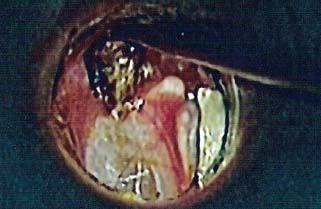
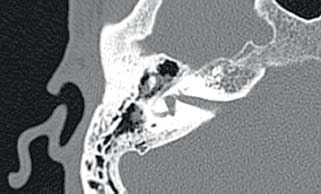
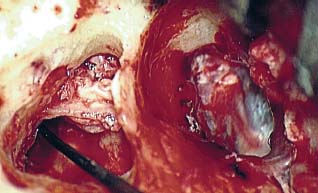
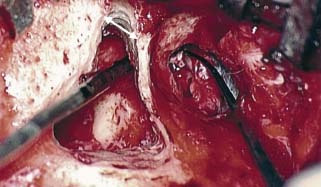
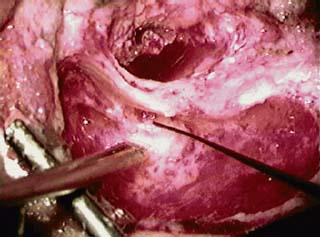
8 Controversies in Cholesteatoma Surgery There are a few areas in cholesteatoma management that remain controversial, where experienced surgeons continue to hold fundamental differences of opinion about treatment options. The canal wall up versus canal wall down debate is chief among these and has endured despite decades of accumulated clinical evidence and shifting viewpoints. The question of second-stage (or “second-look”) surgery has also persisted, and yet uncertainty remains about what to do if recurrent disease is found. Facial nerve monitoring has found a place in the surgical management of cholesteatoma and chronic ear disease, but despite its widespread use, there continues to be disagreement about its value and about its proper method of implementation. Endoscopes, which have gained broad acceptance in other areas of otolaryngology and skull base surgery, have not replaced the microscope in the treatment of ear disease and yet have found some suitable applications in the management of cholesteatoma and in the detection of recurrence. The adequacy of follow-up is a factor that continues to be vital in controlling this disease, because of the persistently high rate of recurrence. These areas of controversy are discussed in detail here. The ideal treatment for cholesteatoma is a single-stage operation with a very low recurrence rate and no need for bowl maintenance. Such a procedure still does not exist, however, and so debate continues about the relative merits of canal wall up (CWU) and canal wall down (CWD) procedures. The controversy between CWU versus CWD surgery reached its pinnacle in the 1960s and ′70s, when vocal and colorful advocates of each position argued, with a certain amount of zealotry, for one approach at the exclusion of the other. As with most trends in medicine, a more balanced view took hold over time, with the recognition that each approach was appropriate in certain situations. The CWD technique (also called open cavity or modified radical mastoidectomy) has a low rate of residual disease that can be detected and treated in the office, and a (theoretically) zero rate of recurrent disease, albeit at the expense of creating a mastoid cavity. CWD therefore results in a “safe” ear. Published studies have shown CWD to have a lower recurrence rate than CWU, with a rate varying from 0% to less than 10% in studies with at least 10 years follow-up. However, the cavity is an alteration of the normal anatomy, a reservoir for accumulation of cerumen and epithelial debris, and a potential area for mucositis and persistent postoperative drainage. Because of the need for periodic cleaning and water precautions, the CWD procedure carries a penalty, especially for children. CWU avoids the problems of an open cavity, but at the cost of a higher rate of recurrent/residual disease. The CWU technique, also called closed cavity, combined approach, and intact canal wall, preserves the posterior bony canal wall and results in a normal ear canal without a cavity. The advantages of CWU are preservation of normal anatomy, more rapid healing, and (as is often claimed), better hearing. The disadvantage of CWU is the possibility of residual cholesteatoma (disease left behind by the surgeon) behind an intact wall, and the risk of recurrence (re-formation of cholesteatoma from a new retraction pocket), usually in the attic beneath the bony scutum. The incidence of residual disease has led many advocates of CWU to recommend a second-stage operation, usually 6 to 18 months after the initial operation, to examine for and treat such a possibility. Except in the eyes of the most dogmatic surgeons, each technique has a proper role in the management of cholesteatoma. The determining factors are the size of the mastoid, the extent of the disease, the presence of a complication (such as labyrinthine fistula), and the reliability of the patient for follow-up. Patients with sclerotic mastoids can be safely and easily treated with CWD, which results in a compact cavity with little risk of recurrence or postoperative drainage. Patients with large, extensively pneumatized mastoids are better treated with CWU when their disease permits, because postoperative cavity maintenance could be problematic. Extensive cholesteatomas are better treated with CWD because of their greater likelihood for recurrence; small, well-localized cholesteatomas can be adequately managed with CWU. Complications are usually better managed by CWD because of the overriding need to create a safe ear. Recurrences too are usually managed with CWD. Unreliable or migratory patients are better treated with CWD. Fig. 8.1 Otomicroscopic view of right ear, showing probing of attic cholesteatoma. Preoperative computed tomographic (CT) scanning is helpful for surgical planning. CT reveals the size of the mastoid, the extent of the disease, and the presence of complications, including labyrinthine fistula, facial nerve or tegmen or sigmoid sinus dehiscence, or disease invasion into difficult areas. Although CT is not mandatory, in most cases it is nice to have. In many, if not most, cases, the decision to leave the canal wall or take the canal wall down can be made at surgery. Patients are usually consented (generically) for tympanomastoidectomy and are told that the procedure may result in a cavity and may require a second operation, depending on the surgical findings. In this “flexible” or “eclectic” approach, the mastoid is approached though a postauricular incision, and a CWU mastoidectomy is performed first. Once the extent of the disease and the difficulty of dissection are determined, the surgeon can decide whether or not to take the canal wall down. Fig. 8.3 Canal wall up procedure, dissecting a cholesteatoma sac from the mastoid antrum. In practice, the two areas that might be most problematic for a CWU approach are the sinus tympani and the anterior epitympanic space. The following case illustrates this approach in practice. A 40-year-old woman complained of intermittent drainage and hearing loss in her right ear. Examination revealed a dry crust over the pars flaccida; gentle debridement revealed a deep retraction pocket with squamous debris and scutum erosion (Fig. 8.1). An audiogram revealed a 40 dB conductive hearing loss. CT revealed soft tissue in the attic and antrum (Fig. 8.2). At surgery, the lesion was first explored through the meatus. By probing the attic retraction pocket, it was seen that the sac extended back to the epitympanum. A CWU mastoidectomy was created with transmastoid atticotomy and facial recess (Fig. 8.3). The sac was incised in the antrum, its contents were evacuated, and it was delivered forward toward the middle ear. The incus was removed, and the malleus head was cut with a nipper. There was found to be more extensive involvement than indicated by the CT, with disease extending toward the anterior epitympanic space (Fig. 8.4). At this juncture, the surgeon has to decide whether the cholesteatoma can be completely removed via a CWU approach, or whether the canal wall should be taken down. In this case, exposure of the anterior epitympanic space was compromised by a low-lying tegmen—the posterior bony canal wall could not be thinned any further without perforating it, so it was decided to take the canal wall down (Fig. 8.5) (another option in this case would be a transcanal atticotomy, removing the scutum to gain anterior exposure, and reconstructing it later with cartilage). Choosing between CWU or CWD is often not a black and white decision, as this case illustrates. If CWU is selected, the patient may need a second-stage (second-look) operation to search for residual disease, or may risk developing a recurrent cholesteatoma through the scutum graft. If CWD is performed, the risk of recurrence will be lower but a cavity will result. Several techniques of canal wall reconstruction have been proposed to mitigate the cavity factor. Reconstruction of the canal wall in mastoid surgery is not a new concept. It may have been introduced by Mosher1 and was later utilized by Wullstein2 and others. Different techniques and materials have since been used in an effort to decrease cavity size and prevent recurrent disease. The musculocutaneous flap described by Silvola and Palva,3 using temporalis muscle and fibrous tissue pedicled to the conchomeatal skin, is a widely popular means of reducing cavity size but often retracts and does not completely eliminate the mastoid bowl (the technique is described in Chapter 5). Bone pate, which is readily available in the surgical field, is commonly used for cavity obliteration, but the material may resorb or become infected. It might never completely solidify, or it may contain squamous elements that can lead to recurrence.4 Mercke5 introduced a technique of mastoid reconstruction reimplanting the posterior canal wall and obliterating the cavity with bone chips. This technique resulted in no recurrent cholesteatoma and reduced rate of residual, dry ear, and intact tympanic membrane (TM), and it improved upon the results obtained with CWU and CWD. Gantz et al,6 in 2005, published a series of 127 patients who underwent a similar procedure, with a recurrence rate of 1.5% at 4 years mean follow-up. This technique improved on the recurrence rate when compared with CWU, but wound infections were relatively common, requiring inpatient intravenous (IV) antibiotics. In addition, the procedure did not eliminate the need for a second-look operation, and the ossicular chain was routinely sacrificed at the first operation, with ossicular reconstruction delayed until the second stage. McElveen and Chung7 introduced a “reversible canal wall down” technique, in which the bony canal wall is removed then refixated using bone cement. Although only five cases were described, the technique appears to provide good surgical exposure but results in a CWU-type cavity with the same potential for recurrence. “Soft wall” reconstruction has also been attempted, first by Smith et al,8 and later by Takahashi et al,9 in which a portion of the posterior canal wall is removed for surgical exposure and later replaced by temporalis fascia and canal skin. Recurrence/residual rates were similar to those for CWD, but retraction of the canal skin into the mastoid bowl occurred in nearly 50% of cases. Dornhoffer10 advocated reconstruction of the canal wall with cartilage from the cimbum concha, and reported an 18% recurrence rate and excellent hearing results with this technique. Although these results are quite acceptable, rigid techniques appear to yield a lower rate of recurrence. Several foreign materials have been used to obliterate the mastoid cavity. Hydroxyapatite (HA) granules and implants have shown favorable results, but HA cement may have a high rate of infection.11 HA canal wall implants failed 25% of cases, usually because of middle ear infection rather than recurrent cholesteatoma.12 Ceravital, a bioactive ceramic glass, has also been successful, but its use is cautioned against in patients with immunologic disorders or diabetes.13 Titanium implants have also been used to reconstruct the canal wall.14Hard materials such as these may eliminate the cavity problem but also may hinder the detection of the recurrent cholesteatoma. Because of the need for meticulous fitting and for vascularized soft tissue coverage, rigid implant materials have not gained widespread use, and concern remains over the potential for infection or extrusion. We have used an alternative technique of reconstructed canal wall mastoidectomy in which the posterior canal wall is removed en bloc and then reused as a free bone graft to obliterate the sinodural angle (Figs. 8.6, 8.7, and 8.8). The surgical exposure is the same as for CWD, so the chance of leaving disease behind is very low. The attic remains exteriorized, thereby preventing recurrent disease from forming in that location. No foreign material or bone pate is used; therefore there is no increased risk of infection and little risk of reimplanting disease. The procedure still results in the creation of a cavity, but the size of the cavity is lessened, both by the reimplantation of cortical bone from the posterior canal, and by maintaining the mastoid tip. The bony reconstruction blocks the sinodural angle and results in a cavity no deeper than the lateral semicircular canal (Fig. 8.8). Using a conchomeatal flap in conjunction with the bony obliteration results in a round bowl with sloping edges, and a functionally small cavity with little surface area to collect debris. Fig. 8.6 Attic cholesteatoma, with a low-lying tegmen. Obliteration techniques carry the potential risk of burying residual disease, and so would not be advisable in cases of giant aggressive cholesteatoma, “cholesteatosis,” or plunging disease. Second-stage surgery is a decision that is made at the time of the initial operation. There are several reasons for choosing to perform a second stage: 1. Invasive cholesteatoma into the mucosa of the middle ear, anterior or posterior epitympanum, antrum, or mastoid 2. Cholesteatoma in a location such that the surgeon questions the success of total removal at the first stage 3. Previous failed surgery for tympanomastoid surgery with chronic otitis media and significant mucosal disease 4. Significant allergic or gastroesophageal reflux The main purpose of second-stage surgery is to search for the presence of residual disease after a CWU operation. The second purpose of second-stage surgery is to reconstruct the hearing mechanism in a staged fashion, at a time when the middle ear is stable and without disease. The second-stage operation was originally recommended as a routine measure to detect residual disease by the proponents of CWU surgery. The main disadvantage of the CWU procedure is the possibility of recidivistic disease, which can either take the form of residual cholesteatoma (an epithelial remnant left in the mastoid cavity, anterior or posterior epitympanic space, or middle ear, separate from the tympanic membrane), or recurrent cholesteatoma (arising from a new tympanic membrane retraction). The former can grow silently behind an intact canal wall and tympanic membrane, and so a second-stage surgery is therefore recommended as a way to detect and treat the growth early, before it has had the opportunity to become destructive. The latter are clinically evident and can be detected without an additional surgical operation. The further rationale for a staged second operation is to perform ossicular reconstruction in a stable middle ear. Cholesteatoma is destructive, and the ossicles are frequently eroded or separated by the disease. Primary ossicular reconstruction does not always meet with uniform success because of the presence of inflammation at the time of the original surgery, as well as the vagaries of healing. The tympanic membrane may become scarred, retracted, or adherent to the middle ear mucosa over time, and this may negatively influence the results of attempted ossicular repair. The odds of success may be improved if ossiculoplasty is undertaken at a later time, when there is no cholesteatoma present and the middle ear is not inflamed. In a stable middle ear, the height of the tympanic membrane relative to the stapes footplate will remain constant during healing, and so an ossicular prosthesis or sculpted incus can be sized and positioned accurately and predictably during surgery and will not be expected to shift or change. The main objection to second-stage surgery is that it requires a separate trip to the operating room, subjecting the patient to additional anesthesia, surgical risk, and cost. It should be noted that many second-stage surgeries may be performed under IV sedation techniques markedly reducing risks of operation. Experience with local anesthesia techniques and IV sedation allows limited risk and immediate results of hearing improvement. Opponents of the second-stage approach will also argue that the second look will be negative in a significant proportion of cases, and that clinical follow-up alone is adequate because residual disease will make itself known in time. In some surgeons’ hands, primary ossicular reconstruction yields good results in the majority of cases so that reconstruction need not be postponed to a later stage. Certain issues remain unresolved about the role of second-stage surgery for cholesteatoma. First, there is disagreement about whether a second-stage operation should be routinely performed after CWU operation, and whether for residual disease or for hearing or both. Although some surgeons still perform a planned second-stage after every case, many will do this selectively in cases where the completeness of excision was not certain (eg, in the sinus tympani, scutum, anterior or posterior epitympanic space, or mucosal invasion), or where the dissection was difficult because the disease was adherent or the anatomy did not allow unhindered exposure. The second stage also offers an opportunity to perform ossicular reconstruction in a “clean” field, as already discussed. A second unresolved question is the optimal timing of the second operation. Most will perform the surgery as early as 6 months or as late as 2 years. Earlier surgery allows for residual disease to be dealt with when it is smaller and well circumscribed. Later surgery allows for maturation of scar tissue and a more stable middle ear space. For instance when the surgeon is trying to reconstruct and enlarge the middle ear cleft, waiting longer allows time for conformity of the middle ear space with the spacer Silastic sheeting enlarging this space. The timing remains a matter of individual preference. A further unresolved question is how to deal with recurrence when it is found. This must be individualized by the situation found at surgery. The most conservative (but radical) approach would be to convert to a CWD mastoidectomy. This approach would virtually eliminate the possibility of further recurrence, but might result in a large, unwieldy cavity and would be overkill in the case of a small residual pearl. It is possible to leave the middle ear cleft by keeping the canal wall over the fallopian canal partially in place where the chorda tympani is located. On the other hand, maintaining an intact canal wall would be risky if extensive or invasive disease is discovered. In such a case, the surgeon may decide on performing a third stage with the suggestion that hearing improvement up to normalized hearing is potentially still possible. Many patients would not look forward to the prospect of a third operation, but in our experience, the surgeon–patient relationship and surgeon experience are key to this decision. The technique of second-stage surgery presents certain options. The most formal method would consist of raising a tympanomeatal flap and reopening the mastoid through a postauricular incision. A more limited method would be to raise the tympanomeatal flap and use a mirror or endoscope to inspect the epitympanum or antrum. This technique might miss disease in the mastoid; however, some surgeons have used a small stab incision to pass a slim 2.7 or 4 mm endoscope into the mastoid. A limited procedure such as this can even be performed under local anesthesia. If extensive residual disease is found, the incision can be opened and a formal revision mastoidectomy performed. Concerns remain about the yield of second-stage surgery and the adequacy of follow-up. In most studies, residual disease will be found in ∼20 to 50% of second-look operations, meaning that the majority of operations will result in a negative exploration. This argues for a selective approach to second-look surgery and makes limited, endoscopically assisted surgery more attractive. Imaging techniques such as CT and magnetic resonance imaging (MRI) have been tried in an effort to avoid routine surgical exploration, but their predictive value is not perfect. CT cannot distinguish between one type of soft tissue and another, so postoperative fibrosis and residual disease might look the same. MRI likewise lacks specificity, although newer protocols such as diffusion-weighted imaging might improve on this. Issues of expense and patient discomfort persist with MRI, however. Not all patients will agree to second surgery, and not all patients will return for regular follow-up, so some cases of residual disease will certainly be missed despite all efforts to be thorough. In a recent study from the New York Eye and Ear Infirmary (pers. comm.), the rate of follow-up after cholesteatoma surgery was around 50%, a rate that raises concern. It is important to realize that the doctor–patient relationship is crucial. Personally, in our preoperative education we talk to patients about their disease and that surgery is done in two stages to look for recurrent or residual cholesteatoma and optimize their hearing back toward normal limits. The facial nerve is at risk during chronic ear surgery, and facial nerve injury remains a dreaded complication. The incidence of facial nerve paralysis during surgery is low, certainly less than 1% in experienced hands, but even so the threat of a facial nerve complication remains a sobering reality. Experienced surgeons recognize the fact that distorted anatomy, congenital anomalies, or extensive inflammatory disease can expose the facial nerve to unexpected surgical trauma even when every precaution is exercised. It is therefore not surprising that the use of facial nerve monitoring has become commonplace during ear surgery. Nerve monitors have become seamless in their design and reliable in their performance, and have shown themselves to be helpful in difficult situations. Nevertheless, monitoring is not a substitute for an intimate knowledge of facial nerve anatomy or for meticulous surgical technique. Facial nerve monitoring should be seen as an adjunct, an extra layer of protection during surgery. The purpose of the facial nerve stimulator/monitor is to help locate the nerve anatomically, to provide live feedback when dissecting on an exposed nerve, and to verify the functional integrity of the nerve by electrical stimulation. Facial nerve monitoring does not replace technical proficiency, anatomical knowledge, or the judgment that comes from surgical experience. The facial nerve monitor provides contextualinformation during surgery that must be interpreted within the situation at hand. The principles of facial nerve monitoring have been outlined in detail in several sources.15,16 A few devices are commercially available for monitoring the facial nerve during surgery. These devices contain two separate electric circuits, one to stimulate the nerve electrically, and the other to monitorthe activity of the facial muscles. The nerve stimulator allows the surgeon to apply a small electrical current to the nerve during surgery. This provides immediate information about the anatomical location and the functional integrity of the nerve. Electrical stimulation is delivered through a probe. Stimulator probes usually operate in a monopolar mode, dispersing current radially from the tip of the stimulator probe (bipolar probes, which emit current between two probe tips, are also available, but they are less often used because the current must be oriented parallel to the nerve).17 Special surgical instruments also exist that can deliver an electric stimulus during dissection.18 Stimulators are usually set to deliver a constant current, and the current level can be selected on the device (constant voltage devices also exist but can deliver excessive current if the tissue resistance is low). Typically, a current level of 1 to 2 mA (mA) will be needed to stimulate the facial nerve through an intact bony covering, whereas a very small current (0.1 mA) will stimulate a bare nerve. Stimulation will spread bidirectionally along the nerve. In the setting of facial nerve injury, the nerve will continue to stimulate distal to the site of the lesion for up to 3 days, even after complete transaction. Stimulation proximal to the site of the lesion will give information as to the degree of injury; brisk stimulation at a low current level implies functional continuity of the nerve, whereas a high stimulation threshold or complete absence of stimulation implies a severe conduction block or a transected nerve. Constant-current stimulation is susceptible to shunting, wherein the current is dissipated by any electrolytic fluid (blood, saline, CSF) in the surgical field; the field should be dried with a suction before attempting to stimulate the nerve. The nerve monitor most commonly employs electromyography (EMG) to detect contraction of the facial muscles during surgery, although there are motion sensors (“strain gauge sensors”) that have a higher threshold for detecting a response. The monitor produces an audible signal (as well as a visual oscilloscopic tracing) in response to facial muscle contraction. The audible signal provides live feedback to the surgeon during the dissection. The facial nerve monitor typically allows for recording from two or more channels during surgery. The usual recording array uses one pair of needle electrodes in the orbicularis oculi muscle and a second pair in the orbicularis oris, plus a common ground (Fig. 8.9). A second ground electrode for the stimulator circuit is inserted into a distant location, usually the shoulder. EMG monitoring produces various types of responses, and the surgeon needs to be familiar with these.15,18 “Burst” responses are brief, nonrepetitive signals that occur in response to brief mechanical stimulation of the nerve (Fig. 8.10A). During surgery, burst responses will occur when one brushes against an exposed nerve with a blunt instrument. They serve as an immediate warning to the surgeon and do not usually forebode permanent injury unless the disturbing activity is forceful or sustained. “Train” responses are repetitive signals that occur in response to events that are longer in duration, such as mechanical traction on the nerve (Fig. 8.10B). The surgeon should cease whatever activity produced the train response to avoid permanent injury. Train responses also occur in response to thermal trauma, such as after saline irrigation. Direct electrical stimulation of the nerve elicits a “pulse” response, a synchronous repetitive signal (Fig. 8.10C). Each recording channel produces a different tone, so that the pulse responses are specific for the upper and lower divisions of the nerve. In cholesteatoma surgery, facial nerve monitoring is most useful when dissecting disease away from an exposed facial nerve. As previously discussed, spontaneous bony dehiscences of the facial canal are common, and the incidence is higher in the presence of erosive disease. When a bare facial nerve is covered by cholesteatoma matrix or by granulation tissue, it is vulnerable to injury during the removal of the disease. The most common site of dehiscence is the tympanic segment of the nerve, just superior to the stapes (Fig. 8.11). The surgical strategy in these cases is to approach the nerve from posteriorly and superiorly, developing a plane of dissection between the intact bone and the mucosal layer, and lifting the matrix away from the surface of the bare nerve using a sharp instrument. Once this plane is established the disease will usually peel away, leaving the nerve sheath intact and undisturbed. In these cases, the nerve monitor will provide audible feedback if there is blunt or sharp disturbance (burst response) or if there is mechanical traction (train response). With careful surgical technique, no response will be elicited. Fig. 8.11 Dissection of a recurrent cholesteatoma from bare facial nerve, tympanic segment, right ear, working from superiorly downward, using the stimulator probe as a dissecting instrument. A second situation for monitoring may occur when one is drilling near the nerve, especially around the mastoid segment or second genu (Fig. 8.12). Although the nerve has a fairly constant anatomical course, variability exists in the medial-to-lateral position of the mastoid segment, approaching the digastric groove. Also, when opening the facial recess, it is desirable to shave away the bone anterior to the facial nerve to gain the best view of the sinus tympani (Fig. 8.13). A third vulnerable area is anterior to the processus cochleariformis (Fig. 8.12), where the nerve may be exposed in the anterior epitympanic space. In these instances, the monitor may give a burst or train response if the drill or dissecting instrument brushes the nerve sheath. Injury may be avoided if the surgeon is aware of the slightly more lateral position of the nerve in this location. In cases with distorted anatomy, or a nerve embedded in disease, the nerve stimulator may be used to map the course of the nerve anatomically. A monopolar stimulator, such as the Prass probe (Medtronic-Xomed Corp., Jacksonville, FL), can be used at a higher setting initially (eg, 2 mA) to verify that it is working, then the current setting can be turned down (0.5 mA) to gain more specific information about the position of the nerve. A bare nerve should stimulate briskly at 0.5 mA or less, whereas a nerve with an intact bony sheath will require more current, typically 2 mA or higher. The stimulator can also be used after a difficult dissection to verify the integrity of the nerve. Stimulation with a supra-threshold current level proximal to the site of concern will produce a brisk response if the nerve is intact. An absent response or a very high threshold may be a reason to explore the nerve proximally and distally and to decompress the bony sheath. Certain caveats should be noted during monitoring. Bipolar or monopolar electrocoagulation causes an artifactual response. This can be defeated using a muting circuit, but monitoring is suspended when muting is in effect. Irrigation can elicit a train response because of thermal stimulation of the nerve. Although this is not clinically significant, it may take several minutes for the response to die down and for normal monitoring to resume. Brief mechanical trauma using blunt dissecting instruments or drill will cause a burst response—this is moderately significant and should be taken as a warning of impending injury if the activity that caused the response is not stopped. An instantaneous trauma to the nerve, however, may not elicit any response if it is very short in duration; therefore it is possible to have a serious injury to the nerve, such as sharp transection, without any signal at all from the nerve monitor. When the patient has preoperative facial nerve paralysis in the presence of cholesteatoma or chronic inflammatory middle ear disease, the facial nerve stimulator/monitor is helpful for assessing the degree and location of injury. However, if the nerve does not respond to stimulation preoperatively, the device will not necessarily signal any further trauma to the nerve that occurs during the course of surgery. Fig. 8.13 Stimulation of the facial nerve in the facial recess (right ear). After dissection of the disease, the exposed area of the nerve is visualized, and the bony covering is carefully removed proximal to the site of involvement (Figs. 8.14A,B). The presence of a response to electrical stimulation indicates that the nerve has functional integrity and will probably recover fully. The absence of a response, on the other hand, implies severe neuropraxia and a poorer prognosis, and the surgeon may want to decompress the nerve more widely to allow for progressive edema. When the patient wakes up with immediate facial paralysis after chronic ear surgery, the patient is usually brought back to surgery for exploration and possible repair of the injured nerve. In this situation, the nerve monitor may be helpful for mapping the exact site of injury and indicating whether the disruption is complete or partial. If the disruption is complete, the nerve edges should be freshened and the nerve reapproximated without tension using fine sutures (eg, 9–0 Prolene, Ethicon, Inc., Somerville, NJ). If the ends of the nerve cannot be brought together without tension, a nerve graft should be placed, using the great auricular nerve or sural nerve as a donor. If there is partial disruption, judgment must be used as to whether to allow the nerve to heal spontaneously or to repair the nerve primarily. If the injury involves less than a 50% cross-sectional area, or if the electrical response is preserved, the frayed edges of the nerve can be brought together with a collagen sheath, and partial recovery of function can be expected over a 6- to 12-month period. If a middle ear tumor is suspected, the nerve monitor plays a valuable role because the anatomy may be altered and facial nerve involvement may not be predicted preoperatively. Middle ear adenoma (Fig. 8.15) may mimic inflammatory ear disease and may erode the bony covering and compress or distort the nerve anywhere along its course. Salivary choristoma (Fig. 8.16) is composed of heterotopic salivary tissue that can occur anywhere along the facial nerve. Facial nerve paralysis has been reported after biopsy of this unusual lesion, and the facial nerve monitor may help to prevent injury.19 Glomus tympanicum tumors arise along the course of the Jacobson nerve within the middle ear cleft; glomus jugulare (Fig. 8.17) and glomus vagale tumors may grow into the middle ear space via the jugular foramen. Removing these tumors usually requires a facial recess approach; jugulare tumors often require facial nerve transposition or a facial bridge technique to the hypotympanum. Facial nerve schwannomas can occur along the entire length of the facial nerve, and while these rarely occur solely in the middle ear, middle ear involvement is common. Removal of a facial nerve schwannoma will usually result in facial nerve paralysis, and primary grafting should be planned. If the diagnosis is not made preoperatively, the facial nerve stimulator can be helpful for confirming the fact that the tumor is of facial nerve origin. Debate continues over whether or not to routinely monitor the facial nerve during chronic ear surgery. Unlike acoustic neuroma surgery, where most surgeons would consider facial nerve monitoring to be indispensable, in chronic ear surgery the issue is less clearly resolved. The incidence of facial nerve paralysis in routine mastoidectomy is low, even in the presence of cholesteatoma. Some surgeons would argue that the surgical anatomy is familiar, and using a monitor does not reduce the chance of facial nerve injury. Indeed, monitoring will not necessarily warn against sudden, penetrating injury, as already discussed. And in inexperienced hands, the nerve monitor may contribute to a false sense of security leading to a surgical misadventure. Proper training in the setup and use of facial nerve monitoring is therefore essential. Further, monitoring has a significant monetary cost—a commercially made unit costs around $20,000.00, the disposable electrodes up to $100.00 for each use, and a disposable stimulator probe even more. Some centers use a technician for live monitoring, adding to the cost. The increased time for setup creates intangible costs as well. On the other hand, it is easy to argue for the potential benefits of facial nerve monitoring. When properly used, the monitor gives valuable information about the position of the nerve, provides feedback to the surgeon during delicate dissection on an exposed nerve, and usually warns of impending injury. These benefits would be difficult to prove “scientifically” because the variables encountered in surgery do not lend themselves easily to quantitative measurement in an outcome study, but most surgeons would agree that the information provided by monitoring could help prevent a facial nerve complication. Because the incidence of facial nerve injury is very low, a very large study would have to be constructed to have sufficient statistical power. Facial paralysis will often result in medicolegal action, even in the absence of negligence or wrongdoing by the surgeon,20 and failing to use a nerve monitor may be perceived as a departure from current “best practices.” In real-world current practice, the facial nerve monitor is probably used by most surgeons in most cases of cholesteatoma surgery. Although some surgeons will employ it selectively, monitoring should probably be set up and used routinely to be most useful because it is impossible to predict when it will really be needed.21 In a policy statement,22 the American Academy of Otolaryngology–Head and Neck Surgery (AAO-HNS) has recognized “the proven efficacy of neurophysiologic monitoring of the facial nerve which may minimize the risk of injury to the nerve during surgical procedures in which the nerve is vulnerable.” But they also state that these “guidelines are not a substitute for the experience and judgment of a physician . . . and in no sense do they represent a standard of care.” This statement strikes a balance between the opinions of surgeons’ pro and con. Endoscopes have proved to be a useful adjunct to surgery of the middle ear and mastoid.23 The advantage provided by the endoscope is the ability to place the surgeon’s eye deep within the surgical cavity, and to be able to look around corners. This provides an advantage in areas such as the facial recess and anterior epitympanic space, which are difficult to visualize directly using the microscope. The disadvantages of the endoscope are the need to switch from the microscope to a different optical system (using a separate video setup or the naked eye), the presence of an object within the surgical field (with the potential to injure the stapes or facial nerve), the generation of heat (another potential source of injury), the loss of three-dimensionality, the loss of a free hand (or the need for an assistant to hold the scope), and the loss of visualization while operating with blood in the surgical field. These disadvantages make it unlikely that the endoscope will replace the microscope in ear surgery, but the enhanced visualization makes the endoscope useful nonetheless. Smaller endoscopes provide a view of recesses within the middle ear space. A 2.7 mm, 30 degree scope can be inserted into the middle ear via a tympanomeatal flap, allowing the surgeon to look in the sinus tympani and epitympanum and reducing the possibility of leaving disease behind. This should translate into a lower rate of residual disease, and some authors have claimed improved results.24,25 The ability to peer around corners may also permit less bone removal during mastoid surgery. It is possible that the endoscope may preclude the need for a facial recess approach for sinus tympani disease, allowing the surgeon to look in the sinus tympani and epitympanum and reducing the possibility of leaving disease behind (Fig. 8.18).26 The endoscope may also allow for a less invasive approach for second-stage surgery.27,28 Passing the endoscope through a small stab incision in the postauricular scar allows the surgeon to rule out residual disease in the mastoid cavity without the need for a large postauricular incision (but of course if disease is found, a formal revision operation will be necessary). An endoscope-only technique has been devised for surgery of the middle ear and attic.29 Using a 4 mm endoscope in the ear canal gives a wider field of view than the microscope can, and allows a complete transcanal atticotomy to be performed. For disease confined to the attic, this may provide thorough surgical removal through a more limited approach. Adequacy of follow-up is an important factor in determining success or failure after cholesteatoma surgery. Follow-up is necessary to determine whether a patient has recurrent or residual disease, to clean and to manage problem cavities, and to assess postoperative hearing status in the short and long term. In clinical practice, however, many factors affect the reliability of follow-up, such as the patient’s distance from the surgeon, (changeable) insurance status, relationship with a referring physician, or moving out of the area. Without regular follow-up, the long-term results of cholesteatoma management are unknowable. For surgeons with out-of-town referral sources, this can be an even greater problem. The tendency of cholesteatoma to recur lends particular importance to this issue. In a patient whose reliability is in question, CWD surgery is often the safest option. CWD has an intrinsically low recurrence rate, and residual disease can be spotted on exam and often dealt with in an office setting. Further, CWD exteriorizes the mastoid, so that complications (ie, spread of infection to the labyrinth, facial nerve, or intracranial structures) are unlikely to occur. Several studies have taken up the issue as to what constitutes adequate length of follow-up. The recurrence rate for cholesteatoma increases with the length of follow-up in both children and adults.30–32 Because cholesteatoma is thought to be more aggressive disease in children, the importance of follow-up is greater in pediatric patients. Recurrent cholesteatoma can form at any time, even years after the original surgical procedure (in contrast to residual disease, which is theoretically present at the conclusion of the surgery). Long-term follow-up studies have reported recurrent disease up to 15 years later, and giant cholesteatomas have been discovered after even longer intervals.33 Although the likelihood of recurrence diminishes with time, there is no end point, practically speaking, at which follow-up is no longer needed. The reporting of results can be influenced by the adequacy of follow-up. This can affect the surgeon’s perceived success rate in treating patients. Most studies traditionally used a standard rate calculation, where the total number of observed recurrences is divided by the total number of years of follow-up for the study. This method, however, fails to account for patients who drop from follow-up, or who have insufficient length of follow-up.34 The use of Kaplan-Meier analysis adjusts for those who are “censored” from the study, and gives a truer estimate of disease recurrence overall.35 1. Canal wall up and canal wall down operations each play an important role in the management of cholesteatoma, and factors including mastoid size, aggressiveness of disease, presence of complications, and reliability of follow-up should be considered in selecting the best approach in a given patient. Reconstructed canal wall techniques may provide a useful compromise in certain cases. 2. Second-stage surgery can be useful in certain patients to rule out recurrent or residual disease and to allow for ossicular chain reconstruction in a stable middle ear. 3. Facial nerve stimulation and monitoring provide an added measure of patient safety but do not replace surgical judgment, anatomical knowledge, or technical skill. 5. Adequacy of follow-up is an important factor in cholesteatoma management because the recurrence rate remains significant, even in the most experienced hands.
♦ Canal Wall Up versus Canal Wall Down
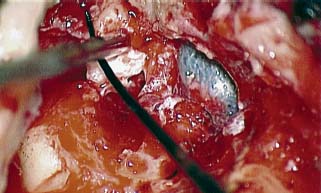
Canal Wall Reconstruction
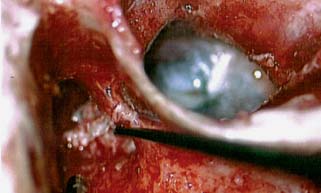
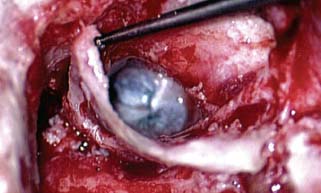
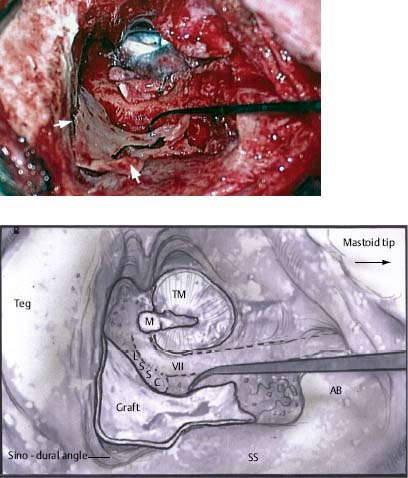
♦ The Timing and Necessity of Second-Stage (Second-Look) Surgery for Cholesteatoma
♦ Facial Nerve Monitoring
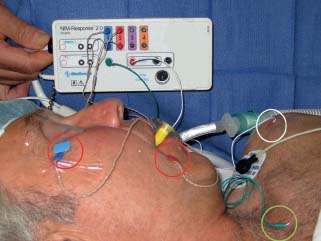
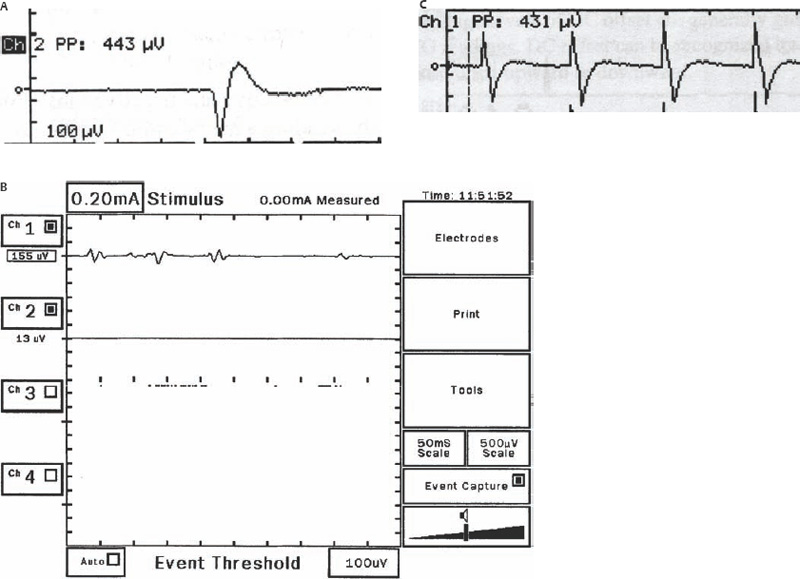
Surgical Applications of Facial Nerve Stimulation/Monitoring
Primary Chronic Ear Surgery
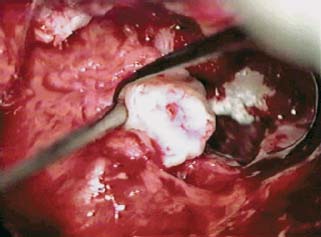
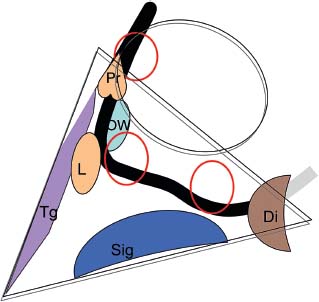
Chronic Ear Disease Presenting with Facial Nerve Paralysis
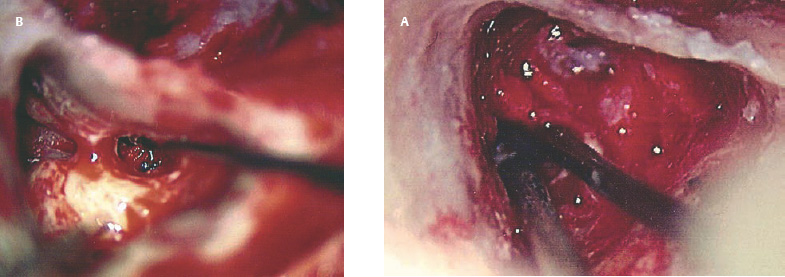
Postoperative Facial Nerve Paralysis
Other Surgical Scenarios
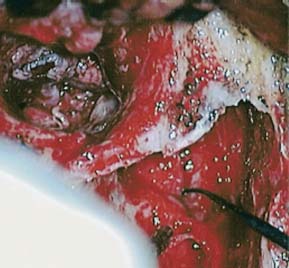
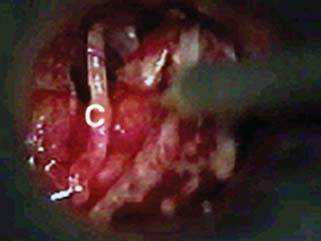
Issues and Controversies Regarding Facial Nerve Monitoring
♦ Endoscopes in Chronic Ear Surgery
♦ Adequacy of Follow-Up
♦ Conclusions
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


