Fig. 4.1
Photograph of lipofibroma located in the midline posteriorly on the surface of the tongue
2.
Choristoma is a benign, well-organized heterotopic growth of tissue that forms in excess in areas in which it normally is not located, i.e., normal tissue that forms in excess in abnormal locations. In the oral cavity, choristomas may consist of a proliferation of an ectopic rest of bone, cartilage, fat, neural or glial tissue, thyroid tissue, respiratory tissue, gastric mucosa, or intestinal mucosa. They may or may not form a mass lesion and are not neoplastic. More common choristomas in the oral cavity include those that occur on the tongue, specifically gliomas, salivary tissue rests, and skin tissue (dermoids, see below) (Fig. 4.2).
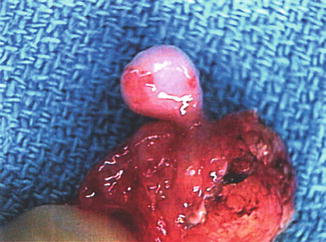

Fig. 4.2
Photograph of ectopic salivary tissue specimen
Teratoma is a benign, haphazardly organized growth of tissue (or tumor) containing all three germ layers (endoderm, mesoderm, and ectoderm), which may be located anywhere in the body, especially in the head and neck. Teratomas may have malignant potential. Unlike hamartomas and choristomas, teratomas may also present as large obstructing masses of the head and neck. Smaller teratomas may be found anywhere within the oral cavity including the tonsil and are treated by surgical excision.
1.
A hairy polyp is similar to a teratoma but consists of cells of only two germ cell lines (mesodermal and ectodermal tissue). These masses tend to originate in the oropharynx or nasopharynx and may stem from a malformed second branchial arch. Hairy polyps are more common in girls and usually are present at birth or within the first few years of life. They may be associated with other malformations of the head and neck, including cleft palate. The treatment for these tumors is complete surgical excision (refer Chap. 3, Fig. 14).
2.
Dermoid is a lesion, which consists of mostly ectodermal tissue that forms a cyst filled with keratin or sebaceous material, and sometimes hair. It is most commonly found in planes of embryonic fusion. Common cutaneous locations include the regions lateral to the eyebrow, and in the midline of the neck, but they may be located anywhere within the oral cavity, including on the tongue or uvula. Treatment is excision.
3.
Congenital epuli arise most often arise from the maxilla or mandible of the newborn. They are composed of cells derived from mesenchyme (granular cells with eosinophilic cytoplasm and small, eccentric nuclei). The lesions are more commonly found in girls and can present as small or very large lesions. Some have been found to regress with time, but they should be excised if they interfere with feeding or if they bleed (Fig. 4.3) [1].
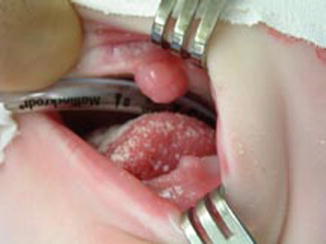

Fig. 4.3
Photograph of infant with two epuli: one on surface of mandible and one on surface of maxilla
Hemangiomas/Vascular Malformations
Definition
Vascular birthmarks are most commonly located in the skin and subcutaneous tissues, but they are also the most common congenital malformations found within the oral cavity and oropharynx. These lesions are usually located within the mucous membranes of the oral cavity and oropharynx, but they may also appear within the hypopharynx and the larynx. They may range in appearance from flat vascular mucosal patches to bulky lesions that can interfere with breathing or swallowing.
Vascular birthmarks are classified as either hemangiomas or vascular malformations. Infantile hemangiomas are the most common benign tumors of infancy. Thirty percent are seen at birth and the remaining form within the first few weeks of life. Eighty percent are focal and solitary in nature. The remaining may be multifocal or segmental. Segmental hemangiomas may involve large areas of skin and often associated with complications and associated malformations. Superficial hemangiomas involve the superficial dermis and consequently appear red in color, whereas deep hemangiomas reside in the deep dermis and subcutis, resulting in a blue color on the surface. Mixed hemangiomas involve both the superficial dermis and the deep dermis/subcutis. Infantile hemangiomas, which are not apparent at birth, must be distinguished from the less-common congenital hemangiomas. These lesions proliferate in utero and are fully formed at birth. They may be further categorized based on their pattern of involution after birth (RICH, rapidly involuting congenital hemangioma and NICH, non- involuting congenital hemangioma).
Vascular malformations are nonproliferative hamartomatous lesions that are usually present at birth and are classified as low-flow lesions (lymphatic, venous, capillary) or high-flow lesions (arteriovenous) [2].
Incidence
Infantile hemangiomas have been reported to occur in 2.5–10 % of infants (12 % of Caucasian infants, 1.4 % of African American infants, and 0.8 % of Asian infants, respectively). Fifty percent are located in the head and neck. Females are affected three times more often than male babies. The incidence is higher in preterm babies (up to 25–30 %), especially in those of extreme low birth weight and in multiple births (independent of birth weight) Vascular malformations are less common, have no gender predilection, and tend to grow proportionally with the affected child.
Heredity
Most hemangiomas and vascular malformations occur sporadically, although hemangiomas have been found to be familial in 12 % of cases. A linkage to chromosome 5q31–33 has been identified, but no specific gene has been implicated in their etiopathogenesis. DNA studies of some families who have multiple vascular malformations suggest that a genetic defect may exist in cells that form the smooth muscle lining of normal blood vessels.
Etiology
Infantile hemangiomas are neoplasms composed of proliferating endothelial cells that eventually form vascular spaces and channels, some filled with blood cells. There is limited basement membrane and as they grow they fill local potential spaces in the oral cavity and oropharynx. They eventually may appear as lobular, red structures extending beyond mucosal membranes. Congenital hemangiomas are composed of normal endothelial cells. Vascular malformations present as true structural anomalies, with normal rates of endothelial cell turnover. They lack muscular support and contain large vascular channels because they do not have smooth muscle lining cells that are normally present in blood vessels.
Multiple hypotheses exist to explain the underlying etiology of infantile hemangiomas, including the theory that they represent immature tissue, possibly similar to placental tissue, endothelial progenitor cells, or mesenchymal stem cells. These cells may maintain their proliferative capacity for a period of time postnatally. Various growth factors, including beta-fibroblast growth factor, vascular endothelial growth factor (VEGF) and proliferating cell nuclear antigen may induce proliferation. However as the endothelial cells differentiate, mast cells appear which produce transforming growth factors and other tissue inhibitors including interferon, which terminate the proliferation. Involution of the hemangioma tends to occur when these cells and factors are present [2, 3].
Associated Malformations
Most hemangiomas occur as isolated lesions, but the following associations have been recognized:
PHACE association represents a neurocutaneous disorder most commonly presenting with large segmental facial hemangiomas and additional structural anomalies of cerebral vessels, eye vessels, and the aorta. The acronym stands for posterior cranial fossa malformation(s) (including Dandy-Walker and Arnold Chiari malformations), large cervicofacial or laryngeal hemangiomas, arterial anomalies of the head and neck (that can involve the carotid and cerebral vessels), coarctation of the aorta and cardiac defects, and eye anomalies. Sternal clefts or other ventral defects may also be present (PHACE(S)). The PHACE association affects female infants in 90 % of cases (Fig. 4.4) [3–5].
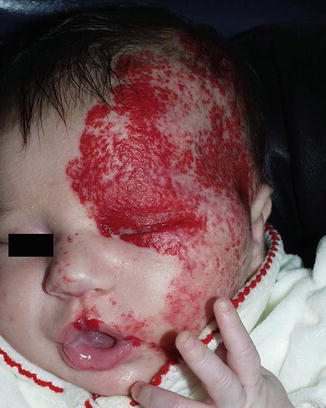
Fig. 4.4
Photograph of infant with PHACE syndrome of left face (Image courtesy of David W. Low, MD, Attending Surgeon, The Children’s Hospital of Philadelphia; Associate Professor, Department of Plastic Surgery, University of Pennsylvania School of Medicine)
Kasabach–Merritt Phenomenon is a rare, life-threatening condition which may occur if one of two specific benign vascular tumors is present (tufted angioma or kaposiform hemangioendothelioma). These lesions tend to be larger and cause thrombocytopenia by trapping and destroying platelets. This phenomenon does not occur in children with common infantile hemangiomas.
Several syndromes associated with vascular malformations include the following:
Sturge–Weber (encephalotrigeminal angiomatosis): A rare congenital, usually unilateral phakomatoses (neural and skin disorder). It is associated with a facial port wine stain (or capillary malformation) located in the region of the first and second divisions of the trigeminal nerve (V1 and V2 correlating to the regions of the forehead and cheek, respectively), along with ipsilateral leptomeningeal and brain angiomas (frequently leading to mental retardation and seizures). Glaucoma may be present caused by excess capillaries near the ophthalmic branch of the trigeminal nerve. This is a noninherited, nonfamilial syndrome (Fig. 4.5).
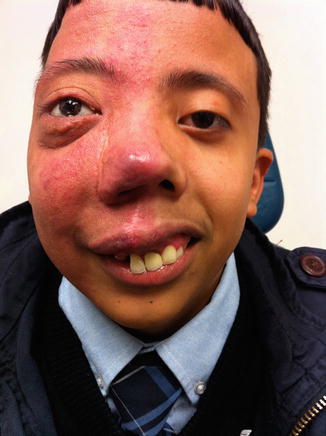
Fig. 4.5
Patient with Sturge Weber demonstrating port wine stain (capillary hemangioma of right face) and facial deformity due to changes in growth of these bones
Klippel–Trenaunay: Capillary malformation of an extremity associated with deeper venous and lymphatic malformations and skeletal overgrowth.
Parkes–Weber: Capillary malformation of an extremity associated with a deeper arteriovenous malformation.
Osler–Weber–Rendu (hereditary hemorrhagic telangiectasia): An inherited disorder of blood vessels resulting in arterial venous malformations (AVMs) within the skin and mucous membranes. Affected individuals have diffuse cutaneous and visceral telangectasias and small AVMs. It may be associated with epistaxis, oral or gastrointestinal bleeding, hematuria, and CNS hemorrhage. This syndrome may be inherited in an auto-dominant pattern.
Clinical Features
Infantile hemangiomas are rarely visible at birth, but are recognized within days to weeks of delivery. In the oropharynx, these benign vascular proliferative lesions initially appear as flat red or bluish areas of mucosal discoloration. Most undergo rapid proliferation in the first 6–12 months of life, with the majority of proliferation occurring within the first 5 months. After reaching a plateau, hemangiomas slowly regress, or involute, a process that may continue for many years (50 % by 5 years, 70 % by 7 years and 90 % by 9 years of age) (Table 4.1). Complications of proliferative hemangiomas include ulceration and bleeding, airway obstruction, visual compromise, feeding impairment, and, in very large lesions, high-output congestive heart failure. Hemangiomas of the lip, neck, and diaper area are most prone to ulceration. Facial hemangiomas may cause cosmetic disfigurement and may necessitate surgical reconstruction after involution (Fig. 4.6).
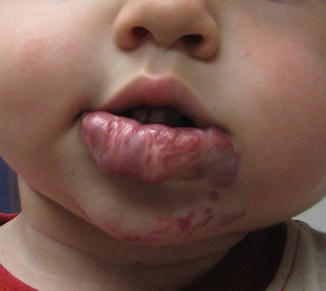
Table 4.1
Comparisons of characteristics of infantile hemangiomas vs. vascular malformations
Infantile hemangioma | Vascular malformation | |
|---|---|---|
Onset | Only one-third present at birth Most appear within first few weeks of life | Present at birth Not always obvious |
Growth pattern | Rapid in first year Regress over early childhood | Grow proportionately to child Never regress |
Gender | Affects girls more than boys (3:1) | Girls and boys equally affected |
Color/consistency | Red, less often bluish Raised with distinct border Doughy texture | Purple to blue Flatter, often submucosal Usually soft |

Fig. 4.6
Photograph of oral and lip hemangioma in infant (Image courtesy of David W. Low, MD, Attending Surgeon, The Children’s Hospital of Philadelphia; Associate Professor, Department of Plastic Surgery, University of Pennsylvania School of Medicine)
Children who are at higher risk of developing complications from their hemangiomas include those with large hemangiomas, multiple cutaneous hemangiomas, or segmental hemangiomas. Infants with more than five cutaneous hemangiomas are more likely to have visceral hemangiomas, including those of the liver, gastrointestinal tract, liver, lungs, and brain. Larger hemangiomas and segmental hemangiomas are more likely to cause cosmetic disfigurement and are more likely to ulcerate and bleed as they grow. Airway lesions involving the subglottis, larynx, or oral cavity may be present in up to 29 % of those with segmental hemangiomas of the face (including patients who do not have PHACE(S) syndrome), especially in those with hemangiomas that are located within the beard or mandible distribution [5]. In most instances, the complications described above are apparent within the first 6 months of life during the phase of rapid proliferation (Fig. 4.7a) [3].
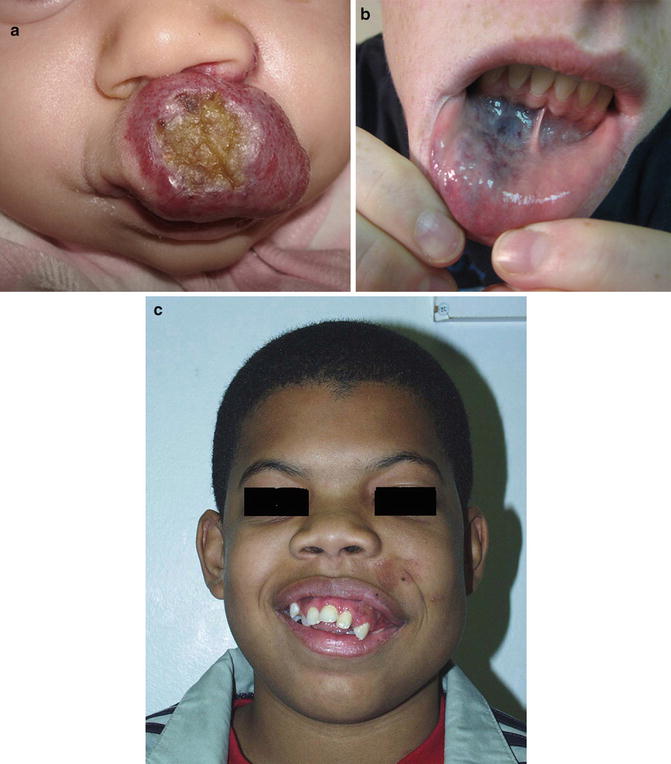

Fig. 4.7
Photographs demonstrating the variation in appearance of facial birthmarks. (a) Upper lip ulcerating hemangioma. (b) Venous malformation of lower lip. (c) Arterial venous malformation distorting left maxilla in young boy (Images courtesy of David W. Low, MD, Attending Surgeon, The Children’s Hospital of Philadelphia; Associate Professor, Department of Plastic Surgery, University of Pennsylvania School of Medicine)
Unlike hemangiomas, vascular malformations are usually present at birth and do not undergo rapid proliferation or involution. Low flow vascular malformations typically demonstrate growth that is proportional to that of the patient. Venous malformations are soft, easily compressible, and are usually blue or purple in color (Table 4.1). Although usually small and asymptomatic, venous malformations of the oropharynx, oral cavity, or tongue may expand to compromise the airway, interfere with speech or cause failure to thrive secondary to poor feeding (Fig. 4.7b).
Capillary malformations (also known as port wine stains) usually present at birth as pink, macular discoloration of the skin. With time, untreated capillary malformations may darken and hypertrophy. Patients with capillary malformations of the head and neck may also have involvement of the oropharyngeal or oral mucosa (Fig. 4.7b).
Arteriovenous malformations (AVMs) are high-flow congenital vascular anomalies that may be found within the tongue or oral/oropharyngeal submucosa. The blood vessels appear as a tangled mass of arteries and veins without an interposed capillary bed. Although most are present at birth, they often remain undetected for many years. On physical examination, arteriovenous malformations frequently appear similar to venous malformations, but they may be associated with an audible bruit, a palpable thrill, or visible pulsations. Complications of AVMs include potentially life-threatening hemorrhage. Larger AVMs may extend to include surrounding soft tissue and bone causing distortion of the face and oral cavity (Fig. 4.7c).
Diagnosis
In most cases, the diagnosis of hemangioma or vascular malformation is made by correlating the pattern and timing of emergence of the lesion along with the physical findings. In the oral cavity and oropharynx, both direct exam and flexible nasopharyngoscopy and laryngoscopy are helpful to determine the extent of the lesion. Magnetic resonance imaging (MRI) with gadolidium contrast-enhanced computed tomography (CT) may be warranted to further determine the extent and depth of the lesion (Fig. 4.8). Biopsy is indicated in patients when the growth appears atypical or when malignancy is suspected. Angiography may be helpful in the assessment of AVM.
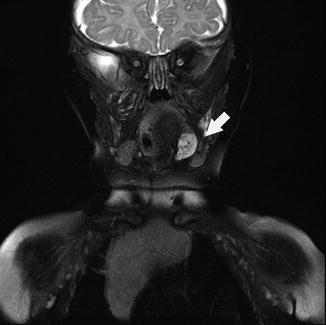

Fig. 4.8
Coronal MRI of head demonstrating left tongue hemangioma
Patients with multiple cutaneous hemangiomas (more than 5 before age 6 months) may benefit from abdominal ultrasound or MRI to rule out the presence of visceral hemangiomas. Those with suspected PHACE(S) syndrome should undergo MRI/MRA (magnetic resonance angiography) of the brain and intracranial vasculature, assessment of the upper airway, cardiac evaluation, and ophthalmologic examination.
Management
Most hemangiomas may simply be observed so long as there are no functional or cosmetic concerns. Pulsed dye laser therapy may be effective for treatment of flat (less than 1 mm) superficial hemangiomas, for healing and pain control of ulcerated hemangiomas, and for the elimination of residual post-involutional telangiectasias. Intralesional corticosteroids may be useful for the suppression of small, localized hemangiomas, but their use should be avoided in the periocular region due to the risk of retinal artery occlusion and blindness. Systemic therapy may be required in patients at risk for functional impairment (e.g., airway obstruction or visual compromise) or for the suppression of lesions in cosmetically sensitive regions. Corticosteroids (2–5 mg/kg/day of oral prednisolone) have been the most commonly used first-line systemic drug for the treatment of proliferative hemangiomas. Adverse effects of corticosteroid administration include irritability, gastritis, immunosuppression, hypertension, adrenal suppression, and growth suppression, all of which are reversible following taper of the medication. Recently, encouraging results have been reported in children treated with propranolol, although the mechanism whereby this nonselective beta-blocker suppresses proliferation of hemangiomas remains uncertain. Potential adverse effects of beta blocker therapy include hypotension, bradycardia, hypoglycemia, bronchospasm, and congestive heart failure. Surgical resection may be indicated in patients who have lesions that may be life-threatening such as may occur when they are located within the oral cavity or airway (especially the subglottis) or for those who fail medical therapy.
Superficial capillary malformations usually respond well to pulsed dye laser therapy. Symptomatic or disfiguring lymphatic, venous, and arteriovenous malformations may require surgical excision, interventional radiologic treatment (sclerotherapy or embolization), or a combination of both).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


