CHAPTER 114 Diagnosis and Management of Tracheal Neoplasms
Overview
Primary tracheal tumors are relatively uncommon with an estimated annual incidence of two cases per 1 million people.1,2 Therefore an understanding of these neoplasms is based predominantly on small case series from large referral centers. Tracheal tumors are categorized as either primary or secondary depending on whether they arise within the trachea or secondarily involve the trachea from metastatic disease or local invasion. Primary tracheal tumors are further subdivided into benign or malignant lesions. In adults 90% of primary tracheal tumors are malignant, but in children 80% are benign.1 Primary tracheal tumors are most often treated with multimodality therapy including surgical resection and radiation.3 Currently, the disease-specific and all-cause mortality for primary malignant tracheal tumors are 73% and 79%, respectively.4
Historical Review
Morgagni described the first primary tracheal neoplasm, a fibroma, in 1761. Over 100 years would pass before experimental attempts at tracheal resection with primary end-to-end reconstruction would be described.6 Kuester performed the first human tracheal resection and primary reconstruction in 1884. Before the 1960s, it was generally believed that tracheal resection was limited to four tracheal rings, about 2 cm, because greater resections would place undue tension on the anastomosis and prevent adequate healing.7 Because of the presumed limits of resection and primary anastomosis, efforts at prosthetic replacement of the trachea were undertaken. In 1950 Belsey7 reported his initial attempt with tracheal reconstruction after resection of an adenoid cystic carcinoma using fascia lata reinforced with stainless wire. Pearson and colleagues8 used heavy Marlex mesh for tracheal replacement in seven patients and reported excellent function of the airway in three patients for several years. Neville and colleagues9 reported extensive experience with the use of a solid silicone tube to replace the trachea; others, however, have reported unacceptable morbidity and mortality with this nonporous material.10 Over the past several decades, a variety of other foreign materials, nonviable tissue, autogenous tissue, tissue engineering, and transplantation techniques have been attempted, but successful tracheal replacement remains elusive.11
Fortunately, the development of techniques for tracheal mobilization12–14 made it possible to perform extensive tracheal resections, involving 50% of the tracheal length, with primary reconstruction. Using these techniques, tracheal replacement is uncommon. For the rare occasion when primary anastomosis cannot be performed and replacement is necessary, promise lies with tracheal allotransplantation, but long-term immunosuppression requirements will obviously limit its potential uses.15
Primary Tracheal Tumors
Primary tumors of the trachea are uncommon, but a variety of benign and malignant neoplasms have been described (Table 114-1). In recent reports, men and women are equally affected by tracheal tumors, and the peak age of diagnosis is 50 to 59 years. Primary tumors can originate from any layer of the tracheal wall and histologically are classified as either epithelial or mesenchymal tumors. Although primary tumors can be found anywhere in the trachea, the proximal and distal thirds of the trachea are most frequently affected, as is the membranous wall. A few large surgical series of primary tracheal tumors have been reported (Table 114-2).16–19
Table 114-1 Classification of Tracheal Tumors
| Epithelial Neoplasms | Mesenchymal Neoplasms |
|---|---|
| Benign | Benign |
| Squamous cell papilloma | Fibroma |
| Papillomatosis | Hemangioma |
| Pleomorphic adenoma | Granular cell tumor |
| Schwannoma | |
| Malignant | Neurofibroma |
| Squamous cell carcinoma | Fibrous histiocytoma |
| Adenoid cystic carcinoma | Pseudosarcoma |
| Carcinoid | Hemangioendothelioma |
| Mucoepidermoid carcinoma | Leiomyoma |
| Adenocarcinoma | Chondroma |
| Small-cell undifferentiated carcinoma | Chondroblastoma |
| Lipoma | |
| Secondary Malignancies | Malignant |
| Invasion by adjacent malignancy | Leiomyosarcoma |
| Metastases | Chondrosarcoma |
| Paraganglioma | |
| Spindle-cell sarcoma | |
| Non-neoplastic Tumors | Lymphoma |
| Tracheobronchopathia osteochondroplastica | Malignant fibrous histiocytoma |
| Amyloidosis | Rhabdomyosarcoma |
| Inflammatory pseudotumor | Melanoma |
From McCarthy MJ, Rosado-de-Christenson ML. Tumors of the trachea. J Thorac Imag. 1995;10:180.
Staging
Neither an American Joint Committee on Cancer nor a Union Internationale Centre le Cancer staging system currently exists for primary tracheal tumors. However, a tumor node metastasis (TNM) system has been proposed on the basis of a single institution’s retrospective review of 99 patients with primary tracheal tumors. T1 and T2 tumors were defined as tumors confined within the trachea that are less than 2 cm or greater than 2 cm, respectively. T3 tumors extend beyond the trachea but do not involve any surrounding structure. T4 tumors extend into surrounding structures such as the esophagus or great vessels. In addition, patients are either N0 or N1 and M0 or M1 depending on the absence or presence of lymph node or distant metastasis, respectively.20
Benign Primary Tracheal Tumors
Uncommon in adults, benign primary tracheal tumors are usually well circumscribed, round, soft, and smaller than 2 cm. On chest computed tomography (CT) scanning, these lesions typically are seen as smooth, homogeneous tumors that do not extend through the tracheal wall. The presence of calcium within the lesion suggests a benign histology.21
Squamous Papilloma
Squamous cell papillomas are superficial, sessile, or papillary masses consisting of a connective tissue core covered by squamous epithelium. In children, squamous papillomas are the most common tracheal neoplasm. In adults, these lesions are rare and associated with heavy cigarette smoking. Although these lesions can be found throughout the respiratory tract, approximately 90% to 95% are isolated to the larynx.12,22,23 Eleven percent of the patients had papillomatosis in the trachea in addition to the larynx, but only 1.2% had disease isolated to the trachea. Presence of the disease below the larynx has been linked to the presence of a tracheostomy.22 Papillomatosis is caused by human papillomavirus (HPV) types 6 and 11,23 transmitted from mother to fetus during childbirth.24 Despite being a benign tumor, papillomatosis frequently recurs and is difficult to completely eradicate. Fortunately, it usually regresses spontaneously after puberty. Numerous therapies have been used including microdébridement, nebulized or intralesional injection of cidofovir, PDT, pulsed dye laser, and indole-3-carbinol.25,26 Recently, the carbon dioxide laser has shown promise in the treatment of papillomatosis.7 Adjuvant treatment with α-interferon has been used.27
Papillomas undergo malignant transformation to squamous cell carcinoma and verrucous carcinoma, a phenomenon apparently more commonly seen with HPV 11,28 that occurs with an incidence of 1.6% to 4%.25,29
Granular Cell Tumor
Granular cell tumors were first described by Abrikosoff in 192630 as a myogenic tumor, but more currently these tumors are understood to be neurogenic in origin. There appears to be no gender predilection, but the majority of granular cell tumors occur in African Americans. They rarely are seen in children. Approximately 50% of granular cell tumors occur in the head and neck region and 10% appear in the larynx, but the cervical trachea is rarely affected.31 Multicentric tumors occur in 20% of patients, and these tumors tend to be biologically more aggressive.32 Granular cell tumors are not encapsulated and tend to invade locally, despite being benign. Seventy-three percent of these tumors are intraluminal, and the remainder are either extraluminal or mixed.33 Malignant degeneration occurs in 1% to 2% of all granular cell tumors, but this has never been reported in children.34,35 Bronchoscopic resection of tumors smaller than 8 mm has been recommended.34 Granular cell tumors larger than 8 mm have a high likelihood of transmural involvement and recurrence following bronchoscopic resection, and these should be managed with segmental tracheal resection.
Chondroma
Chondromas are the most common benign mesenchymal tracheal tumors, occurring most often in older men.17–1936 These cartilaginous tumors are hard, broad-based, and covered by intact mucosa. The most common site of origin is the internal aspect of the posterior cricoid lamina,37 but they are evenly distributed throughout the length of the trachea. Calcification is radiographically seen in 75% of patients, although this will not distinguish chondromas from malignant chondrosarcomas, into which chondromas may degenerate on the basis of anecdotal evidence.38 Endoscopic resection may be used for palliation but leads to recurrence, and thus segmental tracheal resection is recommended.
