CHAPTER 85 Diagnostic Imaging and Fine-Needle Aspiration of the Salivary Glands
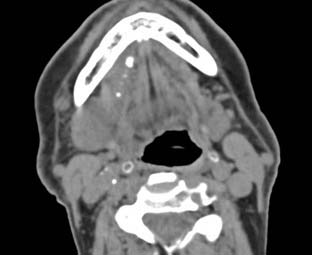
Sialolithiasis (Fig. 85-1) can be seen in any salivary gland but is most commonly associated with the submandibular gland secondary to the more mucinous saliva and the horizontal trajectory of the submandibular duct. Symptoms of acute pain and swelling in the region of a salivary gland would raise suspicion for sialolithiasis, and a CT should be ordered because CT is the modality with the greatest sensitivity for calcification in an obstructive stone.1
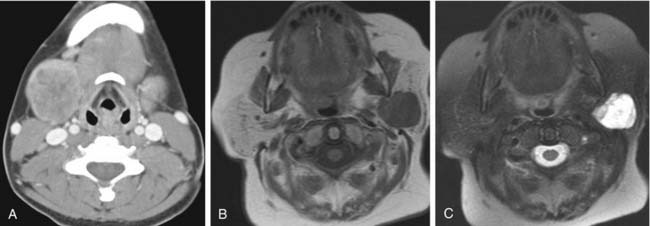
Neoplasms can occur in any salivary gland tissue. The likelihood of malignancy is largest in the smallest glands. Lesions of the minor salivary glands, for example, have a 50% to 80% chance of being malignant. In the parotid gland, only 20% to 30% of lesions will be malignant, whereas 40% to 60% of submandibular gland lesions will be malignant. In the parotid, therefore, 70% to 80% of lesions will be benign. Of these benign lesions, 80% will be pleomorphic adenomas (Fig. 85-2).2 Although imaging can give clues as to the pathologic diagnosis (i.e., smooth borders are more likely benign and infiltration of adjacent structures is a sign of malignancy), the role of imaging is primarily to identify the lesion and describe its association with adjacent structures.
Anatomy
Parotid Glands
The paired parotid glands are the largest of the salivary glands. They are located in the parotid spaces wrapping around the posterior aspects of the mandibular rami bilaterally. Each gland is divided into a superficial lobe and a deep lobe with the posterior facial vein (retromandibular vein) present as the radiographic landmark between the two lobes. The main trunk of the facial nerve exits the skull base at the stylomastoid foramen and pierces the posterior fascial border of the parotid space lateral to the surface of the posterior belly of the digastric muscle and then courses within the parotid gland lateral to the retromandibular vein. High-resolution MR techniques can show the facial nerve and its relationship to the parotid duct, vascular structures, and, if present, intraparotid tumors.3 On contrast-enhanced CT or when high-resolution images are not performed, the location of the facial nerve is approximated by the retromandibular vein. Approximately 80% of the parotid lies in the superficial lobe anterior and inferior to the external auditory canal extending down to the angle of the mandible. The deep lobe extends through the stylomandibular tunnel formed anteriorly by the ramus of the mandible and posteriorly by the stylomandibular ligament. The location of a mass in the parotid gland must be defined as superficial, deep, or extraglandular to best plan the surgical approach in order to limit damage to the facial nerve. The parotid duct (Stensen’s duct) extends anteriorly from the superficial lobe of the parotid gland around the masseter muscle and anterior to the buccal fat where it proceeds medially to pierce the buccinator muscle. The duct then briefly runs caudally before exiting the papilla at the buccal mucosa opposite the second maxillary molar.
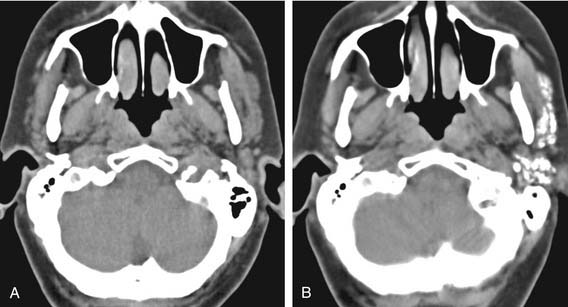
Frommer,4 in his study of anatomic dissections, found that approximately 21% of parotid glands have an accessory gland. The accessory component is typically located superficial to the masseter muscle just cranial to the main parotid duct (Fig. 85-3). One excretory duct from the accessory parotid gland joins with Stensen’s duct. The accessory parotid gland is subject to the same pathologies that affect the parotid gland. The rate of malignancy is estimated to be between 30% and 50%, with mucoepidermoid the most common lesion. Pleomorphic adenoma is the most common benign neoplasm of the accessory parotid gland.
The parotid glands contain many lymph nodes that drain the palatine tonsil, posterior palate, root of the nose, eyelids, frontotemporal scalp, external auditory meatus, and tympanic cavity. This network of nodes drains into the level IIA and IIB nodes of the internal jugular chain and spinal accessory chain.5
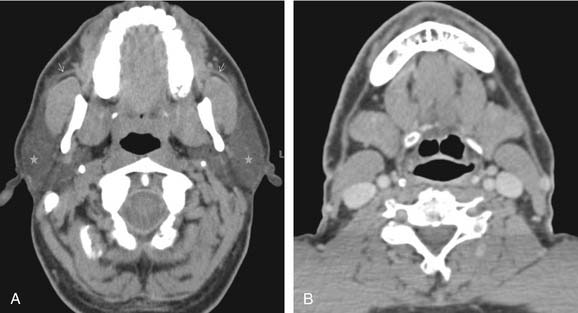
The parotid gland is interspersed with fat. On CT, the attenuation of the normal gland is lower than muscle and greater than fat. The greater the fat content of the gland, the easier the detection of a parenchymal soft tissue nodule. On MRI, the gland is intermediate to high signal on T1-weighted imaging and low to intermediate on T2-weighted imaging, reflecting its fat content. Flow voids are detectable on T2-weighted imaging due to vessels within the gland. Septations of lower T1 signal are seen in the gland due to interstitial tissues, ducts, and facial nerve branches. The intraparotid facial nerve can be visualized, especially on high-resolution imaging3,6,7; the nerve does not normally enhance. Contrast will reveal homogeneous enhancement of the normal parotid glandular tissue (Fig. 85-4).
Submandibular Glands
The paired submandibular glands are visible posterior to the angle of the mandible largely within the submandibular space with a small deep portion curling up cranially around the posterior border of the mylohyoid muscle. The submandibular duct (Wharton’s duct) exits the deep lobe extending anteriorly and superiorly, medial to the sublingual gland in the sublingual space terminating at the sublingual papilla in the floor of the mouth. The lingual nerve and hypoglossal nerve are closely associated with the duct as it courses anteriorly through the fat-filled sublingual space. The lymphatic drainage from the glands is into the submandibular nodes (level IB).5
Sublingual Glands
The small, paired sublingual glands are positioned up against the sublingual depressions of the inner surface of the mandible just superior to the mylohyoid muscles bilaterally. Approximately 20 ducts (ducts of Rivinus) drain into the floor of the mouth. The lymphatic drainage from the sublingual gland is into the submental and submandibular lymph nodes (levels IA and IB).5
Minor Salivary Glands
The minor salivary glands are numerous and are present in the deeper submucosal planes of the oral cavity (especially the lip and hard and soft palates), paranasal sinuses, pharynx, external auditory canal, nose, and paranasal sinuses. Though high-resolution MR imaging has been shown to reveal these small glands in the labia,8 these minor salivary glands are not typically identified on imaging unless they are pathologically enlarged or involved with tumor, of which they are an important tissue of origin.
Magnetic Resonance Imaging
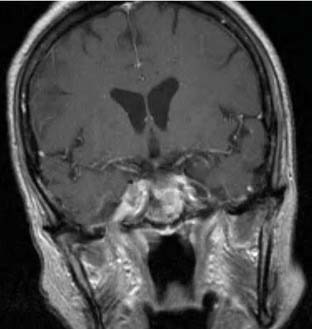
MRI is the modality of choice for the evaluation of salivary neoplasms. Because there is such distinct soft tissue differentiation on MRI, most lesions are identifiable on T1-weighted images, especially in the parotid gland with the fatty background. With the addition of postcontrast imaging on fat-saturated sequences, salivary lesions are easily identified.9 The adjacent soft tissues are just as well defined, allowing for the assessment of perineal spread, intracranial extension, or marrow invasion. After fat saturation, the previously hyperintense fat on T1-weighted imaging becomes hypointense and the soft tissue mass stands out within the gland. Intravenous gadolinium contrast is helpful to further define salivary lesions and, if present, enhancement along the meninges, within the marrow of the mandible or maxilla or within the foramina of the skull base, all of which would suggest spread of tumor along these routes. Enhancement within the stylomastoid foramen (cranial nerve VII), foramen ovale (cranial nerve V-III), foramen rotundum (cranial nerve V-II), vidian canal (vidian nerve), or the pterygopalatine fossa suggests perineural spread, and MRI is the best modality to investigate this (Fig. 85-5).
Benign lesions are typically well-defined simple or lobulated lesions. Pleomorphic adenomas will have low T1 and high T2 signal intensities similar to water but with solid enhancement best seen on fat-suppressed T1WI after gadolinium. Keep in mind that although pleomorphic adenomas are thought of as benign lesions, malignant degeneration within a pleomorphic adenoma (carcinoma ex pleomorphic adenoma or malignant mixed tumor) may be as high as 25% if left untreated and may, in fact, occur in a patient known to have a previously resected pleomorphic adenoma.2 For this reason, pleomorphic adenomas are most commonly removed when they are diagnosed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree