Chapter 46 Diabetic Retinopathy
Genetics and Etiologic Mechanisms
![]() For additional online content visit http://www.expertconsult.com
For additional online content visit http://www.expertconsult.com
Introduction
Diabetic retinopathy is the leading cause of blindness among individuals between 25 and 74 years of age in the industrialized world. It affects three out of four diabetic patients after 15 years of disease duration. Chronic hyperglycemia is the primary factor leading to the development of diabetic retinopathy and other complications of the disease. The importance of long-term glycemic control has been conclusively established in the landmark clinical trials including the Diabetes Control and Complications Trial (DCCT),1 and the UK Prospective Diabetes Study (UKPDS).2,3 However, the mechanisms by which elevated blood sugar levels lead to the development of diabetic retinopathy and the anatomic changes visible histopathologically remain to be fully elucidated. This chapter will review the biochemical and molecular pathways believed to be responsible for the development of diabetic retinopathy and will highlight recent developments in genetics that provide insight into the influential role of genetic susceptibility.
Anatomic lesions
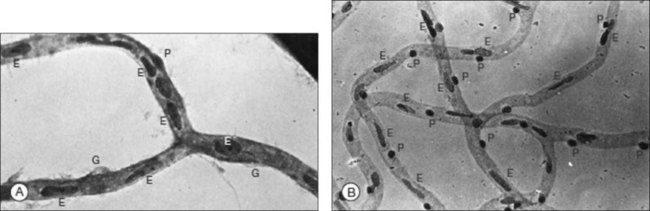
Loss of pericytes
Loss of pericytes is one of the earliest and most specific signs of diabetic retinopathy. This finding was first described by Cogan, Kuwabara and coworkers after examining trypsin-digested retinal vasculature flat mounts from diabetic human subjects.4–6 Since their initial report, their findings have been confirmed by various investigators.2,7,8 In humans and canines, trypsin digestion of retinal vasculature flat mounts reveals the loss of pericytes as evidenced by the development of pericyte ghosts, empty aneurysmal spaces bulging from the capillary walls that lack the darkly staining nucleus of a viable pericyte (Fig. 46.1A). Pericytes are normally identifiable in these spaces by their nuclei which stain darkly and are spaced regularly along the capillary wall, producing the appearance of “bumps on a log” (Fig. 46.1B).
Pericytes are contractile cells that play an important role in microvascular autoregulation.9 Loss of pericytes leads to alterations of vascular intercellular contacts and impairment of the inner blood–retina barrier. These effects result in the venous dilation and beading that is visible clinically. Loss of intercellular contacts also appears to promote endothelial cell proliferation resulting in the development of microaneurysms.10 Loss of pericytes appears to be especially significant in the development of diabetic retinopathy, although pericyte loss has also been reported in diabetic peripheral neuropathy leading to neuronal ischemia.11 The mechanism by which hyperglycemia leads to pericyte degeneration remains largely unknown. The two leading hypotheses implicate the aldose reductase pathway and platelet-derived growth factor-beta (PDGF-β).
Akagi et al. reported the localization of the enzyme aldose reductase in retinal capillary pericytes but not in endothelial cells in human specimens using immunohistochemistry techniques.12 These findings would be consistent with the specific loss of capillary pericytes observed in diabetic retinopathy and other microvascular complications of diabetes.13 Two other groups, however, were unable to identify the presence of aldose reductase in rodent and canine retinal capillaries using immunohistochemistry techniques.14,15 In addition, a third group reported the presence of aldose reductase activity in cultured bovine retinal capillary pericytes as well as retinal capillary endothelial cells. They also found aldose reductase activity in cultured monkey retinal pericytes.16 These conflicting reports probably result from species-specific differences in aldose reductase expression and highlight the need to use caution when interpreting animal models of disease.
PDGF-β has been found to be critical in the recruitment of pericytes in the vasculature of various tissues and organs.17 It is well documented that endothelial cells express PDGF-β,18–21 and in vitro pericytes are known to express PDGF-β receptors and respond to PDGF-β.22,23 Lindahl et al. reported that in the PDGF-β-deficient mouse model, pericytes fail to develop in developing capillaries during angiogenesis.24 Subsequent studies using PDGF-β- and PDGF receptor-β (PDGFR-β)-deficient mice found that while PDGF-β/PDGFR-β-independent induction of pericyte precursors may occur, the expansion of pericytes is dependent on an intact PDGF-β/PDGFR-β paracrine signaling pathway.25 Ablation of either PDGF-β or PDGFR-β led to identical phenotypes in these mice.26 These studies suggest that endothelial cell-derived PDGF-β promotes the co-migration of PDGFR-β-expressing pericytes along sprouting new vessels, and the interruption of the PDGF-β/PDGFR-β results in the observed deficiency of pericytes in capillaries. Since PDGF-β has been found to be critical in the recruitment of pericytes during angiogenesis, it has been suggested that PDGF-β may play an important role in maintaining pericyte viability in mature vasculature, although no studies have confirmed this hypothesis.
