Complications of Uveitis and Their Management
Debra A. Goldstein
Michael Horsley
Lawrence J. Ulanski II
Howard H. Tessler
Management of uveitis is frequently complicated by the development of cataract, glaucoma, macular edema, hypotony, and band keratopathy. This chapter addresses each of these entities and highlights current methods available for their treatment. Other complications, such as rhegmatogenous and nonrhegmatogenous retinal detachments, are addressed elsewhere.
Cataract
Cataract is a frequent complication of uveitis, occurring in up to 57% of patients with pars planitis, 78% of patients with Fuchs iridocyclitis and 83% of those with juvenile idiopathic arthritis (JIA).1,2,3,4,5,6,7,8,9 The incidence in other forms of uveitis is also high and seems to be related to both the location and duration of inflammation, as well as to the use of corticosteroid therapy. The contribution of steroids to cataractogenesis is well known from the data on nonuveitic patients receiving steroid therapy, as well as from experimental data.10 Any route of corticosteroid administration may result in cataract formation; inhaled steroids have been reported to cause both posterior subcapsular and nuclear cataract.11 In addition, recent reports suggest that the prevalence of cataract in pars planitis may be as low as 10% if steroid use is limited or replaced by other forms of treatment, such as cryotherapy or immunosuppressive therapy.12 However, most of the complications of uveitis are more difficult to treat than is cataract; thus, the fear of cataract formation should not prevent the judicious use of corticosteroids.
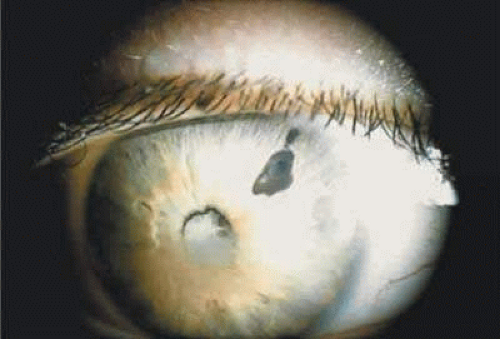
When a cataract develops in a uveitis patient, the management is more complex than in the nonuveitic patient. The presence of posterior synechiae, pupillary membranes, and inflammation may make the surgery more difficult, and the postoperative course may be stormy (Fig. 1). Much controversy still exists regarding the best method of managing the uveitis patient with a cataract. Ideally, absolute control of inflammation should be maintained for at least 3 months preceding surgery; however, there are exceptions to this rule, as in the case of lens-induced uveitis.13
The choice of surgical technique is also controversial, but it is probably not as important as the perioperative management. The patient should be treated preoperatively with topical steroid drops (e.g., prednisolone acetate) at least four times a day for at least 3 days prior to surgery. Also, pretreatment with oral prednisone up to 1 mg/kg/day for at least 3 days is strongly recommended if a patient’s medical status permits. The steroid may be slowly tapered postoperatively as the inflammatory status dictates.
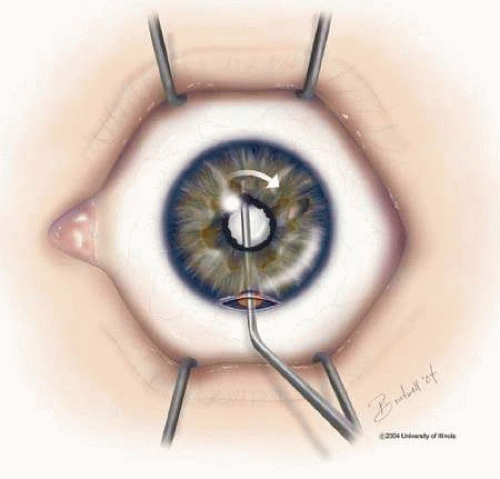
Adequate pupillary dilation is often difficult to achieve preoperatively because of posterior synechiae and pupillary membranes. If there are 360 degrees of posterior synechiae a surgical peripheral iridectomy may be required. The posterior synechiae can then be lysed with the aid of a cyclodialysis spatula; access maybe gained to the superior synechiae through the peripheral iridectomy (Fig. 2). If adequate pupillary dilation is still not achieved, straight or curved long-handled retinal scissors can be used to fashion small membranotomies or sphincterotomies (1 to 2 per clock hour), and viscoelastic material can then be used to dilate the pupil. A Beehler dilator, which both stretches the pupil and creates small microsphincterotomies circumferentially, can also be used to provide adequate mydriasis for safe phacoemulsification.14 Care must be taken to avoid engaging this device in the anterior capsule, leaving a traumatic tear. Finally, iris hooks may provide sufficient dilation without the need for sphincterotomies (Fig. 3). Recent evidence shows that this method also offers superior pupillary stabilization over the Beehler method or pupil-dilator ring but requires the most surgeon time.15 The lens is then removed with the use of either phacoemulsification or standard extracapsular (ECCE) techniques. Although visual acuity is similar for both, phacoemulsification is the preferred method for cataract extraction in most patients due to decreased incidence of postoperative inflammation as compared to ECCE.16
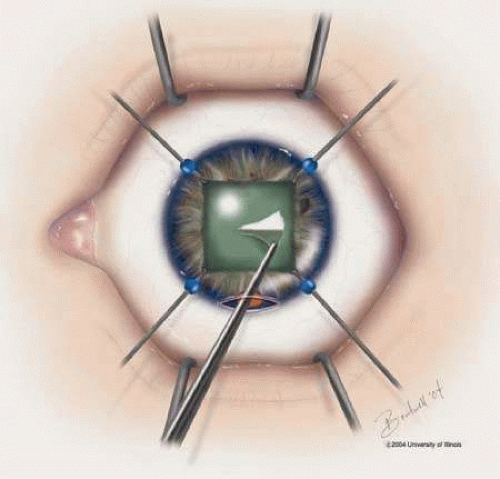 Fig. 3. Iris hooks inserted for pupil dilation and indocyanine green or trypan blue dye used to stain the anterior capsule prior to capsulotomy. |
Implantation of an intraocular lens (IOL) in patients with uveitis is now well accepted. There is much evidence that an IOL can be safely implanted in cases of Fuchs iridocyclitis,17,18,19,20,21 and pars planitis.12,21,22,23 Past literature has favored lensectomy combined with vitrectomy without lens implantation in JIA due to the risks of hypotony and a more challenging postoperative course (Fig. 4).8,24,25,26,27 However, recent reports have demonstrated good long-term visual prognosis from IOL implantation in this population when preoperative as well as postoperative inflammation is minimized.28,29,30
The use of an IOL in noninfectious uveitis of other etiologies is standard, but an IOL should probably be avoided in a patient with inflammation that is unable to be adequately controlled preoperatively. A foldable one-piece acrylic lens may be placed in the capsular bag, or a three-piece lens inserted in the ciliary sulcus. There has been much deliberation concerning the most appropriate IOL material. For capsule placement, acrylic lenses have decreased postoperative complication rates and more favorable visual prognosis compared with polymethyl methacrylate (PMMA), silicone, and heparin-coated PMMA lenses.31,32 Hydrophilic lenses have shown better capsular biocompatibility than PMMA lenses but also show a tendency toward early posterior capsule opacification, leading some to recommend a hydrophobic acrylic IOL due to its improved visual outcomes and reduced complication rates.31,32,33,34 However, this is still controversial, as posterior capsule opacification formation may be related not only to lens material, but optic edge design. Placement of the IOL in the capsular bag rather than the ciliary sulcus is preferred by many surgeons, because it is felt that this may lessen the risk of inflammation secondary to iris-haptic contact. However, no increase in inflammation or intraocular pressure was reported with ciliary sulcus IOL fixation in one series. In fact, localization of the IOL within the ciliary sulcus was found to reduce posterior synechiae development in this series.35 We routinely place the IOL in the capsular bag, using sulcus fixation only if in-the-bag placement is deemed unsafe. Anterior chamber lenses should not be placed in uveitis patients. If there is inadequate support for a sulcus lens, a sutured PCIOL may be placed, or the patient left aphakic.
Occasionally, even in eyes with little or no preoperative inflammation, significant membranes form around an IOL (Fig. 5). This can occur even in the absence of significant anterior chamber cell and flare in the postoperative period.36 Although these cocoonlike membranes can be lysed with the Nd:YAG laser, they tend to reform. Therefore, it is imperative to be vigilant, especially in the first 6 to 12 weeks after surgery, aggressively treating any signs of inflammatory deposits on the IOL, even in the absence of anterior chamber reaction. If laser membranectomy is required, vigorous anti-inflammatory treatment is necessary to prevent re-formation of the membrane. In severe cases, vitrectomy and removal of the IOL may be required and may assist in inflammation control.37
 Fig. 5. Patient with pars planitis with giant-cell precipitates and inflammatory membrane encasement of the intraocular lens. Note peripheral iridectomy. |
It has been suggested that pars plana vitrectomy may be warranted when substantial vitreous cell and debris might preclude good postoperative visual acuity.8,12,19,38 Although we do not routinely perform pars plana vitrectomy on uveitis patients with cataracts, this procedure is considered for eyes with significant vitreous debris or inflammation, or with intractable macular edema associated with chronic uveitis.39 The postoperative management is similar to that of nonuveitis patients, except that inflammation is usually more severe and prolonged, often requiring depot steroid injections and systemic anti-inflammatory treatment.
The results of cataract surgery in uveitis patients vary according to the preoperative diagnosis, but overall the prognosis is favorable.30 Recent improved visual outcomes may be partly due to the use of trypan blue 1% and indocyanine green (ICG) 0.5% dyes, which have been shown to be safe and effective tools to aid in the visualization of the anterior capsule for creation of capsulorhexus (Fig. 3).40 Trypan blue is commercially available as a ready to use diluted dye, but ICG must be prepared by the surgeon. The ICG 0.5% is prepared by dissolving 25 mg of ICG powder in 5 mL of sterile water; 1 mL of the dissolved ICG is then aspirated and mixed with 9 mL of sterile balanced salt solution. The ICG or trypan blue is used to stain the anterior capsule under an air bubble, which is then replaced with viscoelastic prior to anterior capsulotomy.
Patients with Fuchs iridocyclitis generally do well, with most obtaining a visual acuity of 20/40 or better.17,18,19,41,42,43 However, a good outcome should not be assumed, and postoperative vigilance should be maintained, especially when the patient has coexisting glaucoma or a hyphema, or conversion to ECCE technique is required.44 Patients with pars planitis also do well, with 60% to 82% achieving a visual acuity greater than 20/40.12,37 Most pars planitis patients who fail to achieve good visual acuity do so because of cystoid macular edema (CME).8,12,13 In these patients, more aggressive anti-inflammatory treatment, both preoperatively and postoperatively, has resulted in improved outcomes.22
Traditionally, patients with JIA have not had as good an outcome as those with Fuchs iridocyclitis or pars planitis. A visual acuity greater than 20/40 was generally achieved in only about 60% of patients.24 However, with aggressive preoperative control of inflammation, visual acuity greater than 20/40 has been reported in 75% to 100% of JIA patients.19,29 The most common postoperative complication for JIA patients after cataract extraction is posterior capsular opacification, being reported in 70% to 100% of eyes.29,27,45,46 Finally, those patients with idiopathic and other forms of nongranulomatous anterior uveitis tend to do well postoperatively, with almost 80% achieving a visual acuity of 20/40 or better, again provided inflammation is well controlled preoperatively.13
Glaucoma
Elevated intraocular pressure (IOP) is a frequent complication of uveitis, occurring in up to one fourth of patients with chronic inflammation.47,48,49 Therefore, it is essential to measure the IOP of the uveitis patient at each visit. Elevated IOP can be seen in any type of uveitis but is most common in Fuchs iridocyclitis, Vogt-Koyanagi-Harada syndrome, JIA-associated iridocyclitis, and iritis secondary to toxoplasmosis, herpes simplex, and herpes zoster.9,50 Glaucoma occurring with chronic uveitis can be either secondary open angle or closed angle, or a combination of both types. Careful gonioscopy of the uveitis patient with elevated IOP is therefore critical. Despite aggressive treatment, glaucoma may result in visual field defects in a significant proportion of uveitic patients.51
Open-angle glaucoma is the most common type of elevated IOP in patients with chronic uveitis.52 The chamber angle may become blocked by debris and inflammatory cells. Alternatively, the trabecular meshwork (TM) itself may become inflamed, reducing outflow facility. This is the proposed mechanism of elevated IOP in Posner-Schlossman syndrome (glaucomatocyclitic crisis). It has also been postulated that the endothelial cells of the TM are capable of phagocytosis. During periods of inflammation these cells may ingest debris, migrate off the trabecular beams, and eventually may be lost. This loss of endothelial cells would then be reflected in a decreased outflow facility and an increased IOP. An alternate theory has proposed that an increase in the aqueous humor protein level could lead to a blockage of the TM, creating a susceptibility to the development of glaucoma in uveitic patients.53
Another mechanism that may be responsible for increased IOP in uveitis patients is the so-called steroid response. This elevated IOP can occur with any route of steroid administration, but it is seen most often after topical use; it is least likely with systemic treatment. The IOP rise seems to be dependent on the type, frequency, and duration of treatment as well as on patient characteristics. It is seen in up to 30% of the general population after 3 to 4 weeks of treatment with topical steroids, and in a higher percentage of patients with a prior history of glaucoma. The IOP rise in glaucoma patients tends to be faster and of a greater magnitude than in nonglaucomatous eyes. The incidence is also high in patients with a family history of glaucoma, suggesting a genetic basis for the response.54,55,56,57,58,59 Other risk factors for steroid-induced glaucoma include increased age, type I diabetes, connective tissue diseases, and high myopia.60,61,62,63,64
Children may be more at risk for secondary glaucoma than adults.65 If it is not possible to obtain an IOP in the office in a child, an exam under anesthesia may be required.
The mechanism responsible for steroid-induced ocular hypertension has not been completely elucidated but does seem to result from either decrease in outflow facility54,55,56,57,58,66 or an increase in aqueous production.67 Outflow obstruction may result from a stabilization of lysosomal membranes, which in turn could cause an increased deposition of glycosaminoglycans in the TM, because lysosomal enzymes are, at least in part, responsible for glycosaminoglycan breakdown.68 While the genetic component is not completely understood, myocilin, a 55-kDa protein, also referred to as the TIGR or TM-inducible glucocorticoid response gene product, has been proposed to be involved in steroid-induced ocular hypertension due to its high expression in TM cells when exposed to glucocorticoids.69 The precise role of the myocilin gene remains elusive, but its mutation has been linked to both juvenile- and adult-onset primary open-angle glaucoma, and in human recombinant models it appears to increase the outflow resistance in the anterior segment.70,71
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree