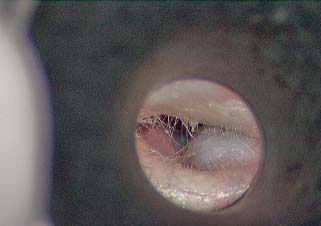
12 Complications of the Surgery The complications of cholesteatoma surgery relate to recurrence, residual, or recidivism of the disease, infection, cavity management, and loss of the specialized functions of the ear. Recurrence, residual, and recidivism are known complications of cholesteatoma surgery and are discussed in detail in Chapter 8. The frequency of these is somewhat dependent on the severity of the disease (cholesteatoma and infection) at the time of surgery. Recidivism is not always considered a surgical complication because it results from the aggressive nature of the disease, but it can be prevented (or its incidence lessened) by good surgical technique, so the surgeon should regard its occurrence as a complication of form. Postoperative infection may be a complication of surgery as in surgical procedures for other diseases, or because of the inherent nature of cholesteatoma, where infection is often present at the time of surgery, even in the absence of suppuration. Cholesteatoma harbors bacteria and often presents with purulent discharge. Despite the wish to operate on the ear while it is dry, it is sometimes necessary to remove the disease to bring the infection under control, and it is also possible to develop postoperative infection from dormant bacteria in the lesion. Chronic otitis media without cholesteatoma is likewise associated with chronic mucositis, osteitis, and perforation that allows for outside contamination. Postoperative infection manifests itself as pain, swelling of the auricle or the postauricular wound, purulent discharge, and occasionally fever. Sometimes the infection is not obvious until the packing is removed 1 week postop when pus underneath the packing and a fetid odor are noted. Once discovered, culture and Gram stain should be taken, all nonresorbable packing removed, and the patient started on oral antibiotics and drops. Gram-negative organisms (Pseudomonas, Proteus, Escherichia coli) and Staphylococcus aureus (sometimes methicillin resistant) are most common. Ciprofloxacin is a good initial choice of drug until cultures return. The chronic draining cavity can be a problem after canal wall down mastoidectomy. This may be caused by persistent granulation tissue, either along the lines of the meatal incisions, where new skin has failed to heal in, or at the base of the cavity, where persistent mastoid cells remain filled with mucosa rather than squamous epithelium. Treatment of the draining ear is by suctioning or swabbing wet material in the office setting (sometimes indecorously referred to as “toilet”), debriding any accumulated cerumen and squamous debris, and applying topical agents. A variety of medicaments have been devised for this purpose, but boric acid powder, applied through an atomizer, seems to work well as a bactericidal and drying agent. Other agents include wet or dry Gentian violet, a combination of antimicrobial agents in powder form such as CSTF (chloramphenicol, sulfa, tolnaftate, and Fungizone [Bristol-Myers Squibb, New York, NY]) powder, or 3% acetic acid drops. Acidification of the ear is initially attempted and then the antimicrobial agents are utilized if acidification does not reverse the process. Patches of granulation tissue can be lightly cauterized with silver nitrate (for larger areas) or 50% trichloroacetic acid (TCA). Care should be taken not to apply cauterizing agents to an exposed facial nerve. The draining ear may require frequent office visits at first, but the condition usually settles down over time in the majority of cases.1 Occasionally, the draining cavity can become refractory despite these measures, and revision surgery can be considered. The aim in these cases is to remove pockets of infected tissue, remove retained secreting air cells, lowering the posterior canal wall, removal of superior bony canal wall to the non-aircell tegmen, to provide drainage and aeration of the cavity and provide an adequate conchomeatoplasty to prevent retention of disease.2 A computed tomographic (CT) scan may be useful if focal osteitis is suspected. Sometimes a recurrent cholesteatoma may be discovered only during revision surgery because occasionally epithelial healing of the cavity will close over deep retraction areas, especially surrounding the otic capsule (superior to the superior semicircular canal or posterior semicircular canal). Revision surgery should address each of these elements. The principles should be followed as in primary canal wall down surgery (as outlined in Chapter 4, Creating a Manageable Cavity), namely, creating a round, shallow cavity with beveled edges, lowering the facial ridge, eliminating the mastoid tip, and creating a wide meatus. The latter cannot be overemphasized—a generous meatoplasty including removal of much of the conchal and occasional antihelical cartilage is important to long-term success. In addition, all cellular bone should be exenterated, and the depth of cavity polished with a coarse diamond burr. Sometimes a split-thickness skin graft is useful to resurface the floor of the cavity,3 but we have not often found this to be necessary. Fig. 12.1 A blue-domed cyst of the mastoid cavity (cholesterol granuloma). Cholesterol granuloma cysts are blue-domed mucosal cysts that form in the floor of the antrum or in the sinodural angle. If they are asymptomatic, these can simply be followed. Occasionally, though, they cause discharge or pain and should be removed. Usually they can be incised, evacuated, and marsupialized in an office setting, but they do tend to re-collect (Fig. 12.1). Meatal stenosis is usually an error of technique. The meatoplasty is an important step in canal wall down surgery. The technique of meatoplasty was reviewed in Chapter 5. The success of meatoplasty depends upon resecting (or at least breaking the spring of) the conchal cartilage, lengthening the canal incisions to create a conchomeatal flap, and using stay sutures to ensure meatal patency.4 When restenosis occurs, the meatoplasty might need to be revised. In these cases, a postauricular approach should be used, a strip of conchal cartilage is taken, and stay sutures should be placed between the subcutaneous tissue of the conchomeatal flap and the perichondrium of the concha. These sutures are important for keeping the meatus open. Nonresorbable packing should also be placed but should not be relied upon. Long-term stents can be used in stubborn cases. In canal wall up surgery, a posterior and superior bony canalplasty is often performed to better visualize the disease process. In these patients it is helpful to perform a limited meatoplasty by making an extended superior soft tissue canal incision and removing soft tissue and sometimes a rim of superior conchal cartilage. The intraoperative packing is important to maintain this opening, and Silastic sheeting may be used against the bone with interior packing. Reapproximating the periosteal incisions at the end of the procedure is also important to prevent meatal collapse. Medial stenosis can also occur. In canal wall up surgery, this is caused by subcutaneous fibrosis of distal canal skin, or by adhesions from the posterior canal skin to the anterior canal wall, or to “blunting” of the tympanic membrane in the anterior sulcus. Stenosis can be prevented by carefully redraping the skin flaps at the end of the surgical procedure, and by using nonadherent packing in the canal lumen. Blunting is a special and frequent problem that comes about when an overlay graft technique is used,5 or when the anterior rim of the tympanic membrane is lacking and the tympanic membrane graft is allowed to ride up the anterior bony canal. Carefully and accurately placing the graft under the anterior bony annulus may help prevent this. Once blunting has occurred, it will usually resist attempts at correction by revision surgery. Correction of blunting requires revision of the tympanoplasty by an experienced surgeon. After the anterior canal skin is incised and elevated, a fascial graft should be placed medial to the bony annulus of the ear canal, and a sliding graft of canal skin or partial-thickness skin graft is used to reconstruct the anterior and superior canal wall skin.6 Packing is important in these cases, and it is preferable to use Silastic sheeting followed by careful internal packing. Medial stenosis can also occur after canal wall down surgery. This usually takes the form of a membranous partition that forms across the attic or antrum. At times this is a useful development in that it limits the functional size of the cavity (by acting as a “soft wall” reconstruction), but it does create a potential space for recurrent disease to form that cannot be accessed through the meatus (an example of this is illustrated in Chapter 10, Fig. 10.9B). A thin membranous stenosis can often be lysed in the office using local anesthetic, but Gelfoam packing (Pfizer, Inc., New York, NY) in large strips should be placed for at least 2 weeks to prevent it from reforming. Residual cholesteatoma that forms behind a membranous partition will usually have to be removed in the operating room. Conductive hearing loss is not uncommon after surgery, even in patients who have good hearing at the time of initial presentation. It is often the case that the cholesteatoma–ossicular complex transmits sound as a solid mass even with incus and stapes erosion. Conductive hearing loss should not be regarded as a true complication because removal of the incus or separation of the ossicular chain cannot be avoided in many cases, and the success of ossicular reconstruction is never certain. As discussed in Chapter 6, preservation of an intact ossicular chain should not take precedence over complete removal of disease. If the disease recurs, the eventual hearing outcome will almost surely decline. Postoperative scarring with fibrosis of the middle ear, reduction of the size of the middle ear cleft because of lowering the posterior bony canal wall to the fallopian canal, and reducing the middle ear space can potentially prevent future middle ear hearing reconstruction. Staging surgery is accepted when there is intact canal wall surgery in extensive cholesteatoma. Staging is also performed in patients with significant middle ear disease and eustachian tube dysfunction. At the first stage Silastic sheeting is frequently utilized to prevent middle ear fibrosis and help maintain a middle ear space. Second stage surgery may be performed under intravenous sedation, and the middle ear is entered, Silastic removed, and ossicular reconstruction performed. When it is necessary to remove the posterior canal wall, it is frequently possible to lower the canal wall just to the level of the chorda tympani inlet, still removing the sinus tympani or hypotympanum disease, therefore preserving a middle ear space for immediate or second-stage reconstruction. At a second stage the sheeting is removed and it is hoped that a middle ear cleft is retained. Then ossicular reconstruction may be performed under intravenous sedation technique. A hearing aid can be offered to the patient once the ear is dry and stable. Bone-anchored hearing aid (BAHA, Cochlear Americas, Centennial, CO) is an option in chronically draining ears that cannot accept a hearing aid.7 A BAHA provides excellent hearing results when the middle ear cannot be reconstructed and when bone conduction is good, but it requires a second operation to implant a titanium post and the use of an external processor. Round window implantation with the Vibrant device (MedEl, Durham, NC) is a newer option for hearing rehabilitation. This technique, in which a floating-mass transducer is placed against the round window membrane, introduced by Colletti et al8 as a means of restoring hearing by bypassing the middle ear conductive mechanism, produced very encouraging initial hearing outcomes, although other authors have reported mixed results.9 Sensorineural hearing loss or deafness is fortunately not common after ear surgery. Preoperative sensorineural loss may be a feature of chronic middle ear infection, caused by the spread of inflammation to the inner ear. High-frequency postoperative sensorineural hearing loss occasionally occurs and may be undetected by the patient, but complete loss of hearing is a fairly serious complication. The incidence is increased when a fistula of the inner ear is present (labyrinthine or cochlear) from the disease, and especially in the presence of active infection. If a fistula is present at the time of surgery, the matrix should be kept on the fistula and canal wall down surgery performed, or rarely a second-stage surgery is performed with the matrix intact. At the second stage bony regrowth will occur. Iatrogenic opening of the inner ear may occur if the semicircular canal is opened during drilling, or if the round window membrane is perforated during middle ear dissection, or if the annular ligament is broken while the surgeon is manipulating the stapes. When these events occur and are recognized during the surgery, immediate sealing of the inner ear with a fascial graft will usually prevent a significant cochlear loss.10 The pathogenesis of sensorineural hearing loss in these cases may be toxic or suppurative labyrinthitis, perilymph leak, or inner ear fibrosis. When labyrinthitis occurs postoperatively, steroids may forestall the progression of sensorineural hearing loss, but once this has occurred it is probably irreversible. High-frequency sensorineural hearing loss may result from vibrational trauma during drilling, particularly if the ossicular chain is intact. There is a potential for neurosensory hearing loss secondary to acoustic trauma of drilling (temporary or permanent threshold shift). Suction irrigation with drill sound may generate 100 dB levels for several minutes of drilling near the cochlea. There have been studies that have examined the pathophysiology of this phenomenon.11,12 Vibrations transmitted through the skull bones are probably not sufficient to cause a permanent threshold shift, but drilling directly on the ossicular chain might be. To prevent this, care should be taken to avoid touching the drill to the ossicles in the attic (the cholesteatoma may provide a protective cushion), reducing the bone to an eggshell thickness that can be manually removed with a curette. Incus erosion is common in the presence of cholesteatoma, but sometimes an intact ossicular chain can be found even when the cholesteatoma is large. The status of the incudostapedial joint can be determined at the start of the case by raising a tympanomeatal flap, and it may be safer to separate the incudostapedial joint before drilling.10 Tinnitus in the early postoperative period may be related to middle ear swelling and often recovers when the surgical packing is removed. Patients who have tinnitus preoperatively may experience louder tinnitus postoperatively. Fortunately this is usually transient. High-pitched tinnitus is usually a manifestation of cochlear loss and may result from vibrational trauma. Tinnitus can be a vexing problem for patients and does not have an outright cure. Ambient masking is helpful, and a masking device may be beneficial for some patients. Benzodiazepines can alleviate anxiety and insomnia and often lessen the symptom. Tricyclic antidepressants (nortriptyline, amitriptyline) are also helpful and may act centrally to suppress tinnitus, rather than by an antidepressant effect. Dizziness and imbalance are not frequent. Like sensorineural loss, vestibular symptoms may arise from labyrinthitis or perilymph leakage. The chance of vertigo increases if an inner ear fistula is present. Vestibular symptoms may also result from surgical trauma to the inner ear, such as excessive manipulation of the stapes. Postoperative vertigo is usually transient and can be managed with vestibular suppressant medications such as meclizine, promethazine, or diazepam. Unilateral vestibular loss that is permanent generally recovers as central compensation takes hold. Imbalance will subside gradually and completely as activity is increased. Vestibular rehabilitation physical therapy may be helpful in cases that are slow to recover. Dysgeusia arises from surgical trauma to the chorda tympani nerve. Patients may complain of a metallic or altered taste on that side of the tongue, or dryness of the mouth. Its occurrence cannot be predicted. The chorda tympani is often enveloped by the cholesteatoma and must be sacrificed, and yet dysgeusia is not inevitable.13 Stretch injury of the nerve may be more common than transection, and may result in greater symptoms.14,15 Dysgeusia is usually temporary but may take 3 months or longer to resolve. In cases of bilateral chorda tympani sacrifice it may be permanent. Facial paralysis is a dreaded complication that can arise from surgical trauma, the effect of local anesthesia, or the spread of infection to the nerve. It is more likely to occur when there is bony dehiscence of the fallopian canal, altered nerve anatomy, or extensive disease. The role of facial nerve monitoring and stimulation and the surgical technique of dissecting disease from the nerve have previously been covered. Proper technique demands an intimate knowledge of the course and anatomical position of the facial nerve. Monitoring is not a substitute for this.16 Most facial nerve injuries are first detected in the recovery room, not during the surgery.17 If the paralysis is incomplete, then the nerve is probably not transected, and the function will probably recover. Note that complete (passive) eye closure may persist for > 24 hours after a facial nerve transection, and it is important not to be misled by this.18 Partial or delayed facial nerve weakness should be treated with high-dose steroids, and both have a good prognosis for full recovery. If a patient awakens from surgery with complete facial paralysis, the surgeon must consider where in the course of the operation an injury could have occurred (it is good surgical practice to identify the facial nerve in every otologic procedure).19 If the disease was dissected off a bare nerve, the surgeon should have visualized the nerve and known the condition of the nerve at the end of the dissection. If the nerve was known to be intact and not mechanically traumatized, and if the nerve stimulator produced a good response with stimulus applied proximal to the site of the dissection, then it can be presumed that there was a neuropraxic injury with a very good chance of full spontaneous recovery. In most cases, however, the status of the nerve is not that certain, and urgent reexploration is advisable. In general, a transmastoid approach should be used (even if the original operation was a tympanoplasty), and the entire nerve should be skeletonized from the first genu to the stylomastoid foramen.20 The area of nerve exposure should be inspected carefully. If the nerve appears intact but is edematous, a limited decompression of the bony canal should be conducted, using a small diamond drill, removing the bony covering both proximal and distal to the site of injury. The nerve stimulator should be applied proximal to the injury, starting at a low current level (0.05 mA) and increasing gradually to 2 mA until a response is obtained. Absence of response indicates severe neuropraxia and an uncertain prognosis. If the nerve is found to be disrupted, a limited decompression should similarly be undertaken, and the nerve stimulated proximally. If a response to stimulation is obtained, the nerve should be covered with a collagen sheath. If there is no response, the nerve should be carefully inspected. If less than 50% of the diameter of nerve is involved, the frayed edges should simply be reapproximated. If there is greater than 50% involvement, primary repair should be undertaken, either using fine sutures (9–0 nylon or Prolene, Ethicon, Inc., Somerville, NJ) through the epineurial sheath or coapting the ends in the bony fallopian canal. If a section of the nerve is missing, an interposition graft should be taken from the great auricular nerve. The time course of return of function will be days to weeks for a neuropraxic injury, 6 to 12 months for a severed nerve in the middle ear. Neuropraxia will generally recover fully, whereas a nerve repair will usually result in partial recovery (House-Brackmann grade 3). It is very important to counsel the patient and family of the expected outcome, and to remain involved in the patient’s care during the postoperative period.21 Eye protection is important to prevent corneal dryness, and a lid-loading gold weight procedure22 may be beneficial if a long period of recovery is likely. The sigmoid sinus, internal jugular vein, and internal carotid artery are susceptible to surgical injury. The neurotologic skull base surgeon usually possesses firsthand experience in approaching and handling these structures, but the general otolaryngologist who operates on the ear may not. It is important therefore to know what to do if a vascular injury does arise. The sigmoid sinus is usually identified anatomically during mastoidectomy. There is a thin plate of bone overlying the sinus, and in cholesteatoma surgery this should be maintained. A cutting burr can be used to skeletonize the sigmoid provided the surgeon keeps a very light hand, but a diamond burr is safer. The sinus is a dural structure, and removing the bony covering does not necessarily lead to venous injury, but this may happen if the sinus is thin or bulging. Lacerating the sigmoid sinus causes profuse venous bleeding, of course, and the surgeon should be equipped to deal with this. The first measure is to control the bleeding with external pressure, using oxidized cellulose (Surgicel, Ethicon, Inc., Somerville NJ) gauze, a neurosurgical cottonoid, or even finger pressure. The bleeding can then be definitively controlled using an extraluminal plug of Gelfoam (Pfizer, Inc., New York, NY) soaked in thrombin. Using a 5F Baron suction in the nondominant hand to divert the blood, the Surgicel is slowly peeled away until the bleeding site is visualized. If it is very small it can be controlled with bipolar cautery. If it is 1 or 2 mm in size, a Gelfoam-thrombin plug can be gently inserted into the opening. If it is larger than 2 mm, a square of Gelfoam-thrombin can be placed extraluminally and held in place by sandwiching it between the edges of the bony opening and the sinus wall.23 This requires gently separating the dura from the bone with a blunt duckbill elevator prior to placing the packing. A still larger tear, should it ever occur, might require obliterating the sinus with extraluminal Surgicel packing, but this situation should be avoided if at all possible. Intraluminal packing should be avoided because it can embolize toward the heart; if this is ever necessary, the internal jugular vein should be ligated in the neck.20 The jugular bulb can also bleed if treated roughly, and bleeding from the jugular is handled in the same way. The jugular is encountered in two places—in the hypotympanum through the middle ear, and in the retrofacial cell tract through the mastoid. The jugular bulb is highly variable in position. A high jugular bulb can be injured during seemingly innocuous maneuvers, such as elevating a tympanomeatal flap, if it lies in a vulnerable location. The internal carotid artery is not often encountered during surgery for chronic ear disease, and injury to the carotid is rare. The carotid can be reliably found medial to the eustachian tube in the protympanum. The bony wall there is thin or can be absent, placing the carotid at risk in that location. The carotid has a thick muscular wall. Surgical injury when it occurs results from penetrating trauma; sharp instrumentation should therefore be avoided in that area. A laceration of the carotid artery will cause dramatic pulsatile bleeding. Temporary control can be gained with extraluminal pressure; definitive control will require an endovascular procedure performed by an interventional neuroradiologist. In a dire emergency, ligation of the internal carotid artery in the neck might be required. Cerebrospinal fluid (CSF) leakage is an uncommon but potentially serious complication because it can lead to meningitis. CSF leakage can result from removal of cholesteatoma matrix from thin dura or from surgical trauma during drilling. The tegmen is the most vulnerable site. Spontaneous dural dehiscences occur in at least 20% of chronic ears, and small encephaloceles are even more vulnerable to surgical trauma. CSF leakage can also occur spontaneously in association with arachnoid granulations, intracranial hypertension, meningoencephaloceles, or congenital inner ear anomalies. Once a CSF leak is discovered, it is imperative to repair it. Small dural tears can usually be repaired through a transmastoid approach, as described in Chapter 6. A multilayer repair using fascia and conchal cartilage in the epidural space will usually be successful. Occasionally it may be necessary to use one of the manufactured onlay synthetic dural repair substances, which are conformable and resorbable membrane with collagen. (ie, Durepair, Medtronic Sofamor Danek, Inc., Memphis, TN; DuraMatrix Stryker, Kalamazoo, MI; DuraGen, Integra NeuroScience, Plainsboro, NJ) Rarely, the mastoid cavity may have to be packed with abdominal fat in a canal wall up procedure. It is very important when obliterating the mastoid to be certain that no residual disease remains. In a canal wall down, the area of leakage will be open to the outside. The option exists of obliterating the ear with fat and oversewing the meatus, but this will compromise hearing as well as postoperative surveillance. Hydroxyapatite bone cement can also be used but must be completely covered with a soft tissue flap prior to closure. Large encephaloceles, multiple dural tears, or leaks that are inaccessible from below can be repaired via transcranial route. This can be done through an extradural approach using a middle fossa craniotomy (or minicraniotomy), elevating the dura bluntly until the area of the dural tear is reached. An oversized fascia graft can then be sandwiched into the epidural space, and a piece of