11 Complications of the Disease Acute and chronic otitis media, with or without cholesteatoma, consists of inflammation of the pneumatized spaces of the temporal bone, which include the eustachian tube, middle ear, and mastoid air cell system. By convention, the definition of a complication of otitis media is the spread of infection outside the temporal bone. The most commonly used classification includes intratemporal (or “aural”) and intracranial complications (Table 11.1). Intratemporal complications include mastoiditis, labyrinthitis, facial paralysis, and petrositis. Intracranial complications include meningitis, brain abscess, epidural abscess, subdural abscess, sigmoid (or lateral) sinus thrombosis, and otitic hydrocephalus. It is commonly understood that complications of otitis media have become quite rare since the advent of antibiotics. However, because of their low incidence, complications are sometimes overlooked, or their diagnosis is delayed because of lack of recognition. For example, children with facial paralysis secondary to otitis media are sometimes mistakenly treated for Bell palsy, without attention given to treating the middle ear suppuration. It is therefore imperative that otologists who treat inflammatory middle ear disease be familiar with all the complications and their proper management. Many of the complications of otitis media nowadays are seen in poorer urban centers,1 or in immigrants from developing countries.
Aural Complications | Intracranial Complications |
Mastoiditis | Meningitis |
Labyrinthitis | Epidural abscess |
Facial paralysis | Subdural abscess |
Petrositis | Brain abscess |
Sigmoid sinus thrombosis | |
Otitic hydrocephalus |
♦ Mastoiditis
Mastoiditis occurs when there is coalescence of mastoid air cells. Mastoiditis is the process of liquefactive necrosis of bone. It can occur after acute or chronic otitis media, but the chronic form usually involves cholesteatoma. Otitis media is a suppurative infection of the middle ear. It passes through stages of hyperemia, exudation, suppuration, and (usually) resolution. In the rare event that the suppurative process does not resolve, liquefactive necrosis of bone can occur and an abscess can form. In the most common scenario, the pus erodes the mastoid cortex near the MacEwan triangle and forms a subperiosteal abscess; rarely it can erode through other weak points in the temporal bone—anteriorly at the zygomatic root, or inferiorly through the digastric ridge into the deep spaces of the neck (Bezold abscess).2
Cholesteatoma permits the formation of coalescent mastoiditis by blocking the egress of pus from the mastoid into the middle ear. The so-called attic-antral block causes pus to accumulate in the mastoid under pressure and erode the cortical bone to escape into the subperiosteal or neck spaces. Acute mastoiditis associated with cholesteatoma is relatively rare, occurring in approximately one of 40 cases of mastoiditis.3
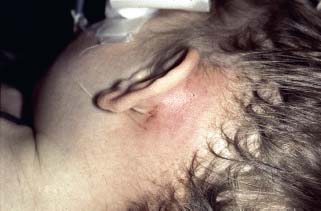
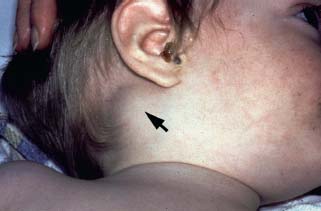
Acute mastoiditis presents in an acutely ill patient, often with fever and systemic signs. Mastoiditis that develops after acute otitis media classically occurs 2 weeks after the acute infection, but mastoiditis that develops from chronic suppurative otitis media can occur at any time in the course of the disease.4 The clinical signs of a subperiosteal abscess include a tender, fluctuant swelling in the postauricular sulcus, protrusion of the auricle, and “sagging” of the posterior canal wall (Fig. 11.1). A Bezold abscess, less commonly seen, presents with tenderness, redness, swelling, and stiffness of the neck (Fig. 11.2).
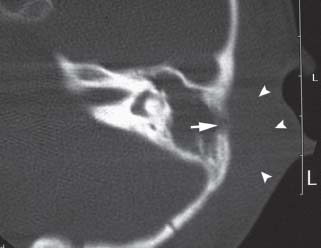
The radiologic hallmark of mastoiditis is coalescence, or loss of the bony septations of the mastoid (N.B.: radiologists will often read opacification of the mastoid air cells as “mastoiditis”; while this is technically correct in the radiologic sense, acute or serous otitis media routinely causes diffuse opacification of the mastoid air cells, and this is not equivalent to “mastoiditis” in the clinical sense). Plain films of the mastoid will demonstrate coalescence, but these have become scarcer as computed tomographic (CT) scanners have become ubiquitous. On CT, coalescence will appear as loss of bony septations (not different in appearance from any cholesteatoma); the abscess will appear as a dome-shaped fluid collection in the subperiosteal space, accompanied by a focal interruption of the bony cortex (Fig. 11.3).
The bacteriology of acute mastoiditis usually consists of pyogenic organisms, of which Streptococcus pneumoniae is most common. Chronic suppurative otitis media is more likely to be caused by Pseudomonas sp., other gram-negative rods. Staphylococcus aureus is also occasionally seen, and methicillin-resistant S. aureus (MRSA) is increasing in prevalence and can be difficult to eradicate.
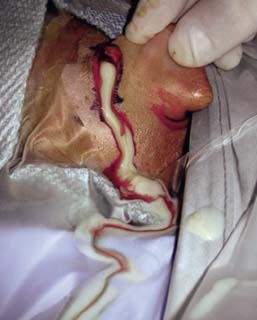
Treatment of mastoiditis consists of culture of the ear canal drainage, intravenous fluids and antibiotics, and urgent surgery to drain the abscess and remove its source. The surgical drainage of a subperiosteal abscess is performed through a postauricular incision (Fig. 11.4). The periosteum is usually very thick and edematous, and tented by the abscess. A stab incision of the periosteum usually releases pus under pressure, and the fluid should be evacuated and cultured, and the wound irrigated thoroughly. The periosteal incision is lengthened, and the mastoid cortex exposed. The cortex can be opened with a cutting burr. There may be more bleeding than usual, so constant suction-irrigation is advisable. The mastoidectomy is then completed and the cholesteatoma removed. If there is too much inflammation to do this safely, then after the mastoid is decorticated, the wound can be closed loosely over a Pen-rose drain and the completion mastoidectomy postponed for a week or two until the inflammation subsides.
Recent attention has been given to the nonsurgical management of acute mastoiditis, but this remains a matter of controversy.5 It is not clear that all these cases meet the criteria for acute mastoiditis—some cases of postauricular swelling may reflect periosteitis rather than a florid abscess. In any case, if cholesteatoma is present, surgical therapy is not optional and should be instituted as soon as the patient is stable to undergo the procedure.
♦ Labyrinthitis
Labyrinthitis is extension of inflammation to the inner ear and manifests clinically as sensorineural hearing loss and vestibular impairment. According to Schuknecht,6 infectious labyrinthitis can be toxic or suppurative. Toxic, or serous, labyrinthitis is caused by bacterial toxins that enter the fluids of the inner ear. The portal of entry may be the round or oval window membrane, or through an acquired fistula. The degree of cochlear and vestibular impairment is variable, as is the degree of recovery. Milder cases are characterized pathologically by endolymphatic hydrops and proteinaceous precipitates in the endolymph; more severe cases have degeneration of the sense organs, and so loss of cochlear and vestibular function may be permanent. Sometimes toxic labyrinthitis is diagnosed retrospectively, when sensorineural hearing loss occurs in the setting of chronic ear disease.
Suppurative labyrinthitis is characterized by polymorphonuclear cells in the perilymph and later in the endolymph. Necrosis of the membranous labyrinth can ensue, and the infection can spread intracranially. Fibrous, and later bony, obliteration of the labyrinth (labyrinthitis ossificans) can occur over time.
Chronic labyrinthitis is characterized by progressive destruction of the bony labyrinth, endolymphatic hydrops, proteinaceous exudates, and later degeneration of the sense organs and labyrinthine sclerosis.
Labyrinthine fistula can be caused by cholesteatoma and can be the portal of infection into the labyrinth (see Chapter 6).
Sensorineural hearing loss can be a late manifestation of chronic labyrinthitis in the presence of cholesteatoma. Sensorineural hearing loss has been found to occur with higher incidence in chronic otitis media than in the general population.7,8
♦ Facial Nerve Paralysis
The facial nerve is a hearty nerve with a myelin sheath, and so paresis caused by infection is not common. The facial nerve is protected by a bony shell in the middle ear. Natural dehiscences occur in at least 8% of cases,9 but the incidence is higher in the presence of cholesteatoma.10–12 Paresis of immediate onset usually signifies suppuration against a bare nerve. Paresis of gradual onset is the result of pressure against the nerve, usually by cholesteatoma.
The first circumstance requires urgent surgery—the goal should be to drain the pus. If the eardrum is intact and the patient has acute otitis media, a wide myringotomy should be performed and the middle ear fluid should be cultured and antibiotics started. This condition has a good prognosis provided the treatment is done as soon as possible after the onset of the problem.13 Myringotomy is usually sufficient treatment, unless the facial paralysis occurs in the setting of acute mastoiditis (bony coalescence with subperiosteal abscess); in this situation, mastoidectomy will be necessary to provide adequate surgical drainage.
On the other hand, when the onset of facial paresis is gradual, or if facial paralysis occurs in the setting of chronic suppurative otitis media, the prognosis is not as good. In these cases, surgery is indicated, with the goal of removing the infected focus from against the nerve (ie, to perform a tympanomastoidectomy to remove the cholesteatoma). In this event, the prognosis for recovery of facial nerve function is variable and depends on the length of time the paresis has been present before surgery. Paralysis that is present for 1 week or less usually recovers;14 paralysis of greater than 1 year does not.13 Paresis that develops slowly will usually take weeks to recover. If the paresis is long-standing, fibrosis will develop that will prevent the nerve from recovering completely.
Three cases are presented to illustrate these principles. In the first, a small child came to the emergency room with earache and fever and facial paralysis of acute onset. Examination revealed a red, bulging tympanic membrane. In a crying child, it is relatively easy to assess the status of the facial nerve (Fig. 11.5). He was started on antibiotics and a wide myringotomy was performed to evacuate pus from the middle ear. The infection resolved and the facial paralysis recovered fully within a few days.
The second case is an elderly woman who presented with gradual onset facial paralysis. She had a history of mastoid surgery in that ear 20 years earlier, but the details were not known. Examination revealed complete (grade VI) facial paralysis and drainage from the ear (Fig. 11.6). At surgery, a small pars tensa cholesteatoma was discovered medial to the incus and pressing against an exposed tympanic segment of the facial nerve. A canal wall down mastoidectomy was performed with removal of the incus along with the disease. The ear healed well and the facial nerve recovered completely within 3 months.
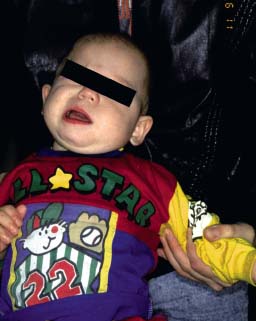
Fig. 11.5 Acute facial paralysis, left side, as a complication of acute otitis media in a child.
The third case is a 50-year-old man with chronic ear drainage and facial paralysis of gradual onset over 1 or 2 weeks. He had a radical mastoid cavity in his other ear for a history of cholesteatoma. At surgery, there was a tympanic membrane perforation and abundant granulation tissue with pus in the middle ear and mastoid. A canal wall up mastoidectomy with facial recess was performed, and the granulation tissue was completely removed, dissecting it off the exposed facial nerve (Fig. 11.7). Cultures showed S. aureus, and the patient was treated with ciprofloxacin orally and ofloxacin drops postoperatively. The paralysis recovered completely over a 3-week period. However, the ear drainage later recurred and the hearing did not improve.
♦ Petrositis
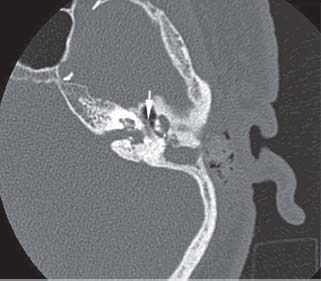
This rare complication occurs when the petrous apex air cells become infected, causing a triad of symptoms, termed Gradenigo syndrome, consisting of purulent ear discharge, retrobulbar pain (from trigeminal nerve irritation), and lateral rectus palsy (caused by sixth nerve involvement in the Dorello canal). The petrous apex cells are often pneumatized, and suppurative infection can become trapped there if there is outflow obstruction to the middle ear (Fig. 11.8). This situation might occur in the presence of giant cholesteatoma (“petrous temporal bone cholesteatoma”), or in cholesteatosis where the extensive disease involves the petrous apex cells (see Chapter 6). The treatment consists of surgical evacuation of the cholesteatoma from all the involved areas. The petrous apex is relatively difficult to approach surgically; this area communicates with the mastoid through infralabyrinthine, retrolabyrinthine, and supralabyrinthine cell tracts. The bony pathway created by the erosive disease will usually provide surgical access; in very advanced cases, a labyrinthectomy might be necessary to evacuate all the disease.15
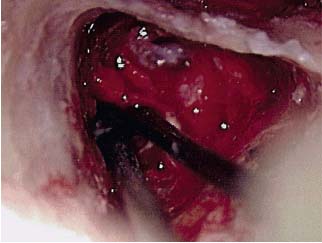
Fig. 11.7 Dissection of granulation tissue off the facial nerve, right ear.
Primary cholesteatomas of the petrous apex can also occur. 16 These are actually epidermoid tumors and are not related pathogenetically to the acquired cholesteatomas that are the principal subject of this book. These uncommon tumors appear radiologically as solid tumors of the petrous apex. They can present with any of the features of petrous apicitis, or with an intracranial mass effect, or as an incidental radiographic finding. On CT they appear as expansile lesions that erode the bony cortex. On magnetic resonance imaging (MRI), they have imaging characteristics that follow cerebrospinal fluid (CSF); they are hypodense on T1-weighted images, hyperintense on T2, and do not enhance with gadolinium. Surgical access can be gained through a transcochlear, posterior fossa or middle cranial fossa approach. These lesions are difficult to eradicate and readily regrow if they are not completely resected.
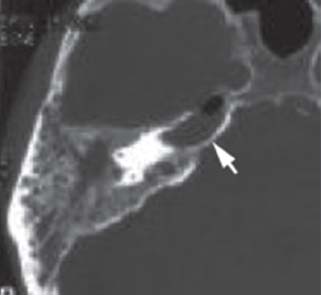
Fig. 11.8 Acute petrositis, opacification and air-fluid level of the right petrous apex (arrow).
♦ Intracranial Complications
Intracranial spread of infection from the mastoid can cause meningitis, brain abscess, epidural abscess, or subdural empyema. Spread of infection to the sigmoid sinus can cause septic thrombophlebitis of that structure (“lateral sinus thrombosis”), in addition to intracranial infection. Venous thrombosis may also result in increased intracranial pressure, or “otitic hydrocephalus.” Intracranial complications often may cause headache or fever without focal neurological signs. Although CT scanning is instrumental in determining the extent of disease in the mastoid, MRI is most useful in demonstrating intracranial complications.17
Meningitis can be otogenic in origin, occurring more commonly with acute otitis media than with chronic otitis or cholesteatoma,18 but it is not always clear that the middle ear process is causal.19 Infection can spread intracranially through bony channels or hematogenously.20 The diagnosis is made by lumbar puncture; CSF culture and sensitivity are mandatory, and treatment is by immediate administration of intravenous antibiotics. Pneumococcus has surpassed Haemophilus as the most common organism.18 Bedside myringotomy can be performed adjunctively if there is a bulging tympanic membrane, but if mastoiditis is present this should be treated by urgent mastoidectomy.20 Meningitis can lead to permanent sensorineural hearing loss and cochlear ossification, so steroid administration is recommended, especially in children.
Brain abscess classically presents with focal neurological signs or seizures, but nowadays the majority cause only headache or fever and the abscess is discovered on CT scan21 (Fig. 11.9). Otogenic brain abscesses occur either in the cerebellum or in the temporal lobe and develop by either hematogenous or contiguous spread. Neurosurgical consultation should be routinely sought. The treatment of the abscess consists of prolonged medical treatment with culture-appropriate antibiotics, in addition to single or repeated aspiration, or occasionally excision of the lesion.22 The ear pathology can be addressed surgically when the patient’s neurological status is stabilized. Needle aspiration can occasionally be performed through the mastoidectomy.23 Small abscesses may be treated with antibiotics alone. Streptococcus sp., anaerobes, and Enterobacter are the most commonly isolated organisms.21,24 Interestingly the ear and abscess cultures do not always concur.21
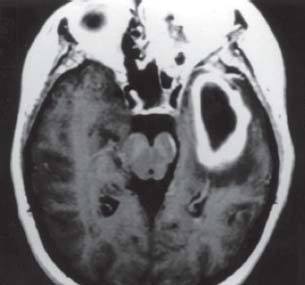
Fig. 11.9 A large brain abscess of the left temporal lobe, resulting from chronic otitis media.
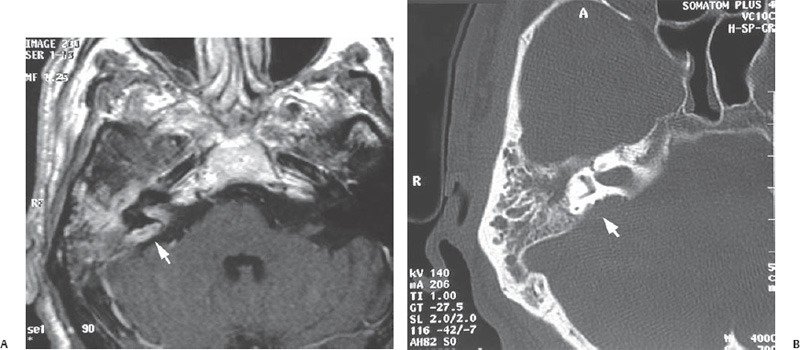
Epidural abscess occurs when there is erosion of the tegmen or posterior fossa bony plate by disease in the mastoid. Epidural granulation tissue is a fairly common finding during mastoidectomy for chronic ear disease, but a true collection of pus in the epidural space is relatively rare. Epidural abscess appears to be more common than brain abscess in the pediatric age group.15 The patient will have headache, fever, and focal meningeal signs. MRI will demonstrate the abscess (Fig. 11.10A),17 and CT will show the site of bony erosion (Fig. 11.10B). The abscess can be evacuated through a transmastoid approach by removing the overlying bone.
Lateral sinus thrombosis is a rare complication in which a septic thrombus forms in the lateral venous (sigmoid) sinus. The incidence has become very low in the antibiotic era, and the clinical presentation has changed. The classically described syndrome of headache, otorrhea, and spiking (“picket fence”) fevers is not often seen; instead patients are likely to present with headache and neurological findings including VI or VII nerve palsies or signs of increased intracranial pressure, without fever or ear complaints.25 Lateral sinus thrombosis can be seen in the clinical context of acute otitis media, with or without mastoiditis, or chronic otitis media, with or without cholesteatoma.
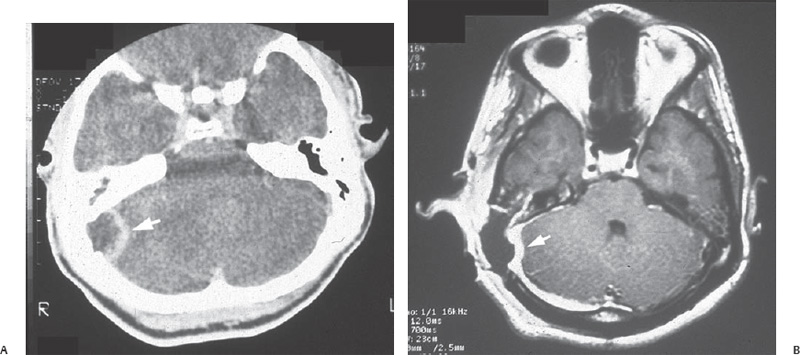
Imaging studies are helpful and may have eliminated the need for needle aspiration of the sinus in making the diagnosis. 26 CT of the temporal bones with contrast will demonstrate enhancement of the soft tissues around the sinus and a filling void within the lumen, termed a delta sign (Fig. 11.11A).27 MRI will display increased signal intensities on T1- and T2-weighted images indicating venous stasis (Fig. 11.11B), and gadolinium-enhanced MRI may outline the thrombus. Magnetic resonance venography will demonstrate the absence of flow. Doppler ultrasonography can demonstrate occlusion of the lateral sinus or extension of a thrombus along the internal jugular vein. MRI will also demonstrate any concurrent intracranial pathology; in one series, brain abscess, epidural abscess, and subperiosteal abscess were found to coexist in patients with lateral sinus thrombosis.26
Lateral sinus thrombosis should be regarded as a grave complication, and immediate treatment should be initiated with hospitalization and intravenous antibiotics. Mastoidectomy should be performed and the sigmoid sinus unroofed. Classically, the sinus is aspirated with a 25 gauge needle, and if no blood or pus is retrieved, the sinus is filleted open and the septic clot evacuated. Venous backbleeding can be controlled with extraluminal packing with oxidized cellulose or sclerosants.28 Syms et al have recently advocated nonsurgical management of the thrombosed sinus, contending that with intravenous antibiotics and low-dose heparin the sinus will eventually recanalize.26 Conservative management of the sinus was also successful in other series.25,27 Manolidis and Kutz, on the other hand, have argued that because of the increased prevalence of antibiotic resistance, evacuation of the thrombosed sinus is essential.28 The use of anticoagulation is controversial, but it is probably warranted if thrombosis extends to the torcula, because thrombosis of the superior sagittal sinus is a grave complication.
Otitic hydrocephalus is a rare and poorly understood condition, characterized by signs of increased intracranial pressure (headache, nausea, papilledema), usually developing insidiously.29 Occasionally the cause will be lateral sinus thrombosis, as proven by CT/MRI, and this should be treated accordingly. In other cases, the otogenic source will be in question. Otitic hydrocephalus is treated with steroids, acetazolamide, and sometimes serial lumbar puncture for CSF drainage.
♦ Conclusion
Complications of otitis media and cholesteatoma have become rare but continue to occur sporadically and must be recognized and treated promptly and aggressively. Mastoiditis occurs when there is bony coalescence; though early cases might successfully be treated with antibiotics alone, the presence of a subperiosteal abscess almost always necessitates surgery, and cholesteatoma when present must always be removed. Labyrinthitis can take on different degrees of aggressiveness; permanent loss of sensorineural and vestibular function often results despite treatment. Facial nerve paralysis requires wide myringotomy in the setting of purulent otitis media, and surgical mastoidectomy with nerve exploration when cholesteatoma is present, in addition to antibiotics and steroids. Petrositis is rare and demands surgical drainage, with the route selected according to the patient’s anatomy. Intracranial complications are grave and require aggressive medical treatment. Surgical drainage is needed for lateral sinus thrombosis and epidural abscess, and for removal of cholesteatoma. Brain abscess requires neurosurgical consultation; these are treated with repeated aspiration, or nonoperatively in selected cases.
References
1. Greenberg JS, Manolidis S. High incidence of complications encountered in chronic otitis media surgery in a U.S. metropolitan public hospital. Otolaryngol Head Neck Surg 2001;125 (6):623–627
2. Smouha EE, Levenson MJ, Anand VK, Parisier SC. Modern presentations of Bezold’s abscess. Arch Otolaryngol Head Neck Surg 1989;115 (9): 1126–1129
3. Shaffer HL, Gates GA, Meyerhoff WL. Acute mastoiditis and cholesteatoma. Otolaryngology 1978;86 (3 Pt 1):ORL394–ORL399
4. van den Aardweg MTA, Rovers MM, de Ru JA, Albers FW, Schilder AG. A systematic review of diagnostic criteria for acute mastoiditis in children. Otol Neurotol 2008;29 (6):751–757
5. Niv A, Nash M, Peiser J, et al. Outpatient management of acute mastoiditis with periosteitis in children. Int J Pediatr Otorhinolaryngol 1998; 46 (1-2):9–13
6. Schuknecht HF. Pathology of the Ear. 2nd ed. Malverne, PA: Lea and Febiger; 1993:211–217
7. da Costa SS, Rosito LP, Dornelles C. Sensorineural hearing loss in patients with chronic otitis media. Eur Arch Otorhinolaryngol 2009;266 (2): 221–224
8. Eisenman DJ, Parisier SC. Is chronic otitis media with cholesteatoma associated with neurosensory hearing loss? Am J Otol 1998;19 (1):20–25
9. Bayazit YA, Ozer E, Kanlikama M. Gross dehiscence of the bone covering the facial nerve in the light of otological surgery. J Laryngol Otol 2002;116 (10):800–803
10. Moody MW, Lambert PR. Incidence of dehiscence of the facial nerve in 416 cases of cholesteatoma. Otol Neurotol 2007;28 (3):400–404