Leiomyoma
Tracheal leiomyomas originate from the smooth muscle of the tracheal wall and most commonly arise from the membranous portion of the lower third of the trachea.39 These tumors typically grow as smoothly contoured, broad-based polypoid masses. Intraluminal growth can cause symptomatic airway obstruction. Death from bleeding has been reported during bronchoscopic excision, and this treatment modality has been recommended only for patients with pedunculated tumors.40 Segmental tracheal resection is recommended for leiomyomas in order to prevent local recurrence following incomplete resection.
Hemangioma
Hemangiomas of the airway occur in adults and children. In adults, cavernous hemangiomas develop in the larynx, whereas capillary hemangiomas originate in the subglottic trachea. Tracheal hemangiomas occur more often in young children and are actually the most common obstructive subglottic mass in this patient population.32 These lesions are asymptomatic at birth, but most will cause stridor within the first 6 months of life. Cutaneous hemangiomas are present in half of the affected infants.
In the airway, capillary hemangiomas develop within the submucosa and are covered by normal respiratory epithelium. They are typically located on the posterolateral aspect of the subglottic trachea, are smooth, bluish in color, and project into the airway from a sessile base. Most of these spontaneously regress by 2 to 3 years of age and close observation is advocated when possible. In highly symptomatic patients, a tracheostomy may be required temporarily. Regression following treatment with steroids, interferon, or laser ablation has been reported, although surgical resection may be associated with less stricture formation.41
Miscellaneous Benign Primary Tumors
A variety of other benign tracheal tumors have been reported including adenomas, myoepithelial cell tumors, lipomas, fibromas, schwannomas, neurilemomas, hemangiomas, paragangliomas, fibrous histiocytomas, neurofibromas, chondroblastomas, benign mucoepidermoid tumors, angiofibromas, xanthomas, myoblastomas, hamartomas, glomus tumors, intratracheal goiters, and chemodectomas. All combined, these tumors represent less than 10% of all primary tracheal tumors. Treatment for these diverse tumors is generally surgical excision, although some may be successfully managed with endoscopic resection.39
Malignant Primary Tracheal Neoplasms
In adults, the majority of primary tracheal tumors are malignant. Typically, malignant lesions have a shorter duration of symptoms than benign lesions, are larger in size, and exhibit an irregular surface. In most series, approximately 75% of all primary malignant tracheal neoplasms are histologically either squamous cell carcinoma or adenoid cystic carcinoma.3,18,19,42,43 Historically, malignant tumors were much more common in men than women and squamous cell histology was most prevalent. More recently, however, the incidence of tracheal squamous cell carcinomas has declined, while the incidence of adenoid cystic carcinomas has increased. At the same time, the distribution between genders has nearly equalized.1,4,19 The most frequent presenting symptoms are cough (72%), dyspnea (66%), hemoptysis (39%), stridor (39%), and hoarseness (31%), but the presence or absence of these symptoms has no prognostic implication.44
Squamous Cell Carcinoma
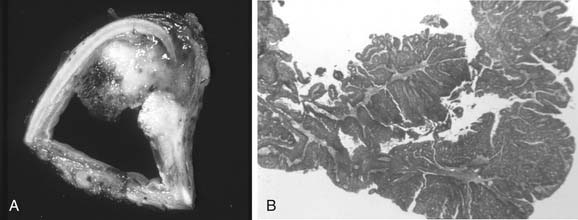
Until the 1980s, squamous cell carcinomas were the most common primary malignancy of the trachea, but the incidence of this disease has decreased significantly since then (Fig. 114-1).4,44,45 Men are twice as likely to develop this cancer than are women, and the mean age of patients is 60 years.46 A history of cigarette smoking is found in 90%, and synchronous or metachronous respiratory tract malignancies occur in 30% of these patients.46 These tumors demonstrate exophytic growth with ulceration. Sputum cytology results are positive for cancer in nearly one half of patients. The lateral tracheal wall is often involved by these tumors, although when a squamous cell carcinoma involving the membranous tracheal wall is found, invasion from a primary esophageal tumor should always be considered.
Squamous cell cancers of the trachea are lethal, biologically aggressive tumors that grow rapidly and metastasize early. In a large series of squamous cell and adenoid cystic tracheal tumors by Gaissert and colleagues,42,46 33% of patients had tumors that were unresectable at presentation, and this percentage declined over the past 4 decades. Of those with resected squamous cell tumors, 27% had positive mediastinal lymph nodes. Hospital mortality averaged 7% in the series reviewing a 4-decade experience but improved to 3% in the last decade. Median survival was 38 months for patients with resectable tumors but only 8.8 months for unresectable patients. The 5-year survival rates for these two groups were 39.1% and 7.3%, respectively. The presence of invasive carcinoma at the resection margins correlated with poorer prognosis, although the number of patients in this category was too small to reach statistical significance. Mediastinal lymph node involvement had the strongest prognostic implication following surgical resection in this review, but this finding was consistent among other case series. The 5-year survival for patients with negative mediastinal lymph nodes was 48.4% compared with 12.5% of patients with positive lymph nodes.46 Conversely, a large French review found that the presence of positive lymph nodes had no impact on long-term survival, which had an overall 5-year survival rate of 47%.19
Radiation therapy has been used to palliate patients with unresectable squamous cell carcinomas. In a series by Grillo and Mathisen,17 the median survival of patients with tracheal squamous cell carcinomas treated with radiation alone was 10 months, which was greater than survival among untreated patients. In addition, better survival has been observed in patients treated with surgery and radiation compared with either surgery alone or radiation with or without chemotherapy.4 Similarly, Jeremic and colleagues47 reported a series of tracheal squamous cell carcinomas treated with 60 Gy of radiation. The median survival was 24 months, and 5-year survival was 27%. The presence of metastasis in the mediastinal lymph nodes portended a poor survival.47
Adenoid Cystic Carcinoma
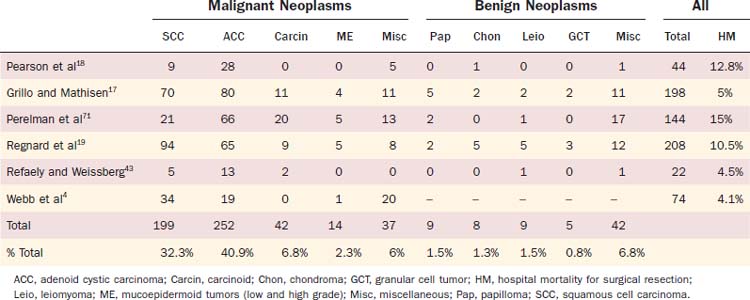
Adenoid cystic carcinomas, previously referred to as cylindromas, were first described by Billroth in 1859. These tumors can be seen in all age groups with equal distribution between men and women, but the peak incidence occurs in the fourth or fifth decade. They usually arise in the trachea or main bronchi and thus are frequently symptomatic, although many patients experience symptoms for more than a year before the diagnosis is secured. There is no association with sex, race, or cigarette smoking. Adenoid cystic carcinomas are nonencapsulated, slow-growing, low-grade malignant tumors that originate from the epithelium of the glands lining the mucosa of the respiratory tract. Adenoid cystic carcinomas are known to push mediastinal structures aside rather than invade them (Fig. 114-2). Despite growing slowly, these tumors unfortunately show a predilection for perineural, submucosal, and distant metastatic spread. Surgical resection margins are frequently microscopically positive well away from grossly apparent tumor. In these circumstances, adjuvant radiation therapy is used and commonly associated with long-term survival.46,48 Lymphatic metastases are relatively uncommon (13% to 19%), but hematogenous metastases develop in 44%.
Treatment consists of surgical resection with preoperative radiation frequently given for large tumors. Surgical resection is possible in only 75% of patients at the time of their initial presentation due to extensive radial and longitudinal growth.46 The indolent but persistent nature of adenoid cystic carcinomas conveys long-term survival rates of 33% and 10% at 5 and 10 years, respectively, in unresectable patients.46 Survival is improved following surgery to 54% and 33% at 5 and 10 years.46 Although controversial because of the prolonged survival seen even with unresectable disease, pulmonary metastases are not considered a contraindication to tracheal resection in otherwise operable patients. Mean survival in patients with pulmonary metastases was only 37 months.19,48 Unfortunately, late local recurrences often occur, leading to death in patients thought to be completely resected. Overall mean and median survival rates as high as 85 and 60 months, respectively, have been reported.48
Adenoid cystic carcinomas are responsive to radiation therapy.42 Radiation therapy usually commences approximately 4 weeks after surgical resection. Before initiating radiation, bronchoscopy is performed to assess adequacy of healing. Regnard and colleagues19 reported adjuvant radiotherapy significantly improved survival in patients with an incomplete resection (positive margins).
Other Malignant Primary Tracheal Tumors
Besides adenoid cystic carcinoma and squamous cell carcinomas, a variety of other malignant tracheal tumors have been reported. All combined, these tumors represent approximately 25% of primary tracheal tumors.3,18,19 In a recent large, single-institution review of all primary tracheal lesions, 90 of 357 patients had histology that was neither squamous cell nor adenoid cystic carcinoma.3 Of this diverse group, carcinoids and mucoepidermoid carcinomas are the most frequent. Less commonly seen malignant primary tracheal tumors are seen in Table 114-1. Survival at 10 years was 94% for benign tumors. Long-term survival is also frequently observed in patients with uncommon malignant tracheal tumors following resection. Gaissert and colleagues3 reported a 10-year survival rate of 83% for carcinoid tumors and 5-year survival of 60% for bronchogenic tumors, 100% for mucoepidermoid tumors, and 78% for sarcomas.
Carcinoid Tumors
Carcinoid tumors develop from the argyrophilic Kulchitsky’s cells that are present in the airway mucosa. Carcinoids are considered part of a spectrum of neuroendocrine tumors that include small cell lung cancers. These tumors have been subcategorized as neuroendocrine grade I (typical carcinoid), grade II (atypical carcinoid), and grade III (small or large cell undifferentiated neuroendocrine carcinoma).49 Typical carcinoids, which occur 10 times more frequently than atypical carcinoids, grow slowly and infrequently metastasize.3 Atypical carcinoids demonstrate aggressive biologic behavior and are characterized by malignant histologic features including nuclear abnormalities, mitotic activity, and necrosis. The majority of patients with atypical carcinoids have lymph node or distant metastases at the time of diagnosis. Small cell carcinoma of the trachea is rare and usually unresectable when diagnosed.
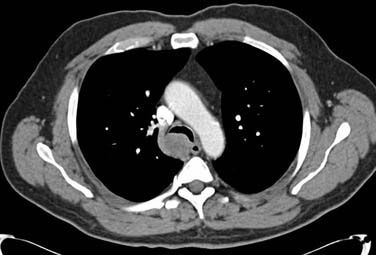
The diagnosis of carcinoid tumors depends on histologic confirmation that can often be obtained by bronchoscopic biopsy. These tumors tend to bleed profusely, so biopsy in the operating room using a rigid bronchoscope may be preferred. The typical bronchoscopic appearance has been described as “mulberry-like” (Fig. 114-3).
The treatment of choice for tracheal carcinoids without mediastinal lymph node involvement is surgical resection, which has been associated with an 83% 10-year survival rate.4 Aggressive atypical carcinoids are often treated as small cell lung cancers, and responses to chemotherapy and radiation therapy have been seen. Small cell carcinoma of the trachea has an extremely poor prognosis despite management with combined chemotherapy and radiation.
Mucoepidermoid Carcinoma
At bronchoscopy, mucoepidermoid carcinomas of the trachea appear as pink, polypoid masses that can be confused with a carcinoid tumor. Mucoepidermoid carcinomas are derived from minor salivary gland tissue of the proximal tracheobronchial tree. On the basis of mitotic activity, level of necrosis, and nuclear pleomorphism, these tumors are classified as low or high grade. Low-grade tumors behave in a benign fashion, whereas high-grade mucoepidermoid carcinomas progress rapidly. Bronchoscopic biopsy provides the diagnosis and optimal treatment requires surgical resection. Long-term survival is common (100% 5-year survival) when these tumors are completely resected, but unresectable tumors have a poor prognosis with no patients surviving 5 years.4 Adenosquamous carcinomas are pathologically similar to mucoepidermoid carcinomas and have an aggressive course.