cartilage, split calvarial bone, or hydroxyapatite disc can be used to repair the tegmen from above. These measures will control most CSF leaks. In cases of profuse or recurrent leaks, a lumbar drain can be inserted for 3 to 5 days to reduce CSF pressure and allow the defect to seal, and an intradural repair can be performed via temporal craniotomy by the neurosurgeon, although not usually necessary. Cholesteatoma surgery is challenging, and even the most experienced surgeon may encounter complications. The neurosensory organs and vascular structures of the temporal bone require delicate and precise surgical technique. Preparedness in knowing how to deal with problems in the operating room and in the postoperative period is essential to maximizing surgical outcomes. 1. Kos MI, Castrillon R, Montandon P, Guyot JP. Anatomic and functional long-term results of canal wall-down mastoidectomy. Ann Otol Rhinol Laryngol 2004;113 (11):872–876 2. Jackson CG, Schall DG, Glasscock ME III, Macias JD, Widick MH, Touma BJ. A surgical solution for the difficult chronic ear. Am J Otol 1996;17 (1): 7–14 3. Ramsey MJ, Merchant SN, McKenna MJ. Postauricular periostealpericranial flap for mastoid obliteration and canal wall down tympanomastoidectomy. Otol Neurotol 2004;25 (6):873–878 4. Paparella MM, Meyerhoff WL. “How I do it”—otology and neurology: a specific issue and its solution. Meatoplasty. Laryngoscope 1978;88 (2 Pt 1):357–359 5. Sheehy JL, Glasscock ME III. Tympanic membrane grafting with temporalis fascia. Arch Otolaryngol 1967;86 (4):391–402 6. Hough JV. Revision tympanoplasty including anterior perforations and lateralization of grafts. Otolaryngol Clin North Am 2006;39 (4): 661–675, v 7. Macnamara M, Phillips D, Proops DW. The bone anchored hearing aid (BAHA) in chronic suppurative otitis media (CSOM). J Laryngol Otol Suppl 1996;21:38–40 8. Colletti V, Carner M, Colletti L. TORP vs round window implant for hearing restoration of patients with extensive ossicular chain defect. Acta Otolaryngol 2009;129 (4):449–452 9. Beltrame AM, Martini A, Prosser S, Giarbini N, Streitberger C. Coupling the Vibrant Soundbridge to cochlea round window: auditory results in patients with mixed hearing loss. Otol Neurotol 2009;30 (2):194–201 10. Palva T, Kärjä J, Palva A. Immediate and short-term complications of chronic ear surgery. Arch Otolaryngol 1976;102 (3):137–139 11. Urquhart AC, McIntosh WA, Bodenstein NP. Drill-generated sensorineural hearing loss following mastoid surgery. Laryngoscope 1992;102 (6):689–692 12. Migirov L, Wolf M. Influence of drilling on the distortion product otoacoustic emissions in the non-operated ear. ORL J Otorhinolaryngol Relat Spec 2009;71 (3):153–156 13. Clark MPA, O’Malley S. Chorda tympani nerve function after middle ear surgery. Otol Neurotol 2007;28 (3):335–340 14. Gopalan P, Kumar M, Gupta D, Phillipps JJ. A study of chorda tympani nerve injury and related symptoms following middle-ear surgery. J Laryngol Otol 2005;119 (3):189–192 15. Michael P, Raut V. Chorda tympani injury: operative findings and postoperative symptoms. Otolaryngol Head Neck Surg 2007;136 (6): 978–981 16. Prass RL. Iatrogenic facial nerve injury: the role of facial nerve monitoring. Otolaryngol Clin North Am 1996;29 (2):265–275 17. Green JD Jr, Shelton C, Brackmann DE. Surgical management of iatrogenic facial nerve injuries. Otolaryngol Head Neck Surg 1994;111 (5):606–610 18. Graham MD. Prevention and management of iatrogenic facial palsy. Am J Otol 1984;5 (6):513 19. Glasscock ME III, Wiet RJ, Jackson CG, Dickins JR. Rehabilitation of the face following traumatic injury to the facial nerve. Laryngoscope 1979;89 (9 Pt 1):1389–1404 20. Weber PC. Iatrogenic complications from chronic ear surgery. Otolaryngol Clin North Am 2005;38 (4):711–722 21. Pulec JL. Iatrogenic facial palsy: the cost. Ear Nose Throat J 1996;75 (11): 730–736 22. Bojrab DI. ___ Insights in Otolaryngology 1989;4 (4):1–5 23. Brackmann DE. Iatrogenic injuries in temporal bone surgery. In: Britton BH, ed. Common Problems in Otology. St. Louis: Mosby; 1991:115–119
♦ Recurrence, Residual, Recidivism
♦ Infection
♦ Meatal Stenosis
♦ Medial Stenosis
♦ Hearing Loss (Conductive and Neurosensory)
♦ Tinnitus
♦ Dizziness and Imbalance
♦ Dysgeusia
♦ Facial Paralysis
♦ Vascular Injury
♦ Cerebrospinal Fluid Leak
♦ Conclusion
References
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


