are less susceptible to patient motion than conventional CT, although image noise is slightly increased.86,87 Thin sections can provide information about specific regions, such as the anterior commissure.88 Moreover, volumetric spiral data permit multiplanar and 3D reconstructions.87,89,90 The amount of intravenous contrast may be reduced compared with dynamic CT.91 Iodinated contrast is administered intravenously to distinguish nonenhancing lymph nodes from enhancing vessels. Images are obtained during slow inspiration. If the images are performed during suspended inspiration, adduction of the true vocal cords may cause the airway to appear falsely narrowed. Scans are occasionally obtained during phonation or during modified Valsalva’s maneuver to distend the pyriform sinuses and improve visualization of the aryepiglottic folds.
3D CT is currently being used for radiotherapy planning. Slices are obtained in the conventional axial orientation. Using special computer software, selected anatomic structures are then traced and reformatted into a 3D wire diagram depicting the tumor and key adjacent tissues that can then be manipulated to reveal the optimal radiation port. This technique limits extraneous collateral radiation to other organs such as the salivary glands.92,93 CT is also useful for guiding percutaneous biopsies.94–97
MR techniques may vary depending on scanner type and available hardware and software. Surface coils are essential to adequately image the larynx.42,98 Sagittal, transverse, and coronal T1-weighted images best display anatomic relationships. T1-weighted and T2-weighted transverse images further define the signal characteristics of the tissues. Sections are usually 3 to 5 mm thick. Gradient moment nulling, flow compensation, cardiac gating, and presaturation pulses minimize motion artifacts.74 Fast spin-echo T2-weighted technique has replaced conventional spin-echo technique and offers relatively short acquisition times.99–102
Gadolinium-enhanced images improve delineation of margins in many lesions,103–107 although lesions embedded in fat may be obscured.108,109 Another potential pitfall is that healthy aerodigestive mucosa enhances, therefore possibly obscuring small mucosal tumor volumes.54 Fat-suppression techniques improve conspicuity of soft tissue lesions embedded in fat by selectively diminishing the hyperintensity of fat on T1-weighted images.108,110 Gadolinium-enhanced, fat-suppressed T1-weighted images are especially useful in staging nodal disease.111 Agents are typically well-tolerated.104,112 Newer agents such as gadobenate dimeglumine are improving lesion conspicuity compared with equivalent doses of older agents. A word of caution is necessary, however. Recently, several cases of nephrogenic systemic fibrosis/nephrogenic fibrosing dermopathy (NSF/NFD) have been reported in patients with renal failure receiving gadolinium compounds, especially in high doses. (It is worth noting that the use of double and triple doses of contrast, sometimes valuable in intracranial disease, has little value in neck imaging.113) NSF/NFD, a rare and somewhat mysterious complication, causes a progressive sclerosis of the skin and connective tissue and may lead to death. Currently, recommendations include calculating a glomerular filtration rate for patients with renal insufficiency or frank failure and avoiding the use of contrast agents in the impaired patient.
Fat suppression may produce several annoying artifacts, however, such as signal loss at bone-air interfaces, inhomogeneous suppression at the base of skull compared with the infrahyoid regions, and frank failure of suppression at fat-air interfaces producing aberrant hyperintense signal.114 Moreover, some techniques may be misleading because nonfatty tissue with T1 values similar to fat (e.g., proteinaceous fluid, methemoglobin, intensely enhancing soft tissue) may also be suppressed.115
MR techniques and applications such as magnetization transfer improves the contrast between lesions and background tissue.116–120 Reconstruction algorithms permit simultaneous 3D depiction of the tumor volume embedded within the surrounding soft tissues. This display assists the transformation of two-dimensional information into a 3D format and allows the examiner to “dissect” the display to a desired tissue depth.121,122 Diffusion-weighted MRI, nanoparticle-enhanced MRI, and perfusion MRI are also currently being explored.123
Open-bore units are being used to guide biopsies.124,125 These units are useful for the claustrophobic patient, who would otherwise be unable to tolerate the confining space of conventional magnets. Unfortunately, many of these scanners lack sufficient magnet strength to optimally display difficult or complex anatomic relationships.
Special applications such as MR spectroscopy have been evaluated for certain head and neck tumors.126 MR spectroscopy measures and graphically displays tissue metabolites.127 Choline (Cho), a precursor to phosphatidylcholine, is involved in membrane turnover. An elevation of the Cho spectral peak within tissue implies cellular turnover and may serve as a potential tumor marker.128,129 MR spectroscopy may prove useful by using metabolic data to map tumors and complement anatomic data from conventional imaging.130 Although MR spectroscopy has advanced rapidly on the intracranial application front, initial enthusiasm for head and neck lesions has dampened. Formidable technical issues such as artifacts from paranasal sinuses, airway, neck fat, and blood vessels have made consistently reliable spectra difficult. Therefore widespread use in head and neck awaits significant refinement.
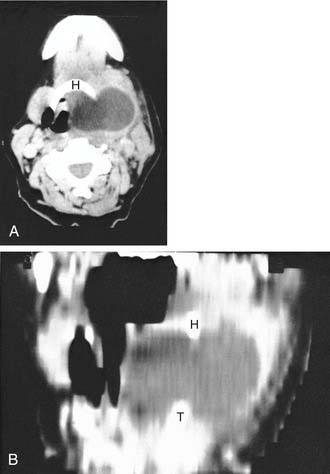
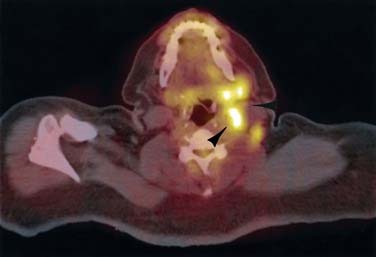
Combined positron emission tomography (PET) CT is a new technique in which near simultaneously acquired PET and CT information is superimposed. This image fusion has the advantage of comparing superior sensitive functional PET data with the superior anatomic data offered by CT (Fig. 106-18). Promising applications include evaluation of post-therapy patients for evidence of recurrent tumor, analysis of ambiguous lymph nodes and soft tissue masses, and verification of biopsy sites.123,131,132