Capillary basement membrane thickening
Thickening of capillary basement membranes is a well-documented lesion of diabetic retinopathy, visible on electron microscopy. Additional electron microscopic findings include deposition of fibrillar collagen and “Swiss cheese” vacuolization of the otherwise homogenous pattern of basement membrane collagen. The biochemical mechanism leading to basement membrane thickening remains unknown but studies suggest a role for the aldose reductase and the sorbitol pathway.27–30 Nondiabetic rats fed a galactose-rich diet for prolonged periods of time develop retinal capillary basement membrane thickening, fibrillar collagen deposition, and Swiss cheese vacuolization. By contrast, rats fed a control diet or a galactose-rich diet along with the aldose reductase inhibitor sorbinil do not develop basement membrane thickening.27,30 However, the observation that basement membrane thickening of renal glomeruli in diabetic and galactosemic rats is not inhibited by aldose reductase inhibitors casts doubt that the aldose reductase pathway is the primary pathway and suggests that basement membrane thickening may be a secondary nonspecific response.31
Glycation of basement membrane collagen by enzymatic32 and nonenzymatic processes33 may be another mechanism that plays a role in basement membrane thickening. Besides type IV collagen, the predominant type of collagen present in the basement membrane, other collagen types, and noncollagen macromolecules, such as laminin,34 entactin, heparan sulfate proteoglycan,35,36 and basement membrane-bound growth factors are also present.35 Glycation appears to alter the structure of the basement membrane by changing the chemical composition and relative amounts of these components. For example, Shimomura/Spiro and Spiro/Spiro reported decreased heparan sulfate proteoglycan in renal glomeruli from diabetic human subjects.37,38 Quantitative electron microscopic immunocytochemical studies have found that retinal and renal glomeruli basement membrane thickening in galactosemic rats is associated with a relative increase in the levels of type IV collagen and laminin, while the relative levels of heparan sulfate proteoglycan remain unchanged.29,31
Microaneurysms
Although pericyte loss is the earliest sign of diabetic retinopathy, it is only observable histologically. The earliest clinically visible sign of diabetic retinopathy is the microaneurysm.39 Microaneurysms appear as grape-like or spindle-shaped dilations of retinal capillaries on light microscopy.4 They can be either hypercellular or acellular. By ophthalmoscopic examination, microaneurysms appear as tiny, intraretinal red dots located in the inner retina. By fluorescein angiography, they appear as punctate hyperfluorescent dots with variable amounts of fluorescein leakage.
Pericyte cell death and loss of vascular intercellular contacts may lead to endothelial cell proliferation and microaneurysm development.24,40,41 Pericytes appear to exert an antiproliferative effect and pericyte loss may explain the development of hypercellular microaneurysms. However, this mechanism does not account for acellular microaneurysms. Acellular microaneurysms may develop from hypercellular microaneurysms that become acellular from endothelial cell and pericyte apoptosis.42
Pericyte loss may also result in weakening of the capillary wall, promoting the development of microaneurysms at the structural weak points. Pericytes contain myofibrils with contractile properties and may act as smooth muscle cells of larger vessels, exerting tone to the vessel wall in order to counteract the transmural pressure. Loss of pericyte tone may result in focal dilation of the vessel wall leading to the development of a microaneurysm. However, the transmural pressure of the capillary bed is low relative to the arterial circulation, and retinal capillary microaneurysms can develop in other diseases in which pericyte loss is not observed.43,44
Capillary acellularity
Complete loss of the cellular elements of the retinal capillary network can be seen as a more advanced microvascular lesion in diabetic retinopathy and other microvascular retinopathies. In clinicopathological correlations with fluorescein angiograms performed shortly before the eye was enucleated for therapeutic purposes or because the patient died and the eye was removed during autopsy, the retinal vascular digests of the enucleated eye showed that acellular capillaries are nonfunctional since they appeared as regions of nonperfusion on angiography.45 The mechanism by which capillaries become acellular is unknown and can be seen in diabetes, other retinal microvascular diseases, or in experimental diabetes or galactosemia in animal models. Since capillary acellularity is not unique to diabetes, a variety of pathogenic mechanisms may result in this nonspecific finding.