11. Lin JC, Ho KY, Kuo WR, Wang LF, Chai CY, Tsai SM. Incidence of dehiscence of the facial nerve at surgery for middle ear cholesteatoma. Otolaryngol Head Neck Surg 2004;131 (4):452–456
12. Selesnick SH, Lynn-Macrae AG. The incidence of facial nerve dehiscence at surgery for cholesteatoma. Otol Neurotol 2001;22 (2):129–132
13. Makeham TP, Croxson GR, Coulson S. Infective causes of facial nerve paralysis. Otol Neurotol 2007;28 (1):100–103
14. Quaranta N, Cassano M, Quaranta A. Facial paralysis associated with cholesteatoma: a review of 13 cases. Otol Neurotol 2007;28 (3): 405–407
15. Isaacson B, Kutz JW, Roland PS. Lesions of the petrous apex: diagnosis and management. Otolaryngol Clin North Am 2007;40 (3):479–519, viii
16. Muckle RP, De la Cruz A, Lo WM. Petrous apex lesions. Am J Otol 1998;19 (2):219–225
17. Dobben GD, Raofi B, Mafee MF, Kamel A, Mercurio S. Otogenic intracranial inflammations: role of magnetic resonance imaging. Top Magn Reson Imaging 2000;11 (2):76–86
18. Migirov L, Duvdevani S, Kronenberg J. Otogenic intracranial complications: a review of 28 cases. Acta Otolaryngol 2005;125 (8):819–822
19. Smith JA, Danner CJ. Complications of chronic otitis media and cholesteatoma. Otolaryngol Clin North Am 2006;39 (6):1237–1255
20. Dubey SP, Larawin V, Molumi CP. Intracranial spread of chronic middle ear suppuration. Am J Otolaryngol 2010;31 (2):73–77
21. Couloigner V, Sterkers O, Redondo A, Rey A. Brain abscesses of ear, nose, and throat origin: comparison between otogenic and sinogenic etiologies. Skull Base Surg 1998;8 (4):163–168
22. Isaacson B, Mirabal C, Kutz JW Jr, Lee KH, Roland PS. Pediatric otogenic intracranial abscesses. Otolaryngol Head Neck Surg 2010;142 (3): 434–437
23. Wanna GB, Dharamsi LM, Moss JR, Bennett ML, Thompson RC, Haynes DS. Contemporary management of intracranial complications of otitis media. Otol Neurotol 2010;31 (1):111–117
24. Tandon S, Beasley N, Swift AC. Changing trends in intracranial abscesses secondary to ear and sinus disease. J Laryngol Otol 2009;123 (3): 283–288
25. Bales CB, Sobol S, Wetmore R, Elden LM. Lateral sinus thrombosis as a complication of otitis media: 10-year experience at the children’s hospital of Philadelphia. Pediatrics 2009;123 (2):709–713
26. Syms MJ, Tsai PD, Holtel MR. Management of lateral sinus thrombosis. Laryngoscope 1999;109 (10):1616–1620
27. Kutluhan A, Kiri M, Yurtta
M, Yurtta V, Kiro
V, Kiro lu AF, Unal O. When can lateral sinus thrombosis be treated conservatively? J Otolaryngol 2004;33 (2): 107–110
lu AF, Unal O. When can lateral sinus thrombosis be treated conservatively? J Otolaryngol 2004;33 (2): 107–110
28. Manolidis SJ, Kutz JW Jr. Diagnosis and management of lateral sinus thrombosis. Otol Neurotol 2005;26 (5):1045–1051
29. Kuczkowski J, Dubaniewicz-Wybieralska M, Przewo ny T, Narozny W, Mikaszewski B. Otitic hydrocephalus associated with lateral sinus thrombosis and acute mastoiditis in children. Int J Pediatr Otorhinolaryngol 2006;70 (10):1817–1823
ny T, Narozny W, Mikaszewski B. Otitic hydrocephalus associated with lateral sinus thrombosis and acute mastoiditis in children. Int J Pediatr Otorhinolaryngol 2006;70 (10):1817–1823
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


