CHAPTER 140 Complications of Temporal Bone Infections
Epidemiology
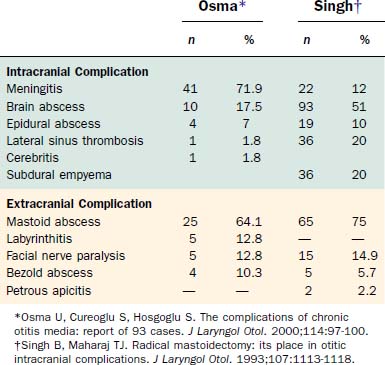
Table 140-1 presents the age distribution of extracranial, intracranial, and combined complications in a large series of patients. Nearly 80% of extracranial complications and 70% of intracranial complications occurred in children in the first 2 decades of life. Extracranial complications, led by postauricular abscess, most commonly occurred in children younger than 6 years.1 In a series of 93 intracranial and extracranial complications of otitis media, 58% were present in patients younger than 20 years.2 Low socioeconomic status and overcrowding confer either greater risk of or diminished resistance to an infection with an extended course and complications. Associations with inadequate health education and limited access to medical care likely contribute to heightened risk of complication. For this reason, most of the current reports of otogenic brain abscesses come from underdeveloped countries. Although the number of cases with immunodeficiency has increased, large series of patients with complications of otitis media have not been reported in patients with human immunodeficiency virus infection or acquired immunodeficiency syndrome, or in patients receiving immunosuppressive therapy after organ transplantation, even though such individuals do have heightened risks of suppurative ear disease.
Table 140-1 Age Distribution of 268 Patients with Complications of Otitis Media between January 1985 and December 1990
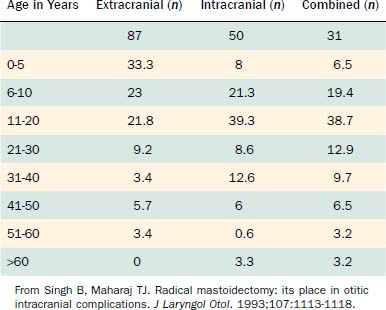
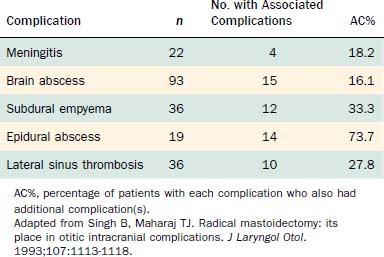
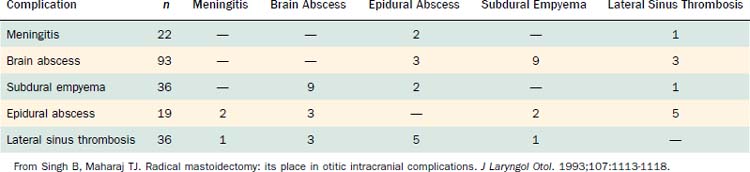
Table 140-2 shows the classification of extracranial and intracranial complications, and Table 140-3 summarizes the relative frequencies of those complications. The dominant extracranial complication is postauricular abscess, and the dominant intracranial complication is meningitis. Complications tend to occur multiply, especially intracranial complications, as shown in Tables 140-4 and 140-5. Although all of the complications originate from infection in the pneumatized spaces of the middle ear and mastoid, the mechanisms by which complications occur in AOM differ from the mechanisms associated with COM. We discuss these two entities separately.
Table 140-2 Classification of Complications of Acute and Chronic Otitis Media
| Extracranial | Intracranial |
|---|---|
| Acute mastoiditis | Meningitis |
| Coalescent mastoiditis | Brain abscess |
| Chronic mastoiditis | Subdural empyema |
| Masked mastoiditis | Epidural abscess |
| Postauricular abscess | Lateral sinus thrombosis |
| Bezold abscess | Otitic hydrocephalus |
| Temporal abscess | |
| Petrous apicitis | |
| Labyrinthine fistula | |
| Facial nerve paralysis | |
| Acute suppurative labyrinthitis | |
| Encephalocele and cerebrospinal fluid leakage |
From Harker LA. Cranial and intracranial complications of acute and chronic otitis media. In: Snow JB, Ballenger JJ, eds: Ballenger’s Otorhinolaryngology Head and Neck Surgery. 16th ed. Hamilton, Ontario: Decker; 2003.
Acute Otitis Media
An estimated 85% of all children experience at least one episode of AOM, making it the most common bacterial infection of childhood.3 Predisposing factors include young age; male sex; receiving bottle feedings; and being exposed to a daycare environment, crowded living conditions, or smoking within the home. Medical conditions such as cleft palate; Down syndrome; and mucous membrane abnormalities such as cystic fibrosis, ciliary dyskinesia, and immunodeficiency states also predispose individuals to otitis media.
After the first few weeks of life, acute suppurative otitis media is caused primarily by three organisms: Streptococcus pneumoniae, Haemophilus influenzae, and Branhamella catarrhalis, composing roughly 30%, 20%, and 10% of isolates.3 Optimal treatment for acute suppurative otitis media with complications includes appropriate antibiotics in addition to myringotomy and placement of a ventilating tube. Tympanocentesis is used primarily to obtain material for culture and sensitivity to identify the offending organism, but it can also reduce the bacterial population. Myringotomy and tube placement also provide material to identify the involved organism. After treatment, the physician should document that the AOM has completely resolved by tympanometry and otoscopy if the tympanic membrane is intact, and by otoscopy if a ventilating tube is in place. If the complication was intracranial, a computed tomography (CT) scan or magnetic resonance imaging (MRI) study should be obtained.
Chronic Otitis Media
COM changes are accompanied by a characteristic bacteriology compared with acute conditions. Harker and Koontz4 cultured 30 cholesteatomas at surgery and isolated at least one anaerobic organism in 67% of the cases, at least one aerobic organism in 70%, and both organisms in 50%. In 57% of the cholesteatomas, multiple organisms were cultured; in 30%, five or more bacteria were identified. Even without clinical infection, anaerobes, such as Propionibacterium acnes, were frequently isolated. An ear with COM is highly likely to harbor multiple bacteria of anaerobic and aerobic types.
Diagnosis
History
Intracranial complications, most notably meningitis, intraparenchymal brain abscess, and subdural empyema, can alter the patient’s level of consciousness. Of patients in the study by Singh and Maharaj,1 15% were drowsy on admission, 18% were stuporous, and 2% presented in a comatose state. Establishing the chronology of this alteration of sensorium helps the physician differentiate among diagnoses of brain abscess, meningitis, and subdural empyema. A brain abscess takes weeks to develop, whereas it takes only a few hours to several days for meningitis and subdural empyema to become fulminant and progress to coma.
Imaging Techniques
CT scanning is essential for all patients suspected to have complications of otitis media. CT is a fast and reliable method for assessing the status of the middle ear and the mastoid air cell system and diagnosing intracranial complications of otitis media.5,6 CT reveals bony details of the middle ear, epitympanic, and mastoid structures, and documents pneumatization versus opacification by inflammatory process. CT can show progressive demineralization and loss of the bony septa of air cells in coalescent mastoiditis, and reveal erosion of the bony plates covering the sigmoid sinus, cerebellum, or tegmen of the middle ear, mastoid, and bony labyrinth itself.
Treatment
A mastoidectomy under these circumstances is hampered by inflammation, and landmarks can be obscured. When no cholesteatoma is associated with the mastoiditis, the external auditory canal wall can be left intact unless visibility is inadequate. An open cavity, canal wall down procedure is preferred in the presence of cholesteatoma.1
Extracranial (Intratemporal) Complications
Acute Mastoiditis
Acute mastoiditis can develop when AOM fails to resolve. According to Luntz and colleagues,7 acute mastoiditis exists when there are signs of AOM on otoscopy and local inflammatory findings over the mastoid process (e.g., pain, erythema, tenderness, auricular protrusion), or when the mastoid inflammatory changes coexist with radiographic or surgical findings of mastoiditis with or without evidence of AOM. Other authors insist on the concomitant presence of AOM, mastoid physical findings, and radiologic findings.8
Luntz and colleagues7 reported results of a multicenter retrospective study of 223 such patients that provided valuable insight into the process. Of patients, 28% were younger than 1 year at diagnosis, 38% were 1 to 4 years old, and 21% were 4 to 8 years old. Although one third of patients experienced signs and symptoms of AOM immediately preceding the mastoiditis, two thirds did not. Thirty percent of the patients had a history of recurrent AOM, and 5% (all of whom had recurrent AOM) had a prior episode of acute mastoiditis.
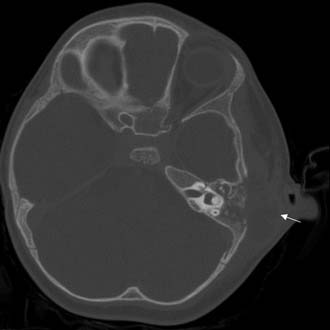
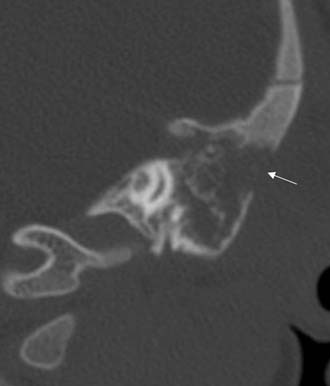
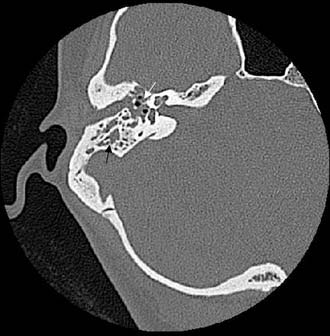
One third of the patients exhibited symptoms for 48 hours or less before diagnosis, and another third had symptoms for 2 to 6 days before presenting with acute mastoiditis. Spontaneous tympanic membrane perforation occurred in less than one fourth of patients; the tympanic membrane was bulging or erythematous in two thirds. Twenty-two percent of the patients presented with complications on admission, the most common of which was subperiosteal abscess (Fig. 140-1), followed by meningoencephalitis and occasional cases of other complications.
Bak-Pedersen and Ostri9 reviewed the records of 79 patients who underwent acute cortical mastoidectomy for acute mastoiditis. All patients had erythema and swelling and pain over the mastoid associated with a current or recent episode of AOM that did not improve after 24 to 48 hours of intravenous antibiotics. The average age in the series was 16 months, and the average duration from the onset of disease (AOM) until admission for acute mastoiditis was 9 days. Only one third of the patients exhibited an asymptomatic interval between AOM symptoms and the mastoiditis.
Van Zuijlen and colleagues10 pointed out that in countries such as the Netherlands, where it is unusual to prescribe antibiotics as first-line treatment for AOM, the incidence of acute mastoiditis is considerably higher than in countries where antibiotics are routinely prescribed for AOM. Lower overall costs and reduced incidence of allergic reactions resulting from withholding antibiotics in routine AOM must be weighed against heightened risks of mastoiditis and other complications.
Coalescent Mastoiditis
Pathology
Initially, hyperemia and edema of the mucoperiosteal lining of the mastoid air cells block the narrow aditus and disrupt aeration. The mucous membrane thickens, and impaired ciliary function prevents normal middle ear drainage through the eustachian tube. The serous exudate becomes purulent as inflammatory cells accumulate. Continued inflammation, hyperemia, and accumulation of purulent debris result in venous stasis, localized acidosis, and decalcification of the bony septa. The osteoclastic activity in the inflamed periosteum softens and decalcifies the bony partitions, causing the small air cells to coalesce into a larger cavity.11
Pathophysiology
As the infection grows, elevated pressure within the mastoid may extend the infection beyond the confines of the mastoid. In the presence of intense inflammation and infection, phlebitis and periphlebitis are common and spread the infection to the adjacent meninges, sigmoid sinus, cerebellum, and temporal lobe.12 Infection may extend to the meninges, sigmoid sinus, labyrinth, or facial nerve. The most common pathway for infection to extend beyond the mastoid is through the lateral cortex behind the ear. Less commonly, it can extend to the soft tissues in the upper portion of the neck (see section on Bezold’s abscess) and, rarely, to the soft tissue anterior and superior to the auricle either by direct extension through eroded bone or by phlebitis and periphlebitis. Go and coworkers13 found only 8 of 118 patients in whom the mastoiditis had caused an intracranial complication.
Diagnosis
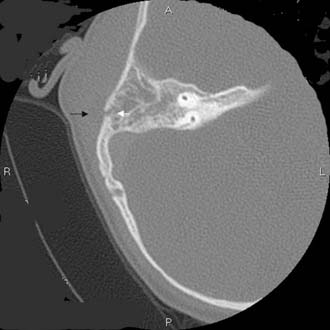
The clinician should order a complete blood count and hematologic studies followed by CT scanning, which can establish the diagnosis by documenting the breakdown of the bony cell walls and opacification of the pneumatized spaces (Figs. 140-2 and 140-3). If any suggestion of an intracranial complication exists, the clinician should obtain an enhanced MRI scan.
Chronic Mastoiditis
Etiology
Chronic mastoiditis can occur in association with a long-standing tympanic membrane perforation, with cholesteatoma, or as a complication from an infection after placement of a middle ear ventilating tube. As noted previously, ventilating tube mastoiditis tends to occur in young children who have experienced water contamination and have undergone cultures and treatment with multiple antibiotic drops and oral antibiotics. Mastoiditis with tympanic membrane perforation occurs when an episode of AOM with perforation pursues a course of chronic infection, rather than resolving or developing into coalescent mastoiditis. Chronic mastoiditis of this type can also begin when a long-established uninfected central perforation becomes infected and extends to the mastoid (Fig. 140-4).