Breakdown of blood–retina barrier
Breakdown of the blood–retina barrier is an important pathophysiologic feature of diabetic retinopathy that leads to the development of macular edema, the leading cause of vision loss in diabetic patients. One mechanism by which the function of this barrier becomes altered involves opening of the tight junctions between vascular endothelial cell processes.46,47 These tight junctions, also known as zonula occludens, appear as a pentalaminar structure on electron microscopy, consisting of two outer and one central electron-dense layer sandwiching two electron-lucent layers, giving the appearance of two “fused” plasma membranes. Using tracers such as lanthanum chloride or horseradish peroxidase, electron microscopy can be used to demonstrate that these molecules cannot pass in the presence of an intact tight junction. However, when the tight junctions are open, they become permeable to these tracer molecules.47 Several important proteins are involved with the formation and function of tight junctions, with ZO-1 (zonula occludens) and occludin being the best characterized. In the presence of histamine, the expression of ZO-1 in cultured retinal endothelial cells is reduced in a dose-dependent manner.48 Culturing in astrocyte-conditioned medium increases expression of ZO-1 while high glucose decreases expression of ZO-1.49 These in vitro results have also been supported by in vivo studies. Reduced expression and anatomic distribution of occludin was found in experimental diabetes.50 Likewise, in experimentally diabetic rats and in diabetic humans, antihistamines reduce leakage of fluorescein into the vitreous.51,52
Vascular endothelial growth factor (VEGF) has been found to be an important mediator leading to the breakdown of the inner blood–retina barrier. Before the discovery of its well-documented role in promoting neovascularization, VEGF was found to increase the permeability of vessels, leading to its alternative name “vascular permeability factor”.53 The mechanism by which VEGF leads to the breakdown of the inner blood–retina barrier appears to involve alteration of endothelial cell tight junctions. Intravitreal injection of VEGF in rats increased the production of the free radical nitric oxide and led to phosphorylation of ZO-154,55
Another important factor promoting retinal vascular permeability involves the kallikrein–kinin system. Proteonomic studies of the vitreous from patients with advanced diabetic retinopathy have identified components of the kallikrein kinin system, including plasma kallikrein, factor XII, and kininogen.56,57 In rodent models, activation of plasma kallikrein in the vitreous has been found to increase retinal vascular permeability.58 Likewise, inhibition of the kallikrein kinin system reduces retinal vascular leakage caused by diabetes and hypertension.58,59 The mechanism by which the kallikrein kinin system promotes vascular permeability probably involves bradykinin. Bradykinin, via nitric oxide, induces vasorelaxation of retinal arterioles.60 Intravenous infusion of bradykinin results in dilation of retinal arterioles and venules, an effect that is reduced by indomethacin and the cyclooxygenase-2 selective inhibitor nimesulide.61 Bradykinin is also a neuropeptide with a direct effect on glia and neurons, which can release vasoactive factors that affect blood flow and vascular permeability.62
Biochemical mechanisms in the pathogenesis of diabetic retinopathy
Although diabetic retinopathy does not manifest the classic “rubor, tumor, calor, and dolor” features of an inflammatory disease and lacks a prominent infiltration of inflammatory cells, there are characteristics that imply a chronic low-grade inflammatory component. In the retinas of diabetic rats, increased activation of leukocytes with increased amounts of inflammatory cytokines and adhesion molecules have been observed. The upregulation of these molecules enhances leukocyte adhesion to retinal capillary walls, leading to increased capillary stasis, occlusion, and ultimately hypoxia as seen in diabetic retinopathy.63–65 Clinically, it has been observed that intravitreal corticosteroid injections decrease diabetic macular edema, often with improvement of visual acuity.66,67 The mechanism by which intravitreal corticosteroids reduce macular edema in diabetes and other etiologies, like retinal vein occlusions, is unclear, but the anti-inflammatory effects of corticosteroids support an inflammatory component to the development of at least diabetic macular edema as well as macular edema from retinal vein occlusions. Likewise, there is growing evidence that the kinin–kallikrein system, which plays an important role in the inflammatory cascade by acting on phospholipase and promoting the release of arachidonic acid and the production of prostaglandins, contributes to the development of diabetic macular edema.68
The aldose reductase theory
Elevation of intracellular glucose levels can cause increased activation of the aldose reductase pathway. Also known as the polyol pathway or the sorbitol pathway, the aldose reductase pathway is a series of intracellular reactions that involves the enzymes aldose reductase and sorbitol dehydrogenase.69,70 Aldose reductase uses the reduced form of nicotinamide adenine dinucleotide phosphate (NADPH) as a cofactor to reduce many aldose sugars into their respective sugar alcohols. Glucose is reduced to sorbitol, which is then oxidized into fructose by sorbitol dehydrogenase. However, sorbitol may build up to high intracellular levels because the sorbitol dehydrogenase reaction is slow and the accumulating sorbitol does not easily cross the plasma membrane into the extracellular space. In normoglycemic conditions, the aldose reductase pathway is nonoperative because glucose is a poor substrate for aldose reductase due to its high binding constant (kM). However, in the setting of hyperglycemia as seen with uncontrolled diabetes, the aldose reductase pathway becomes activated once the other enzymatic pathways of glucose metabolism become saturated. Lens epithelium expresses high levels of aldose reductase and accumulation of sorbitol is believed to lead to the development of a cataract in diabetes.71 Osmotic stress has been proposed as the mechanism by which elevated intracellular sorbitol leads to the pathologic changes seen in diabetes.72 However, the levels of sorbitol in vascular cells are in the nanomolar range, which is orders of magnitude less than other glucose metabolites, which have ranges in the micromolar and millimolar range.73
A different mechanism that may account for the role of aldose reductase involves the cellular redox balance. Increases in the utilization of aldose reductase in the hyperglycemic state of diabetes will result in a decline in intracellular NADPH that alters the cellular redox balance. Reduction of intracellular NADPH may also decrease the production of nitric oxide in endothelial cells.74 Similarly, the increased use of sorbitol dehydrogenase can lead to an increase in the NADH/NAD+ ratio that alters the cellular redox balance and may lead to oxidative stress and cellular damage.75
Of interest to animal models of diabetes, galactose is reduced by aldose reductase into galactilol. However, sorbitol dehydrogenase cannot oxidize galactilol, resulting in the rapid intracellular accumulation of galactilol. In the setting of chronic galactosemia, diabetic-like vascular basement membrane changes,27,28 and pericyte loss, development of microaneurysms, and capillary acellularity76 have been reported. When these experiments were repeated and the animals were also treated with aldose reductase inhibitors, it was reported that the development of diabetic-like retinopathy changes was slowed, although most animals developed some degree of changes.30,77–79 While aldose reductase inhibitors were reported to slow or prevent some of the pathologic changes in animal models of diabetes, the aldose reductase inhibitor sorbinil was found not to be effective in humans in the Sorbinil Retinopathy trial.80 The lack of efficacy of aldose reductase inhibitors in human trials may, however, reflect dose-limiting side-effects of the drug that may have precluded it from achieving therapeutic levels in the tissues of interest.
Advanced glycation endproduct (AGE) theory
Accelerated aging by nonenzymatic glycation and crosslinking of proteins has been proposed as a mechanism to explain the complications of diabetes.81 Advanced glycation endproducts (AGEs) is the collective name given to proteins, lipids, and nucleic acids that undergo irreversible modification by reducing sugars or sugar-derived products. The series of chemical reactions that lead to the formation of AGEs is called the Maillard reaction. The Maillard reaction is responsible for the “browning” of tissue seen with aging as well as the “browning” of food during cooking. The initial chemical reaction is known as early glycation and involves reversible nonenzymatic binding of a sugar to amino acid groups on proteins, lipids, or nucleic acids. They form Schiff bases which can undergo rearrangement to form more stable Amadori products. Glycosylated hemoglobin (HbA1c) and fructosamine are well-known examples of Amadori products used clinically as markers of glycemic control. Although they are not AGEs, they can undergo further reactions to eventually lead to the formation of AGEs. Formation of AGEs may directly damage cells by impairing the function of a variety of proteins,82 including both extracellular proteins like collagen83 and intracellular proteins.84,85
The cellular effect of AGEs is also mediated by its binding to receptors, namely receptor for AGE (RAGE). RAGE is a multiligand transmembrane receptor that is part of the immunoglobulin superfamily of proteins.86,87 When bound to AGEs, it initiates a cascade of signal transduction involving at least p21ras, p44/p42 mitogen-activated protein kinase (MAPK), nuclear factor-kappa B (NF-κB), and protein kinase C (PKC).88–93 Activation of these intracellular kinases can subsequently lead to cell dysfunction.94 Other receptors have also been reported to bind to AGEs, such as the macrophage scavenger receptor, P60, P90, and galectin-3.94–96 Aminoguanidine is an inhibitor of AGE formation and has been reported to block the development of many of the microvascular complications of diabetes in animal models.97–100 The effects of aminoguanidine cannot be automatically attributed to the blockade of the AGE pathway, however, since aminoguanidine also has parallel action as an inhibitor of inducible nitric oxide synthase and oxidants.101 Owing to limits secondary to toxicity, clinical trials in humans have been inconclusive; in animal models, however, the use of soluble RAGE to block binding of AGEs to RAGE has been found to prevent many of the effects of hyperglycemia.102
Reactive oxygen intermediates (ROI) theory
One of the oldest theories proposes that chronic hyperglycemia leads to the complications of diabetes by increasing oxidative stress. The usual metabolic pathway of glucose is through glycolysis and the tricarboxylic acid cycle, which takes place in the mitochondria and yields reducing equivalents used to drive the synthesis of adenosine triphosphate via oxidative phosphorylation. However, byproducts of oxidative phosphorylation include free radicals, such as superoxide anion, whose production is increased by high levels of glucose.103 Free radicals can damage mitochondrial DNA104 as well as cellular proteins105 and are also produced by the autoxidation of glucose. Elevated oxidative stress also reduces nitric oxide levels,106,107 promotes leukocyte adhesion to the endothelium and decreases the barrier function of endothelial cells,108 and damages cellular proteins.109 In diabetic mice that overexpress Cu2+/Zn2+ superoxide dismutase, they develop less mesangial expansion compared to wild-type diabetic mice, suggesting that oxidative stress promotes at least some complications of diabetes.110 Oxidative stress can also activate PKC by increasing the formation of diacylglycerol (DAG).111
There is some evidence of increased oxidative stress in diabetic patients. It has been reported that diabetic patients have lower levels of antioxidants, such as vitamin C, vitamin E, and glutathione,112–114 although these results have not been unequivocally reproduced by other researchers.115 However, other markers of oxidative stress, such as oxidized low-density lipoprotein116 and urinary isoprostanes, are elevated in diabetic patients.117 In animal models of diabetes, the use of antioxidants has blocked the development of some of the microvascular complications of diabetes.118–122 One clinical trial reported that high doses of vitamin E (>1000 IU/day) and lipoic acid improved retinal blood flow and creatinine clearance in diabetic patients.123 However, most studies evaluating antioxidants to prevent the complications of diabetes in people have been unsuccessful.124
Protein kinase C (PKC) theory
PKC is a ubiquitous enzyme that appears to promote the development of many of the complications of diabetes without the involvement of the aldose reductase pathway. It has been observed that diabetes and galactosemia can produce elevation of DAG within cells of the retina and aorta in dogs despite treatment with the aldose reductase inhibitor sorbinil.125,126 Activation of PKC occurs through the activation of phospholipase C, which leads to an increase in intracellular Ca2+ and DAG, which in turn results in the activation of PKC.127 Hyperglycemia can result in pathological activation of PKC. Elevated glucose levels result in activation of the glycolytic pathway and lead to increased levels of intracellular glyceraldehyde-3-phosphate. Glyceraldehyde-3-phosphate can promote the de novo synthesis of DAG through glycerol-3-phosphate, which in turn activates PKC.128 Activation of PKC can also be mediated by AGE92 and ROI.111
Elevated levels of DAG and PKC activity has been detected in the tissues of animals with diabetes.129 The pathologic effects of PKC activation that cause vascular damage are mediated through increased vascular permeability,130 disruption of nitric oxide regulation,131,132 increased leukocyte adhesion to vessel walls,133 and changes in blood flow.134 In fact, the effects of PKC on retinal blood flow as measured by fluorescein video angiography, as well as glomerular filtration rate and albumin excretion rate, were improved in a dose-dependent fashion with ruboxistaurin (LY333531), a PKC-β inhibitor.135 VEGF136 and endothelin137 can also activate PKC, which in turn can promote the expression of growth factors like VEGF138 and transforming growth factor-beta (TGF-β).139 PKC activation can influence other signally pathways, such as MAPK or NF-κB.140
PKC inhibition by ruboxistaurin has been reported to block many of the vascular abnormalities in endothelial cells and contractile cells from the retina, arteries, and renal glomeruli.141 In animal models of diabetes, ruboxistaurin protected against or reversed many of the early vascular changes seen with retinopathy, nephropathy, and neuropathy.135,136,142,143 However, a prospective clinical trial of ruboxistaurin did not meet its primary outcome (progression to sight-threatening diabetic macular edema or application of focal/grid photocoagulation for diabetic macular edema) at 30 months although there was a significant reduction of progression to sight-threatening diabetic macular edema when considered alone.144 An open-label extension of the Protein Kinase C Diabetic Retinopathy Study 2 (PKCDRS-2) reported that over a 6-year study period patients with the greatest ruboxistaurin exposure (~5 years) had less sustained moderate vision loss (>15-letter decline) compared to those in the original placebo group (~2 years of ruboxistaurin use).145
Insulin receptors and glucose transporters
In certain types of cells, such as adipocytes and skeletal muscle cells, insulin is required to transport glucose from the extracellular fluid across the plasma membrane into the cytoplasm. This action requires a specific receptor for insulin on the plasma membrane. Although it has been commonly stated that the microvascular complications of diabetes do not occur in tissues in which insulin is required for the transport of glucose into cells, insulin receptors have been reported on the pericytes and endothelial cells of the retinal microvessels.146 There is no evidence, however, that the retinal microvascular insulin receptors are required for glucose transport, although insulin does enhance glycogen synthesis from radiolabeled glucose in retinal microvascular pericytes and endothelial cells and aortic smooth-muscle cells, but not in aortic endothelial cells.146 Insulin in physiologic concentrations (as low as 10 ng/ml) stimulated [3H]-thymidine incorporation into retinal microvascular pericytes and endothelial cells and aortic smooth-muscle cells but not aortic endothelial cells.146 It is noteworthy that, in these experiments, such low concentrations of insulin produced an effect, because unphysiologically high (e.g., 1 mg/ml) concentrations of insulin will stimulate proliferation of many types of cultured cells. However, since microvascular endothelial cells and pericytes do not normally proliferate in the mature retina,147 the importance of these results for normal retinal vascular physiology is unclear. Additionally, these results indicate that there are metabolic differences between microvascular endothelial cells and the endothelial cells of larger vessels, so that translation of results from one type of vascular endothelial cell to another must be done with great caution.
There are at least five different types of facilitated cell membrane glucose transporters, designated GLUT1, GLUT2, GLUT3, GLUT4, and GLUT5, that appear to be most important for the intracellular transport of glucose in tissues like the retina that do not require insulin. Of these, GLUT1 appears to be the most prevalent in the retina,148–150 occurring in microvascular and macrovascular endothelial cells and on RPE cells, as well as in the Müller cells. GLUT2 localization has been reported by immunocytochemistry at the apical ends of the Müller cells of the rat retina, facing the interphotoreceptor matrix,151 while GLUT3 has been reported by similar techniques to be localized to the plexiform layers of the rat152 and human150 retina. An initial report using light microscopic immunocytochemistry in human eyes148 also reported GLUT1 in the nerve fiber layer of the retina and in photoreceptor cell bodies, but GLUT1 was absent from retinal neovascular proliferations in the eyes of diabetic subjects. Subsequently, these investigators used quantitative immunogold electron microscopic immunocytochemistry to examine GLUT1 localization in the eyes of two nondiabetic subjects and three diabetic subjects with little or no retinopathy.149 In approximately half of the retinal microvessels from the diabetic subjects there was no quantitative difference in GLUT1 immunoreactivity by comparison with microvessels from the two normal individuals. However, in the other half of the retinal microvessels studied from diabetic individuals, these investigators reported an increase of GLUT1 immunoreactivity of about 18-fold over normal on the luminal plasma membranes. If these findings can be confirmed in a much larger number of eyes, such upregulation could be a mechanism that initiates glucose-mediated cellular damage by permitting a much greater influx of glucose into cells.
Whether galactose also enters cells by one or more of these facilitated glucose transporters has not been directly tested. However, the fact that rats fed a 50% galactose diet – which also contains the normal amount of glucose – double their food intake by comparison with normal rats, but nevertheless gain weight at only 60–70% of the rate of the normal animals,153 suggests that the excessive amount of galactose competes with glucose for the transport sites, thereby limiting the entry of glucose into cells and diminishing glucose-requiring cellular energy metabolism. However, galactose can participate in other cellular pathways along with glucose, including protein glycation/advanced glycation endproduct formation and synthesis of DAG to activate PKC.126 Whether glucose or galactose can upregulate the mRNAs governing synthesis of any of the GLUT proteins in a fashion such that this upregulation persists long after cessation of the hyperglycemic or galactosemic state has not yet been explored.
Genetic factors in the pathogenesis of diabetic retinopathy
There is good evidence that diabetic retinopathy has a genetic predisposition.154 Diabetic Retinopathy Study (DRS) data indicated that only 50% of nonproliferative diabetic retinopathy (NPDR) patients developed PDR, and many diabetic patients never developed diabetic retinopathy (DR). Twin studies of DR also lend support to this notion.155 Some investigators have also suggested that aldose reductase gene polymorphisms may be associated with risk of DR.156–162 There appears to be considerable value in further investigation of genetic factors related to the pathogenesis of the more severe forms of diabetic retinopathy: severe nonproliferative and proliferative retinopathy, as well as macular edema. Nearly all individuals with type 1 diabetes, and most with type 2 disease, will demonstrate some of the lesions of early retinopathy with sufficient disease duration, but only 50% or less will develop proliferative disease.163,164 Like clinically evident diabetic nephropathy, which similarly affects fewer than 50% of all diabetic subjects regardless of the duration of their diabetes, this suggests genetic factors, in addition to chronic hyperglycemia, are likely involved in the development of these severe forms of retinopathy.
Several studies have explored the relationship between human leukocyte antigen (HLA) antigens, expressed on cell surfaces (and customarily tested with leukocytes withdrawn by venipuncture), and the presence, or severity, of diabetic retinopathy. Rand et al.,165 used a case–control design and found a strong association (relative risk, 3.74) between proliferative retinopathy and the presence of HLA-DR phenotypes 4/0, 3/0, and X/X (neither 3 nor 4). Subjects who had HLA-DR phenotypes 3/4, 3/X, and 4/X had no increased risk of proliferative retinopathy as compared with “control” diabetic subjects, matched for age, sex, and diabetes duration, but without retinopathy. This question was also investigated in a group of 425 subjects with insulin-dependent diabetes who were randomly selected from a much larger population-based study.166 After adjustments were made for duration of diabetes, glycemic control, hypertension, and nephropathy, these authors also found a significantly increased risk of proliferative diabetic retinopathy in subjects with the HLA DR4+ DR3 phenotype.
More recent studies have investigated other aspects of the genetics of diabetic retinopathy. Of particular note is a report from the DCCT research group,167 which examined familial clustering of severe diabetic retinopathy (Early Treatment Diabetic Retinopathy Study (ETDRS) score >47, i.e., severe preproliferative disease) among families of DCCT subjects with multiple diabetic members. Significant associations were found when the correlation of retinopathy severity among family members was investigated in several different ways. However, a less strong familial clustering of diabetic nephropathy was found. This is surprising since evidence from other studies has demonstrated considerable familial clustering of diabetic nephropathy, a complication of diabetes that is now considered to have a strong genetic component.168–171 With increased sophistication in molecular biology, a number of investigators have examined several genetic loci for abnormalities that might be related to a hereditary susceptibility to complications of diabetes. Although these studies are not definitive, one of these claimed that a mutation in the aldose reductase gene conferred increased susceptibility to early-onset diabetic retinopathy in patients with type 2 diabetes.172
Although many genes and proteins of vascular growth have been studied in association with PDR, few if any definitive predisposing genes for PDR have been identified. Below is a brief summary of several studies performed on candidate genes. One of the best known and most well-studied genes is the VEGF gene.173–175 VEGF refers to 2 families of proteins created by alternate splicing of exon 8 in the VEGF gene176 and is an important mediator of ischemia-induced vascularization and neovascularization. Current research of VEGF has focused on the role that certain single nucleotide polymorphisms (SNP) may play in contributing either a risk or protective effect in patients.176–178 Recently, three SNPs in the promoter and 5’UTR regions of the gene (C(-7)T, C(-634)G, T(-1498), and G(-1190)A) were studied for frequency differences between patients with and without PDR.177 These studies have looked at different genetic populations, focusing on Japanese and Indian patients. The results of these genetic studies are at times confusing, as a disease-associated SNP in one population may not confer a risk in another population. For example, in the Japanese population, the CC genotype at the C(-634)G region was significantly associated with PDR, whereas CG genotype at the same site was found to be risk-associated in the Indian population. In addition, disease duration, age, and sex must also be taken into account.
Ramprasad and coworkers in 2007 described work evaluating the role of SNPs within the receptor for advanced glycation end products (RAGE) in PDR.179 At least 20 different polymorphisms have been studied within the RAGE gene. Interaction between the receptor RAGE and its associated glycation ligands plays a role in initiating a proinflammatory cascade, and this interaction has been studied in many disorders of chronic inflammation, including peripheral vascular disease and PDR. Research in these SNPs has shown disease association with the NPDR disease phenotype, yet this association needs to be replicated in other independent cohorts. More recently, Balasubbu et al.180 analyzed the association of nine candidate genes (RAGE, PEDF, AKR1B1, EPO, HTRA1, ICAM, HFE, CFH, and ARMS2) but only found a significant association with diabetic retinopathy in one locus (rs2070600 in RAGE → reduces risk).
Proliferative diabetic retinopathy (PDR) and endstage renal disease (ESRD) are two of the most common and severe microvascular complications of diabetes. There is a high concordance in the development of PDR and ESRD in diabetic patients, as well as strong familial aggregation of these complications, suggesting a common underlying genetic mechanism. However, the precise gene(s) and genetic variant(s) involved remain largely unknown. Erythropoietin (EPO) is a potent angiogenic factor observed in the diabetic human and mouse eye. By a combination of case–control association and functional studies, Tong et al.181 demonstrated that the T allele of SNP rs1617640 in the promoter of the EPO gene is significantly associated with PDR and ESRD. The study was performed in three European-American cohorts (Utah: P = 1.91×10-3; GoKinD: P = 2.66×10-8; Boston: P = 2.1×10-2). The EPO concentration in human vitreous was 7.5-fold higher in normal subjects with the TT risk genotype than in those with the GG genotype. Computational analysis suggests that the risk allele (T) of rs1617640 creates a matrix match with the EVI1/MEL1 or AP1 binding site, accounting for an observed 25-fold enhancement of luciferase reporter expression as compared to the G allele. These results suggest that rs1617640 in the EPO promoter is significantly associated with PDR and ESRD and suggest EPO as a potential pathway mediating severe diabetic microvascular complications.181
Genome-wide association studies (GWAS) of diabetic retinopathy have also been pursued. In a Taiwanese population, Huang et al. found genetic associations for susceptibility for development of diabetic retinopathy in five loci, including PLXDC2 and ARHGAP22, which are genes implicated in endothelial cell proliferation and capillary permeability.182 In addition, one group recently published their preliminary results of a GWAS on a small (286 total) Mexican-American diabetic cohort. They identified 32 SNPs (in 11 regions) with a nominal association for severe diabetic retinopathy, though none was located in traditional candidate genes for diabetes or diabetic retinopathy.183 These findings have yet to be replicated, and as noted above, the cohort was small. In two large type I diabetes cohorts, several novel genetic loci associated with sight-threatening complications due to diabetic retinopathy, were identified, including rs10521145 in the intron of CCDC101, a histone acetyltransferase.184
Catalyzed by the rapid advances in whole-genome SNP genotyping technologies and the construction of reference haplotype maps, genetic variants associated with ~300 traits have successfully been mapped by GWAS in the past few years, using a P-value threshold of 10-5. GWAS is based on the hypothesis that common diseases are mainly caused by common genetic variants in the population (Common Disease Common Variants, CD-CV), each having relatively weak effects.185 This assumption appears to be generally true for many genetic traits such as age-related macular degeneration.186–188 Another hypothesis is Common Disease Rare Variants (CD-RV), or that common diseases are due to the presence of many rare variants in the same genes or pathways, each having relative strong effects. There is mounting evidence suggesting that CD-RV is also true for many diseases.189–192 In diseases such as cancer, coronary atherosclerosis, and Parkinson disease, nonsynonymous variants in the disease genes were found more frequently in the disease samples compared to the controls. In addition, a higher proportion of these are predicted to be damaging.189,193,194 For example, Cohen et al. sequenced genes in individuals at risk for coronary disease.189 They found that 16% of the individuals in the disease group had novel nonsynonymous variants in candidate genes, in contrast to only 2% for individuals not at risk. Causal variants under the CD-RV hypothesis are difficult to detect in GWAS, because they are in very weak linkage disequilibrium with the tagging SNPs included in genotyping assays. Deep resequencing in the case and control population is required to uncover such variants.195 Due to the high cost of large-scale resequencing, only a very limited number of candidate genes have been screened.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree